Abstract
A novel series of 7,8-dichlorobenzofuro[3,2-c]quinoline-6,9,10(5H)-triones was obtained regioselectively in good yields. The products were formed by the reactions of the 4-hydroxy-2(1H)-quinolinones with 3,4,5,6-tetrachloro-1,2-benzoquinone in tetrahydrofuran as the solvent. Infrared, nuclear magnetic resonance (two-dimensional nuclear magnetic resonance), mass spectra and elemental analysis were used to elucidate the structures of new compounds.

Introduction
2-Quinolones have considerable interest as anticancer
1
and antiviral
2
reagents. 4-Substituted 3-phenyl-2-quinolones exhibit high affinity in binding to the glycine site of the N-methyl-

Examples of natural occurring heterocycles having a furanopyridine moiety.
Previously, we prepared ethyl pyrano[3,2-c]quinoline-4-carboxylates (I, Figure 2), in good yields. 8 The reactions of 4-hydroxy-1-phenylquinolin-2(1H)-one with 2-(2-oxo-1,2-dihydroindol-3-ylidene)malononitrile produced spiro(indoline-3,4′-pyrano[3,2-c]quinoline)-3′-carbonitriles (II, Figure 2) 9 in addition to our synthesis of 2,3-bis-(4-hydroxy-2-oxo-1,2-dihydroquinolin-3-yl)succinates and arylmethylene-bis-3,3′-quinoline-2-ones (III, Figure 2). 10

Representative examples of our previous work.
N-2,3-Bis(6-substituted-4-hydroxy-2-oxo-1,2-dihydro-quinolin-3-yl)naphthalene-1,4-diones and their substituted triethylammonium salts (IV, Figure 2) were also synthesized as potential antineoplastic reagents. 11 Moreover, we demonstrated on the design and synthesis of fused naph-thofuro[3,2-c]quinoline-6,7,12-triones (V, Figure 2) and pyrano[3,2-c]quinoline-6,7,8,13-tetraones (VI, Figure 2), which have shown inhibition towards extracellular-signal-regulated kinase (ERK). 12
In general, quinolones and their fused analogues are in high demand for pharmaceuticals. Therefore, the development of simple and mild methods for synthesizing quinolone derivatives is always needed. Consequently, we have an interest in investigating the reaction of 4-hydroxy-2(1H)-quinolinones
Results and discussion
The reaction of 4-hydroxy-2(1H)-quinolinones

Reactions of 2-quinolones
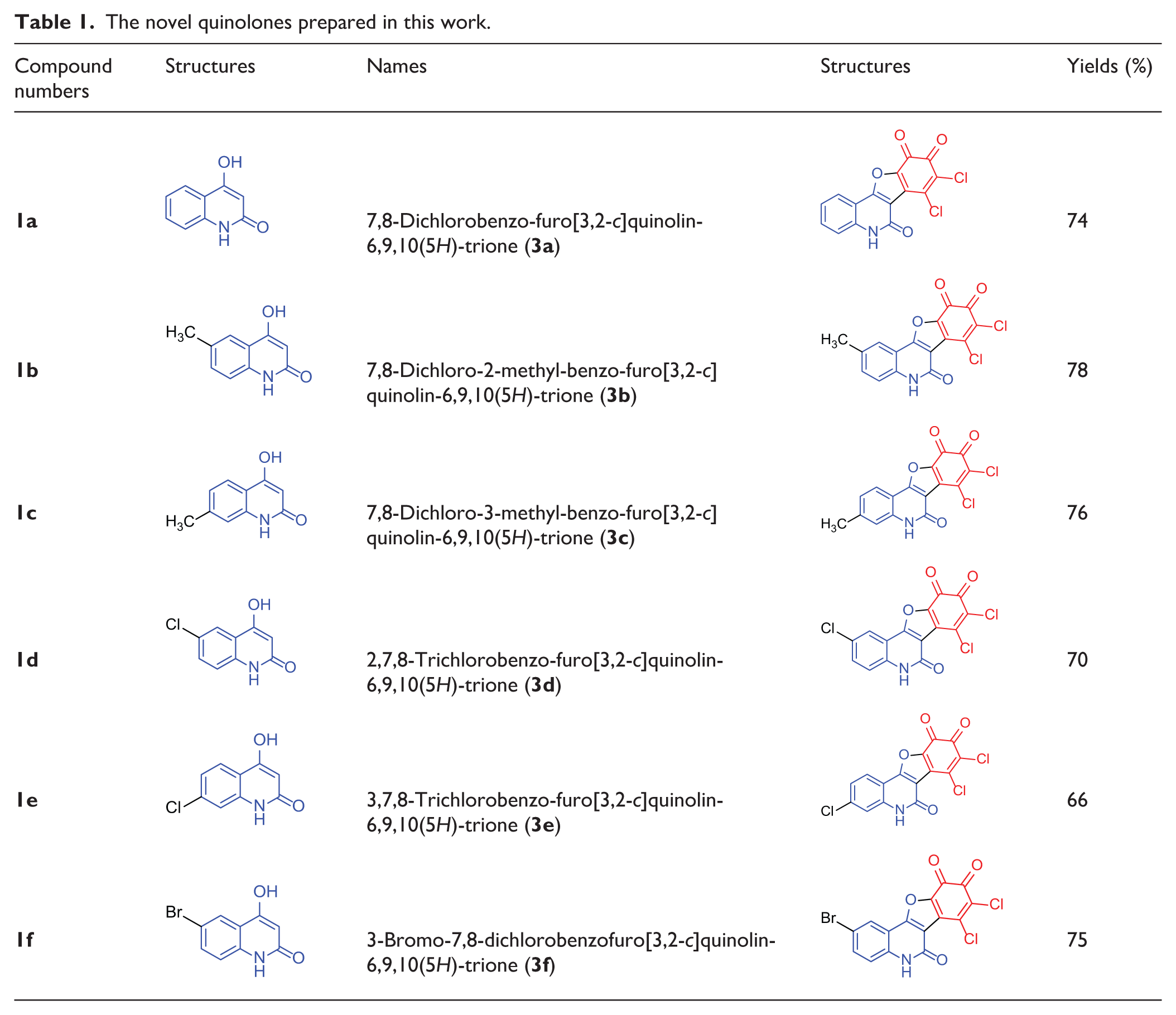
The structures of the products were established by including infrared (IR), mass spectra, nuclear magnetic resonance (NMR) spectra and elemental analyses. Table 1 summarizes the results obtained.
The novel quinolones prepared in this work.
The structure of product

