Abstract
New oxidovanadium(IV) complexes of composition [VO(AcSHA)2]
Keywords

Introduction
The ever-increasing research interest in oxidovanadium(IV) complexes is due to their prodigious biological and medicinal properties,1–4 such as insulin-mimetic,5–8 DNA binding and cleavage activity,9–11 anticancer, 12 antioxidant,13–15 antitrypanosomiasis,16,17 antileishmaniasis, 18 antiamebiasis, 19 antituberculosis, 20 and anti-HIV 21 activity inducing apoptosis, inhibiting cell proliferation and preventing the metastasis of cancer against various cell lines.22–24 The model studies related to vanadium biochemistry providing information on vanadium metabolism, toxicity, detoxification, and catalytic activity have been reported. 25 The bis(maltolato) (BMOV), bis(ethylmaltolato) (BEOV), and bis(allixinato)oxidovanadium(IV) are some of the most studied insulin-enhancing oxidovanadium(IV) complexes.26–28 A drug containing vitamin A and vanadium(IV) salt having insulin-mimetic antidiabetic properties has been reported. 8 Oxidovanadium(IV) complexes as YopH tyrosine phosphatase inhibitors, a virulence factor produced by pathogenic species of Yersinia, have also been described. 29 The exponential progress in the medicinal chemistry of biologically relevant vanadium complexes for therapeutic applications is evident from numerous reports.30–33
On the contrary, hydroxamic acids with the functionality RCC(O)N(RN)OH (RC = alkyl/aryl; RN = H or alkyl/aryl) are an important and key class of weak organic acids that have been well studied as bioligands. The distinctive chelating ability34,35—due to keto–enol tautomerism exhibited by the hydroxamic and hydroximic forms 36 —and the vast spectrum of pharmacological, toxicological, and pathological properties of hydroxamic acids have fascinated researchers in the design of new complexes of biological relevance.37–39
Biologically significant acetylated hydroxamic acids synthesized from salicylhydroxamic acid using various acetylating agents and different methodologies have been reported.40,41 The reaction of salicylhydroxamic acid with acetyl chloride in the presence of pyridine and ethyl acetate forms acetylsalicylhydroxamic acid (AcSH2A) where AcSH2A = C6H4(OH)(CONHOCOCH3), and the acetyl group is located on the hydroxamic acid group. The vanadium hydroxamate complexes derived from acetohydroxamic acid (aceto-HAha), benzohydroxamic acid (HBha), N-phenylbenzohydroxamic acid, 2-hydroxypyridine N-oxide, 42 nicotinohydroxamato, 4-nitrobenzohydroxamate, and 2-chloro-4-nitrobenzohydroxamate ligands are also well-documented.43,44
As a part of our continuing work on vanadium hydroxamates,45–47 we herein report the synthesis of new bis(acetylsalicylhydroxamato) and the mixed-ligand (acetylacetonato)(acetylsalicylhydroxamato)oxidovanadium(IV) complexes using two different vanadium precursors along with their characterization by the Fourier-transform infrared spectroscopy (FTIR), ultraviolet–visible spectroscopy (UV-Vis), electron spin resonance (ESR), and mass spectral techniques. The electrochemical and thermal behavior of the complexes has also been studied. The antimicrobial and antioxidant potential of the complexes has been assayed against some pathogenic bacteria and fungi by minimum inhibitory concentration (MIC) method and DPPH (1,1-diphenyl-2-picrylhydrazyl) free radical scavenging method, respectively.
Results and discussion
The reactions of VOSO4.5H2O and VO(acac)2 with 2 equiv. of AcSH2A in absolute ethanol in separate experiments gave dark blue complexes of composition [VO(AcSHA)2]
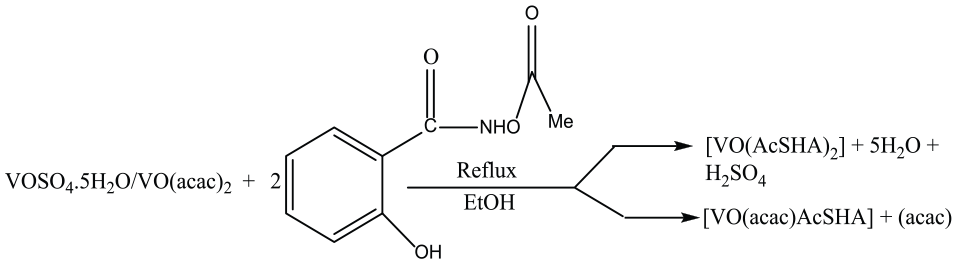
Synthesis of oxidovanadium(IV) complexes.
The molar conductance values of the complexes measured in methanol were 3.92 and 3.90 Scm2 mol−1, respectively, suggesting their non-electrolytic nature.
48
At the room temperature, magnetic moment of 1.70 and 1.72 BM (Bohr magneton) for complexes
Infrared spectra
A comparison of the infrared (IR) spectra of the complexes with that of the ligand supports their formation. Free AcSH2A exhibited bands due to ν(OH)phenolic, ν(N–H), ν(C=O)acetyl, ν(C=O)hydroxamic, ν(C–N), and ν(N–O) modes at 3423, 3345, 1773, 1645, 1348, and 954 cm−1, respectively, whereas the complexes [VO(AcSHA)2] and [VO(acac)(AcSHA)] displayed the respective bands at 3274, 1770, 1603, 1371, and 907 cm−1 and 3392, 1770, 1660–1626, 1348–1342, and 917 cm−1, as presented in Tables 1 and 2, respectively.
IR spectral data of the ligand (AcSH2A) and the complexes

