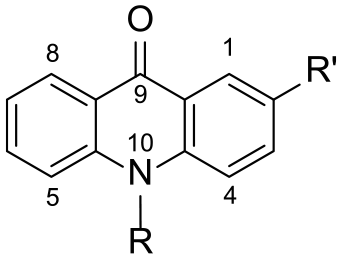
Preparation of compounds 2i and 3i
A solution of acridone (1 mmol) in N,N-dimethylformamide (10 mL) was added to a stirred suspension of sodium hydride (3 mmol) in N,N-dimethylformamide (10 mL). The mixture was heated at 80 °C for 15 min, treated with propyl iodide (for 2i) or 1-bromobutane (for 3i) (5 mmol), and heated at 80 °C for an additional 0.5 h. The reaction mixture was cooled and then water was added and the mixture was extracted with ethyl acetate. The combined organic layer was washed with brine and water, dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was purified by silica gel column chromatography.
10-(3′-N-Piperidinopropyl)acridone (
2a
):
23
(67%) yellow solid, m.p. 135.0 °C−136.0 °C; FTIR, νmax (cm−1): 1634, 1598, 1489, 1462, 1377, 1262, 1177, 753, 674; 1H NMR (300 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.5 Hz, 2H), 7.80−7.60 (m, 4H), 7.29 (t, J = 7.6 Hz, 2H), 4.47 (t, J = 8.4 Hz, 2H), 2.55−2.40 (m, 6H), 2.10−2.00 (m, 2H), 1.75−1.60 (m, 4H), 1.60−1.45 (m, 2H); 13C NMR (75 MHz, CDCl3): δ 178.0, 141.9 (2C), 133.8 (2C), 127.9 (2C), 122.4 (2C), 121.1 (2C), 114.8 (2C), 55.7, 54.8 (2C), 44.1, 26.1 (2C), 24.8, 24.4; MS (EI): m/z (relative intensity): 320 (M+, 8), 208 (4), 180 (4), 124 (10), 98 (100); HRMS (APCI): m/z calcd for C21H25N2O [M + H]+: 321.1961; found: 321.1954.
10-(4′-N-Piperidinobutyl)acridone (
3a
):
23
(96%) yellow solid, m.p. 115.5 °C−116.0 °C; FTIR, νmax (cm−1): 1633, 1598, 1490, 1461, 1377, 1264, 1177, 752, 732, 673; 1H NMR (400 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.2 Hz, 2H), 7.74−7.60 (m, 4H), 7.31−7.25 (m, 2H), 4.37 (t, J = 7.6 Hz, 2H), 2.50−2.40 (m, 6H), 2.00−1.91 (m, 2H), 1.80−1.70 (m, 2H), 1.70−1.60 (m, 4H), 1.60−1.55 (m, 2H); 13C NMR (100 MHz, CDCl3): δ 178.0, 141.7 (2C), 133.8 (2C), 127.8 (2C), 122.4 (2C), 121.1 (2C), 114.8 (2C), 57.9, 54.6 (2C), 45.9, 26.1 (2C), 24.6, 24.5, 23.6; MS (EI): m/z (relative intensity): 334 (M+, 44), 208 (10), 138 (21), 98 (100); HRMS (APCI): m/z calcd for C22H27N2O [M + H]+: 335.2118; found: 335.2125.
10-(5′-N-Piperidinopentyl)acridone (
4a
): (77%) yellow solid, m.p. 121.5 °C−122.5 °C; FTIR, νmax (cm−1): 1632, 1597, 1490, 1460, 1376, 1290, 1262, 1176, 753, 673; 1H NMR (300 MHz, CDCl3): δ 8.57 (dd, J = 8.1, 1.5 Hz, 2H), 7.74−7.68 (m, 2H), 7.47 (d, J = 8.7 Hz, 2H), 7.28 (t, J = 7.5 Hz, 2H), 4.32 (t, J = 8.4 Hz, 2H), 2.38−2.31 (m, 6H), 2.00−1.89 (m, 2H), 1.70−1.44 (m, 10H); 13C NMR (75 MHz, CDCl3): δ 177.9, 141.7 (2C), 133.8 (2C), 127.9 (2C), 122.4 (2C), 121.1 (2C), 114.5 (2C), 59.2, 54.7 (2C), 46.1, 27.0, 26.7, 26.0 (2C), 25.0, 24.4; MS (EI): m/z (relative intensity): 348 (M+, 30), 208 (13), 154 (14), 98 (100); HRMS (APCI): m/z calcd for C23H29N2O [M + H]+: 349.2274; found: 349.2281.
10-(6′-N-Piperidinohexyl)acridone (
5a
): (77.5%) yellow solid, m.p. 81.0 °C−82.0 °C; FTIR, νmax (cm−1): 1631, 1598, 1492, 1460, 1377, 1289, 1263, 1178, 756, 674; 1H NMR (400 MHz, CDCl3): δ 8.59 (dd, J = 8.0, 1.6 Hz, 2H), 7.78−7.73 (m, 2H), 7.50 (d, J = 8.8 Hz, 2H), 7.30 (t, J = 7.2 Hz, 2H), 4.36 (t, J = 8.0 Hz, 2H), 3.00−2.77 (m, 6H), 2.00−1.75 (m, 8H), 1.66−1.40 (m, 6H); 13C NMR (100 MHz, CDCl3): δ 177.9, 141.7 (2C), 134.0 (2C), 128.0 (2C), 122.5 (2C), 121.2 (2C), 114.5 (2C), 57.9, 53.7 (2C), 45.8, 27.1, 26.8, 26.4, 24.5, 23.5 (2C), 22.7; MS (EI): m/z (relative intensity): 362 (M+, 5), 208 (12), 149 (25), 98 (98), 86 (100); HRMS (APCI): m/z calcd for C24H31N2O [M + H]+: 363.2431; found: 363.2438.
10-(3′-N-Morpholinopropyl)acridone (
2b
):
23
(78.5%) yellow solid, m.p. 114.0 °C−114.5 °C; FTIR, νmax (cm−1): 1633, 1598, 1491, 1462, 1378, 1290, 1263, 1178, 1115, 754, 674; 1H NMR (300 MHz, CDCl3): δ 8.59 (dd, J = 8.0, 1.2 Hz, 2H), 7.75−7.62 (m, 4H), 7.29 (t, J = 7.4 Hz, 2H), 4.49 (t, J = 7.5 Hz, 2H), 3.80 (t, J = 4.5 Hz, 4H), 2.55−2.47 (m, 6H), 2.12−2.02 (m, 2H); 13C NMR (75 MHz, CDCl3): δ 178.0, 141.8 (2C), 133.8 (2C), 128.0 (2C), 122.4 (2C), 121.2 (2C), 114.6 (2C), 67.0 (2C), 55.4, 53.8 (2C), 43.7, 24.3; MS (EI): m/z (relative intensity): 322 (M+, 6), 279 (8), 222 (12), 208 (10), 180 (9), 100 (100); HRMS (APCI): m/z calcd for C20H23N2O2 [M + H]+: 323.1754; found: 323.1750.
10-(4′-N-Morpholinobutyl)acridone (
3b
):
23
(73%) yellow solid, m.p. 143.5 °C−144.5 °C; FTIR, νmax (cm−1): 1632, 1600, 1493, 1462, 1378, 1265, 1116, 731, 703; 1H NMR (400 MHz, CDCl3): δ 8.57 (dd, J = 8.0, 1.6 Hz, 2H), 7.74−7.69 (m, 2H), 7.58 (d, J = 8.4 Hz, 2H), 7.30−7.26 (m, 2H), 4.37 (t, J = 8.3 Hz, 2H), 3.78 (t, J = 4.8 Hz, 4H), 2.55−2.47 (m, 6H), 2.02−1.93 (m, 2H), 1.82−1.73 (m, 2H); 13C NMR (100 MHz, CDCl3): δ 177.9, 141.7 (2C), 133.8 (2C), 127.9 (2C), 122.4 (2C), 121.2 (2C), 114.6 (2C), 67.0 (2C), 57.6, 53.7 (2C), 45.8, 24.4, 23.2; MS (EI): m/z (relative intensity): 336 (M+, 10), 293 (14), 222 (9), 208 (23), 180 (15), 142 (14), 100 (100); HRMS (APCI): m/z calcd for C21H25N2O2 [M + H]+: 337.1911; found: 337.1908.
