Experimental
All chemicals were purchased (Aladdin or Sinopharm Chemical Reagent Co., Ltd) for the reactions are analytical grade and were used without further purification. Solvents were dried and stored using standard procedures. Highly moisture- or oxygen- sensitive reactions were performed under a nitrogen atmosphere. Melting points were taken on a Gallenkamp melting point apparatus without calibration. Flash column chromatography and thin-layer chromatography (TLC) inspections were performed on Aladdin silica gel (200–300 mesh) and silica gel plates (GF254), respectively. The 1H NMR and 13C NMR spectra were recorded on Bruker AM-500 MHz, Bruker AM-600 MHz spectrometers. high-resolution mass spectra (HRMS) were obtained on a Bruker APEXII47e spectrometer.
(E)-Ferulic acid ethyl ester (3)
To an oven-dried round-bottom flask (500 mL) charged with vanillin (2) (38.48 g, 252.88 mmol), monoethyl malonate (50.11 g, 379.32 mmol) and piperidine (4.0 g, 50.58 mmol) was added pyridine (100 mL) under air at room temperature. The reaction solution was stirred for 24 h at 100 °C. The resulting solution was cooled to 0 °C and hydrochloric acid was added dropwise until pH was 1–2. The mixture was allowed to stand for 24 h at −10 °C, and then filtered and dried. The residue was purified by flash column chromatography (petroleum/EtOAc = 6/1) to afford 3: white solid: m.p. 60–61 °C (Lit.
22
59–61 °C); yield: 55.08 g (98%); 1H NMR (500 MHz, CDCl3), δ 7.61 (d, J = 16 Hz, 1H, ArCH=CH), 7.06 (dd, J = 8 Hz, 2 Hz, 1H, ArH), 7.03 (d, J = 2 Hz, 1H, ArH), 6.91 (d, J = 8 Hz, 1H, ArH), 6.29 (d, J = 16 Hz, 1H, ArCH=CH), 5.92 (s, 1H, ArOH), 4.25 (q, J = 7 Hz, 2H, CH2CH3), 3.92 (s, 3H, OCH3), 1.33 (t, J = 7 Hz, 3H, CH2CH3); 13C NMR (125 MHz, CDCl3), δ 167.5 (C=O), 147.8, 146.7, 145.0, 127.5, 123.1, 115.8, 114.6, 109.5, 60.4, 55.8, 14.40. The spectral data are consistent with the literature.
23
Ethyl (E)-3-(3-methoxy-4-(methoxymethoxy)phenyl)acrylate (4)
To an oven-dried round-bottom flask (250 mL) charged with a solution of 3 (10 g, 45.0 mmol) in acetone (80 mL) was added anhydrous K2CO3 (18.66 g, 135.0 mmol) under air at room temperature. The reaction mixture was stirred for 1 h at room temperature and MOM-Cl (5.43 g, 67.49 mmol) was added dropwise at 0 °C. After stirring for 6 h, the reaction was quenched with water and the aqueous layer was extracted with ethyl acetate (3 × 20 mL). The combined organic extracts were washed with water (3 × 10 mL) and brine (3 × 10 mL), and dried (MgSO4), and the solvent was removed in vacuo. The crude product was purified by flash chromatography (petroleum/EtOAc = 6/1) to give 4: white solid; m.p. 50–53 °C; yield 11.74 g (98 %); 1H NMR (500 MHz, CDCl3), δ 7.62 (d, J = 16 Hz, 1H, ArCH=CH), 7.14 (d, J = 8 Hz, 1H, ArH), 7.08–7.06 (m, 2H, ArH), 6.32 (d, J = 16 Hz, 1H, ArCH=CH), 5.26 (s, 2H, CH3OCH2O), 4.25 (q, J = 7 Hz, 2H, CH2CH3), 3.91 (s, 3H, OCH3), 3.51 (s, 3H, CH3OCH2O), 1.33 (t, J = 7 Hz, 3H, CH2CH3); 13C NMR (125 MHz, CDCl3), δ 167.2 (C=O), 150.0, 148.4, 144.4, 128.8, 122.2, 116.5, 115.8, 110.2, 95.1, 60.4, 56.3, 55.9, 14.3. HRMS (ESI): M/Z [M]+ calcd for C14H18O5: 266.1154; found: 266.1159.
