Abstract
The kinetics of the aquation reaction of the [VO(ida)(bipy)]·2H2O (VO(ida)(bipy)) complex (where ida = iminodiacetate anion and bipy = 2,2’-bipyridine) promoted by [Fe(H2O)6]3+ ions were investigated in aqueous solutions. Spectrophotometric studies were carried out at different temperatures in the range of 293.15–313.15 K. The concentration of the [Fe(H2O)6]3+ (Fe3+) ions was kept within the range of 2 × 10–4 to 8 × 10–4 mol L–1, and the concentration of VO(ida)(bipy) was 1 × 10–3 mol L–1. The values of the observable reaction rate constants were calculated based on the Glint computer program. Furthermore, the mechanism for the aquation of VO(ida)(bipy), induced by Fe(III) ions, has been proposed.
Introduction
Aquation is a type of substitution reaction. During the aquation process, the ligand is substituted by water molecules. Recently, interest in the kinetics of the complexation reaction of transition metal compounds has increased.1–8 For many coordination compounds, the aquation requires the introduction of a special promoter. The substitution reaction of complex compounds with special promoters may occur in acidic (the promoters are H+ ions) or basic (the promoters are OH– ions) media or may be induced with Fe3+ ions.1–8
Polycarboxylic anions are a group of organic compounds which are used in coordination chemistry as ligands,1–20 because they stabilize metal ions in solutions 14 and, moreover, they are noncytotoxic. 15 Furthermore, the polycarboxylate coordination compounds of cobalt(II), nickel(II),12,16,18 oxidovanadium(IV),15,17 and copper(II)19,20 have interesting antioxidant, antibacterial, and antifungal activities.
The kinetics of the aquation reaction of oxydiacetate complex compounds of nickel(II), cobalt(II), and chromium(III) induced by Fe(III) ions have been known in the literature.6–8 Knowledge of the mechanisms of substitution in coordination compounds is important to understand processes involving the activity of enzymes activated by metal ions or the transport of metal ions across the cell membrane. 21
In this work, for the first time, we try to describe the kinetics of the aquation reaction of oxidovanadium(IV) complex. Consequently, the aquation of [VO(ida)(bipy)]·2H2O (VO(ida)(bipy)) (Figure 1) induced by Fe3+ ions has been investigated. The values of the reaction rate constants were calculated for the spontaneous step of the reaction as well as for the step of reaction induced by Fe3+ ions. Finally, the mechanism for the aquation of VO(ida)(bipy), induced by Fe(III) ions, has been proposed.
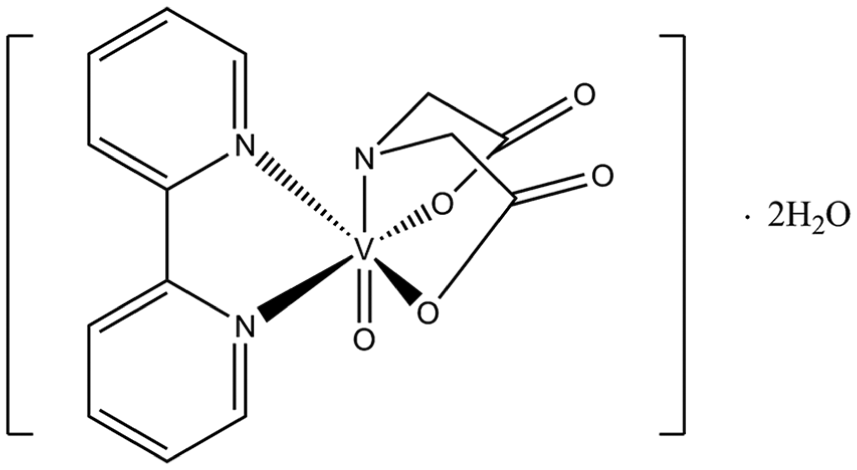
2,2’-Bipyridyl(iminodiacetato)oxidovanadium(IV) dihydrate.