10-(5′-N-Morpholinopentyl)acridone (
4b
): (75%) yellow solid, m.p. 116.5 °C−117.0 °C; FTIR, νmax (cm−1): 1633, 1597, 1490, 1460, 1376, 1290, 1261, 1176, 1116, 753, 673; 1H NMR (400 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.6 Hz, 2H), 7.75−7.70 (m, 2H), 7.48 (d, J = 8.7 Hz, 2H), 7.29 (t, J = 7.6 Hz, 2H), 4.34 (t, J = 8.2 Hz, 2H), 3.74 (t, J = 4.6 Hz, 4H), 2.50−2.37 (m, 6H), 2.02−1.91 (m, 2H), 1.70−1.54 (m, 4H); 13C NMR (100 MHz, CDCl3): δ 177.9, 141.7 (2C), 133.8 (2C), 128.0 (2C), 122.5 (2C), 121.2 (2C), 114.4 (2C), 66.9 (2C), 58.7, 53.8 (2C), 46.0, 27.0, 26.2, 24.7; MS (EI): m/z (relative intensity): 350 (M+, 4), 332 (9), 319 (15), 307 (19), 264 (20), 222 (18), 208 (41), 180 (20), 100 (100); HRMS (APCI): m/z calcd for C22H27N2O2 [M + H]+: 351.2067; found: 351.2065.
10-(6′-N-Morpholinohexyl)acridone (
5b
): (77%) yellow solid, m.p. 95.5 °C−96.0 °C; FTIR, νmax (cm−1): 1634, 1598, 1490, 1460, 1376, 1290, 1262, 1176, 1116, 753, 673; 1H NMR (400 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.5 Hz, 2H), 7.74−7.69 (m, 2H), 7.48 (d, J = 8.7 Hz, 2H), 7.31−7.26 (m, 2H), 4.32 (t, J = 8.2 Hz, 2H), 3.73 (t, J = 4.6 Hz, 4H), 2.47–2.42 (m, 4H), 2.36 (t, J = 7.3 Hz, 2H), 1.98−1.89 (m, 2H), 1.62−1.52 (m, 4H), 1.50−1.43 (m, 2H); 13C NMR (100 MHz, CDCl3): δ 177.9, 141.7 (2C), 133.8 (2C), 127.9 (2C), 122.4 (2C), 121.1 (2C), 114.5 (2C), 66.9 (2C), 58.9, 53.8 (2C), 46.0, 27.2, 27.1, 26.8, 26.5; MS (EI): m/z (relative intensity): 364 (M+, 4) 346 (23), 333 (37), 321 (32), 277 (16), 222 (18), 208 (43), 100 (100); HRMS (APCI): m/z calcd for C23H29N2O2 [M + H]+: 365.2224; found: 365.2225.
10-(3′-N-Pyrrolidinopropyl)acridone (
2c
): (69%) yellow solid, m.p. 70.5 °C−71.5 °C; FTIR, νmax (cm−1): 1633, 1598, 1490, 1461, 1377, 1290, 1263, 1177, 753, 674; 1H NMR (300 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.2 Hz, 2H), 7.77−7.68 (m, 2H), 7.64 (d, J = 8.7 Hz, 2H), 7.29 (t, J = 7.5 Hz, 2H), 4.50 (t, J = 7.7 Hz, 2H), 2.75−2.62 (m, 6H), 2.22−2.11 (m, 2H), 1.92−1.85 (m, 4H); 13C NMR (75 MHz, CDCl3): δ 178.0, 141.8 (2C), 133.9 (2C), 127.9 (2C), 122.4 (2C), 121.2 (2C), 114.7 (2C), 54.2 (2C), 52.9, 43.9, 26.4, 23.5 (2C); MS (EI): m/z (relative intensity): 306 (M+, 13), 208 (6), 180 (6), 110 (7), 84 (100); HRMS (APCI): m/z calcd for C20H23N2O [M + H]+: 307.1805; found: 307.1819.
10-(4′-N-Pyrrolidinobutyl)acridone (
3c
): (93%) yellow solid, m.p. 108.5 °C−109.5 °C; FTIR, νmax (cm−1): 1633, 1597, 1490, 1461, 1378, 1264, 1177, 752, 673; 1H NMR (400 MHz, CDCl3): δ 8.57 (dd, J = 8.0, 1.6 Hz, 2H), 7.73−7.68 (m, 2H), 7.59 (d, J = 8.7 Hz, 2H), 7.30−7.25 (m, 2H), 4.37 (t, J = 8.3 Hz, 2H), 2.62−2.53 (m, 6H), 2.03−1.94 (m, 2H), 1.86−1.73 (m, 6H); 13C NMR (100 MHz, CDCl3): δ 177.9, 141.7 (2C), 133.8 (2C), 127.9 (2C), 122.4 (2C), 121.1 (2C), 114.7 (2C), 55.3, 54.1 (2C), 45.9, 25.7, 24.9, 23.5 (2C) ppm; MS (EI): m/z (relative intensity): 320 (M+, 25), 208 (9), 180 (11), 124 (21), 84 (100); HRMS (APCI): m/z calcd for C21H25N2O [M + H]+: 321.1961; found: 321.1962.
10-(5′-N-Pyrrolidinopentyl)acridone (
4c
): (73%) yellow solid, m.p. 113.0 °C−114.0 °C; FTIR, νmax (cm−1): 1633, 1597, 1490, 1460, 1377, 1290, 1263, 1176, 753, 673; 1H NMR (400 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.4 Hz, 2H), 7.75−7.70 (m, 2H), 7.49 (d, J = 8.7 Hz, 2H), 7.29 (t, J = 7.4 Hz, 2H), 4.34 (t, J = 8.2 Hz, 2H), 2.58−2.50 (m, 6H), 2.01−1.91 (m, 2H), 1.85−1.78 (m, 4H), 1.74−1.56 (m, 4H); 13C NMR (100 MHz, CDCl3): δ 177.9, 141.8 (2C), 133.8 (2C), 128.0 (2C), 122.5 (2C), 121.2 (2C), 114.5 (2C), 56.3, 54.3 (2C), 46.1, 28.6, 27.1, 25.0, 23.4 (2C); MS (EI): m/z (relative intensity): 334 (M+, 13), 208 (11), 180 (8), 140 (17), 84 (100); HRMS (APCI): m/z calcd for C22H27N2O [M + H]+: 335.2118; found: 335.2106.
10-(6′-N-Pyrrolidinohexyl)acridone (
5c
): (71%) yellow solid, m.p. 76.0 °C−77.0 °C; FTIR, νmax (cm−1): 1630, 1596, 1491, 1460, 1377, 1263, 1177, 755, 674; 1H NMR (300 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.5 Hz, 2H), 7.77−7.72 (m, 2H), 7.50 (d, J = 8.7 Hz, 2H), 7.30 (t, J = 7.5 Hz, 2H), 4.34 (t, J = 8.0 Hz, 2H), 3.08 (br s, 4H), 2.88 (t, J = 7.9 Hz, 2H), 2.10−1.81 (m, 8H) 1.63−1.45 (m, 4H); 13C NMR (75 MHz, CDCl3): δ 177.9, 141.7 (2C), 134.0 (2C), 127.9 (2C), 122.4 (2C), 121.2 (2C), 114.5 (2C), 55.6, 53.7 (2C), 45.8, 27.0, 26.8, 26.5, 26.4, 23.3 (2C); MS (EI): m/z (relative intensity): 348 (M+, 13), 208 (8), 180 (9), 110 (11), 84 (100); HRMS (APCI): m/z calcd for C23H29N2O [M + H]+: 394.2274; found: 349.2283.
10-(3′-(N-Pyrrole)propyl)acridone (
2d
): (78%) yellow solid, m.p. 154.5 °C−155.5 °C; FTIR, νmax (cm−1): 1632, 1598, 1491, 1461, 1291, 1264, 1177, 732, 673; 1H NMR (300 MHz, CDCl3): δ 8.54 (dd, J = 8.0, 1.6 Hz, 2H), 7.68−7.60 (m, 2H), 7.26 (t, J = 7.0 Hz, 2H), 7.13 (d, J = 8.8 Hz, 2H), 6.78 (t, J = 2.0 Hz, 2H), 6.30 (t, J = 2.1 Hz, 2H), 4.25 (t, J = 8.1 Hz, 2H), 4.15 (t, J = 6.2 Hz, 2H), 2.40−2.26 (m, 2H); 13C NMR (75 MHz, CDCl3): δ 177.9, 141.4 (2C), 134.0 (2C), 128.0 (2C), 122.4 (2C), 121.3 (2C), 120.8 (2C), 114.0 (2C), 109.1 (2C), 46.8, 43.1, 28.9; MS (EI): m/z (relative intensity): 302 (M+, 41), 222 (62), 208 (35), 180 (20), 152 (13), 81 (100); HRMS (APCI): m/z calcd for C20H19N2O [M + H]+: 303.1492; found: 303.1494.
