Abstract
A series of Zn(II) and Co(II) complexes have been synthesized by template condensation of bis(benzil)-4-chloro-1,2-phenylenediamine and bis(benzil)-4-fluoro-1,2-phenylenediamine, respectively, with 4-chloro-1,2-phenylenediamine and 4-fluoro-1,2-phenylenediamine in the presence of transition metal chlorides. The complexes have been characterized by physicochemical and spectroscopic techniques. Based on these studies, an octahedral geometry has been proposed around the metal ions. The newly synthesized ligands and their metal complexes have been screened for antimicrobial and anticancer activities. The in vitro cytotoxicity towards the HeLa cell line has been evaluated by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay.
Macrocyclic complexes have been synthesized by template condensation followed by their characterization by various spectral techniques.

Introduction
The research field dealing with macrocyclic metal complexes is very broad, due in part to their potential interest in a number of interdisciplinary areas that include bioinorganic chemistry, catalysis and magneto chemistry. The design and study of well-arranged metal-containing macrocycles is an interesting field of chemistry and has attracted the attention of both inorganic and bioinorganic chemists in recent years. 1 The field of macrocyclic chemistry of metals is developing very rapidly because of its importance in the area of coordination chemistry. 2 Macrocyclic compounds have attracted increasing interest due to their mixed soft–hard donor character, versatile coordination behaviour and in the understanding of molecular processes.3,4 The multiple roles played by naturally occurring macrocycles in biological systems are well known. The chemistry of synthetic macrocyclic compounds is also of great importance due to their use as pigments and dyes, magnetic resonance imaging (MRI) contrast agents and as models for naturally occurring macrocycles. 5 Many macrocyclic compounds have been reported showing antibacterial and antifungal activities.6,7 A large number of Schiff bases and their complexes have been studied because of their interesting and important properties, such as their ability to reversibly bind to oxygen and to act as bactericidal, and fungicidal agents.8,9 Schiff bases are potential anticancer drugs and, when administered as their metal complexes, the anticancer activity of those complexes is enhanced in comparison to the free ligands. A large number of transition metal complexes with high anticancer activity have been reported including these of Co(II) and Zn(II). Many efforts have been made for the development of new and effective anticancer metal complexes by varying the coordination modes, metals or ligands. Schiff base macrocycles of transition and inner-transition metal complexes have been prepared extensively for their wide applications.10–17 In addition, the in vitro antitumor activity against human breast cancer and human hepatocarcinoma cell lines has been studied.18–20
In this paper, we report the synthesis and spectral characterization of new macrocyclic transition metal complexes. An evaluation of their antimicrobial and anticancer activities has also been performed in order to understand the pharmaceutical applications of these metal complexes.
Results and discussion
The prepared macrocyclic complexes are coloured solids and monomeric in nature. The complexes are soluble in dimethylformamide (DMF), dimethylsulfoxide (DMSO), acetonitrile and chloroform.
Synthesis of ligands (bis(benzil)-4-chloro-1,2-phenylenediamine and bis(benzil)-4-fluoro-1,2-phenylenediamine)
For the preparation of the ligands, ethanolic solutions (40 mL) of benzil (20 mmol, 4.20 g) and either 4-chloro-1,2-phenylenediamine (10 mmol, 1.42 g) or 4-fluoro-1,2-phenylenediamine (10 mmol, 1.26 g) were mixed in a 2:1 molar ratio. The reaction mixture was heated under reflux for 4–6 h on a ratio head. The volume of the reaction mixture was concentrated to half and the remaining solution was cooled and kept in a desiccator overnight for evaporation of excess solvent. Recrystallization of the coloured crystalline products was performed using the same solvent and the crystals were dried in vacuo. Metal complexes of Pd(II) and Cr(II) with the above synthesized ligands have been reported,21,22 and the present work seeks to expand the range of metal complexes of these ligands by looking at complexation of Co(II) and Zn(II). The synthetic route towards the ligands is shown in Scheme 1, and the analysis and physical properties of these ligands are given in Table 1.

Synthetic route for the preparation of ligands.
The physicochemical properties and analytical data of the ligands and their complexes.