Ethyl 2-bromo-3-methoxy-3-(3-methoxy-4-(methoxymethoxy)phenyl)propanoate (5)
To an oven-dried round-bottom flask (100 mL) charged with a well-stirred solution of 4 (2.0 g, 7.51 mmol) in dry methanol (20 mL) was added NBS (1.47 g, 8.26 mmol) and iodine (0.19 g, 0.75 mmol) at −10 °C under a nitrogen atmosphere. After stirring for 10 h, the resulting solution was quenched with saturated NaHSO3 solution and extracted with EtOAc (3 × 20 mL). The combined organic extracts were washed with saturated NaHCO3 solution (3 × 20 mL) and brine (3 × 20 mL), and dried (MgSO4), and the solvent was removed in vacuo. The crude product was purified by flash chromatography (petroleum/EtOAc = 3/1) to yield 5: yellowish oil; yield 2.77 g (98%); 1H NMR (500 MHz, CDCl3), δ 6.96 (d, J = 8 Hz, 1H, ArH), 6.76–6.72 (m, 2H, ArH), 5.05 (s, 2H, CH3OCH2O), 4.32 (d, J = 10 Hz, 1H, CHBr), 4.10 (q, J = 7 Hz, 2H, CH2CH3), 4.05 (d, J = 10 Hz, 1H, ArCH), 3.70 (s, 3H, OCH3), 3.32 (s, 3H, CH3OCH2O), 3.03 (s, 3H, ArCHOCH3), 1.34 (t, J = 7 Hz, 3H, CH2CH3); 13C NMR (125 MHz, CDCl3), δ 168.5 (C=O), 149.7, 146.8, 130.8, 121.0, 115.6, 110.5, 95.2, 83.8, 61.7, 57.2, 55.9, 55.6, 47.4, 13.8; HRMS (ESI) calcd for C15H2179BrO6: 376.0522; found: 376.0535. The NMR data are similar with that reported in Bar’s