10-(4′-(N-Pyrrole)butyl)acridone (
3d
): (66%) yellow solid, m.p. 145.0 °C−146.0 °C; FTIR, νmax (cm−1): 1633, 1603, 1594, 1491, 1458, 1377, 1172, 752, 726, 673; 1H NMR (300 MHz, CDCl3): δ 8.56 (d, J = 8.0, 1.6 Hz, 2H), 7.72−7.65 (m, 2H), 7.35 (d, J = 8.7 Hz, 2H), 7.30−7.22 (m, 2H), 6.69 (t, J = 2.1 Hz, 2H), 6.19 (t, J = 2.1 Hz, 2H), 4.25 (t, J = 8.2 Hz, 2H), 4.00 (t, J = 6.8 Hz, 2H), 2.08−1.84 (m, 4H); 13C NMR (75 MHz, CDCl3): δ 177.9, 141.6 (2C), 133.9 (2C), 128.0 (2C), 122.4 (2C), 121.3 (2C), 120.4 (2C), 114.4 (2C), 108.5 (2C), 49.0, 45.6, 28.6, 24.4; MS (EI): m/z (relative intensity): 316 (M+, 40), 222 (23), 208 (100), 180 (31), 152 (16), 122 (49), 80 (46); HRMS (APCI): m/z calcd for C21H21N2O [M + H]+: 317.1648; found: 317.1640.
10-(5′-(N-Pyrrole)pentyl)acridone (
4d
): (63%) yellow solid, m.p. 109.0 °C−110.0 °C; FTIR, νmax (cm−1): 1630, 1597, 1492, 1458, 1375, 1279, 1174, 749, 720, 699; 1H NMR (300 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.6 Hz, 2H), 7.74−7.68 (m, 2H), 7.43 (d, J = 8.7 Hz, 2H), 7.29 (t, J = 7.2 Hz, 2H), 6.67 (t, J = 2.0 Hz, 2H), 6.18 (t, J = 2.0 Hz, 2H), 4.29 (t, J = 8.2 Hz, 2H), 3.94 (t, J = 7.0 Hz, 2H), 1.96−1.87 (m, 4H), 1.57−1.47 (m, 2H); 13C NMR (75 MHz, CDCl3): δ 177.9, 141.7 (2C), 133.8 (2C), 128.0 (2C), 122.5 (2C), 121.2 (2C), 120.4 (2C), 114.4 (2C), 108.1 (2C), 49.2, 45.8, 31.2, 26.8, 24.1; MS (EI): m/z (relative intensity); 330 (M+, 35), 236 (26), 222 (46), 208 (100), 195 (17), 180 (40), 149 (27), 80 (43); HRMS (APCI): m/z calcd for C22H23N2O [M + H]+: 331.1805; found: 331.1804.
10-(6′-(N-Pyrrole)hexyl)acridone (
5d
): (70%) yellow solid, m.p. 82.0 °C−83.0 °C; FTIR, νmax (cm−1): 1632, 1597, 1490, 1460, 1376, 1289, 1262, 1176, 753, 723, 673; 1H NMR (300 MHz, CDCl3): δ 8.56 (dd, J = 8.0, 1.5 Hz, 2H), 7.73−7.76 (m, 2H), 7.42 (d, J = 8.7 Hz, 2H), 7.27 (t, J = 7.7 Hz, 2H), 6.65 (t, J = 2.1 Hz, 2H), 6.15 (t, J = 2.1 Hz, 2H), 4.27 (t, J = 8.1 Hz, 2H), 3.90 (t, J = 7.0 Hz, 2H), 1.94−1.77 (m, 4H), 1.58−1.36 (m, 4H); 13C NMR (75 MHz, CDCl3): δ 177.9, 141.6 (2C), 133.9 (2C), 128.0 (2C), 122.4 (2C), 121.2 (2C), 120.4 (2C), 114.4 (2C), 108.0 (2C), 49.4, 45.9, 31.5, 27.1, 26.6, 26.5; MS (EI): m/z (relative intensity): 344 (M+, 80), 264 (36), 222 (50), 208 (100), 196 (36), 180 (40), 80 (38); HRMS (APCI): m/z calcd for C23H25N2O [M + H]+: 345.1961; found: 345.1952.
10-(3′-(N-Diethylamino)propyl)acridone (
2e
):
24
(69%) yellow solid, m.p. 100.5 °C−101.0 °C; FTIR, νmax (cm−1): 1633, 1598, 1490, 1461, 1376, 1291, 1265, 1178, 752, 673; 1H NMR (300 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.5 Hz, 2H), 7.75−7.62 (m, 4H), 7.28 (t, J = 7.8 Hz, 2H), 4.45 (t, J = 8.0 Hz, 2H), 2.70−2.60 (m, 6H), 2.10−2.00 (m, 2H), 1.13 (t, J = 7.1 Hz, 6H); 13C NMR (75 MHz, CDCl3): δ 178.0, 141.8 (2C), 133.8 (2C), 127.9 (2C), 122.4 (2C), 121.1 (2C), 114.7 (2C), 50.2, 46.9 (2C), 44.3, 25.4, 11.7 (2C); MS (EI): m/z (relative intensity): 308 (M+, 9), 208 (7), 180 (6), 166 (9), 86 (100); HRMS (APCI): m/z calcd for C20H25N2O [M + H]+: 309.1961; found: 309.1966.
10-(4′-(N-Diethylamino)butyl)acridone (
3e
):
24
(94%) yellow solid, m.p. 63.0 °C−64.0 °C; FTIR, νmax (cm−1): 1633, 1598, 1490, 1461, 1378, 1290, 1264, 1177, 752, 674; 1H NMR (400 MHz, CDCl3): δ 8.57 (dd, J = 8.0, 1.5 Hz, 2H), 7.74−7.69 (m, 2H), 7.58 (d, J = 8.7 Hz, 2H), 7.28 (t, J = 7.5 Hz, 2H), 4.36 (t, J = 8.3 Hz, 2H), 2.67−2.57 (m, 6H), 2.00−1.90 (m, 2H), 1.82−1.72 (m, 2H), 1.10 (t, J = 7.2 Hz, 6H); 13C NMR (100 MHz, CDCl3): δ 178.0, 141.7 (2C), 133.8 (2C), 127.9 (2C), 122.4 (2C), 121.1 (2C), 114.7 (2C), 52.1, 46.6, 46.0 (2C), 24.8, 24.1, 11.3 (2C); MS (EI): m/z (relative intensity): 322 (M+, 19), 208 (14), 180 (9), 112 (13), 86 (100); HRMS (APCI): m/z calcd for C21H27N2O [M + H]+: 323.2118; found: 323.2107.
10-(5′-(N-Diethylamino)pentyl)acridone (
4e
):
24
(85%) yellow solid, m.p. 61.5 °C−62.5 °C; FTIR, νmax (cm−1): 1630, 1597, 1492, 1459, 1378, 1290, 1263, 1178, 751, 671; 1H NMR (300 MHz, CDCl3): δ 8.55 (d, J = 8.1 Hz, 2H), 7.70 (t, J = 7.0 Hz, 2H), 7.46 (d, J = 8.7 Hz, 2H), 7.26 (t, J = 7.5 Hz, 2H), 4.29 (t, J = 8.1 Hz, 2H), 2.60−2.45 (m, 6H), 2.00−1.85 (m, 2H), 1.70−1.50 (m, 4H), 1.05 (t, J = 7.2 Hz, 6H); 13C NMR (75 MHz, CDCl3): δ 177.9, 141.7 (2C), 133.9 (2C), 127.9 (2C), 122.4 (2C), 121.1 (2C), 114.6 (2C), 52.6, 47.0, 46.1 (2C), 27.0, 26.9, 24.9, 11.6 (2C); MS (EI): m/z (relative intensity): 336 (M+, 3), 208 (5), 180 (4), 126 (6), 86 (100); HRMS (APCI): m/z calcd for C22H29N2O [M + H]+: 337.2274; found: 337.2278.
10-(6′-(N-Diethylamino)hexyl)acridone (
5e
):
24
(79%) yellow solid, m.p. 150.0 °C−151.0 °C; FTIR, νmax (cm−1): 1632, 1597, 1490, 1460, 1376, 1289, 1262, 1176, 754, 674; 1H NMR (400 MHz, CDCl3): δ 8.56 (dd, J = 8.0, 1.6 Hz, 2H), 7.75−7.69 (m, 2H), 7.48 (d, J = 8.7 Hz, 2H), 7.28 (t, J = 7.7 Hz, 2H), 4.32 (t, J = 8.1 Hz, 2H), 2.81 (q, J = 7.2 Hz, 4H), 2.71−2.66 (m, 2H), 1.96−1.87 (m, 2H), 1.75−1.65 (m, 2H), 1.63−1.54 (m, 2H), 1.50−1.42 (m, 2H), 1.20 (t, J = 7.2 Hz, 6H); 13C NMR (100 MHz, CDCl3): δ 177.8, 141.6 (2C), 133.9 (2C), 127.8 (2C), 122.3 (2C), 121.1 (2C), 114.5 (2C), 52.0, 46.6 (2C), 45.9, 27.0 (2C), 26.5, 25.4, 10.1 (2C); MS (EI): m/z (relative intensity): 350 (M+, 23), 335 (9), 321 (9), 208 (23), 180 (13), 86 (100); HRMS (APCI): m/z calcd for C23H31N2O [M + H]+: 351.2431; found: 351.2430.