Important IR frequencies (cm−1) of the ligand (AcSH2A) and the complexes

The nonobservance of the band due to the ν(OH) mode in complexes
Electronic spectra
The UV-Vis spectra of the ligand (AcSH2A) recorded in absolute ethanol displayed an absorption band at 319 nm attributed to an intra-ligand π → π* transition. Complexes
UV-Vis spectral data of the ligand (AcSH2A) and the complexes

Mass spectra
The electrospray ionization (ESI) mass spectrum of complex
Mass spectral data of the complex [VO(AcSHA)2]

The other structurally important fragment ions at m/z (%) 451 (21.7), 390 (8.6), 354 (60.8), 262 (34.7), and 234 (20) corresponded to [VO(AcSHA)2 − 6H]+, [2AcSHA], [2AcSHA − 2CH3 − 4H]+, [VO(AcSHA)]+, and [AcSHA + K]+, respectively, as clearly shown in Scheme 2 (see Figure S2 in the supporting information).

Mass spectral fragmentation pattern of complex
The mass spectrum of complex
Mass spectral data of complex [VO(acac)(AcSHA)]

The other fragment ions at m/z (%) 230 (25.4) and 202 (21.8) corresponded to [AcSHA + K+ − 4H]+ and [VO(acac) + K+ − 3H]+, respectively (Scheme 3) (see Figure S3 in the supporting information).

Mass spectral fragmentation pattern of complex
ESR spectra
At the room temperature, X-band ESR spectra of complexes
ESR spectral data of oxidovanadium(IV) complexes

The observed order
Cyclic voltammetry
The free ligand (AcSH2A) displayed one reductive wave and one oxidative wave at −0.61 and −0.51 V, respectively. The complex
Cyclic voltammetric data of ligand (AcSH2A) and oxidovanadium(IV) complexes