Experimental analysis
Synthesis
[VO(ida)(bipy)]·2H2O was synthesized based on the known method described in the literature. 13 The 2,2’-bipyridyl(iminodiacetato)oxidovanadium(IV) complex was prepared by the reaction of portions of VO(acac)2 (5.30 g) and bipy (3.12 g) with the solution of H2ida (2.66 g) in 100 mL of water. The mixture in a round-bottomed flask was heated for about 1 h at 50°C with magnetic stirring. Then, after several days, dark brown crystals of VO(ida)(bipy) were obtained.
Elemental analyses of the 2,2’-bipyridyl(iminodiacetato)oxidovanadium(IV) complex were performed with the Vario EL analyzer Cube CHNS. Calculations: C, 43.08%; H, 4.36%; N, 10.77%. Found: C, 43.08%; H, 4.36%; N, 10.74%.
Kinetic measurements
The kinetic measurements of the aquation reaction of VO(ida)(bipy) have been carried out spectrophotometrically. Based on the recorded ultraviolet–visible (UV–Vis) spectra of the substrate, the promoter, and the products of the aquation reaction, a wavelength for kinetic studies was chosen based on the largest differences in the absorbance between the substrate and the product. Thus, the progress of the aquation reaction of VO(ida)(bipy), promoted by Fe3+ ions, was monitored at 510 nm. This spectral wavelength is not sensitive to absorption of [Fe(ida)(H2O)3]+. The product of the aquation - [VO(bipy)(H2O)3]2+ is responsible for the absorption at the wavelength at which the reaction studied was spectrophotometrically monitored. The studies have been carried out at five temperatures within the range of 293.15–313.15 K. The concentration of the [Fe(H2O)6]3+ ions was kept within the range of 2 × 10–4 to 8 × 10–4 mol L–1, and the concentration of [VO(ida)(bipy)] was constant and was equal to 1 × 10–3 mol L–1. The ionic strength was 1 mol L–1 (Na+, NO3–), while the pH of the solution was adjusted to 2.5. The values of the observable reaction rate constants as well as the values of the reaction rate constants for the spontaneous step of reaction as well as for the step of reaction induced by Fe3+ ions were calculated.
Instrumentation
The kinetic measurements were carried out using the UV–Vis Lambda 45 PerkinElmer spectrophotometer with the system of temperature control, with a scan accuracy of 1 nm and 1-nm slit width at a 120.00 nm min–1 scanning rate. The values of the observable rate constants were calculated using the “Glint” program based on global analysis. 22
Results and discussion
The kinetics of the aquation of the VO(ida)(bipy) complex induced by Fe3+ ions were studied at five temperatures, within the range of 293.15–313.15 K, in the five promoter concentrations in the range of 2 × 10–4 to 8 × 10–4 mol L–1, whereas the concentration of the coordination compound, that is, VO(ida)(bipy), was constant (1 × 10–3 mol L–1). Fe3+ acts as a promoter because [Fe(ida)(H2O)3]+ has the very high stability constant (logβ1 is 10.72). 23 The values of the observable reaction rate constants (kobs) increased with the Fe3+ concentration and the temperature (Table 1), which was consistent with the formation of the proposed intermediate complex (Figure 2). The values of the observable rate constants were calculated using the “Glint” program by the global analysis method based on the A → B model reaction, which may be described using equation (1)
The promoter concentration in the 2 × 10–4 to 8 × 10–4 mol L–1 range of the Fe3+ ions dependence of the observable rate constants kobs (s–1) of the aquation reaction investigated at five temperatures (together with standard deviations).