10-(3′-(N-Dimethylamino)propyl)acridone (
2f
):
25
(78%) yellow solid, m.p. 86.0 °C−86.5 °C; FTIR, νmax (cm−1): 1634, 1597, 1490, 1461, 1377, 1291, 1261, 1178, 753, 674; 1H NMR (400 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.6 Hz, 2H), 7.75−7.69 (m, 2H), 7.62 (d, J = 8.7 Hz, 2H), 7.29 (t, J = 7.6 Hz, 2H), 4.46 (t, J = 7.7 Hz, 2H), 2.48 (t, J = 6.6 Hz, 2H), 2.33 (s, 6H), 2.11−2.03 (m, 2H); 13C NMR (100 MHz, CDCl3): δ 178.0, 141.8 (2C), 133.9 (2C), 127.9 (2C), 122.4 (2C), 121.2 (2C), 114.7 (2C), 56.4, 45.5 (2C), 43.9, 25.4; MS (EI): m/z (relative intensity): 280 (M+, 100), 236 (46), 222 (51), 209 (43), 180 (31) 166 (40), 152 (28), 58 (74); HRMS (APCI): m/z calcd for C18H21N2O [M + H]+: 281.1648; found: 281.1642.
10-(4′-(N-Dimethylamino)butyl)acridone (
3f
): (95%) yellow solid, m.p. 96.5 °C−97.5 °C; FTIR, νmax (cm−1): 1632, 1596, 1490, 1460, 1378, 1290, 1264, 1177, 1043, 753, 674; 1H NMR (400 MHz, CDCl3): δ 8.57 (dd, J = 8.0, 1.5 Hz, 2H) 7.72 (m, 2H), 7.55 (d, J = 8.7 Hz, 2H), 7.28 (t, J = 7.2 Hz, 2H), 4.37 (t, J = 8.3 Hz, 2H), 2.44 (t, J = 7.2 Hz, 2H), 2.31 (s, 6H), 2.01−1.91 (m, 2H), 1.80−1.71 (m, 2H); 13C NMR (100 MHz, CDCl3): δ 177.9, 141.7 (2C), 133.8 (2C), 127.9 (2C), 122.4 (2C), 121.1 (2C), 114.6 (2C), 58.8, 45.9, 45.3 (2C), 24.9, 24.6; MS (EI): m/z (relative intensity): 294 (M+, 5), 222 (3), 208 (3), 180 (3), 100 (4), 71 (4), 58 (100); HRMS (APCI): m/z calcd for C19H23N2O [M + H]+: 295.1805; found: 295.1797.
10-(5′-(N-Dimethylamino)pentyl)acridone (
4f
): (71%) yellow solid, m.p. 65.0 °C−65.5 °C; FTIR, νmax (cm−1): 1632, 1591, 1492, 1459, 1376, 1261, 1181, 1037, 754, 673; 1H NMR (400 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.4 Hz, 2H), 7.73 (m, 2H), 7.49 (d, J = 8.7 Hz, 2H), 7.29 (t, J = 7.8 Hz, 2H), 4.34 (t, J = 8.1 Hz, 2H), 2.36 (t, J = 6.6 Hz, 2H), 2.28 (s, 6H), 2.01−1.91 (m, 2H), 1.68−1.55 (m, 4H); 13C NMR (100 MHz, CDCl3): δ 177.9, 141.7 (2C), 133.8 (2C), 128.0 (2C), 122.5 (2C), 121.1 (2C), 114.4 (2C), 59.4, 46.0, 45.4 (2C), 27.3, 27.0, 24.7; MS (EI): m/z (relative intensity): 308 (M+, 4), 208 (3), 180 (3), 149 (4), 114 (11), 58 (100); HRMS (APCI): m/z calcd for C20H25N2O [M + H]+: 309.1961; found: 309.1958.
10-(6′-(N-Dimethylamino)hexyl)acridone (
5f
): (95%) yellow solid, m.p. 102.5 °C−103.0 °C; FTIR, νmax (cm−1): 1629, 1594, 1494, 1461, 1380, 1291, 1265, 1179, 748, 670; 1H NMR (400 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.6 Hz, 2H), 7.72 (m, 2H), 7.48 (d, J = 8.7 Hz, 2H), 7.29 (t, J = 7.4 Hz, 2H), 4.33 (t, J = 8.0 Hz, 2H), 2.35−2.30 (m, 2H), 2.26 (s, 6H), 2.00−1.90 (m, 2H), 1.63−1.44 (m, 6H); 13C NMR (100 MHz, CDCl3): δ 177.9, 141.7 (2C), 133.8 (2C), 128.0 (2C), 122.5 (2C), 121.1 (2C), 114.5 (2C), 59.5, 46.1, 45.3 (2C), 27.5, 27.1 (2C), 26.8; MS (EI): m/z (relative intensity): 322 (M+, 5), 208 (4), 180 (4), 167 (6), 149 (19), 58 (100); HRMS (APCI): m/z calcd for C21H27N2O [M + H]+: 323.2118; found: 323.2113.
10-(3′-(N-Trimethylammonium)propyl)acridone (
2g
): (99% from 2f) yellow solid, m.p. 266.0 °C−267.0 °C; FTIR, νmax (cm−1): 1608, 1590, 1497, 1460, 1267, 1179, 1020, 759, 673; 1H NMR (300 MHz, DMSO-d6): δ 8.37 (d, J = 7.9 Hz, 2H), 8.00−7.80 (m, 4H), 7.38 (t, J = 7.4 Hz, 2H), 4.51 (t, J = 7.7 Hz, 2H), 3.70 (t, J = 7.8 Hz, 2H), 3.12 (s, 9H), 2.30−2.15 (m, 2H); 13C NMR (75 MHz, DMSO-d6): δ 176.5, 141.3 (2C), 134.4 (2C), 126.9 (2C), 122.6 (2C), 121.5 (2C), 115.9 (2C), 62.3, 52.5 (3C), 42.2, 20.7; MS (EI): m/z (relative intensity): 280 ([M – CH3]+, 7), 208 (7), 142 (17), 127 (7), 58 (100); HRMS (ESI): m/z calcd for C19H23N2O [M]+:295.1805; found: 295.1796.
10-(4′-(N-Trimethylammonium)butyl)acridone (
3g
): (87% from 3f) yellow solid, m.p. 260.0 °C−261.0 °C; FTIR, νmax (cm−1): 1627, 1607, 1594, 1492, 1457, 1261, 1174, 754, 676; 1H NMR (400 MHz, DMSO-d6): δ 8.35 (dd, J = 8.1, 1.3 Hz, 2H), 7.89−7.81 (m, 4H), 7.35 (t, J = 6.6 Hz, 2H), 4.51 (t, J = 8.2 Hz, 2H), 3.48−3.38 (m, 2H), 3.43 (s, 9H), 2.07−1.97 (m, 2H), 1.84−1.75 (m, 2H); 13C NMR (100 MHz, DMSO-d6): δ 176.9, 141.8 (2C), 134.7 (2C), 127.3 (2C), 122.1 (2C), 121.8 (2C), 116.2 (2C), 65.5, 52.9 (3C), 45.2, 24.1, 19.9; MS (EI): m/z (relative intensity): 294 ([M – CH3]+, 4), 208 (6), 142 (13), 69 (36), 58 (100); HRMS (APCI): m/z calcd for C20H25N2O [M]+: 309.1961; found: 309.1951.
10-(5′-(N-Trimethylammonium)pentyl)acridone (
4g
): (61% from 4f) yellow solid, m.p. 263.0 °C−263.5 °C; FTIR, νmax (cm−1): 1627, 1607, 1596, 1494, 1459, 1378, 1265, 1178, 756, 674; 1H NMR (400 MHz, DMSO-d6): δ 8.35 (d, J = 7.8 Hz, 2H), 7.85−7.81 (m, 4H), 7.36−7.30 (m, 2H), 4.50 (f, J = 7.8 Hz, 2H), 3.36−3.34 (m, 11H), 1.85−1.46 (m, 2H), 1.72−1.63 (m, 2H), 1.60−1.51 (m, 2H), 1.41−1.32 (m, 2H); 13C NMR (100 MHz, DMSO-d6): δ 176.9, 141.9 (2C), 134.7 (2C), 127.2 (2C), 122.1 (2C), 121.7 (2C), 116.4 (2C), 65.6, 52.7 (3C), 45.3, 26.9, 23.2, 22.5; MS (EI): m/z (relative intensity): 296 ([M – 27]+, 12), 268 (9), 185 (9), 69 (63), 55 (100); HRMS (APCI): m/z calcd for C21H27N2O [M]+: 323.2118; found: 323.2109.