Cyclic voltammogram of the free ligand (AcSH2A).
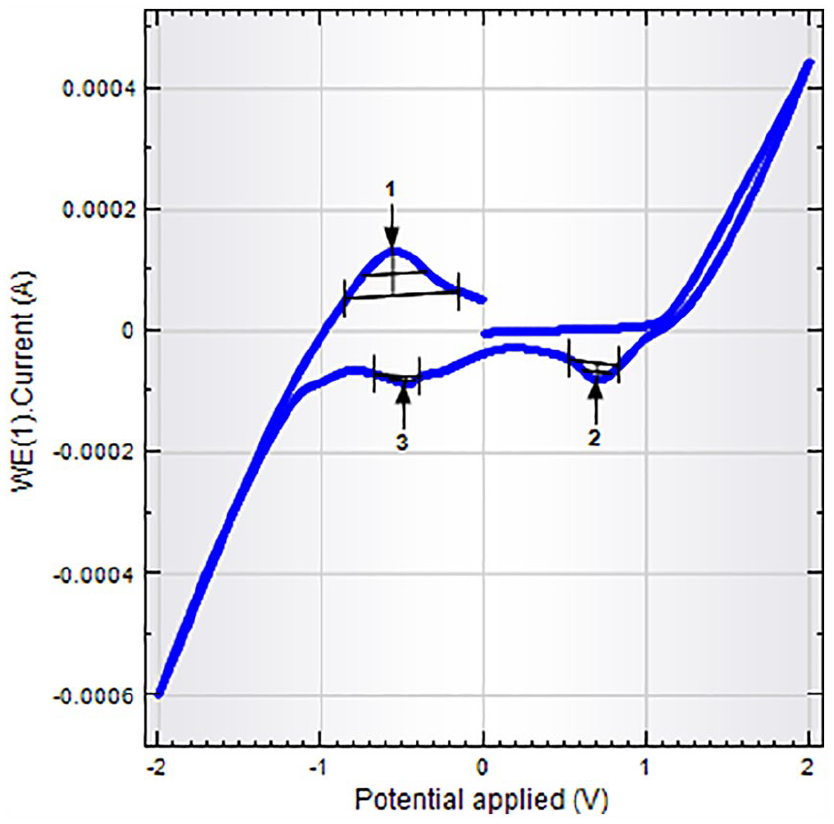
Cyclic voltammogram of the complex

Cyclic voltammogram of the complex
The peak-to-peak separation value (ΔEp = 79 mV) is indicative of a single-step pseudo-reversible one-electron transfer process and also adjudged from the peak current ratio Ipa/Ipc. The electrode process can be represented as