19
work.
Ethyl (Z)-3-methoxy-3-(3-methoxy-4-(methoxymethoxy)phenyl)acrylate (5′)
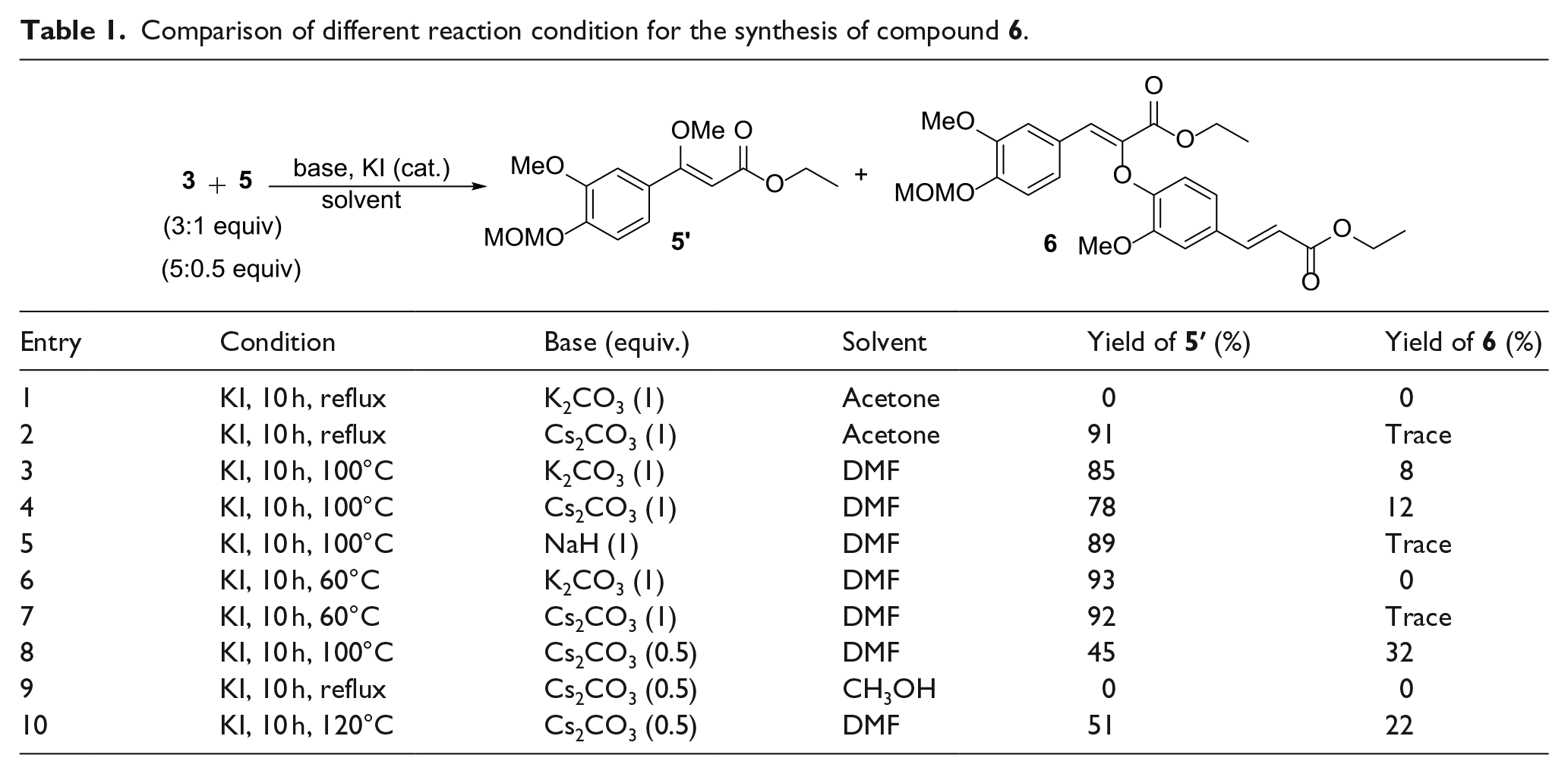
To an oven-dried round-bottom flask (100 mL) charged with 3 (888.96 mg, 4.0 mmol) in dry DMF (3 mL) was added Cs2CO3 (1.30 g, 4.0 mmol) under a nitrogen atmosphere at room temperature. After stirring for 1 h, catalytic amounts of KI (10%) was added and a solution of 5 (754.46 mg, 2.0 mmol) in dry DMF (2 mL) was added dropwise at 40 °C. The reaction mixture was stirred for 6 h at 40 °C under a nitrogen atmosphere. The resulting mixture was treated with water (20 mL) and extracted with EtOAc (3 × 50 mL). The combined organic layer was washed with water (3 × 20 mL) and brine (3 × 20 mL), and dried (MgSO4), and the solvent was removed in vacuo. The crude product was purified by flash chromatography (petroleum/EtOAc = 10/1) to yield 5′: colorless oil; yield 533.35 mg (90%); 1H NMR (500 MHz, CDCl3), δ 7.16–7.11 (m, 2H, ArH), 7.06 (s, 1H, ArH), 5.52 (s, 1H, CHC=O), 5.26 (s, 2H, CH3OCH2O), 4.20 (q, J = 7 Hz, 2H, CH2CH3), 3.90 (s, 3H, OCH3), 3.85 (s, 3H, ArCOCH3), 3.52 (s, 3H, CH3OCH2), 1.31 (t, J = 7 Hz 3H, CH2CH3). HRMS (ESI): M/Z [M]+ calcd for C15H20O6: 296.1260; found: 296.1268.