10-(6′-(N-Trimethylammonium)hexyl)acridone (
5g
): (99% from 5f) yellow solid, m.p. 103.0 °C−104.0 °C; FTIR, νmax (cm−1): 1627, 1607, 1596, 1494, 1459, 1378, 1265, 1178, 756, 674; 1H NMR (400 MHz, DMSO-d6): δ 8.35 (d, J = 7.8 Hz, 2H), 7.85−7.81 (m, 4H), 7.36−7.30 (m, 2H), 4.48 (t, J = 7.8 Hz, 2H), 3.36−3.34 (m, 11H), 1.85−1.46 (m, 2H), 1.72−1.63 (m, 2H), 1.60−1.51 (m, 2H), 1.41−1.32 (m, 2H); 13C NMR (100 MHz, DMSO-d6): δ 176.9, 141.9 (2C), 134.7 (2C), 127.2 (2C), 122.1 (2C), 121.7 (2C), 116.3 (2C), 65.8, 52.7 (3C), 45.5, 27.1, 26.1, 25.9, 22.4; MS (EI): m/z (relative intensity): 322 ([M – CH3]+, 4), 208 (9), 180 (4), 142 (13), 58 (100); HRMS (APCI): m/z calcd for C22H29N2O [M]+: 337.2274; found: 337.2265.
10-(3′-N-Azidopropyl)acridone (
2h
): (89%) yellow solid, m.p. 78.5 °C−79.5 °C; FTIR, νmax (cm−1): 2094, 1632, 1595, 1490, 1461, 1377, 1290, 1261, 1177, 751, 673; 1H NMR (300 MHz, CDCl3): δ 8.55 (d, J = 7.8, 2H), 7.75−7.69 (m, 2H), 7.50 (d, J = 8.7 Hz, 2H), 7.31−7.24 (m, 2H), 4.44 (t, J = 7.8 Hz, 2H), 3.58 (t, J = 6.0 Hz, 2H), 2.19−2.10 (m, 2H); 13C NMR (75 MHz, CDCl3): δ 177.8, 141.6 (2C), 134.0 (2C), 128.1 (2C), 122.5 (2C), 121.4 (2C), 114.2 (2C), 48.9, 43.0, 26.6; MS (EI): m/z (relative intensity): 278 ([M]+, 59), 250 (13), 208 (100), 195 (34), 180 (53), 166 (28); HRMS (APCI): m/z calcd for C16H15N4O [M + H]+: 279.1240; found: 279.1238.
10-(4′-N-Azidobutyl)acridone (
3h
): (95%) yellow solid, m.p. 112.5 °C−113.5 °C; FTIR, νmax (cm−1): 2094, 1632, 1596, 1491, 1460, 1379, 1290, 1263, 1175, 753, 673; 1H NMR (400 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.6 Hz, 2H), 7.76−7.71 (m, 2H), 7.48 (d, J = 8.7 Hz, 2H), 7.32−7.27 (m, 2H), 4.38 (t, J = 8.1 Hz, 2H), 3.45 (t, J = 6.6 Hz, 2H), 2.08−1.90 (m, 2H), 1.88−1.80 (m, 2H); 13C NMR (100 MHz, CDCl3): δ 177.9, 141.6 (2C), 134.0 (2C) 128.0 (2C), 122.4 (2C), 121.3 (2C), 114.3 (2C), 51.0, 45.4, 26.2, 24.5; MS (EI): m/z (relative intensity): 292 ([M]+, 66), 220 (59), 208 (100), 195 (20), 180 (43), 152 (23); HRMS (APCI): m/z calcd for C17H17N4O [M + H]+: 293.1397; found: 293.1387.
10-(5′-N-Azidopentyl)acridone (
4h
): (72%) yellow solid, m.p. 79.0 °C−80.0 °C; FTIR, νmax (cm−1): 2085, 1634, 1594, 1489, 1458, 1377, 1263, 1171, 755, 672; 1H NMR (400 MHz, CDCl3): δ 8.58 (t, J = 7.9 Hz, 2H), 7.73 (t, J = 8.2 Hz, 2H), 7.47 (d, J = 8.7 Hz, 2H), 7.29 (t, J = 7.4 Hz, 2H), 4.34 (t, J = 7.6 Hz, 2H), 3.36 (t, J = 6.3 Hz, 2H), 2.00−1.90 (m, 2H), 1.78−1.60 (m, 4H); 13C NMR (100 MHz, CDCl3): δ 177.8, 141.7 (2C), 133.9 (2C), 128.0 (2C), 122.5 (2C), 121.2 (2C), 114.3 (2C), 51.2, 45.8, 28.7, 26.8, 24.1; MS (EI): m/z (relative intensity): 306 ([M]+, 27), 208 (100), 196 (44), 180 (44), 166 (19), 152 (22); HRMS (APCI): m/z calcd for C18H19N4O [M + H]+: 307.1553; found: 307.1545.
10-(6′-N-Azidohexyl)acridone (
5h
): (96%) yellow solid, m.p. 77.0 °C−78.0 °C; FTIR, νmax (cm−1): 2093, 1634, 1598, 1491, 1460, 1376, 1290, 1262, 1176, 754, 673; 1H NMR (300 MHz, CDCl3): δ 8.58 (dd, J = 8.0, 1.5 Hz, 2H), 7.75−7.70 (m, 2H), 7.47 (d, J = 8.7 Hz, 2H), 7.29 (t, J = 7.8 Hz, 2H), 4.33 (t, J = 8.1 Hz, 2H), 3.32 (t, J = 6.5 Hz, 2H), 2.00−1.90 (m, 2H), 1.70−1.50 (m, 6H); 13C NMR (75 MHz, CDCl3): δ 177.9, 141.7 (2C), 133.9 (2C), 128.0 (2C), 122.4 (2C), 121.2 (2C), 114.4 (2C), 51.3, 45.9, 28.8, 27.1, 26.5, 26.4; MS (EI): m/z (relative intensity): 320 ([M]+, 78), 292 (15), 208 (98), 180 (100), 166 (54), 152 (60), 140 (47); HRMS (APCI): m/z calcd for C19H21N4O [M + H]+: 321.1710; found: 321.1699.
10-(N-propyl)acridone (
2i
):
26
(74%) yellow solid, m.p. 130.0 °C−131.0 °C; FTIR, νmax (cm−1): 1634, 1597, 1490, 1458, 1377, 1290, 1263, 1174, 747, 670; 1H NMR (400 MHz, CDCl3): δ 8.59 (dd, J = 8.0, 1.6 Hz, 2H), 7.75−7.70 (m, 2H), 7.50 (d, J = 8.7 Hz, 2H), 7.31−7.26 (m, 2H), 4.31 (t, J = 8.3 Hz, 2H), 2.03−1.93 (m, 2H), 1.17 (t, J = 7.5 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 178.0, 141.8 (2C), 133.8 (2C), 127.9 (2C), 122.4 (2C), 121.2 (2C), 114.5 (2C), 47.7, 20.5, 11.1; MS (EI): m/z (relative intensity): 237 ([M]+, 54), 208 (100), 180 (13), 166 (8), 152 (8), 140 (5); HRMS (APCI): m/z calcd for C16H16NO [M + H]+: 238.1226; found: 238.1231.
10-(N-butyl)acridone (
3i
):
26
(70%) yellow solid, m.p. 98.5 °C−99.0 °C; FTIR, νmax (cm−1): 1633, 1596, 1489, 1459, 1375, 1290, 1259, 1175, 751, 671; 1H NMR (400 MHz, CDCl3): δ 8.59 (dd, J = 8.0, 1.7 Hz, 2H), 7.75−7.70 (m, 2H), 7.50 (d, J = 8.7 Hz, 2H), 7.31−7.26 (m, 2H), 4.34 (t, J = 8.3 Hz, 2H), 1.97−1.88 (m, 2H), 1.65−1.55 (m, 2H), 1.09 (t, J = 7.4 Hz, 3H); 13C NMR (100 MHz, CDCl3): δ 178.0, 141.7 (2C), 133.8 (2C), 128.0 (2C), 122.4 (2C), 121.1 (2C), 114.5 (2C), 45.9, 29.2, 20.2, 13.8; MS (EI): m/z (relative intensity): 251 ([M]+, 41), 208 (100), 180 (21), 166 (9), 152 (8), 140 (6); HRMS (APCI): m/z calcd for C17H18NO [M + H]+: 252.1383; found: 252.1377.