The E1/2 value of 519 mV may be assigned to metal-centered oxidation of VIV to VV lying within the range reported for related complexes. 55 Based upon physicochemical and various spectral techniques, a distorted square pyramidal geometry in both the mononuclear complexes has been proposed around the vanadium (Figures 4 and 5).
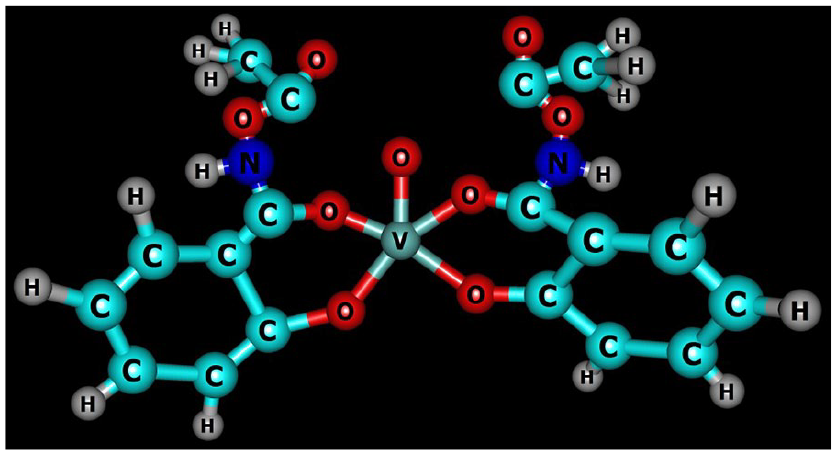
Chemcraft structure of the complex
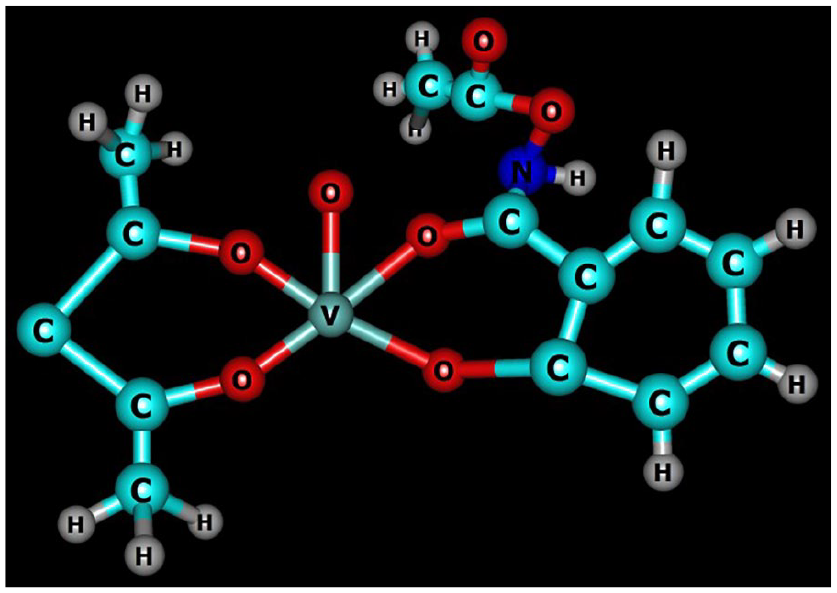
Chemcraft structure of the complex
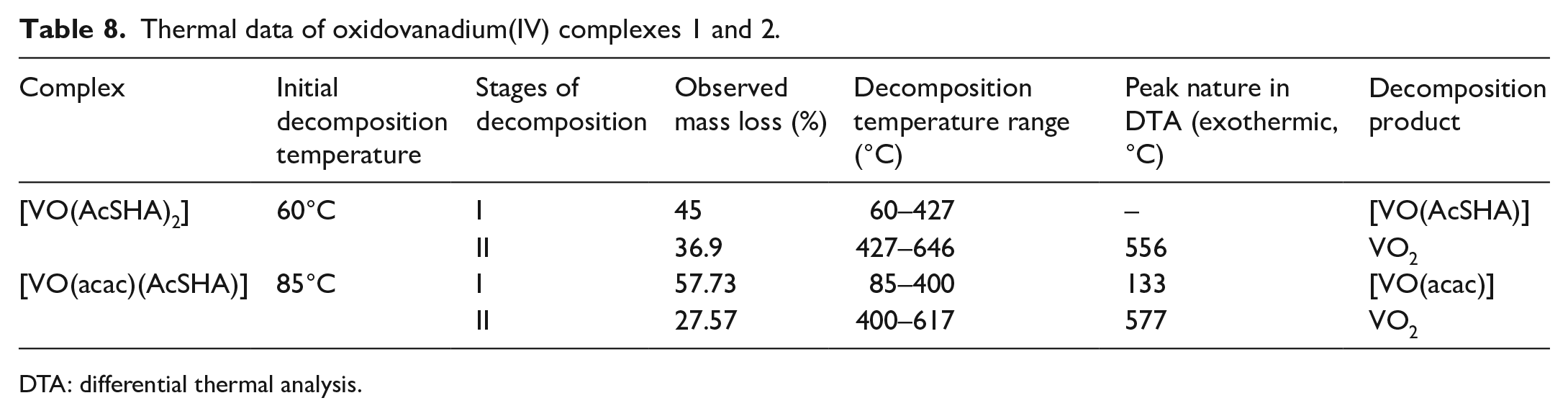
Thermogravimetric studies
The complexes