The structures of the two possible regioisomers
The mechanism of the reaction may involve a Michael addition at C-3 of the quinolone to the α-carbon of its quinone
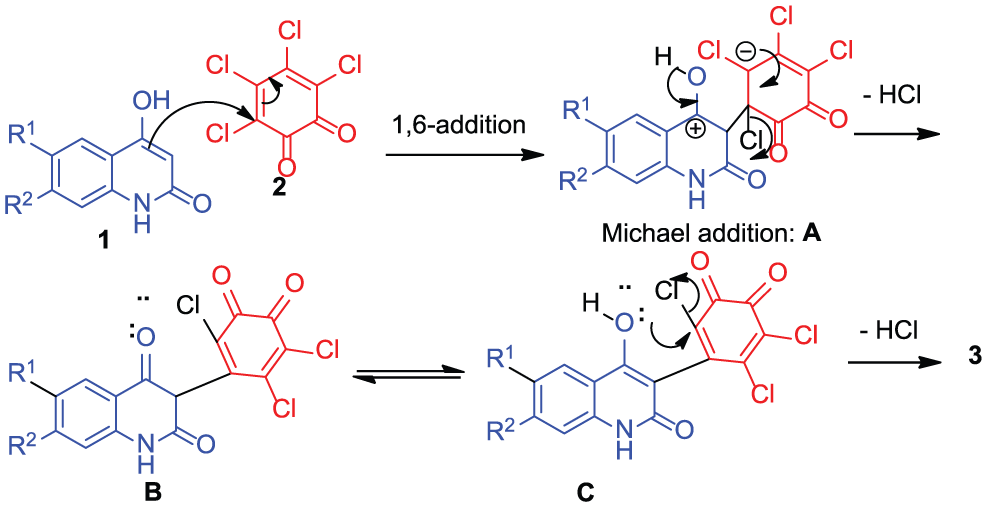
Mechanism describes the formation of compounds
Conclusion
In this study, a series of novel 7,8-dichloro-benzofuro[3,2-c]quinoline-6,9,10(5H)-triones was successfully regioselectively prepared.
Experiment
Materials and methods
Melting points were measued on a Gallenkamp melting point apparatus (Weiss-Gallenkamp, Loughborough, UK) and are uncorrected. The IR spectra were obtained with a FT device at the Minia University, Egypt. Elemental analyses were obtained at the Microanalytical Centre at the Cairo University, Egypt (a Bruker AV-400 spectrometer; 400 MHz for 1H, 100 MHz for 13C and 40.55 MHz for 15N) was used to measure NMR spectra. The new compounds were dissolved in dimethyl sulphoxide (DMSO)-d6; and the chemical shifts are expressed in δ (ppm), versus internal tetramethylsilane (TMS) = 0 for 1H and 13C, and external liquid ammonia = 0 for 15N. A Finnigan Fab spectrometer (70 eV) device was used to record mass spectra at the Institute of Organic Chemistry, Karlsruhe University, Karlsruhe, Germany. Thin layer chromatography (TLC) was carried out on silica aluminum sheets (Kieselgel 60) with PF254 indicator at λmax = 254 nm.
Starting materials
Compounds
Products
A suspension of compounds
7,8-Dichlorobenzofuro[3,2-c]quinolin-6,9,10(5H)-trione (
7,8-Dichloro-2-methyl-benzofuro[3,2-c]quinolin-6,9,10(5H)-trione (
7,8-Dichloro-3-methyl-benzofuro[3,2-c]quinolin-6,9,10(5H)-trione (
2,7,8-Trichlorobenzofuro[3,2-c]quinolin-6,9,10(5H)-trione (
3,7,8-Trichlorobenzofuro[3,2-c]quinolin-6,9,10(5H)-trione (
2-Bromo-7,8-dichlorobenzofuro[3,2-c]quinolin-6,9,10(5H)-trione (
Supplemental Material
Supplm_Data_19-12-2019 – Supplemental material for Regioselective synthesis of new 7,8-dichlorobenzofuro[3,2-c]quinoline-6,9,10(5H)-triones from reactions of 4-hydroxy-2-quinolones with 3,4,5,6-tetrachloro-1,2-benzoquinone
Supplemental material, Supplm_Data_19-12-2019 for Regioselective synthesis of new 7,8-dichlorobenzofuro[3,2-c]quinoline-6,9,10(5H)-triones from reactions of 4-hydroxy-2-quinolones with 3,4,5,6-tetrachloro-1,2-benzoquinone by Ashraf A Aly, Alaa A Hassan, Nasr K Mohamed, Lamiaa E Abd El-Haleem and Stefan Bräse in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank DFG and the Egyptian Mission for providing fellowships to Prof. Ashraf A. Aly and to Mrs Lamiaa E. Abd El-Haleem to enable them to perform analytical measurements at the Karlsruhe Institute of Technology, Institute of Organic Chemistry, 76131 Karlsruhe, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