Diphenylamine-2,4′-dicarboxylic acid (
8
):
27
A mixture of 2-bromobenzoic acid (6) (2.0 g, 9.95 mmol), 4-aminobenzoic acid (7) (1.37 g, 9.95 mmol), potassium carbonate (1.38 g, 9.95 mmol) and copper powder (60 mg, 3% w/w) in N,N-dimethylformamide (10 mL) was refluxed for 4 h. The mixture was cooled and slowly added to aqueous HCl (1:1) (50 mL). The precipitate was collected, washed with hot water, and dried to provide the title compound.
9(10H)-Acridone-2-carboxylic acid (
9
):
27
Diphenylamine-2,4′-dicarboxylic acid (8) (2.57 g, 10.0 mmol) in concentrated H2SO4 (5 mL) was heated at 100 °C for 4 h. The reaction mixture was cooled and poured into ice water. The precipitate was filtered and washed with hot water. The solid was redissolved in 5% aqueous sodium hydroxide and filtered. The filtrate was diluted with an equal volume of ethanol and acidified with acetic acid. The precipitate was filtered off, washed with water, and dried to yield 9 (1.70 g, 71% from 2-bromobenzoic acid). m.p. > 300 °C; FTIR, νmax (cm−1): 3387, 1634, 1595, 1576, 1399, 1136; 1H NMR (300 MHz, DMSO-d6): δ 12.16 (s, 1H), 8.85 (s, 1H), 8.22 (t, J = 8.3 Hz, 2H), 7.76 (t, J = 7.5 Hz, 1H), 7.59 (d, J = 8.5 Hz, 2H), 7.30 (t, J = 7.5 Hz, 1H); 13C NMR (75 MHz, DMSO-d6): δ 176.9, 167.2, 143.3, 140.9, 133.9, 133.4, 128.6, 126.1, 124.0, 121.8, 120.9, 119.7, 117.7, 117.6; MS (EI): m/z (relative intensity): 239 ([M]+, 100), 222 (32), 195 (24), 139 (28), 97 (45), 83 (44); HRMS (ESI): m/z calcd for C14H10NO3 [M + H]+: 240.0655; found: 240.0652.
10-(4′-N-Bromobutyl)acridone-2-(4-bromobutyl)carboxylate (
10a
) and 9(10H)-Acridone-2-(4-bromobutyl)carboxylate (
10b
): A solution of 9 (500 mg, 2.09 mmol) in N,N-dimethylformamide (25 mL) was added to a stirred suspension of sodium hydride (250 mg, 6.25 mmol) in N,N-dimethylformamide (10 mL). The mixture was heated at 80 °C for 15 min, treated with 1,4-dibromobutane (1.5 mL, 12.71 mmol) and heated at 80 °C for an additional 1 h. The reaction mixture was cooled and water was added and the mixture was extracted with ethyl acetate. The combined organic layer was washed with brine and water, dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was purified by silica gel column chromatography to provide 10a (245 mg, 23%) and 10b (226 mg, 29%).
10a
: Yellow solid, m.p. 107.5 °C−108.0 °C; FTIR, νmax (cm−1): 1716, 1639, 1610, 1596, 1482, 1297, 1246, 1139, 754; 1H NMR (300 MHz, CDCl3): δ 9.16 (s, 1H), 8.84 (d, J = 7.9 Hz, 1H), 8.31 (d, J = 8.9 Hz, 1H), 7.76 (t, J = 8.0 Hz, 1H), 7.51 (d, J = 9.0 Hz, 2H), 7.34 (t, J = 7.6 Hz, 1H), 4.45−4.35 (m, 4H), 3.60−3.50 (m, 4H), 2.20−1.90 (m, 8H); 13C NMR (75 MHz, CDCl3): δ 177.5, 165.8, 144.2, 141.5, 134.4, 134.3, 130.5, 128.1, 122.9, 122.8, 122.3, 121.6, 114.7, 114.6, 64.1, 45.5, 33.2, 32.5, 29.5, 29.3, 27.4, 25.6; MS (EI): m/z (relative intensity): 507 ([M]+, 36), 386 (98), 356 (12), 306 (11), 252 (100), 224 (15), 165 (16); HRMS (ESI): m/z calcd for C22H24Br2NO3 (M + H)+: 508.0117; found: 508.0125.
10b
: Yellow solid, m.p. 257.5 °C−258.5 °C; FTIR, νmax (cm−1): 1713, 1634, 1593, 1562, 1526, 1478, 1293, 1243, 1130, 756, 676; 1H NMR (400 MHz, DMSO-d6): δ 12.08 (s, 1H), 8.84 (d, J = 2.0 Hz, 1H), 8.25−8.20 (m, 2H), 7.81−7.76 (m, 1H), 7.63−7.56 (m, 2H), 7.31 (t, J = 7.4 Hz, 1H), 4.35 (t, J = 6.4 Hz, 2H), 3.63 (t, J = 6.4 Hz, 2H), 2.00−1.86 (m, 4H); 13C NMR (100 MHz, DMSO-d6): δ 176.7, 165.3, 143.7, 140.8, 134.1, 133.1, 128.6, 126.1, 122.1, 122.0, 120.9, 119.7, 117.9, 117.7, 63.9, 34.7, 29.1, 27.1; MS (EI): m/z (relative intensity): 373 ([M]+, 44), 239 (96), 222 (100), 194 (42), 166 (23), 139 (29); HRMS (ESI): m/z calcd for C18H17BrNO3 [M + H]+: 374.0386; found: 374.0389.
10-(4′-N-Piperidinobutyl)acridone-20[(N-piperidino)butyl]carboxylate (
11a
): A solution of 10a (1 mmol) and piperidine (5 mmol) in acetonitrile was stirred at room temperature for 24 h. The reaction mixture was then concentrated under reduced pressure, the residue was treated with water and then extracted with dichloromethane. The combined organic layer was washed with saturated aqueous sodium hydrogen carbonate and water, dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was purified by silica gel column chromatography to yield 11a in 97% yield as a yellow solid: m.p. 104.5 °C−105.5 °C; FTIR, νmax (cm−1): 1703, 1639, 1602, 1479, 1251, 1137, 758; 1H NMR (300 MHz, acetone-d6): δ 9.04 (s, 1H), 8.41 (d, J = 7.8 Hz, 1H), 8.26 (d, J = 8.7 Hz, 1H), 7.96 (d, J = 8.1 Hz, 1H), 7.94 (d, J = 7.2 Hz, 1H), 7.80 (t, J = 7.6 Hz, 2H), 7.34 (t, J = 7.3 Hz, 1H), 4.52 (t, J = 8.2 Hz, 2H), 4.37 (t, J = 6.6 Hz, 2H), 2.50−2.30 (m, 12H), 2.10−1.35 (m, 20H); 13C NMR (75 MHz, acetone-d6): δ 177.5, 166.2, 145.4, 142.7, 135.1, 134.4, 130.2, 128.0, 123.6 (2C), 122.8, 122.4, 117.0 (2C), 65.6, 59.4, 58.1, 55.3 (2C), 55.2 (2C), 46.8, 27.6, 27.0 (2C), 26.9 (2C), 25.4, 25.3, 24.8, 24.2, 23.6; MS (EI): m/z (relative intensity): 368 (6), 284 (6), 185 (14), 129 (47), 98 (93), 83 (91), 55 (100); HRMS (APCI): m/z calcd for C32H44N3O3 [M + H]+: 518.3377; found: 518.3379.
9(10H)-Acridone-2-[(N-piperidino)butyl]carboxylate (
11b
): A solution of 10b (1 mmol) and piperidine (5 mmol) in N,N-dimethylformamide was stirred at room temperature for 24 h. Water was added to the mixture and the solvent was removed under reduced pressure. The precipitate was collected, washed with water, and then dichloromethane to give 11b in 92% yield as a yellow solid: m.p. 143.5 °C−144.5 °C; FTIR, νmax (cm−1): 1711, 1627, 1594, 1575, 1525, 1475, 1275, 1241, 763; 1H NMR (300 MHz, DMSO-d6): δ 12.17 (s, 1H), 8.84 (d, J = 2.1 Hz, 1H), 8.25−8.19 (m, 2H), 7.81−7.75 (m, 1H), 7.65−7.58 (m, 2H), 7.33 (t, J = 7.2 Hz, 1H), 4.35−4.31 (m, 2H), 2.65−2.55 (m, 6H), 1.80−1.42 (m, 10H); 13C NMR (75 MHz, DMSO-d6): δ 176.7, 165.3, 143.7, 140.8, 134.1, 133.0, 128.6, 126.1, 122.1, 121.9, 120.9, 119.6, 117.9, 117.7, 64.4, 57.1, 53.3 (2C), 26.1 (2C), 24.4, 23.1, 21.9; MS (EI): m/z (relative intensity): 378 ([M]+, 23), 284 (2), 252 (10), 138 (24), 98 (100); HRMS (APCI): m/z calcd for C23H27N2O3 [M + H]+:379.2016; found: 379.2024.