Thermal decomposition pattern of complex
A broad exothermic peak at 556.0°C in differential thermal analysis (DTA) curve corroborated thermal decomposition in thermogravimetry. The mass loss of 57.73% in complex
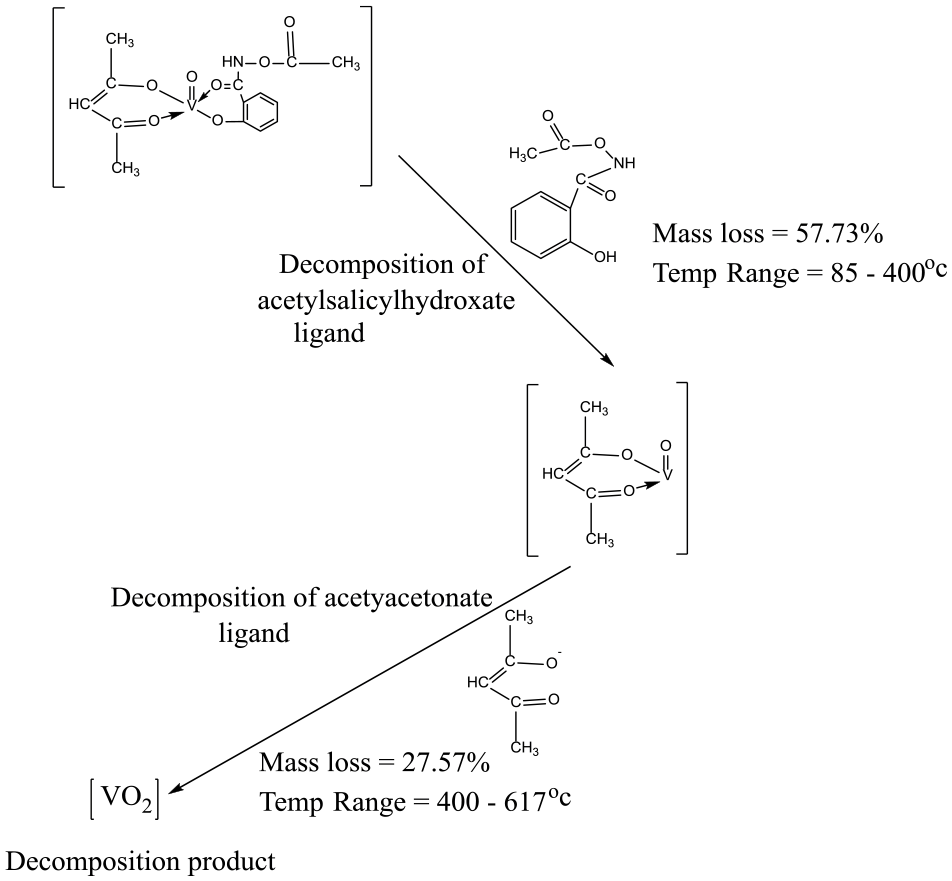
Thermal decomposition pattern of complex
The two-step decomposition in the thermogravimetric analysis (TGA) is accompanied by an exothermic inflection at 133°C and a broad peak at 577°C in the DTA curve (Table 8 and Figures 6 and 7).
Thermal data of oxidovanadium(IV) complexes 1 and 2.
DTA: differential thermal analysis.

TGA and DTA curves of complex

TGA and DTA curves of complex
Biological studies
Literature contains numerous reports on the transport of the pharmacologically active vanadium(IV) and (V) complexes as well as the role of the ligand in various redox and uptake processes, wherein the active species formed depend upon the thermodynamic stability of vanadium complexes and the potential of ligand to cross the cell membrane in bodily fluids at physiological pH.57–69 The oxidovanadium(IV) hydroxamates43,45–47,70,71 have drawn considerable attention owing to their interesting biological activity over the last few years. In pursuit of new vanadium-based antimicrobials, the antimicrobial activity of the complexes
Antibacterial activity
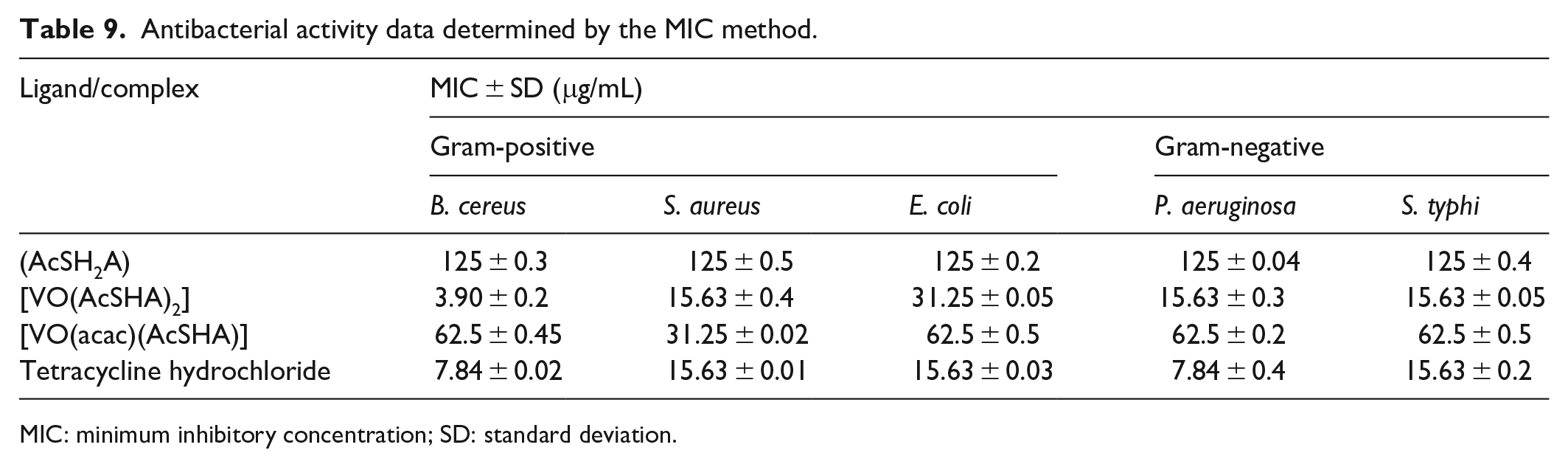
The uncoordinated ligand (AcSH2A) showed inhibitory effects against the test bacteria at MIC 125 µg/mL. The complex
Antibacterial activity data determined by the MIC method.
MIC: minimum inhibitory concentration; SD: standard deviation.

