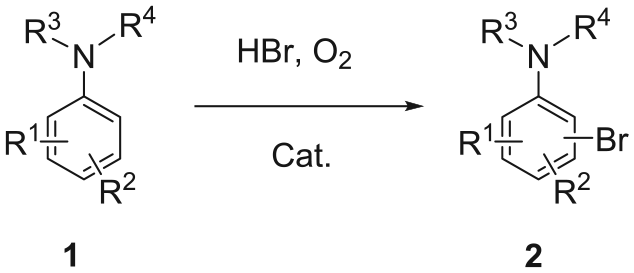
Abstract
An aerobic oxidative bromination of anilines and aryl ketones catalyzed by recyclable 2-methylpyridinium nitrate ionic liquid is achieved in water using hydrobromic acid as the bromine source and molecular oxygen as the oxidant. The catalytic system shows good efficiency and atom economy.

Introduction
Brominated compounds are important intermediates that have been widely used in the synthesis of pharmaceuticals, biologically active compounds, agrochemicals, and other functional chemicals.1,2 The C–Br bond can be easily transformed into C–C and C–heteroatom bonds via various classic reactions such as Stille, Suzuki, Heck, and Sonogashira.3–6 The development of new and “greener” methods for bromination has consistently been one of the topics that chemists have focused on. Oxidative bromination is a promising reaction which can be performed with a combination of bromide anions and a variety of oxidants, 7 among which H2O28,9 and O210–14 are the most environmentally friendly. However, there is a risk of explosion in the case of H2O2 employed as an oxidant, especially at elevated temperatures. Recently, the Wang group released an oxidative bromination with ionic liquid (IL) as both solvent and catalyst. 14 However, they use stoichiometric IL and corrosive bromine as agent, and only anisole derivatives are successfully brominated. In our previous report, 15 a non-metal catalyzed aerobic oxidative bromination of anisoles and phenols was established, which could be carried out smoothly in the presence of a catalytic amount of 2-methylpyridinium nitrate ionic liquid (2-MePy·HNO3). (2-Methylpyridinium nitrate is a white hygroscopic solid that melts while being slightly heated or exposed to humid air, affording a sticky liquid; strictly speaking, it is an “IL” but not an “RTIL” (room-temperature ionic liquid).) We speculated that this system can also be applied to the aromatic bromination of anilines and α-bromination of ketones.
Results and discussion
According to the conditions reported in our previous article, we started our optimization studies for aniline bromination with 0.3 equivalents of 2-methylpyridinium nitrate and hydrobromic acid at 80 ℃; however, only 55% conversion was achieved (see Table S1 in Supplemental Material). The conversion increased when more of the catalyst was added and 1.1 equivalents of the IL, and a temperature of 90 ℃ is needed to obtain the maximum efficiency. The effect of other solvents (common solvent such as H2O, CH2Cl2, tetrahydrofuran (THF), methanol, ethanol, and ethyl acetate (EA)) was subsequently investigated. The results showed that water introduced from hydrobromic acid (40% aqueous solution), rather than other additional solvents, gave the best conversion for the reaction (Table S1 in Supplemental Material). We assume that the reason for the low conversion in these additional solvents is due to the diluted reactants and lower boiling point of these solvents.
Subsequently, we expanded the substrate scope with different substituents on the phenyl ring and nitrogen, and the results are listed in Table 1. Aniline derivatives with an electron-donating group (EDG) or an electron-withdrawing group (EWG) on the phenyl ring could all be monobrominated in good yields (Table 1). For p-substituted anilines (Table 1, entries 4–6, 9, and 10), monobromination occurred at the o-position relative to the amino group. For other anilines, bromination occurred preferentially at the p-position (Table 1, entries 1–3, 7, 8 and 11). Substitution of a hydrogen atom on nitrogen with a methyl did not strongly affect the bromination efficiency (Table 1, entry 11). Normally, dibromination of anilines is more difficult than monobromination because the phenyl ring is slightly deactivated by the existing bromine atom. It is interesting to note that when the amount of hydrogen bromide was raised to 2.8 equivalents, the dibrominated derivative became the major product in good yield (Table 1, entry 12). The stoichiometric use of nitrate does not limit the sustainability of this system. The catalyst can be recycled at least four times without a significant decrease in activity (See Table S2 in Supplemental Material)
Aerobic bromination of aniline derivatives catalyzed by 2-MePy·HNO3. a
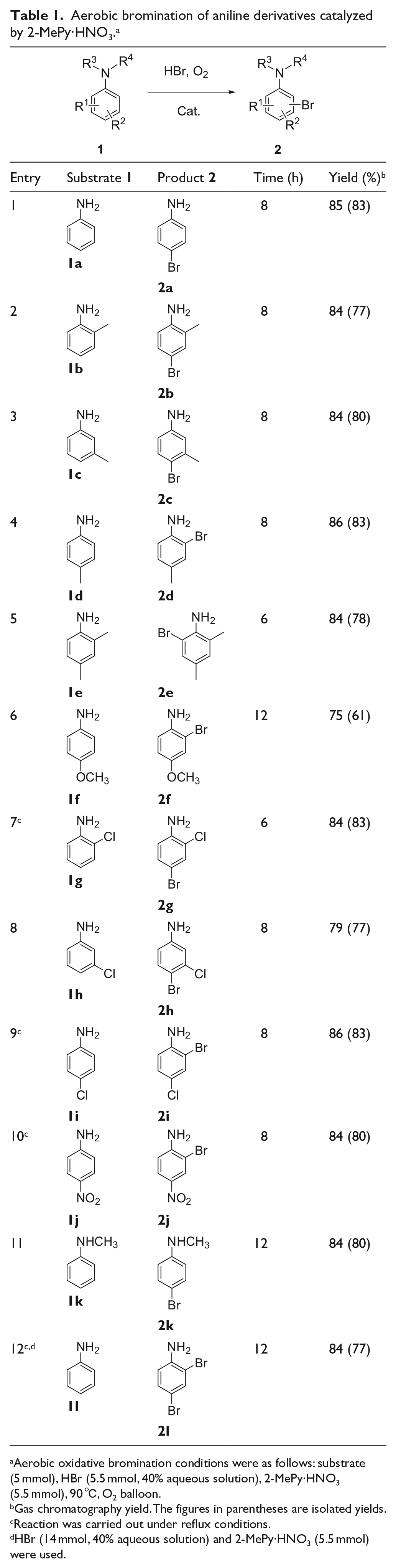
Aerobic oxidative bromination conditions were as follows: substrate (5 mmol), HBr (5.5 mmol, 40% aqueous solution), 2-MePy·HNO3 (5.5 mmol), 90 ℃, O2 balloon.
Gas chromatography yield. The figures in parentheses are isolated yields.
Reaction was carried out under reflux conditions.
HBr (14 mmol, 40% aqueous solution) and 2-MePy·HNO3 (5.5 mmol) were used.
Different from anilines, aryl ketones can be transformed into their corresponding α-brominated derivatives in high yields with 0.1 equivalents of 2-methylpyridinium nitrate and 1.1 equivalents of HBr (40% aqueous solution) at 60 ℃ (see Table S3 in Supplemental Material for optimization of the reaction conditions). There is no significant substituent effect of the phenyl ring on this α-bromination of aryl ketones
Aerobic α-bromination of aryl ketones catalyzed by 2-MePy·HNO3. a