ML1: bis(benzil)-4-chloro-1,2-phenylenediamine; ML2: bis(benzil)-4-fluoro-1,2-phenylenediamine; M.W.: molecular weight; M.P.: melting point.
Synthesis of macrocyclic complexes
Template condensation of the ligands bis(benzil)-4-chloro-1,2-phenylenediamine (ML1) and bis(benzil)-4-fluoro-1,2-phenylenediamine (ML2) with the diamines 4-chloro-1, 2-phenylenediamine and 4-fluoro-1,2-phenylenediamine in the presence of ZnCl2·4H2O and CoCl2·6H2O was carried out under ambient conditions.
Synthesis of [M(C40H26N4Cl3F)] complexes
The ligand ML1 (10 mmol, 5.27 g) was dissolved in methanol (40 mL) and taken into a 100 mL round-bottom flask. Next, a methanolic solution of 4-fluoro-1,2-phenylenediamine (10 mmol, 1.26 g) and the metal chloride (10 mmol) was added to the solution of ligand in a 1:1:1 molar ratio. After addition of all the reagents, the contents were refluxed for about 7–8 h on a ratio head. The volume of the reaction mixture was concentrated to half and the remainder kept in a desiccator at room temperature. The complexes, obtained as solids, were washed with methanol and dried in vacuum.
Synthesis of [M(C40H26N4Cl2F2)] and [M(C40H26N4Cl4)] complexes
A methanolic solution (40 mL) of ligand ML1 (10 mmol, 5.27 g) or ML2 (10 mmol, 5.10 g), 4-chloro-1,2-phenylenediamine (10 mmol, 1.42 g) and 4-fluoro-1,2-phenylenediamine (10 mmol, 1.26 g), in the presence of metal chloride (10 mmol) was mixed in a 1:1:1 molar ratio. The contents were refluxed for 7–8 h on a ratio head and then the volume of reaction mixture was concentrated to half and the remainder kept in a desiccator at room temperature. The complexes, obtained as solids, were washed with methanol and dried under vacuo. 22 The template synthesis of the complexes can result in the formation of mixture of two regioisomers due to different orientations of X-substituted aromatic rings relative to each other, as shown in Scheme 2.

Synthetic route to the macrocyclic metal complexes.
All the complexes were recrystallized from a 1:1 molar solution of methanol and benzene. The purity of the complexes was checked by thin-layer chromatography (TLC).
Infrared spectra
A comparative study of the infrared (IR) spectra of the precursors and their complexes confirmed the formation of macrocyclic complexes with the proposed coordination pattern. The IR spectra of the diamines and ligands exhibit characteristic bands due to vas(NH2) at 3380 cm−1, vs(NH2) at 3250 cm−1 and v(C=O) in the region of 1670–1680 cm−1. However, in the spectra of all the macrocyclic complexes, these bands disappear and strong bands due to coordinated v(C=N) vibrations in the range of 1610–1625 cm−1 appear suggesting the deprotonation and formation of a macrocyclic framework. 21 This contention is further supported by the presence of bands at 1465–1495 and 1355–1390 cm−1, characteristic of a benzil moiety and may be assigned to vasymC6H5 and vsymC6H5, respectively. An important feature is the appearance of a new band of medium intensity in the range of 420–445 cm−1, attributed to ν(M-N), which provides strong evidence for the coordination of the imine nitrogen to the metal ion. 23 The assignments of the absorption frequencies of the ligands and their complexes are listed in Table 2.
IR (cm−1) and 1H NMR (δ ppm) spectral data of the ligands and their corresponding complexes.

IR: infrared; NMR: nuclear magnetic resonance; m: multiplet.
1H NMR spectra
Further evidence for the coordinating mode of the ligands was obtained from 1H nuclear magnetic resonance (NMR) spectra, which were recorded in DMSO-d6. The spectra of the ligands and Zn(II) complexes do not show any signals assignable to primary amino protons. This is strong evidence that the proposed macrocyclic complexes are formed by template reactions. The spectra of the complexes exhibit multiplets in the region of 7.24–8.21 ppm, which are assigned to aromatic protons. The integral intensities of each signal in the 1H NMR spectra of the macrocyclic complexes are found to agree with the number of different types of protons present. Table 2 lists the 1H NMR spectral data of the ligands and their complexes and the spectra are provided in the supporting information.