Bar diagram showing the in vitro antibacterial activity studies of the free ligand and the complexes
The complex
Antifungal activity
The antifungal activity of the free ligand and the complexes
Antifungal activity determined by the MIC method.

MIC: minimum inhibitory concentration; SD: standard deviation.

Bar diagram showing the in vitro antifungal activity studies of the free ligand and the complexes
Antioxidant activity
The antioxidant activity of the free ligand and the complexes
Antioxidant activity data of the free ligand and the complexes

This is in agreement with the fact that large conjugated systems upon chelation with metal ions promote the trapping of free radicals, which results in discoloration from the purple DPPH radical solution to a yellow solution showing scavenging of DPPH radicals by hydrogen donation.
Conclusion
The oxidovanadium(IV) complexes of composition [VO(AcSHA)2] and [VO(acac)(AcSHA)] derived from acetylsalicylhydroxamic acid [C6H4(OH)CONHOCOCH3] have been synthesized and thoroughly characterized by various physicochemical and spectroscopic techniques (FTIR, UV-Vis, ESR) and mass spectrometry. IR data showed the disappearance of the ν(OH)phenolic, lowering of the ν(C=O)hydroxamic, and a shift of the ν(N–O) and ν(C–N) modes to a higher frequency, without any significant change in ν(C=O)acetyl. This indicates that the acetylsalicylhydroxamic acid acts as a bidentate chelating ligand involving bonding through carbonyl and phenolic oxygen atoms (O,O). From spectral studies, a distorted square pyramidal geometry may tentatively be proposed around vanadium. The complexes are electrochemically active and exhibit single-step pseudo-reversible one-electron transfer processes. The thermal behavior of complexes
Experimental details
Materials and methods
All chemicals of reagent grade were used. Vanadyl sulfate (VOSO4.5H2O; Merck) was used as procured. [VO(acac)2] and acetylsalicylhydroxamic acid [C6H4(OH)CONHOCOCH3] were synthesized by reported methods,74,75 and characterized by microanalyses and IR spectroscopy. The vanadium content in the complexes was determined as V2O5 by decomposing the complex with a mixture of concentrated H2SO4 and concentrated HNO3. The carbon, hydrogen, and nitrogen analyses were obtained using the CHN elemental analyzer (FLASH 2000; Thermo Scientific). The molar conductance (10−3 M solution in methanol) of the complexes was obtained using a conductivity bridge (Type CM-82T; Elico) at 25°C ± 0.1°C. At the room temperature, magnetic susceptibility was measured by Gouy’s method, 76 using Hg[Co(NCS)4] as a calibrant. The IR spectra were recorded as KBr pellets using a FTIR spectrophotometer (PerkinElmer). Electronic spectra were recorded using a 100 Bio UV-Vis spectrophotometer (Varian Cary) in absolute ethanol. ESI mass spectra were recorded using a Micromass Quattro-TOF (Waters), with a mass range of 4000 amu in quadrupole and 20,000 amu in Tandem time-of-flight (TOF). At the room temperature, powder X-band ESR spectra were recorded using an E112 ESR spectrometer (Varian). The cyclic voltammetric measurements were performed at 25°C on an Autolab Potentiostat 128N electrochemical analyzer using a single compartmental cell of volume 10–15 mL, comprising a three-electrode system of a Pt-disk as the working electrode, Pt-wire as an auxiliary electrode, and Ag/AgCl electrode as the reference electrode. Potassium chloride (KCl; 0.4 M) was used as the supporting electrolyte in the methanol/H2O (5:95) electrolyte system. Thermograms (TGA and DTA curves) of the complexes were recorded using a Diamond TG/DTA thermogravimetric analyzer (PerkinElmer) at a heating rate of 10°C/min from ambient temperature to 800°C under an N2 atmosphere.
