Abstract
A new lignan, arborlignan A, along with five known substances, neoechinulin A, 4-hydroxy-3,5-dimethoxybenzaldehyde, 3,3′-dimethoxy-4,4′-dihydroxystilbene, β-hydroxypropiovanillone, and coniferyl aldehyde, are isolated from Schefflera arboricola. Their structures are elucidated by mass spectrometry and nuclear magnetic resonance spectroscopy experiments and by comparison with literature data. These compounds are isolated from the genus Schefflera for the first time.
Keywords

Introduction
Schefflera arboricola (Araliaceae), a spineless tree or shrub, is widely distributed in tropical and subtropical areas, mainly in the southwest and southeast regions of China.
1
Several species of this genus are widely used in traditional medicine, such as S. delavayi and S. leucantha.2–4 The main phytochemical constituents reported from S. arboricola were triterpenoids, steroids, sesquiterpenoids, organic acids, benzyl glycosides, lignans, quinones, and diterpenes.5–7 Previous bioactivity studies have confirmed their medicinal properties as analgesic, anti-inflammatory, antitumor, antibacterial, antiviral, and anti-allergic.8–10 As part of our continuing efforts to search for bioactive compounds derived from Schefflera, a new lignan, arborlignan A (
Results and discussion
Arborlignan A (
The 1H nuclear magnetic resonance (NMR) spectrum of compound
1H NMR (500 MHz) and 13C NMR (125 MHz) data for

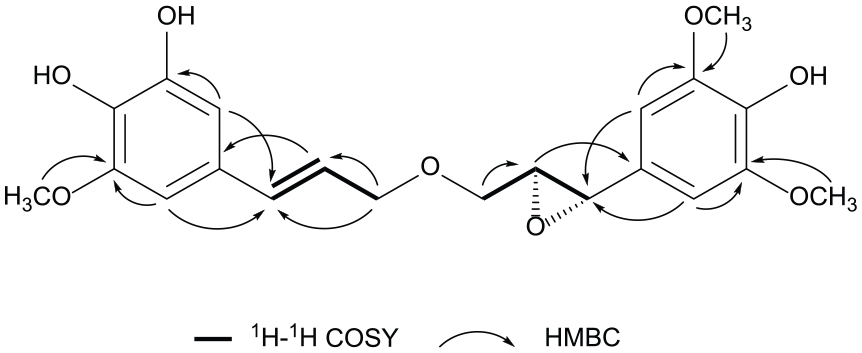
Key 1H-1H COSY and HMBC correlations for
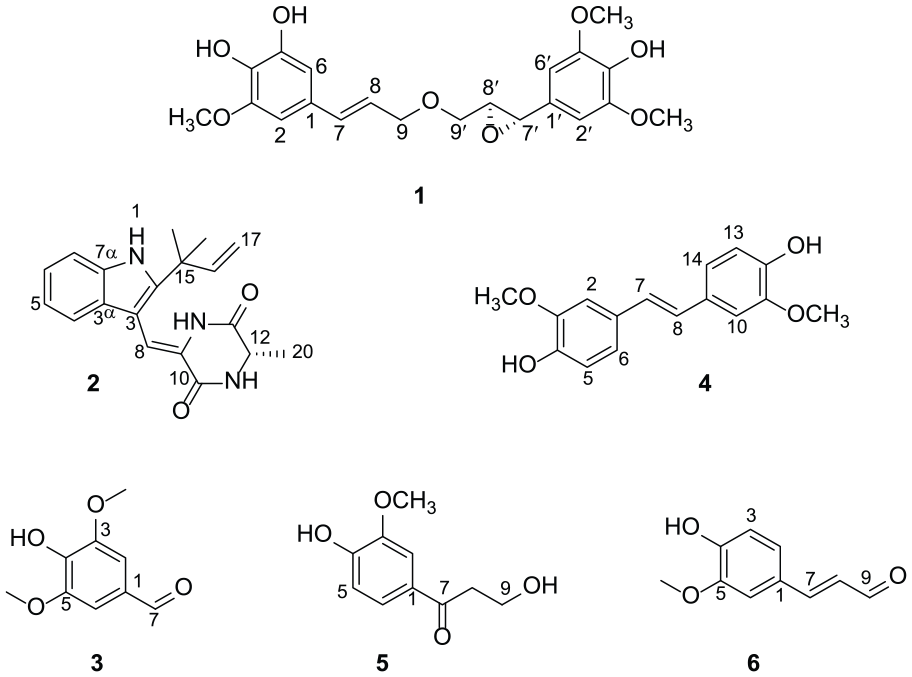
The structures of compounds
Indeed, the right epoxide part of