Electronic spectra
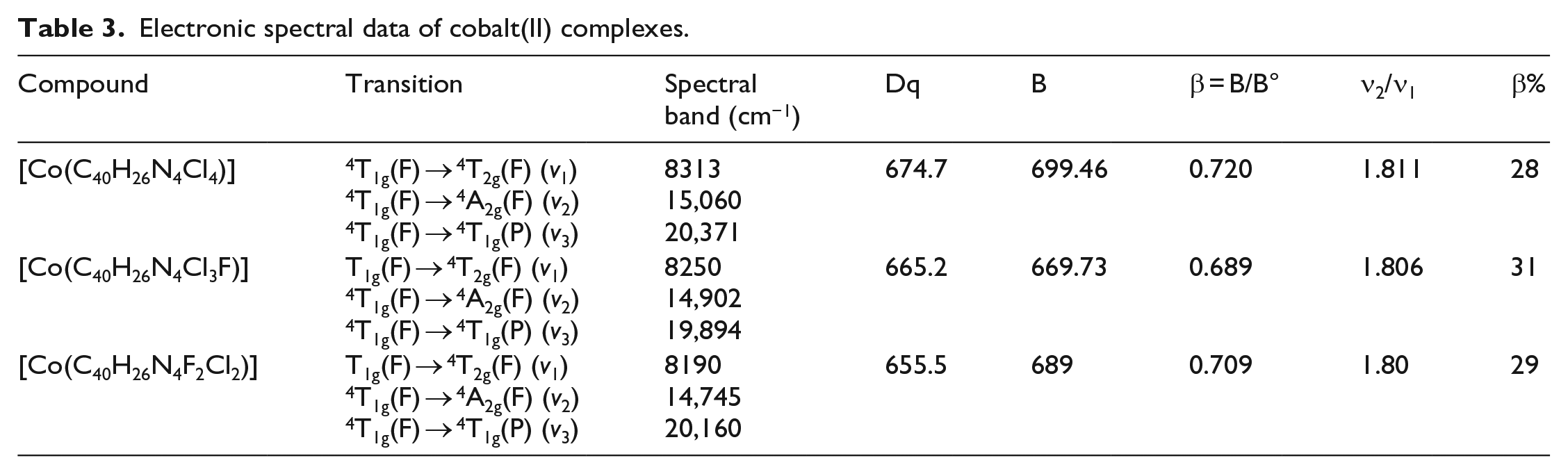
The electronic spectra of the Co(II) complexes were recorded in distilled DMSO. The absorption maxima at 270–335 nm in the spectra of the ligands are due to π → π* electronic transitions which appear almost at the same position in the spectra of the complexes. The spectra of both the ligands ML1and ML2 show a broad band at ~400 nm which can be assigned to the n–π* transitions of the azomethine group, which undergoes a blue shift in the spectra of the complexes due to chelation. 24 The shift of this band in the spectra of the complexes suggests the coordination of nitrogen to the metal atom. The electronic spectra of the macrocyclic Co(II) complexes exhibit three d–d spin-allowed bands in the range of 8000–20,500 cm−1, corresponding to 4T1g(F) → 4T2g(F), 4T1g(F) → 4A2g(F) and 4T1g(F) → 4T1g(P) transitions. Electronic spectral data of Co(II) complexes are shown in Table 3. The v2/v1 was also calculated and is in close agreement with the octahedral geometry around the cobalt(II) ion.
Electronic spectral data of cobalt(II) complexes.
EI mass spectra
The electron impact (EI) mass spectrum of the macrocyclic complex [Zn(C40H26N4Cl3F)] gives a molecular ion peak at m/z = 753.32 amu (calcd m/z = 753.40). The spectrum also shows important fragment ions at m/z 523.76 for [C26H20N4ZnCl2]+, 507.22 for [C25H15N4ZnCl2]+, 375.17 for [C17H15N4ZnCl2]+, 359.21 for [C16H11N4ZnCl2]+, 269.09 for [C14H10N4Cl]+ and 239.05 for [C14H9N2Cl]+. This is in good agreement with the respective theoretical molecular mass of the complexes and also these data confirm the mononuclear nature of the Zn(II) complexes. The mass spectrum of the Zn complex is given in the supporting information.