Synthesis
Synthesis of (AcSH2A)
To pyridine (15 mL) was added salicylhydroxamic acid (5 g, 32.7 mmol), and the reaction mixture was stirred at room temperature to form an orange-colored solution. After cooling to 0°C, acetyl chloride (2.4 mL, 34 mmol) was added. The mixture was stirred at 0°C for 30 min, followed by stirring at room temperature.75,77 To this solution, ethyl acetate (30 mL), 5% HCl (90 mL), and a saturated solution of sodium chloride (120 mL) were added. The aqueous phase was separated from the organic phase through separating funnel and dried over magnesium sulfate. The solvent was removed using a rotary evaporator to afford a cream-colored solid that was recrystallized from ethyl acetate.
Yield = 2.2 g (32%). Decomposition Temp. = 135°C. The formation of acetylsalicylhydroxamic acid [N-(acetoxy)-2-hydroxybenzamide] was established by the elemental analysis, IR, and 1 H nuclear magnetic resonance (NMR) spectroscopy. Anal. calcd for [C6H4(OH)CONHOCOCH3]: C, 55.20; H, 4.90; N, 7.28; found: C, 54.85; H, 4.65; N, 7.00%. IR (KBr disk): 1805, 1795, 1650, 1620, 1200 cm−1. 1 H NMR (MHz CDCl3): δ 2.45 (s, 3H, CH3) and 7–8 (m, 4H, ArH).
Synthesis of [VO(AcSHA)2] from VOSO4.5H2O
To a solution of VOSO4.5H2O (1 g, 3 mmol) in absolute ethanol (10 mL) was added 2 equiv. of AcSH2A (1.5 g, 7 mmol) dissolved in absolute ethanol (10 mL). The reaction mixture was stirred for 1 h and then refluxed for 8–10 h. The excess solvent was removed by distillation. To the concentrate was added petroleum ether (20 mL), and the product was dried under vacuum. A dark blue solid was obtained.
Yield = 1.2 g (67%), Decomposition Temp. = 145°C, Mol. Mass [C18H16N2O9V] = 457, Anal. calcd for [C18H16N2O9V]: C, 50.05; H, 4.35; N, 3.29; V; 14.32; found: C, 49.56; H, 4.00; N, 3.00; V, 13.98%. ΛM (MeOH): 3.92 Scm2 mol−1; µeff (293 K): 1.70 BM.
Synthesis of [VO(acac)(AcSHA)] from [VO(acac)2]
To a solution of VO(acac)2 (0.5 g, 1 mmol) in absolute ethanol (10 mL) was added 2 equiv. of AcSH2A (1 g, 5 mmol) dissolved in absolute ethanol (10 mL). The reaction mixture was stirred for 1 h and then refluxed for 8–10 h. The excess solvent was removed by distillation. To the concentrate was added petroleum ether (20 mL), and the product was dried under vacuum whereupon a dark blue complex was formed.
Yield = 0.50 g (72%), Decomposition Temp. = 157°C, Mol. Mass [C14H15NO7V] = 360, Anal. calcd for [C14H15NO7V]: C, 50.40; H, 4.27; N, 3.25; V; 14.50; found: C, 50.06; H, 3.95; N, 3.05; V, 13.90%. ΛM (MeOH): 3.90 Scm2 mol−1; µeff (293 K): 1.72 BM.
Biological assay
Antibacterial activity
The free ligand AcSH2A and the oxidovanadium(IV) complexes [VO(AcSHA)2] and [VO(acac)(AcSHA)] were screened in vitro for the antibacterial and antifungal activity against the selected Gram-positive bacteria (S. aureus and B. cereus), Gram-negative bacteria (E. coli, P. aeruginosa, and S. typhi), and fungi (R. solani and F. sambucinum) at different concentrations in dimethyl sulfoxide (DMSO) employing the MIC method.78,79 The MIC is the lowest concentration of the antimicrobial agent that prevents the development of visible growth of microbes after overnight incubation. The samples were tested in triplicate.
Method