Ethyl (Z)-2-(4-((E)-3-ethoxy-3-oxoprop-1-en-1-yl)-2-methoxyphenoxy)-3-(3-methoxy-4-(methoxymethoxy)phenyl)acrylate (6)
To an oven-dried round-bottom flask (100 mL) charged with 3 (888.96 mg, 4.0 mmol) in dry DMF (3 mL) was added Cs2CO3 (651.64 mg, 2.0 mol) under nitrogen atmosphere at room temperature. After stirring for 1 h, a catalytic amount of KI of catalytic amounts (10%) was added and the solution of 5 (754.46 mg, 2.0 mmol) in dry DMF (2 mL) was added dropwise at 100 °C. The reaction mixture was stirred for 10 h at 100 °C under a nitrogen atmosphere. The resulting mixture was added water (20 mL) and extracted with EtOAc (3 × 50 mL). The combined organic layer was washed with water (3 × 20 mL) and brine (3 × 20 mL), and dried (MgSO4), and the solvent was removed in vacuo. The crude product was purified by flash chromatography (petroleum/EtOAc = 20/1) to yield 6: white solid; m.p. 91–93 °C; yield 311.36 mg (32%); 1H NMR (600 MHz, CDCl3), δ 7.60 (d, J = 16.2 Hz, 1H, ArCH=CH), 7.42 (d, J = 1.8 Hz, 1H, ArH), 7.36 (s, 1H, ArCH=C), 7.18 (dd, J = 8.4 Hz, 1.8 Hz, 1H, ArH), 7.13 (d, J = 1.8 Hz, 1H, ArH), 7.09 (d, J = 8.4 Hz, 1H, ArH), 6.98 (dd, J = 8.4 Hz, 1.8 Hz, 1H, ArH), 6.76 (d, J = 8.4 Hz, 1H, ArH), 6.31 (d, J = 16.2 Hz, 1H, ArCH=CH), 5.23 (s, 2H, CH3OCH2O), 4.26–4.22 (m, 4H, 2 × CH3CH2), 3.98 (s, 3H, OCH3), 3.76 (s, 3H, OCH3), 3.48 (s, 3H, CH3OCH2O), 1.33 (t, J = 7.2 Hz, 3H, CH2CH3), 1.22 (t, J = 7.2 Hz, 3H, CH2CH3); 13C NMR (150 MHz, CDCl3), δ 167.1 (C=O), 164.4 (C=O), 161.8, 149.6, 149.4, 148.9, 148.2, 144.2, 129.6, 127.5, 121.9, 120.4, 116.9, 116.2, 115.6, 111.0, 109.9, 105.2, 95.1, 60.5, 60.2, 56.4, 56.1, 55.9, 14.3, 14.2; HRMS (ESI): M/Z [M]+ calcd for C26H30O9: 486.1890; found: 486.1898.
2-[4-(2-Carboxyeth-1-en-1-yl)-2-methoxyphenoxy]-3-[3-methoxy-4-(methoxymethoxy)phenyl]prop-2-enoic acid (7)
To an oven-dried round-bottom flask (50 mL) charged with a solution of ester 6 (50.0 mg, 0.103 mmol) in EtOH (1 mL) was added 3 M aqueous solution of NaOH (3 mL). The mixture was stirred and reflux for 6 h. After cooling and concentration, diethyl ether (10 mL) and 3.0 M aqueous acetic acid (6 mL) were added at 0 °C. The organic phase was separated and the aqueous phase was extracted with diethyl ether (3 × 10 mL). The combined organic layers were washed with water (3 × 20 mL) and brine (3 × 20 mL), and dried (MgSO4), and the solvent was removed in vacuo. The crude product was purified by recrystallization (EtOAc/petroleum ether = 3/1) to yield 7: white solid; m.p. 187–189 °C; yield 42 mg (95%); 1H NMR (500 MHz, DMSO-d6), δ 7.51 (d, J = 16 Hz, 1H, ArCH=CH), 7.49 (s, 1H, ArH), 7.43 (s, 1H, ArH), 7.38 (s, 1H, ArCH=C), 7.23 (d, J = 8 Hz, 1H, ArH), 7.12 (d, J = 8 Hz, 1H, ArH), 7.03 (d, J = 8 Hz, 1H, ArH), 6.68 (d, J = 8 Hz, 1H, ArH), 6.47 (d, J = 16 Hz, 1H, ArCH=CH), 5.16 (s, 2H, CH3OCH2O), 3.91 (s, 3H, OCH3), 3.65 (s, 3H, OCH3), 3.34 (s, 3H, CH3OCH2O); 13C NMR (125 MHz, DMSO-d6), δ 173.0 (C=O), 169.3 (C=O), 154.6, 153.9, 152.4, 152.1, 148.9, 143.8, 134.1, 131.6, 131.4, 128.9, 127.3, 122.9, 121.2, 118.6, 118.3, 116.8, 99.7, 61.1, 61.0, 60.4; HRMS (ESI): M/Z [M]+ calcd for C22H22O9: 430.1264; found: 430.1255.