Biological assays
Antimicrobial studies
In the present study, the ligands and their macrocyclic complexes were evaluated for their antimicrobial activity against four bacteria: Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa and Bacillus subtilis, and two fungi: Aspergillus fumigatus and Candida albicans. The results are summarized in Table 4.
Antimicrobial screening data for the ligands and their complexes.
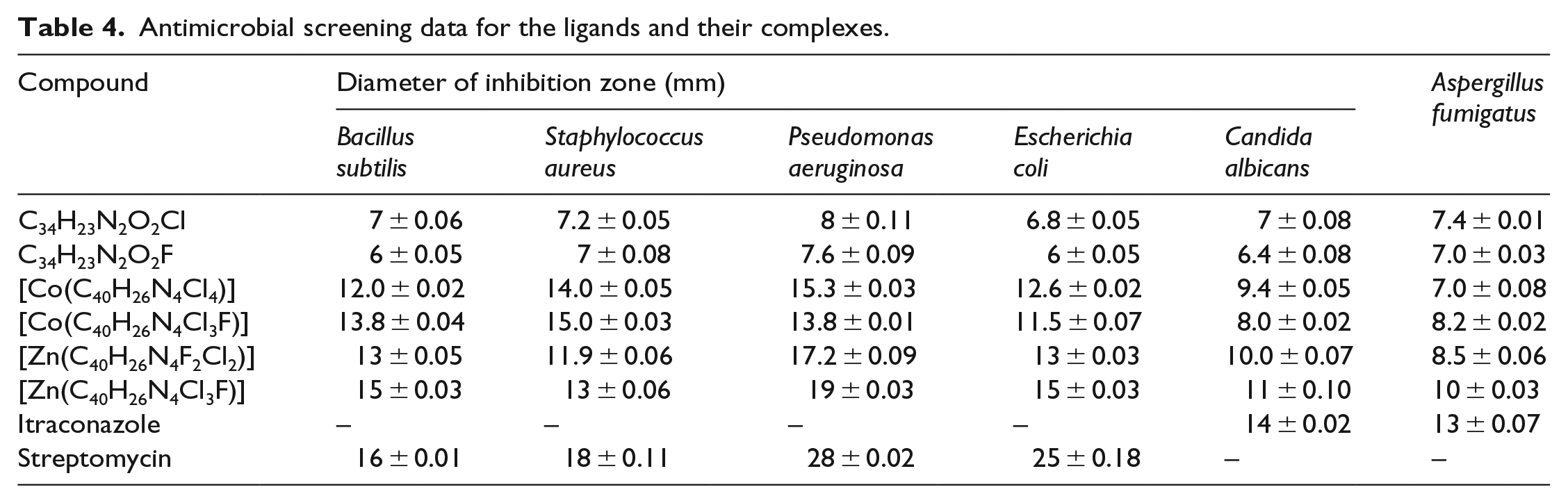
The results were compared with those of the standard drug streptomycin for bacteria and itraconazole for fungi. The ligands and their respective complexes were found to be active against all the fungal and bacterial strains. The results clearly indicate that ML1 shows higher antifungal activity than ML2. The antimicrobial screening data indicate that the metal complexes are more potent antimicrobial agents than the free ligands. The enhanced activity of the macrocyclic metal complexes compared to the free ligands may be explained by Tweedy’s chelation theory, 25 according to which chelation reduces the polarity of the central metal atom because of partial sharing of its positive charge with the ligand, which favours permeation of the complexes through the lipid layer of the cell membrane. 26 This would suggest that chelation could facilitate the ability of a complex to cross a cell membrane. 27
The results of the antimicrobial activity indicate that the activity of the [Zn(C40H26N4Cl3F)] complex against B. subtilis, C. albicans and A. fumigatus is almost similar to the standard drugs. Other complexes of Co(II) and Zn(II) exhibit moderate to good activity against all the bacterial and fungal strains. Therefore, these complexes should be further investigated to assess their potential as future antimicrobial agents. The comparative results for the antimicrobial studies are shown in Figures 1 and 2.