10-(4′-N-Piperidinobutyl)acridone-2-carboxylic acid (
12
): A stirred solution of 11a (248 mg, 0.48 mmol) in ethanol (5 mL) was treated with 2% aqueous NaOH (5 mL) and refluxed for 1 h. The reaction mixture was cooled and concentrated under reduced pressure to remove the solvent. Water was added to the residue and the mixture was neutralized with 5 M HCl. The yellow precipitate was filtered and washed with water to give 12 (180 mg, 99%) as a yellow solid: m.p. 136.0 °C−137.0 °C; FTIR, νmax (cm−1): 3369, 1614, 1593, 1557, 1484, 1376, 1268, 781, 756; 1H NMR (300 MHz, DMSO-d6): δ 8.89 (s, 1H), 8.34 (d, J = 7.6 Hz, 1H), 8.24 (d, J = 8.6 Hz, 1H), 8.00–7.80 (m, 3H), 7.40 (t, J = 6.0 Hz, 1H), 4.55–4.45 (m, 2H), 3.20−2.90 (m, 6H), 2.10−1.90 (m, 2H), 1.90−1.70 (m, 6H), 1.60−1.45 (m, 2H); 13C NMR (75 MHz, DMSO-d6): δ 176.4, 166.7, 143.9, 141.3, 134.7, 134.0, 129.0, 126.8, 123.3, 122.2, 122.0, 120.8, 116.4, 116.3, 55.5, 52.0 (2C), 45.2, 24.2, 22.4 (2C), 21.6, 20.4; MS (EI): m/z (relative intensity): 378 ([M]+, 23), 252 (10), 138 (24), 98 (100); HRMS (APCI): m/z calcd for C23H27N2O3 [M + H]+: 379.2016; found: 379.2015.
10-(4′-N-Piperidinobutyl)acridone-2-methylcarboxylate (
13
): Compound 12 (74 mg, 0.20 mmol) in thionyl chloride (1 mL) was refluxed for 3 h. The reaction mixture was cooled and added dropwise to cold methanol. The solution was then treated with water and extracted with ethyl acetate. The combined organic layer was washed with saturated aqueous sodium hydrogen carbonate and water, dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was purified by silica gel column chromatography to yield 13 (63 mg, 82%) as a yellow solid: m.p. 126.5 °C−127.5 °C; FTIR, νmax (cm−1): 1717, 1643, 1608, 1481, 1297, 1250, 766; 1H NMR (300 MHz, CDCl3): δ 9.18 (s, 1H), 8.54 (d, J = 8.1 Hz, 1H), 8.29 (d, J = 8.7 Hz, 1H), 7.74 (t, J = 7.2 Hz, 1H), 7.64 (d, J = 8.7 Hz, 2H), 7.32 (t, J = 7.2 Hz, 1H), 4.41 (t, J = 8.0 Hz, 2H), 3.96 (s, 3H), 2.60−2.40 (m, 6H), 2.05−1.60 (m, 8H), 1.60−1.45 (m, 2H); 13C NMR (75 MHz, CDCl3): δ 177.7, 166.5, 144.4, 141.7, 134.3, 134.1, 130.5, 128.0, 122.8, 122.7, 122.1, 121.6, 115.1, 115.0, 57.7, 54.5 (2C), 52.1, 46.2, 25.7 (2C), 24.6, 24.2, 23.2; MS (EI): m/z (relative intensity): 392 ([M]+, 21), 167 (3), 149 (9), 138 (19), 98 (100); HRMS (ESI): m/z calcd for C24H29N2O3 [M + H]+: 393.2173; found: 393.2177.
N-(10-(4′-N-Piperidinobutyl)acridone-2-carbonyl)-L-valine benzyl ester (
14
): A solution of 12 (100 mg, 0.26 mmol), L-valine benzyl ester hydrochloride (64.5 mg, 0.26 mmol), HOBt (53.6 mg, 0.40 mmol), and EDCI (76 mg, 0.40 mmol) in N,N-dimethylformamide (10 mL) was stirred at −10 °C for 10 min. The solution was then treated with DIPEA (0.14 mL, 0.78 mmol) and stirred at −10 °C to room temperature for 24 h. Water was added and the mixture was extracted with ethyl acetate. The combined organic layer was washed with saturated aqueous ammonium chloride, brine, saturated aqueous sodium hydrogen carbonate, and water. The organic layer was dried over anhydrous Na2SO4 and concentrated under reduced pressure. The residue was purified by silica gel column chromatography to yield 14 (70%) as a yellow solid: m.p. 89.0 °C−90.0 °C; FTIR, νmax (cm−1): 2934, 1739, 1637, 1608, 1482, 1466, 1264, 1181, 1155, 757; 1H NMR (400 MHz, CDCl3): δ 8.92 (d, J = 2.4 Hz, 1H), 8.60 (dd, J = 8.0, 1.2 Hz, 1H), 8.34 (dd, J = 9.0, 2.0 Hz, 1H), 7.81−7.70 (m, 3H), 7.40−7.32 (m, 5H), 6.93 (d, J = 8.4 Hz, 1H), 5.26−5.18 (m, 2H), 4.86 (t, J = 5.2 Hz, 1H), 4.48−4.43 (m, 2H), 2.50−2.45 (m, 4H), 2.35−2.30 (m, 1H), 2.02−1.96 (m, 2H), 1.82−1.75 (m, 2H), 1.70−1.63 (m, 6H), 1.55−1.45 (m, 2H), 1.02 (t, J = 7.2 Hz, 6H); 13C NMR (100 MHz, CDCl3): δ 177.7, 171.9, 166.3, 143.6, 141.6, 135.3, 134.3, 133.4, 128.6(2C), 128.4 (3C), 127.9, 126.3, 125.8, 122.6, 122.0, 121.2, 115.5, 115.1, 67.0, 57.7, 57.6, 54.4 (2C), 46.1, 31.6, 25.5 (2C), 24.6, 24.0, 23.0, 19.1, 18.0; MS (EI): m/z (relative intensity): 567 ([M]+, 13), 415 (3), 376 (3), 207 (9), 138 (41), 98 (100); HRMS (ESI): m/z calcd for C35H42N3O4 [M + H]+: 568.3170; found: 568.3161.
N-(10-(4′-N-Piperidinobutyl)acridone-2-carbonyl)-L-valine (
15
): To a solution of 14 (30 mg, 0.05 mmol) in methanol (3 mL) was added 10% Pd/C (4 mg, 10% w/w) and the suspension was stirred under a H2 atmosphere at room temperature for 24 h. The reaction was filtered through Celite and eluting with methanol. The filtrate was concentrated under reduced pressure to yield 15 (80%); FTIR, νmax (cm−1): 1632, 1603, 1593, 1481, 1463, 1369, 1260, 761; 1H NMR (300 MHz, DMSO-d6): δ 8.88 (s, 1H), 8.65−8.55 (m, 1H), 8.33 (d, J = 7.2 Hz, 1H), 8.24 (d, J = 8.3 Hz, 1H), 7.92 (d, J = 8.7 Hz, 2H), 7.90−7.70 (m, 1H), 7.40−7.30 (m, 1H), 4.55−4.40 (m, 2H), 4.28 (t, J = 7.1 Hz, 1H), 2.55−2.35 (m, 6H), 2.30−2.15 (m, 1H), 1.85−1.40 (m, 10H), 1.00−0.90 (m, 6H); 13C NMR (75 MHz, DMSO-d6): δ 176.5, 173.4, 165.6, 143.0, 141.4, 134.4, 132.9, 126.8, 126.7, 126.5, 121.8 (2C), 120.7, 116.2, 116.0, 58.9, 56.8, 53.6 (2C), 45.2, 29.8, 25.2 (2C), 24.0, 23.8, 22.2, 19.5, 18.9; MS (EI): m/z (relative intensity): 477 ([M]+, 1), 415 (16), 373 (61), 282 (39), 250 (28), 138 (27), 98 (100), 91 (76); HRMS (ESI): m/z calcd for C28H36N3O4 [M + H]+: 478.2700; found: 478.2693.