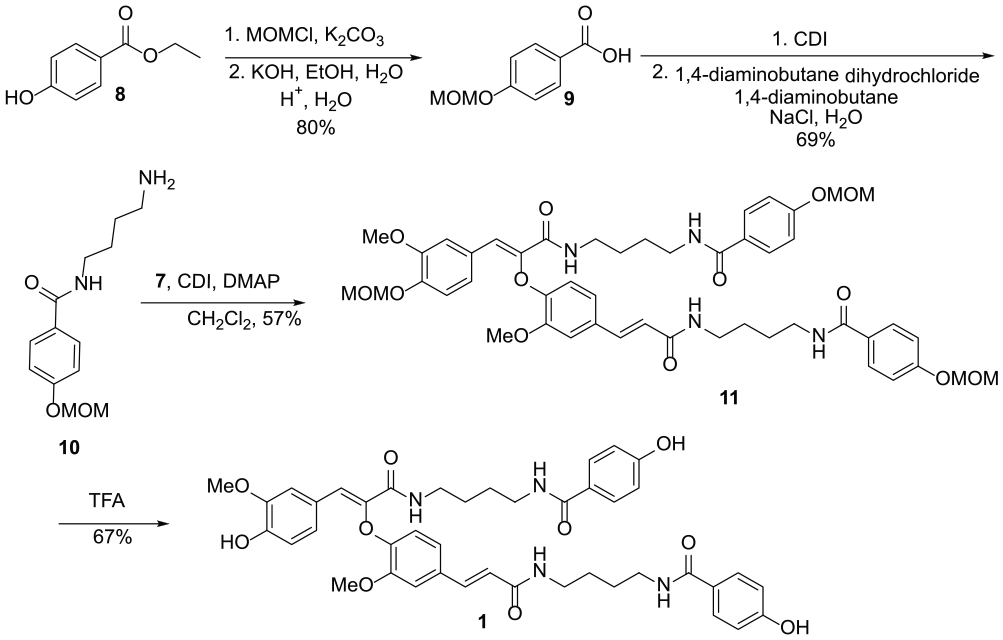
Ethyl 4-(methoxymethoxy)benzoate (9)
To an oven-dried round-bottom flask (100 mL) charged with a solution of 8 (3.32 g, 20 mmol) in acetone (20 mL) was added anhydrous K2CO3 (8.30 g, 60 mmol) at room temperature. The reaction mixture was stirred for 1 h at room temperature and MOM-Cl (3.22 g, 40 mmol) was added dropwise at 0 °C. After stirring for 6 h, the reaction was quenched with water and the aqueous layer was extracted with ethyl acetate (3 × 20 mL). The combined organic layers were washed with water (3 × 10 mL) and brine, and dried (MgSO4), and the solvent was removed in vacuo to give the corresponding protected ester.
To an oven-dried round-bottom flask (50 mL) charged with a solution of the protected ester in EtOH (20 mL) was added a 3.0 M aqueous solution of KOH (10 mL). The mixture was stirred at refluxed for 4 h. After cooling and concentration, 3.0 M aqueous acetic acid (25 mL) was added at 0 °C. The crude white product was precipitated, filtered and washed with water, dried and recrystallized from ethanol to yield compound 9: white powder; m.p. 123–124 °C (Lit.
24
125–126 °C); yield 2.91 g (80%); 1H NMR (500 MHz, CDCl3), δ 8.09 (d, J = 8.7 Hz, 2H, ArH), 7.11 (d, J = 8.7 Hz, 2H, ArH), 5.24 (s, 2H, OCH2OCH3), 3.49 (s, 3H, OCH2OCH3); 13C NMR (125 MHz, CDCl3), δ 170.2 (C=O), 161.3, 132.0, 122.7, 116.0, 94.2, 56.9.