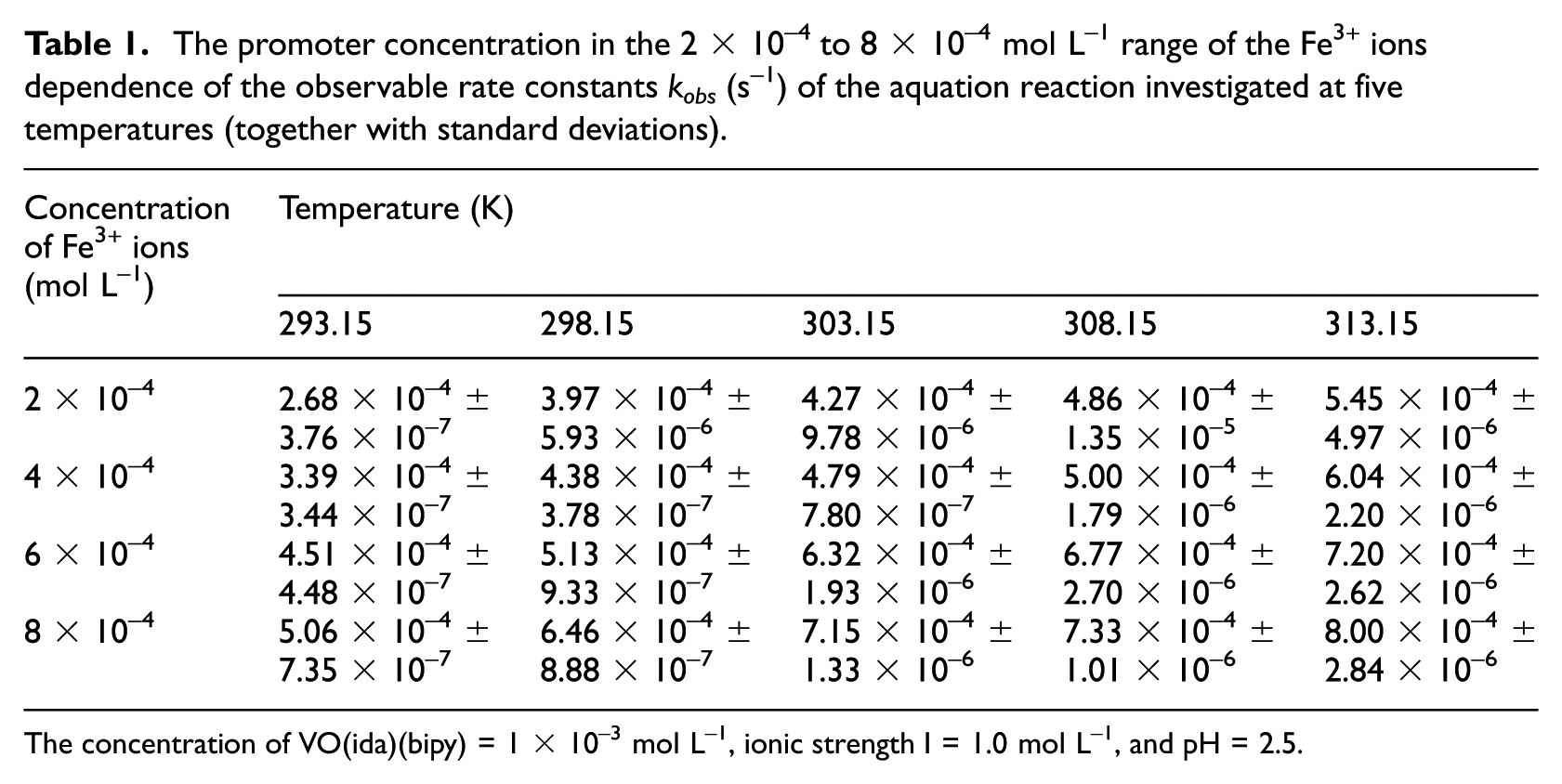
The concentration of VO(ida)(bipy) = 1 × 10–3 mol L–1, ionic strength I = 1.0 mol L–1, and pH = 2.5.