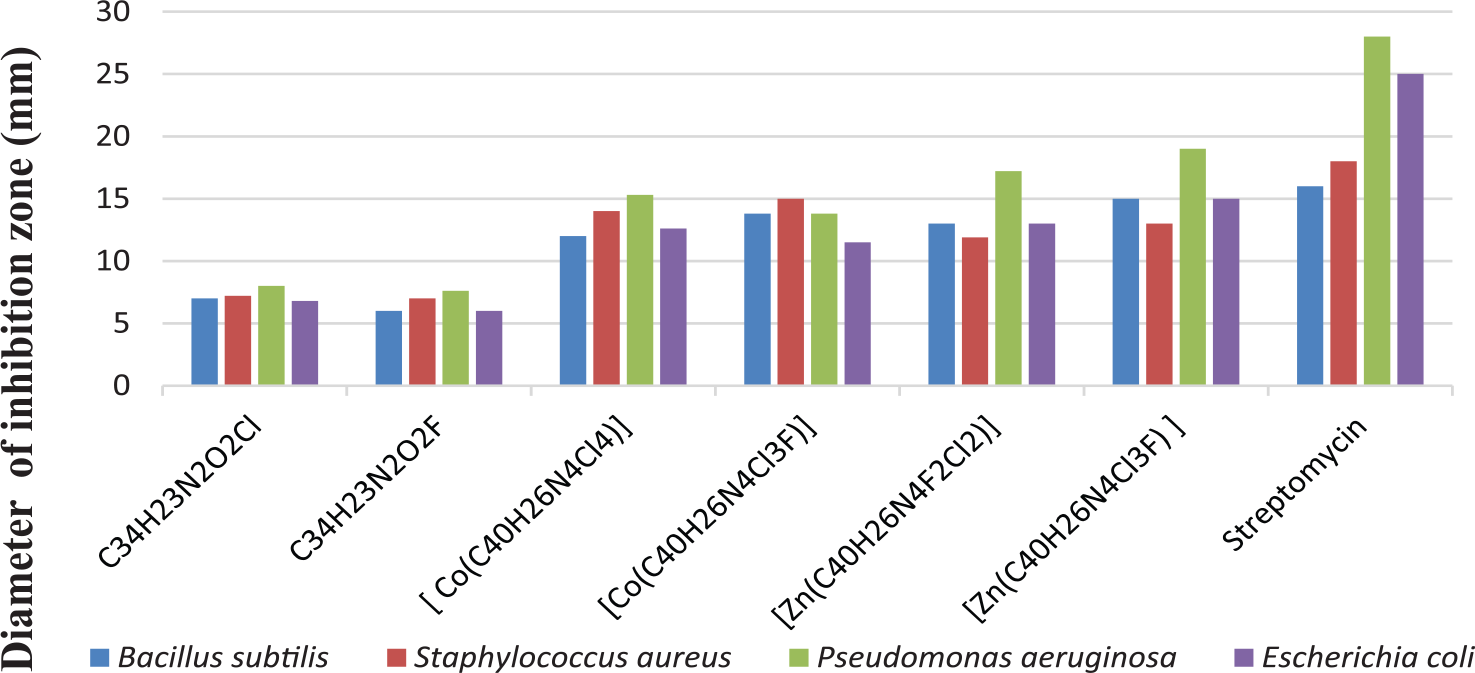
Antibacterial activity of the ligands and their metal complexes towards different bacteria.

Antifungal activity of the ligands and their metal complexes with different fungi.
Anticancer activity
The tested complexes show IC50 values in the range of 19.85–72.80 μg mL−1 towards HeLa cell lines (cervical cancer cell line). The results indicate that complex [Co(C40H26N4Cl4)] showed the highest cytotoxic activity on HeLa cell lines (19.85 μg mL−1). The macrocycle, as well as the type of metal ion, may be the reason for their different anticancer activities. The percentage growth inhibition of the HeLa cell line increases with increasing concentration of the tested compounds. The comparative IC50 values of the macrocyclic metal complexes on the HeLa cell line are shown in Table 5.
Lethal concentration (IC50) of the macrocyclic metal complexes on the HeLa cell line.

