Abstract
Phthalazine and phthalazinone derivatives are important owing to their significant biological activities and pharmacological properties. Herein, a benzoic acid derivative (

Introduction
Phthalazine, also known as benzo-orthodiazine, 2,3-diazonaphthalene, and benzo[d]pyridazine, is a compound in which pyridazine is fused with a benzene ring.
Although, N-containing heterocyclic compounds are widely distributed in nature, the presence of two adjacent nitrogen atoms in the phthalazine ring makes it rare and not very familiar in isolated extracts from living organisms, owing to the fact that it can be constructed from hydrazine which is an uncommon amine source in nature.
Phthalazines, as an important class of bicyclic N-heterocycles, have attracted sizable attention due to their valuable biological and pharmacological activities.1,2
Phthalazines are popular pharmacophores as they are the core chemical motifs in many commercially available drugs such as Azelastin (antihistamine), 3 Vatalanib (vascular endothelial growth factor receptor (VEGFR) inhibitor), 4 and Hydralazine (antihypertensive agent). 5 Phthalazine derivatives are also considered as p38MAP kinase inhibitors, 6 selective binders of gamma-aminobutyric acid (GABA) receptors, 7 cyclooxygenase-2 (COX-2) inhibitors, 8 and high-affinity ligands of voltages gated calcium channels. 9
Like many other isomeric benzodiazines, phthalazine derivatives reveal numerous pharmacological and biological activities such as antimicrobial,10–12 antidiabetic, 13 analgesic,14,15 anticonvulsant,16,17 antitumor,18–20 antiproliferative, 20 antiepileptic,21,22 anti-inflammatory,23,24 and vasorelaxant.25,26
In addition, phthalazines are known as serotonin reuptake inhibitors and are considered as anti-depression agents. 27 Also, they are crucial precursors in the synthesis of many compounds with interesting pharmacological properties like phosphodiesterase inhibitors and blood platelet aggregation inhibitors.27–29
Encouraged by the above-mentioned facts, and in our endeavor to develop novel synthetic approaches to construct new valuable heterocycles for biological screening,30,31 we report herein the synthesis of novel and diverse phthalazine, 1,3,4-oxadiazole, and pyrazole derivatives from readily available and inexpensive scaffolds utilizing short and straightforward synthetic routes.
Results and discussion
Owing to the aforementioned therapeutic properties of phthalazines, aroylation of cumene by phthalic anhydride exploiting Friedel Craft’s reaction conditions was achieved and 2-(4-isopropylbenzoyl)benzoic acid (

Synthesis of o-benzoic acid derivative
Having o-aroylbenzoic acid derivative
Initially, cyclization of 2-(4-isopropylbenzoyl)benzoic acid (

Compounds
According to a reported procedure,
32
phthalazin-1-one derivatives were readily accessible through the condensation reaction of o-benzoyl benzoic acid with hydrazine hydrate in boiling ethanol. Consequently, adopting this procedure and allowing 2-(4-isopropylbenzoyl)benzoic acid (
It is noteworthy that 1-oxophthalazine derivatives
In the same context, investigation of the behavior of benzoxazinone
Another convenient approach for the preparation of phthalazinone derivatives was achieved via the reaction of benzoxazinone derivative
Moreover, 1-oxophthalazine derivatives
Interestingly, changing the solvent and performing the reaction of benzoxazinone derivative
In addition, dihydroisoquinoline derivative
In our attempts to build up additional fused heterocycles, phthalazin-1-one derivative
It is worthwhile to mention that no O-alkylation product was isolated and the reaction most probably took place via SN2 reaction mechanism and this may be attributed to the fact that the nitrogen atom is more nucleophilic than oxygen and therefore the N-alkylation product was the sole product. The structure of compound
In addition, the chemical structure of the latter compound
Refluxing phthalazinone derivative
Driven by the high functionality of the side chain of oxophthalazine acetohydrazide derivative

Reactions of acetic acid hydrazide derivative
In addition, a cyclization reaction took place when acetic acid hydrazide derivative
A new approach was adopted to synthesize more oxadiazole rings attached to the phthalazine moiety in which hydrazide derivative
Likewise, an oxadiazole ring could be attached to our phthalazinone heterocycle by the reaction of hydrazide
Moreover, 2-{2-[4-(4-isopropylphenyl)-1-oxophthalazin-2(1H)-yl]acetyl}-N-phenylhydrazine-1-carbothioamide (
Interestingly, a pyrazole ring could be attached to the phthalazine moiety through the reaction of the hydrazide derivative
A new pyrazole ring attached to the phthalazine ring was obtained through the reaction of hydrazide derivative
Furthermore, acetic acid hydrazide derivative
Antimicrobial assay
Phthalazine and phthalazine hybrid structures, in which a phthalazine moiety is attached to another heterocycle, are considered to be of immense importance owing to their wide spectrum of biological activities.1,2,10 Accordingly, the in vitro antimicrobial activity of the newly synthesized heterocycles was appraised against two Gram-positive bacteria, namely, Bacillus subtilis and Staphylococcus aureus, in addition to two Gram-negative bacteria, namely, Pseudomonas aeruginosa and Escherichia coli.