A sterile flat bottom 96-well microtiter plate was labeled as 1–7, in which wells 1–5 are allotted for the test sample, 6 for the positive control (broth + standard antibiotic (2 μL) + microorganism), and 7 for the negative control (broth + microorganism). Then, 30 µL of media was added to each well, including the positive and negative controls. A stock solution of the sodium salt of Resazurin dye as an indicator (0.02% (wt/mol); Sigma Aldrich) 80 was prepared in distilled water, filtered, sterilized, and stored at 4°C for 1 week. This dye (5 µL) was added to each well. The test samples were added in twofold dilution to wells (1–5). The plates were then wrapped with aluminum foil and incubated at 37°C for 24 h in the case of bacteria and at 28°C for 72 h for fungi. A change in color in the wells of the positive control indicated the proper growth of the isolate, and no change in color of wells of the negative control indicated the absence of contaminants. The results were compared with the standard antibacterial drug tetracycline hydrochloride and the antifungal drug fluconazole.
Statistical analysis
Statistical analysis was performed using the standard t-test, and p < 0.05 was considered significant. The data are represented as standard deviation.
Antioxidant activity
The free radical scavenging activity of the complexes is measured in terms of hydrogen donation or radical scavenging ability using the stable DPPH radical as described by Blois. 81 DPPH is usually used as a substrate to evaluate antioxidant activity. DPPH (1,1-diphenyl-2-picrylhydrazyl) having an unpaired electron displays a strong absorption band at 517 nm, which decreases stoichiometrically upon pairing of the electron.82,83 A change in the absorbance produced in the reaction is widely used to test the ability of molecules to act as free radical scavengers. The newly synthesized oxidovanadium complexes in +4 oxidation state can donate an electron to reduce the DPPH radical to DPPH (α,α-diphenyl-β-picrylhydrazine), and the VO(IV) ion is oxidized to VO(V). The stock solutions of the complexes (0.001 g/mL) were prepared in DMSO, from which different concentrations (20–100 µL) being prepared. A solution of DPPH (0.1 mmol) in methanol was prepared, and 1 mL of this solution was added to each of the test solutions. The mixture was shaken vigorously and incubated for 30 min. The absorbance was measured at 517 nm. All the tests were carried out in triplicate. Ascorbic acid was used as a standard or positive control. The ability to scavenge the DPPH radical was calculated using the following equation

where Acontrol is the absorbance of the control, and Asample is the absorbance of the sample.
Supplemental Material
Supplementary_material_CH-19-0331 – Supplemental material for Synthesis, characterization, and biological properties of oxidovanadium(IV) complexes of acetylsalicylhydroxamic acid (N-acetyloxy-2-hydroxybenzamide) as potential antimicrobials
Supplemental material, Supplementary_material_CH-19-0331 for Synthesis, characterization, and biological properties of oxidovanadium(IV) complexes of acetylsalicylhydroxamic acid (N-acetyloxy-2-hydroxybenzamide) as potential antimicrobials by Bhanu Priya, Abhishek Kumar and Neeraj Sharma in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank the IIT Mandi for recording Fourier-transform infrared spectroscopy (FTIR), mass spectra, and elemental analyses. The authors gratefully acknowledge the support of the Department of Biotechnology, Himachal Pradesh University, Shimla, for providing laboratory facilities to carry out biological studies.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