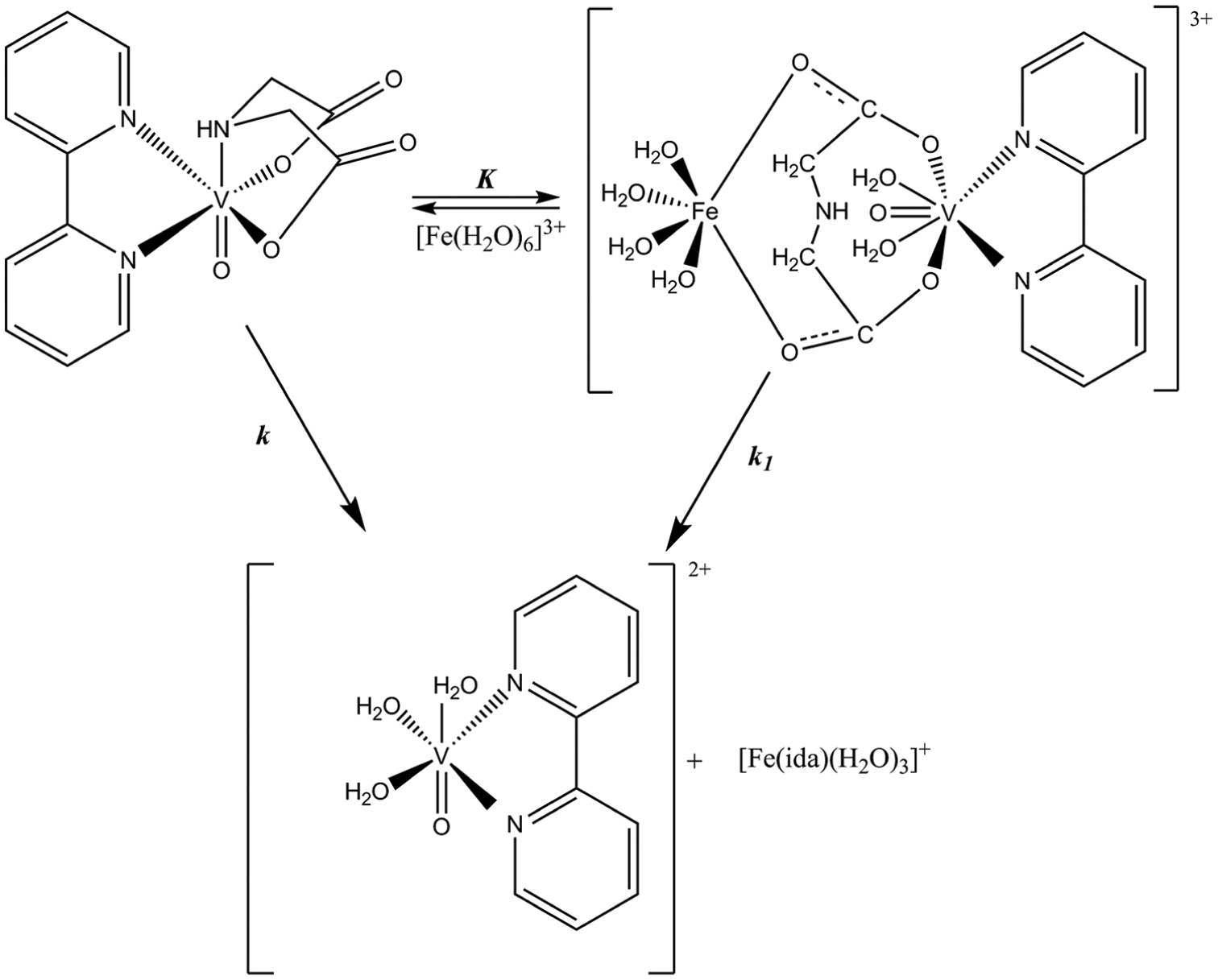
The proposed mechanism of the substitution reaction of the iminodiacetate anion in VO(ida)(bipy) induced by [Fe(H2O)6]3+.
where A is the substrate concentration, B is the product concentration, k is the reaction rate constant, kobs is the observed rate constant, V is the reaction rate, and t is the time period.