In the current in vitro antimicrobial assay, Amoxicillin was employed as a standard and the antibacterial activities of the compounds under investigation were recorded as inhibition diameter zones (IZ) in millimeters (mm) (Table 1).
In vitro antimicrobial activity of the synthesized compounds.
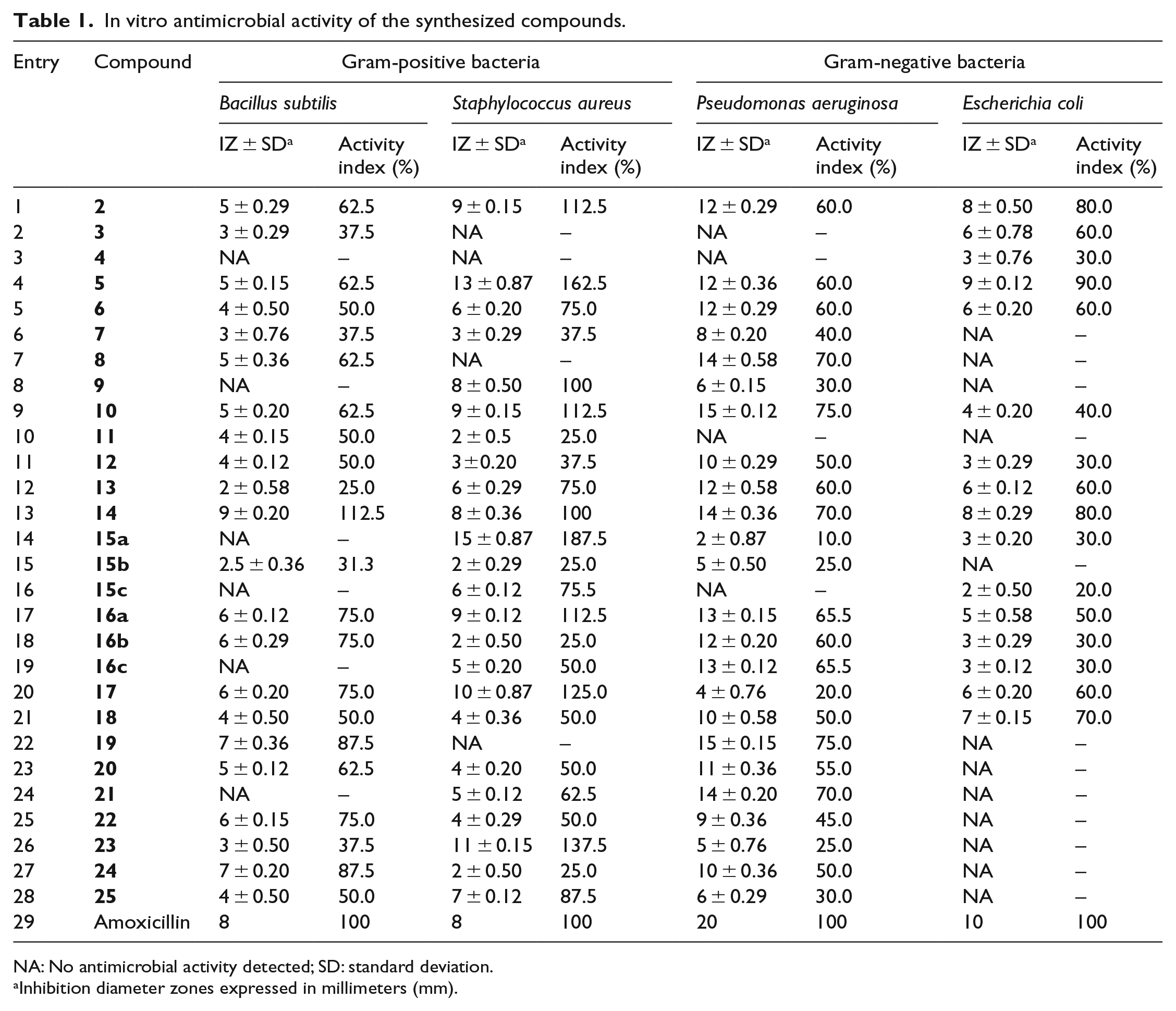
NA: No antimicrobial activity detected; SD: standard deviation.