Anticancer activity of cisplatin as a standard complex
Cisplatin was used as a standard anticancer agent. The observed IC50 value for cisplatin against the HeLa cell line was 4.489 µg. The cytotoxic effect of cisplatin on the HeLa cell line (cervical cancer cell line) is shown in Figure 3.

Cytotoxic effect of the cisplatin on HeLa cell line.
Table 5 shows a comparison of the IC50 values of the Co(II) and Zn(II) complexes with cisplatin, indicating the relatively lower anticancer activity of the metal complexes, which may be because of the unique characteristics such as redox activity, variable coordination modes and nature of the metals and ligands. Metal–ligand interactions also offer a wide range of advantages to metals to interact and coordinate with biological molecules, thus leading to differences in their anticancer potential. The poor solubility and low bioavailability of Co(II) and Zn(II) complexes and their preferential selectivity towards certain cell lines may also contribute to their low IC50 values. Nevertheless, all the complexes show anticancer activity. The cytotoxic properties for all the complexes are shown in Tables S1—S7, Figures 3 and S1–S6 and Images 1–6 provided as supporting information, which show the percentage growth inhibition with different concentrations of the complexes.
Conclusion
We have described the synthesis, characterization and biological activity of Co(II) and Zn(II) macrocyclic complexes. The ligands and their complexes were physicochemically characterized using IR, ultraviolet (UV), 1H NMR and EI mass spectroscopy. Based on analytical and spectral data, an octahedral geometry has been proposed for these macrocyclic complexes. The results revealed the formation of monomeric macrocyclic metal complexes. The antimicrobial assays of the ligands and their complexes were carried out against various bacterial and fungal strains. The enhanced activity of the macrocyclic complexes compared with the parent ligands has been explained based on chelation theory. The [Co(C40H26N4Cl4)] complex exhibits a higher cytotoxicity as compared to other complexes against the HeLa cell line. The results obtained from the bioassays indicate that this class of compounds can be utilized for the design of new antimicrobial and anticancer agents.
Experimental
Materials
All chemicals used were of Analytical Reagent (AR) grade and purchased from Alfa Aesar. Solvents were dried and distilled using appropriate drying agents. All reactions were carried out under normal atmospheric conditions.
Analytical and physical measurements
Kjeldahl and Volhard’s methods were used for estimation of nitrogen and chlorine contents in complexes, respectively. 28 Gravimetric metal estimation was performed and the Rast Camphor method was used for molecular weight determination. 29 Melting points were determined using capillaries in electrical melting point apparatus (Perfit, India). The electronic spectra were recorded on a Shimadzu-1800 UV-Vis spectrophotometer; IR spectra of the ligands and their complexes were recorded using a Shimadzu FTIR-550 spectrophotometer. A JEOL-DELTA2-NMR 400 MHz spectrometer was used for recording the 1H NMR spectra in CDCl3 using tetramethylsilane (TMS) as the internal standard. The purity of the complexes was checked by TLC run in 1:1 benzene–methanol. The analysis and physical properties of these complexes are given in Table 1.
Biological assay
Test microorganism
The antimicrobial properties of all the complexes were evaluated and compared with the standard drug streptomycin for bacteria and itraconazole for fungi. The microorganisms used were E. coli, S. aureus, P. aeruginosa, B. subtilis, A. fumigatus and C. albicans.
In vitro antibacterial activity
All the test compounds were dissolved in DMSO and the concentration of stock solution was 5 mg mL−1. Each well was filled with 50 μL of test compound. Streptomycin was used as a positive control (5 mg mL−1 concentration). Agar plates were prepared for the antibacterial activity. The Mueller–Hinton agar medium and Sabouraud dextrose agar medium are susceptibility test mediums that have been validated by the Clinical and Laboratory Standards Institute (CLSI) for screening the antimicrobial activity by well-diffusion susceptibility testing. Fresh cultures of test isolates of E. coli, S. aureus, P. aeruginosa and B. subtilis were inoculated in peptone water and incubated for 30 min at 37°C. The bacterial and fungal suspensions were compared to 0.5 McFarl and turbidity standard. Microbial cultures were swabbed onto the Mueller–Hinton agar surface. Wells were then filled with 50 μL of different dilutions prepared from stock. The E. coli, S. aureus, P. aeruginosa and C. albicans plates were incubated at 37°C for 24–48 h and the results recorded.