Key NOESY correlations for
The obtained known compounds (Figure 1) are characterized as neoechinulin A (
Conclusion
To the best of our knowledge, compounds
Experimental section
General experimental procedures
UV spectra were obtained in MeOH using a Shimadzu PharmaSpectra UV-1800 instrument. IR spectra were measured on a Thermo Scientific Nicolet iS10 FTIR spectrometer. NMR experiments were conducted on a Bruker Avance III 500 spectrometer. Electron-impact mass spectrometry (EI-MS) was conducted on an Agilent 7890B-5977A. Electrospray ionization mass spectrometry (ESI-MS) was conducted on an Agilent 1200/6320 ion trap XCT LC-MS spectrometer. HR-ESI-MS were performed with an Agilent 6320 LC-Q TOF High Mass. Optical rotations were measured on a Rudolph AUTOPOL® IV polarimeter. Silica gel (200−300 mesh, Qingdao Maine Chemical Factory), and Sephadex LH-20 (Amersham Biosciences) were used for column chromatography. Samples were made visual by spraying with 10% H2SO4/EtOH (v/v) followed by heating. Semipreparative high-performance liquid chromatography (HPLC) was conducted on an Agilent 1100 system using a YMC-Pack ODS C-18 (250 × 10 mm, 5 μm) column with a flow rate of 2 mL/min.
Plant material
The twigs and leaves of S. arboricola were purchased from Quanzhou Zhongqiao Co., Ltd pharmaceutical company in July 2016 in Fujian Province, China.
Extraction and isolation
The dried twigs and leaves of S. arboricola (20 kg) were powdered and extracted with 95% EtOH at 80 °C (three times, 3 h each time). The filtrate was concentrated under reduced pressure to afford a crude extract. This was extracted with petroleum ether, chloroform, ethyl acetate, and n-butanol. The chloroform-soluble fraction (68.9 g) was subjected to silica gel column chromatography, eluting with a gradient of CH2Cl2/EtOAc (from 50:1 to 1:1), to give nine fractions, Fr. 1–9. Fr. 3 was further separated on a silica gel column, eluting with a gradient of PE/EtOAc (from 50:1 to 2:1), to afford Fr. 3.1–3.5. Fr. 3.4 was subjected to silica gel column chromatography, eluting with a gradient of PE/acetone (from 7:1 to 1:1). Finally then compound
Neoechinulin A (
4-Hydroxy-3,5-dimethoxybenzaldehyde (
3,3′-Dimethoxy-4,4′-dihydroxystilbene (
β-Hydroxypropiovanillone (
Coniferyl aldehyde (
Supplemental Material
supporting_information – Supplemental material for A new lignan from Schefflera arboricola
Supplemental material, supporting_information for A new lignan from Schefflera arboricola by Chang-Qing Ye, Jia-Yi Zhang, Zhi-Cheng Ye, Mei-Tian Xiao, Xu-Dong Zhou and Jing Ye in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank the Instrumental Analysis Center of Huaqiao University for analytical support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by grants from the National Natural Science Foundation of China (nos 81903514 and 81703390), the Huaqiao University Support Program for Science and Technology Innovation Young Teachers (no. ZQN-PY515), and the Project for Postgraduates’ Innovative Fund in Scientific Research of Huaqiao University.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