Inhibition diameter zones expressed in millimeters (mm).
As shown in Table 1, the compounds under investigation revealed a wide range of variation in their biological activities.
Compound
Conclusion
In summary, o-aroylbenzoic acid derivative
Experiment
General
Melting points were determined with an electrothermal melting point apparatus and are uncorrected. The reaction times were determined using thin-layer chromatography (TLC) which was performed with fluorescent silica gel plates HF245 (Merck) and the plates were viewed with iodine. Merck silica gel (230–400 mesh) was used for flash chromatography separations. The Microanalytical Center of Cairo University performed the microanalyses. IR (KBr) spectra were recorded on a Pye-Unicam IR spectrophotometer SP 2000 (Faculty of Science, Fayoum University). The mass spectra were run using a Shimadzu-GC-MS-GP 1000 EX instrument by employing the direct inlet system. Nuclear magnetic resonance (NMR) spectra were recorded on a Varian Mercury 300-MHz spectrometer using tetramethylsilane (TMS) as the internal standard at the National Research Center. Chemical shifts (δ) and coupling constants (J) are recorded in parts per million (ppm) and Hertz (Hz), respectively.
2-(4-Isopropylbenzoyl)benzoic acid (2 )
In a 1000 mL three-necked flask equipped with a condenser, a mixture of phthalic anhydride (14.8 g, 0.1 mol) and cumene (15 mL) was treated with then anhydrous aluminum chloride (20.0 g, 0.15 mol) gradually during stirring over a period of half an hour.
32
The reaction mixture was refluxed in a water bath for 2 h and then the mixture was allowed to stand at room temperature overnight. Ice-HCl was added to the mixture and the excess cumene was removed by steam distillation. The collected solid was filtered off, washed well with water, and dried. Recrystallization from ethanol afforded
4-(4-Isopropylphenyl)-1
H
-benzo[d][1,2]oxazin-1-one (3 )
A mixture of compound
4-(4-Isopropyl phenyl)phthalazin-1(2
H
)-one (4 )
Method A: To a solution of
Method B: A mixture of benzoxazine
4-(4-Isopropylphenyl)-1-oxophthalazine-2(1
H
)-carbothioamide (5 )
A mixture of
4-(4-Isopropylphenyl)-1-oxophthalazine-2(1
H
)-carbothiohydrazide (6 )
A mixture of
2-Benzyl-4-(4-isopropylphenyl)phthalazin-1(2H)-one (7 )
A mixture of benzooxazine
1-[4-(4-Isopropylphenyl)-1-oxophthalazin-2(1
H
)-yl]thiourea (8 )
A mixture of benzooxazine
N-[4-(4-isopropylphenyl)-1-oxophthalazin-2(1
H
)-yl]hydrazinecarbothioamide (9 )
A mixture of benzooxazine
7-(4-Isopropylphenyl)-2H-[1,2,4,5]tetrazino[6,1-a]phthalazine-3(4H)-thione (10 )
A mixture of benzooxazine
1,1′-[1-(4-Isopropylphenyl)-4-oxo-3,4-dihydroisoquinoline-3,3-diyl]bis(ethan-1-one) (11 )
To a solution of benzooxazine
Ethyl 2-[4-(4-isopropylphenyl)-1-oxophthalazin-2(1H)-yl]acetate (12 )
A mixture of
2-[4-(4-Isopropylphenyl)-1-oxophthalazin-2(1H)-yl]acetohydrazide (13 )
A mixture of
2-Benzoyl-4-(4-isopropylphenyl)phthalazin-1(2H)-one (14 )
A mixture of phthalazine
Reactions of phthalazine hydrazide derivative 13 with aromatic aldehydes 15a–c
General procedure
A mixture of acetic acid hydrazide
N′-Benzylidene-2-[4-(4-isopropylphenyl)-1-oxophthalazin-2(1H)-yl]acetohydrazide (15a )
Colorless crystals were isolated in 76% yield; m.p. 228–229 °C, IR (KBr): 1579 cm−1 (C=N), 1659 cm−1 (2CO), 2958 cm−1 (CH aliphatic), 3217 cm−1 (NH). 1H NMR (DMSO-d6, 300 MHz, ppm) δ 1.27 (d, J = 6.4 Hz, 6H, 2CH3), 2.98–3.02 (m, 1H, CH), 4.95 (s, 1H, NH), 5.35 (s, 2H, CH2), 7.44 (d, J = 7.5 Hz, 2H, ArH), 7.51 (d, J = 7.5 Hz, 2H, ArH), 7.70–8.40 (m, 9H, ArH), 8.72 (s, 1H, CH); 13C NMR (DMSO-d6, 75 MHz, ppm) δ 23.2, 34.1, 54.1, 126.3, 126.6, 127.5, 127.7, 128.9, 129.1, 129.3, 130.9, 132.6, 134.5, 134.8, 145.7, 146.8, 150.5, 152.9, 155.4, 158.6, 163.3. Anal. calcd for C26H24N4O2: C, 73.56; H, 5.70; N, 13.20; found: C, 73.50; H, 5.73; N, 13.24%. MS (70 eV) m/z (%): 424 (100) (M+), 305 (94), 76 (2).