The antibacterial activity of the common standard antibiotic streptomycin was also recorded using the same procedure. The medium with DMSO as solvent was used as a negative control. The experiments were performed in triplicate.
In vitro antifungal activity
Different concentrations of the test compounds under study were prepared from the stock solution (5 and 2.5 mg mL−1), of which 50 μL was used in each well. Itraconazole was used as a positive control (5 mg mL−1 concentration). The well-diffusion method was used for screening of antifungal activity using Sabouraud dextrose agar medium. A. fumigatus and C. albicans were inoculated in normal saline (0.9%) and kept for 48 h at 28°C. Both the fungi were swabbed on to the Sabouraud dextrose agar surface. Samples (50 μL) from different dilutions prepared from stock (5 and 2.5 mg mL−1 of the compound) were loaded into the respective wells. The antifungal plates were kept for incubation at 28°C for 7 days.
Comparison of the fungal activity of each compound was made with standard itraconazole (5 mg mL−1 (w/v)). The medium with DMSO as the solvent was used as a negative control, whereas the medium with itraconazole was used as a positive control. The experiments were performed in triplicate.
Anticancer activity
Measurement of cell viability and proliferation forms the basis for numerous in vitro assays of a cell population’s response to external factors. The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) cell proliferation assay measures the cell proliferation rate and conversely, when metabolic events lead to apoptosis or necrosis, the reduction in cell viability.
Materials and methods
Dulbecco’s modified eagle medium (DMEM), MTT, trypsin EDTA and phosphate-buffered saline (PBS) were purchased from Sigma chemicals Co., and foetal bovine serum (FBS) was purchased from Gibco. The 25 and 75 cm2 flasks and 96-well plates were purchased from Eppendorf India.
Maintenance of cell lines
The cancer cell lines were purchased from NCCS, Pune and the cells were maintained in DMEM medium supplemented with 10% FBS and the antibiotics penicillin/streptomycin (0.5 mg mL–1) in an atmosphere of 5% CO2/95% air at 37°C.
Preparation of test compounds
For the MTT assay, each test compound was weighed separately and dissolved in DMSO. The final concentration was adjusted to 1 mg mL−1 using medium and the cells were treated with a series of concentrations from 10 to 100 µg mL−1.
Cell viability by MTT assay
Principle
The MTT assay is a colorimetric assay that measures the reduction of yellow MTT by mitochondrial succinate dehydrogenase. The assay depends both on the number of cells present and on the assumption that dead cells or their products do not reduce tetrazolium. MTT enters the cells and passes into the mitochondria where it is reduced to insoluble, dark-purple-coloured formazan crystals. The cells are then solubilized in DMSO and the released solubilized formazan reagent is measured spectrophotometrically at 570 nm.
Procedure
Cell viability was evaluated by the MTT assay with three independent experiments with six concentrations of compounds in triplicate. Cells were trypsinized and the trypan blue assay was performed to assess viable cells in cell suspensions. Cells were counted with a hemocytometer and seeded at a density of 5.0 × 103 cells/well in 100 μL of medium in 96-well plate culture medium and incubated overnight at 37°C. After incubation, the old medium was removed and fresh medium (100 µL) was added with different concentrations of test compounds in representative wells in the 96-well plates. After 48 h, the drug solution was discarded and fresh medium and MTT solution (0.5 mg mL–1) were added to each well and the plates were incubated at 37°C for 3 h. At the end of the incubation time, precipitates formed as a result of the reduction of the MTT salt to the chromophore formazan crystals by the cells with metabolically active mitochondria. The optical density of solubilized crystals in DMSO was measured at 570 nm on a microplate reader.
Supplemental Material
supplementry_data_(2) – Supplemental material for Synthesis, characterization and biochemical behaviour of macrocyclic complexes of Zn(II) and Co(II) metals
Supplemental material, supplementry_data_(2) for Synthesis, characterization and biochemical behaviour of macrocyclic complexes of Zn(II) and Co(II) metals by Nighat Fahmi, Monika Upadhyay, Naveen Sharma and Savita Belwal in Journal of Chemical Research
Footnotes
Acknowledgements
M.U. thanks UPE Non-NET Fellowship.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