N-(10-(4′-N-Piperidinobutyl)acridone-2-carbonyl)-L-valine benzyl amide (
16
): A procedure analogous to the preparation of 14 and starting from 15 and benzyl amine was conducted to obtain compound 16 in 60% yield as a yellow solid: FTIR, νmax (cm−1): 3284, 2931, 1633, 1607, 1481, 1257, 757; 1H NMR (400 MHz, CDCl3): δ 8.89 (d, J = 2.4 Hz, 1H), 8.55 (d, J = 8.0 Hz, 1H), 8.23 (dd, J = 9.0, 2.4 Hz, 1H), 7.78−7.67 (m, 3H), 7.35−7.23 (m, 5H), 7.11 (d, J = 8.4 Hz, 1H), 6.59 (br s, 1H), 4.52−4.39 (m, 5H), 2.49−2.40 (m, 6H), 2.37−2.30 (m, 1H), 2.00−1.92 (m, 2H), 1.85−1.75 (m, 2H), 1.70−1.64 (m, 4H), 1.55−1.47 (m, 2H), 1.10−1.05 (m, 6H); 13C NMR (100 MHz, CDCl3): δ 177.7, 171.1, 166.6, 143.7, 141.7, 137.9, 134.3, 133.2, 128.7 (2C), 128.0, 127.8 (2C), 127.5, 126.2, 126.1, 122.7, 122.0, 121.3, 115.6, 115.2, 59.5, 57.8, 54.6 (2C), 46.2, 43.6, 31.0, 26.0 (2C), 24.6, 24.4, 23.3, 19.5, 18.5; MS (EI): m/z (relative intensity): 566 ([M]+, 12), 368 (10), 207 (17), 138 (40), 98 (100); HRMS (ESI): m/z calcd for C35H43N4O3 [M + H]+: 567.3330; found: 567.3333.
10-(4′-N-Piperidinobutyl)acridone-2-carboxylic acid benzylamide (
17
): A procedure analogous to the preparation of 14 and starting from 12 and benzyl amine was used to obtain compound 17 in 72% yield as a yellow solid: m.p. 77.0 °C−78.0 °C; FTIR, νmax (cm−1): 1632, 1607, 1480, 1299, 1260, 756; 1H NMR (300 MHz, CDCl3): δ 8.92 (s, 1H), 8.47 (d, J = 7.8 Hz, 1H), 8.36 (d, J = 9.0 Hz, 1H), 7.80−7.60 (m, 3H), 7.40−7.20 (m, 5H), 4.66 (d, J = 5.7 Hz, 2H), 4.38 (t, J = 7.2 Hz, 2H), 2.60−2.40 (m, 6H), 2.00−1.60 (m, 8H), 1.55−1.45 (m, 2H); 13C NMR (75 MHz, CDCl3): δ 177.8, 166.2, 143.4, 141.6, 138.2, 133.3, 133.6, 128.6 (2C), 127.8 (2C), 127.4 (2C), 126.7, 125.5, 122.4, 121.9, 121.1, 115.6, 115.1, 57.7, 54.5 (2C), 46.1, 44.1, 25.8 (2C), 24.5, 24.2, 23.2; MS (EI): m/z (relative intensity): 467 ([M]+, 20), 376 (3), 341 (3), 207 (9), 138 (39), 98 (100); HRMS (ESI): m/z calcd for C30H34N3O2 [M + H]+: 468.2645; found: 468.2640.
9(10H)-Acridone-2-methylcarboxylate (
18
):
28
A procedure analogous to the preparation of 13 and starting from 9 was used to obtain compound 18 in 87% yield as a yellow solid: m.p. >300 °C; FTIR, νmax (cm−1): 1717, 1633, 1595, 1575, 1293, 764; 1H NMR (300 MHz, DMSO-d6): δ 12.16 (s, 1H), 8.82 (d, J = 2.1 Hz, 1H), 8.23 (dd, J = 8.1, 1.5 Hz, 1H), 8.18 (dd, J = 8.7, 2.1 Hz, 1H), 7.80-7.74 (m, 1H), 7.61 (d, J = 8.4 Hz, 1H), 7.58 (d, J = 7.5 Hz, 1H), 7.34-7.28 (m, 1H), 3.89 (s, 3H); 13C NMR (75 MHz, DMSO-d6): δ 176.7, 165.7, 143.6, 140.7, 134.0, 132.9, 128.6, 126.0, 122.0, 121.7, 120.9, 119.6, 117.9, 117.7, 52.1; MS (EI): m/z (relative intensity): 253 ([M]+, 100), 222 (84), 195 (20), 167 (29), 149 (76), 97 (37), 57 (50); HRMS (ESI): m/z calcd for C15H12NO3 [M + H]+: 254.0812; found: 254.0807.
N-(9(10H)-Acridone-2-carbonyl)-L-valine benzyl ester (
19
): A procedure analogous to the preparation of 14 and starting from 9 was used to obtain 19 in 73% yield as a yellow solid: m.p. 211.0 °C−212.0 °C; FTIR, νmax (cm−1): 1716, 1642, 1556, 1521, 1479, 1267, 751, 737; 1H NMR (300 MHz, DMSO-d6): δ 11.98 (s, 1H), 8.93 (d, J = 7.5 Hz, 1H), 8.87 (s, 1H), 8.27 (d, J = 8.1 Hz, 1H), 8.20 (d, J = 8.7 Hz, 1H), 7.77 (t, J = 7.5 Hz, 1H), 7.58 (d, J = 6.6 Hz, 2H), 7.39−7.28 (m, 5H), 5.10 (d, J = 4.2 Hz, 2H), 4.37 (t, J = 7.5 Hz, 1H), 2.29−2.22 (m, 1H), 1.00 (d, J = 6.0 Hz, 3H), 0.95 (d, J = 6.9 Hz, 3H); 13C NMR (75 MHz, DMSO-d6): δ 176.9, 171.7, 166.5, 142.7, 140.8, 136.0, 133.9, 132.6, 128.4 (2C), 128.1, 128.0 (2C), 126.5, 126.3, 126.1, 121.7, 120.8, 119.5, 117.6, 117.3, 65.8, 59.0, 29.5, 19.3, 19.2; MS (EI): m/z (relative intensity): 428 ([M]+, 2), 293 (5), 238 (30), 222 (100), 194 (18), 166 (8), 139 (7); HRMS (APCI): m/z calcd for C26H25N2O4 [M + H]+: 429.1809; found: 429.1803.
N-(9(10H)-Acridone-2-carbonyl)-L-valine (
20
): A procedure analogous to the preparation of 15 and starting from 19 was used to obtain compound 20 in 82% yield as a yellow solid: m.p. 200.0 °C (dec.); FTIR, νmax (cm−1): 3273, 1628, 1517, 1474, 1311, 1154, 757; 1H NMR (300 MHz, DMSO-d6): δ 12.11 (s, 1H), 8.82 (s, 1H), 8.57 (d, J = 7.5 Hz, 1H), 8.25 (d, J = 8.1 Hz, 1H), 8.20 (d, J = 8.7 Hz, 1H), 7.76 (t, J = 7.2 Hz, 1H), 7.60 (d, J = 7.2 Hz, 2H), 7.29 (t, J = 7.2 Hz, 1H), 4.30 (t, J = 6.6 Hz, 1H), 2.30−2.20 (m, 1H), 1.05−0.90 (m, 6H); 13C NMR (75 MHz, DMSO-d6): δ 176.9, 173.4, 166.0, 142.6, 140.8, 133.8, 132.4, 126.7, 126.2, 126.1, 121.6, 120.8, 119.5, 117.6, 117.3, 58.8, 29.7, 19.4, 18.9; MS (EI): m/z (relative intensity): 293 ([M – 45]+, 7), 279 (12), 227 (5), 167 (30), 149 (100), 71 (26); HRMS (APCI): m/z calcd for C19H19N2O4 [M + H]+: 339.1339; found: 339.1345.
9(10H)-Acridone-2-carboxylic acid benzylamide (
21
):
29
A procedure analogous to the preparation of 14 and starting from 9 and benzyl amine was used to obtain compound 21 in 67% yield as a yellow solid: m.p. > 300 °C; FTIR, νmax (cm−1): 1622, 1579, 1549, 1523, 1308, 756, 736; 1H NMR (300 MHz, DMSO-d6): δ 12.08 (s, 1H), 9.28 (br t, J = 5.2 Hz, 1H), 8.85 (s, 1H), 8.26 (d, J = 8.0 Hz, 1H), 8.23 (d, J = 8.8 Hz, 1H), 7.76 (t, J = 7.6 Hz, 1H), 7.61 (d, J = 8.0 Hz, 1H), 7.59 (d, J = 7.5 Hz, 1H), 7.40−7.20 (m, 5H), 4.52 (d, J = 5.4 Hz, 2H); 13C NMR (75 MHz, DMSO-d6): δ 176.9, 165.6, 142.6, 140.8, 139.9, 133.8, 132.2, 128.3 (2C), 127.3 (2C), 126.7 (2C), 126.1, 125.8, 121.7, 120.8, 119.6, 117.6, 117.4, 42.7; MS (EI): m/z (relative intensity): 328 ([M]+, 47), 222 (100), 195 (64), 166 (19), 139 (19), 106 (12); HRMS (ESI): m/z calcd for C21H17N2O2 [M + H]+: 329.1284; found: 329.1287.