Aerobic oxidative bromination conditions were as follows: substrate (5 mmol), HBr (5.5 mmol, 40% aqueous solution), 2-MePy·HNO3 (0.5 mmol), O2 balloon, 60 ℃.
Gas chromatography yield. The figures in parentheses are isolated yields.
The proposed mechanism is described as a sequential cascade of double-cycle oxidation–reduction reactions (Scheme 1). Nitrate anions play a critical catalytic role in “activating” molecular oxygen.16–19 The pyridinum cation may synergistically enhance the catalytic activity of nitrate. In our previous report, 2-methylpyridinum shows the best efficiency as counter ion for nitrate. 14

Proposed mechanism of the nitrate-catalyzed aerobic oxidative bromination.
Conclusion
In summary, the 2-MePy·HNO3–catalyzed aerobic oxidative bromination of anilines and ketones has been developed with hydrobromic acid as the bromine source. Anilines and ketones have been converted into their corresponding bromo-compounds in good yields. Compared with existing reports of oxidative brominations in ILs, 14 only catalytic amount of non-metal IL is used in our system with broader substrate scope. The whole procedure avoids the handling of Br2 and toxic volatile organic solvents, and shows good efficiency and atom economy.
Experimental
General procedure
A mixture of substrate
Purification and identification of the product
For 2a -2l
Ten milliliters of saturated Na2CO3 aqueous solution was added at the end of the reaction, and the resulting mixture was extracted with 20 mL EA three times. The combined organic layer was washed with water and dried over Na2SO4; the concentrated residue was purified by column chromatography (silica gel, petroleum ether (PE): EA from 200:1 to 80:1) to afford product
For 4a -4e
Ten milliliters of saturated NaHSO3 aqueous solution was added at the end of the reaction, and the mixture was extracted with 20 mL EA three times. The combined organic layer was then washed with water and dried over Na2SO4; the concentrated residue was purified by column chromatography (silica gel, PE: dichloromethane (DCM) from 50:1 to 30:1) to afford product
Characterization of products
2a:4-Bromoaniline
The compound was obtained as brown solid, yield 0.714 g (83%), m.p. 64–66 oC. δ H (500 MHz, CDCl3; TMS) 7.26–7.23 (m, 2H), 6.58–6.55 (m, 2H), 3.67 (br, 2H).
2j: 2-Bromo-4-nitroaniline
The compound was obtained as brown solid, yield 0.868 g (80%), m.p. 100–102 °C. δ H (500 MHz, CDCl3; TMS) 8.39 (d, J = 2.5 Hz, 1H), 8.04 (dd, J = 8.9, 2.5 Hz, 1H), 6.75 (d, J = 8.9 Hz, 1H), 4.84 (s, 2H).
4e: 2-Bromoindanone
The compound was obtained as yellow sticky oil, yield 0.865 g (82%). δ H (500 MHz, CDCl3; TMS) 7.85 (d, J = 7.5 Hz, 1H), 7.67 (t, J = 7.5 Hz, 1H), 7.47–7.43 (m, 2H), 4.66 (dd, J = 7.5, 3.2 Hz, 1H), 3.84 (dd, J = 18.1, 7.5 Hz, 1H), 3.43 (dd, J = 18.1, 3.2 Hz, 1H).
Supplemental Material
Supporting_Information – Supplemental material for A metal-free aerobic oxidative bromination of anilines and aryl ketones with 2-methylpyridinium nitrate as a reusable ionic liquid
Supplemental material, Supporting_Information for A metal-free aerobic oxidative bromination of anilines and aryl ketones with 2-methylpyridinium nitrate as a reusable ionic liquid by Ming-Fang Li, Jian Wang, Yong-Xin Ke, Song-Cheng Pan, Hong Yin, Wenting Du and Jing-Hua Li in Journal of Chemical Research
Footnotes
Acknowledgements
We are grateful for the support provided by the Zhejiang Provincial Program for Cultivation of High-Level Innovative Health Talents (2014).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.