The values of the first-order rate constants as a function of the [Fe(H2O)6]3+ concentration occur as linear dependences at the five temperatures studied. These results are compatible with equation (2):
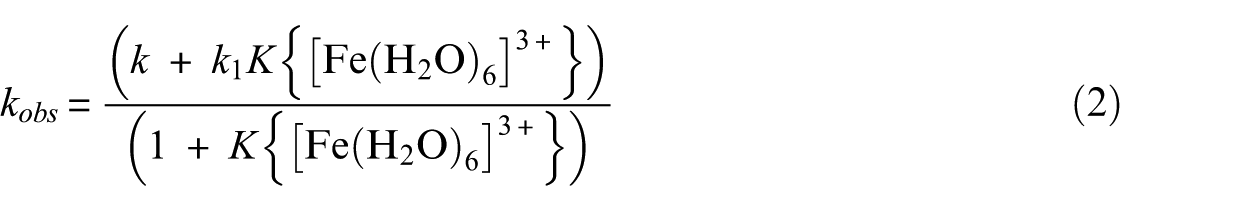
where k is the rate constant of spontaneous reaction in the absence of Fe3+, k1 is the interchange rate constant for a process in which the intermediate undergoes decomposition to give the products, and K is the precursor formation constant describing a fast pre-equilibrium step between the reactants and the intermediate product (a heterodinuclear complex compound with increased coordination number in comparison to VO(ida)(bipy)). However, in the case of the above equation, a limitation is observed because if 1 >> K{[Fe(H2O)6]3+}, this equation is simplified to kobs = k + k1K{[Fe(H2O)6]3+}. Analyzing the dependence of the values of kobs on the Fe3+ concentration (Table 1), it can be concluded that for the Fe3+ concentration equal to 0 mol L–1, the values of the observable rate constants are low, but are not equal to zero. This means that the spontaneous aquation reaction occurs simultaneously with that promoted by Fe3+ ions. However, this process is too slow to be accurately measurable.
VO(ida)(bipy) in aqueous solutions undergoes the aquation reaction induced by Fe3+ ions, resulting in the substitution of the iminodiacetate anion by three water molecules. Based on the analysis of the kinetic relationships, it can be proposed that the investigated reaction proceeds in two parallel paths (Figure 2). The first path is promoter-independent (k) and is likely to proceed according to a dissociative interchange mechanism, where dissociation of the VO(ida)(bipy) is present. The second path consists of two steps. The first one is fast and reversible (K). It depends on the concentration of Fe(III) and results in the formation of a binuclear intermediate complex. In the second, the rate-controlling step, which is the decomposition of the intermediate, takes place (k1), resulting in the formation of the [VO(bipy)(H2O)3]2+ and [Fe(ida)(H2O)3]+ species.
The values of the spontaneous reaction rate constant k and the apparent secondary rate constants
The values of the rate constant k for the spontaneous reaction and the apparent secondary rate constant

The concentration of VO(ida)(bipy) = 1 × 10–3 mol L–1, ionic strength I = 1.0 mol L–1 (NaNO3), and pH = 2.5.
The analysis of the results presented in Table 2 shows that the values of
Conclusion
The aquation of VO(ida)(bipy) has been monitored spectrophotometrically in the presence of the Fe3+ ions as a promoter. It has been found that the values of the observable rate constants kobs increase proportionally to the concentration of the promoter over the entire range of temperatures studied (293.15–313.15 K), which is consistent with the theory of the active complex. The values of the rate constants are lower for the spontaneous step of the aquation than for the step dependent on the promoter. The dissociation of the intermediate binuclear complex has the greatest impact on the rate of the aquation of VO(ida)(bipy) induced by Fe(III) ions. The mechanism for this reaction has been proposed. The first path is promoter-independent and is likely to proceed according to a dissociative interchange mechanism. The second path consists of two steps. In the first step (the reversible part of the aquation reaction dependent on the Fe3+ ions), a dinuclear intermediate product is formed, and in the next step, the products of the reaction are created - [VO(bipy)(H2O)3]2+ and [Fe(ida)(H2O)3]+.