N-(4-Aminobutyl)-4-(methoxymethoxy)benzamide (10)
To an oven-dried round-bottom flask (50 mL) was added 9 (182.18 mg, 1 mmol) and CDI (178.35 mg, 1.1 mmol), and the reaction mixture was mixed with a spatula to start the reaction. The reaction mixture was left at room temperature for 10 min until the solid reaction mixture turned to a pale yellow liquid. To separate round-bottom flask charged with a stirred solution of 1,4-diaminobutane (440.75 mg, 5.0 mmol) in deionized water (20 mL) was added 1,4-diaminobutane dihydrochloride (805.35 mg, 5.0 mmol), and after stirring for 20 min, NaCl (4.0 g, 68.446 mmol) was added. This brine solution was added to the flask containing the acyl imidazole and the mixture was stirred for 1 h at room temperature. The aqueous layer was washed with ethyl acetate to remove the diacylated product. Next 10 mL of a saturated solution of NaOH was added to the aqueous layer which was then extracted with ethyl acetate (3 × 20 mL). The organic layer was washed with water (3 × 10 mL) and brine (3 × 20 mL), and dried (MgSO4), and the solvent was removed in vacuo. The crude product was purified by flash chromatography (dichloromethane/methanol = 3/0.1) to yield 10: yellowish oil; yield 174.08 mg (69%); 1H NMR (500 MHz, CDCl3), δ 7.72 (d, J = 8.5 Hz, 2H, ArH), 6.94 (d, J = 8.5 Hz, 2H, ArH), 5.11 (s, 2H, CH3OCH2O), 3.39 (s, 3H, CH3OCH2O), 3.37–3.29 (m, 2H, CONHCH2), 2.76–2.62 (m, 2H, CH2NH2), 1.65–1.42 (m, 4H, CH2CH2); 13C NMR (125 MHz, CDCl3), δ 167.3 (C=O), 159.6, 128.8, 128.0, 115.7, 94.1, 56.1, 41.0, 39.7, 29.3, 26.9; HRMS (ESI): M/Z [M]+ calcd for C13H20N2O3: 252.1470; found: 252.1469.
N-(4-((E)-3-(3-methoxy-4-(((Z)-1-(3-methoxy-4-(methoxymethoxy)phenyl)-3-((4-(4-(methoxymethoxy)benzamido)butyl)amino)-3-oxoprop-1-en-2-yl)oxy)phenyl)acrylamido)butyl)-4-(methoxymethoxy)benzamide (11)
To an oven-dried round-bottom flask (50 mL) charged with a solution of dicarboxylic acid intermediate 7 (40.0 mg, 0.093 mmol) in dry CH2Cl2 (5 mL) was added CDI (37.67 mg, 0.232 mmol) under a nitrogen atmosphere at room temperature. After stirring for 2 h, a solution of 10 (58.62 mg, 0.232 mmol) and DMAP (1.14 mg, 0.929% mmol) in dry CH2Cl2 (5 mL) was added dropwise to the reaction mixture. The mixture was stirred under a nitrogen atmosphere at room temperature for 6 h. Water (5 mL) was added and the mixture was extracted with CH2Cl2 (3 × 20 mL). The organic layer was washed with water (3 × 10 mL) and brine (3 × 20 mL), and dried (MgSO4), and the solvent was removed in vacuo. The crude product was purified by flash chromatography (dichloromethane/methanol = 3/0.1) to yield 11: colorless oil; yield 47.62 mg (57%); 1H NMR (500 MHz, CDCl3), δ 7.47 (d, J = 15.5 Hz, 1H, ArCH=CH), 7.36 (s, 1H, ArCH=C), 7.16 (d, J = 8.5 Hz, 1H, ArH), 7.07–7.02 (m, 7H, ArH), 6.92 (d, J = 8.5 Hz, 1H, ArH), 6.82 (d, J = 8.5 Hz, 1H, ArH), 6.77–6.40 (m, 4H, ArH), 6.33 (d, J = 15.5 Hz, 1H, ArCH=CH), 5.21 (s, 2H, CH3OCH2O), 5.20 (s, 2H, CH3OCH2O), 5.19 (s, 2H, CH3OCH2O), 3.95 (s, 3H, OCH3), 3.72 (s, 3H, OCH3), 3.48–3.44 (m, 9H, 3 × CH3OCH2O) 3.48–3.44 (m, 2H, CONHCH2), 3.42–3.38 (m, 2H, CONHCH2), 3.34–3.31 (m, 4H, 2 × CONHCH2), 1.70–1.60 (m, 4H, CH2CH2), 1.52–1.39 (m, 4H, CH2CH2); 13C NMR (125 MHz, CDCl3), δ 167.3, 167.1, 166.1, 163.3, 159.8, 149.5, 149.0, 147.6, 145.7, 141.7, 139.6, 131.1, 128.7, 127.9, 126.7, 124.2, 124.1, 121.3, 120.8, 115.9, 115.7, 115.6, 113.1, 111.2, 95.2, 94.2, 56.3, 56.2, 56.0, 55.6, 39.6, 39.3, 39.1, 27.2, 27.1, 26.8, 26.5; HRMS (ESI) calcd for C48H58N4O13: 898.4000; found: 898.4007.