N′-(4-Chlorobenzylidene)-2-(4-(4-isopropylphenyl)-1-oxopht-halazin-2(1H)-yl)acetohydrazide (15b )
Colorless crystals were isolated in 82% yield; m.p. 270–271 °C, IR (KBr): 1580 cm−1 (C=N), 1676 cm−1 (2CO), 2958 cm−1 (CH aliphatic), 3068 cm−1 (CH aromatic), 3205 cm−1 (NH). 1H NMR (DMSO-d6, 300 MHz, ppm) δ 1.28 (d, J = 6.4 Hz, 6H, 2CH3), 3.03–3.21 (m, 1H, CH), 5.41 (s, 2H, CH2), 7.22–7.35 (m, 4H, ArH), 7.42–8.05 (m, 8H, ArH), 8.21 (s,1H, NH), 8.79 (s, 1H, CH); 13C NMR (DMSO-d6, 75 MHz, ppm) δ 23.37, 34.20, 54.2, 126.3, 126.5, 127.2, 127.6, 127.9, 128.9, 129.2, 129.7, 130.2, 132.4, 134.3, 134.7, 135.1, 144.5, 146.8, 150.5, 159.1, 164.1. Anal. calcd for C26H23ClN4O2: C, 68.04; H, 5.05; N, 12.21; found: C, 68.08; H, 5.01; N, 12.23%. MS (70 eV) m/z (%): 458 (82) (M+), 305 (100).
2-[4-(4-Isopropylphenyl)-1-oxophthalazin-2(1H)-yl]-N′-(4-nitrobenzylidene)aceto-hydrazide (15c )
Beige crystals were isolated in 72% yield; m.p. 298–299 °C, IR (KBr): 1584 cm−1 (C=N), 1694 cm−1 (2CO), 2959 cm−1 (CH aliphatic), 3068 cm−1 (CH aromatic), 3446 cm−1 (NH). 1H NMR (DMSO-d6, 300 MHz, ppm) δ 1.28 (d, J = 6.4 Hz, 6H, 2CH3), 2.99–3.05 (m, 1H, CH), 5.70 (s, 2H, CH2), 7.30–7.48 (m, 5H, ArH), 7.60 (s, 1H, NH), 7.65–8.12 (m, 7H, ArH), 8.61 (s,1H, CH); 13C NMR (DMSO-d6, 75 MHz, ppm) δ 23.2, 34.2, 54.2, 124.6, 126.3, 126.6, 127.1, 127.6, 127.9, 128.2, 129.1, 131.2, 132.7, 134.5, 138.9, 145.6, 147.9, 148.7, 150.5, 159.3, 164.2. Anal. calcd for C26H23N5O4: C, 66.51; H, 4.94; N, 14.9; found: C, 66.00; H, 5.10; N, 14.6%. MS (70 eV) m/z (%): 469 (2) (M+), 305 (100).