Mariamide A (1)
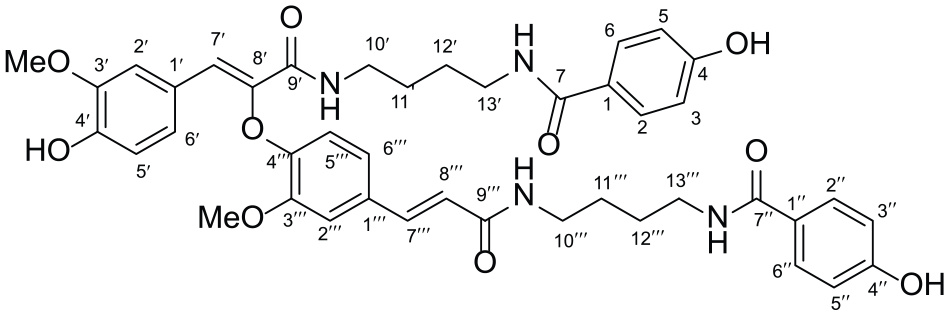
To an oven-dried round-bottom flask (50 mL) charged with a solution of compound 11 (40.0 mg, 0.045 mmol) in dry CH2Cl2 (2 mL) was added TFA (0.11 mL, 1.35 mmol) at room temperature. After stirring for 8 h, the solvent was removed in vacuo. The crude product was purified by flash chromatography (dichloromethane/methanol = 3/0.1) to yield 1: white amorphous powder; yield 22.86 mg (67%) 1H NMR (500 MHz, CD3OD), δ 7.68 (d, J = 8 Hz, 2H, ArH), 7.66 (d, J = 8 Hz, 2H, ArH), 7.41 (d, J = 15.5 Hz, 1H, ArCH=CH), 7.30 (s, 1H, ArH), 7.23 (s, 1H, ArCH=C), 7.23 (s, 1H, ArH), 7.05 (d, J = 8.5 Hz, 1H, ArH), 6.96 (d, J = 8.5 Hz, 1H, ArH), 6.81–6.79 (m, 4H, ArH), 6.75 (d, J = 8.5 Hz, 1H, ArH), 6.70 (d, J = 8.5 Hz, 1H, ArH), 6.44 (d, J = 15.5 Hz, 1H, ArCH=CH), 3.94 (s, 3H, OCH3), 3.68 (s, 3H, OCH3), 3.40–3.36 (m, 2H, CONHCH2), 3.34–3.30 (m, 2H, CONHCH2), 3.27–3.24 (m, 4H, 2 × CONHCH2), 1.63–1.57 (m, 4H, 2 × CH2CH2), 1.49–1.40 (m, 4H, 2 × CH2CH2); 13C NMR (125 MHz, CD3OD), δ 170.1 (C=O), 170.0 (C=O), 168.7 (C=O), 165.7 (C=O), 162.1, 161.9, 150.6, 149.6, 148.9, 147.8, 141.9, 141.2, 132.0, 130.3, 130.2, 126.6, 126.5, 126.3, 125.5, 125.1, 122.2, 121.1, 116.21, 116.19, 116.1, 115.3, 113.7, 112.3, 56.5, 56.0, 40.5, 40.4, 40.32, 40.29, 28.0, 27.9, 27.83, 27.80; HRMS (ESI): M/Z [M]+ calcd for C42H46N4O10: 766.3214; found: 766.3219.