Cyclization of Schiff’s bases 15a–c : general procedure
Compounds
2-[(4-Acetyl-5-phenyl-4,5-dihydro-1,3,4-oxadiazol-2-yl)methyl]-4-(4-isopropylphenyl)phthalazin-1(2H)-one (16a)
Colorless crystals were isolated in 73% yield; m.p. 130–131 °C, IR (KBr): 1581 cm−1 (C=N), 1662 cm−1 (2 CO), 2958 cm−1 (CH aliphatic). 1H NMR (DMSO-d6, 300 MHz, ppm) δ 1.22 (d, J = 6.4 Hz, 6H, 2CH3), 2.04 (s, 3H, CH3), 2.98–3.02 (m, 1H, CH), 5.01 (s, 2H, CH2), 7.03 (s, 1H, CH oxadiazole), 7.21–7.51 (m, 5H, ArH), 7.52–8.38 (m, 8H, ArH); 13C NMR (DMSO-d6, 75 MHz, ppm) δ 23.1, 23.5, 34.2, 46.4, 98.9, 126.3, 126.6, 126.9, 127.1, 127.6, 127.8, 128.9, 129.3, 130.1, 130.9, 132.7, 134.3, 134.6, 145.7, 150.5, 153.7, 157.7, 159.3. Anal. calcd for C28H26N4O3: C, 72.09; H, 5.62; N, 12.01; found: C, 72.05; H, 5.60; N, 12.05%. MS (70 eV) m/z (%): 466 (100) (M+), 424 (91), 277 (3).
2-[(4-Acetyl-5-(4-chlorophenyl)-4,5-dihydro-1,3,4-oxadiazol-2-yl)methyl]-4-(4-isopropylphenyl)phthalazin-1(2H)-one (16b )
Colorless crystals were isolated in 79% yield; m.p. 140–141 °C, IR (KBr): 1583 cm−1 (C=N), 1661 cm−1 (2 CO), 2959 cm−1 (CH aliphatic). 1H NMR (DMSO-d6, 300 MHz, ppm) δ 1.25 (d, J = 6.4 Hz, 6H, 2CH3), 2.07 (s, 3H, CH3), 2.98–3.05 (m, 1H, CH), 5.28 (s, 2H, CH2), 7.11 (s, 1H, CH oxadiazole), 7.27 (d, J = 7.5 Hz, 2H, ArH), 7.32 (d, J = 7.5 Hz, 2H, ArH), 7.49–7.73 (m, 4H, ArH), 7.98–8.38 (m, 4H, ArH); 13C NMR (DMSO-d6, 75 MHz, ppm) δ 23.2, 23.4, 34.2, 46.3, 99.3, 126.3, 126.5, 127.3, 127.5, 127.8, 128.8, 129.2, 129.6, 130.1, 132.7, 134.0, 134.5, 136.4, 146.2, 150.5, 154.1, 158.2, 159.6. Anal. calcd for C28H25 ClN4O3: C, 67.13; H, 5.03; N, 11.18; Cl, 7.08; found: C, 67.15; H, 5.00; N, 11.22; Cl, 6.88%. MS (70 eV) m/z (%): 500 (3) (M+), 73 (100).
2-[(4-Acetyl-5-(4-nitrophenyl)-4,5-dihydro-1,3,4-oxadiazol-2-yl)methyl]-4-(4-isopropylphenyl)phthalazin-1(2H)-one (16c )
Beige crystals were isolated in 72% yield; m.p. 216–217 °C, IR (KBr): 1581 cm−1 (C=N), 1662 cm−1 (2 CO), 2962 cm−1 (CH aliphatic). 1H NMR (DMSO-d6, 300 MHz, ppm) δ 1.26 (d, J = 6.4 Hz, 6H, 2CH3), 2.05 (s, 3H, CH3), 2.98–3.05 (m, 1H, CH), 5.34 (s, 2H, CH2), 7.08 (s, 1H, CH oxadiazole), 7.22 (d, J = 7.5 Hz, 2H, ArH), 7.30 (d, J = 7.5 Hz, 2H, ArH), 7.41–7.68 (m, 4H, ArH), 7.88–8.27 (m, 4H, ArH); 13C NMR (DMSO-d6, 75 MHz, ppm) δ 23.3, 23.4, 34.2, 46.4, 99.2, 124.6, 126.4, 126.7, 126.9, 127.2, 127.5, 127.7, 129.3, 130.8, 132.6, 134.5, 142.1, 145.8, 148.1, 150.5, 154.0, 157.9, 159.7. Anal. calcd for C28H25 N5O5: C, 65.74; H, 4.93; N, 13.69; found: C, 65.71; H, 4.97; N, 13.65%. MS (70 eV) m/z (%): 511 (52) (M+), 469 (100), 277 (1).
4-(4-Isopropylphenyl)-2-[(5-thioxo-4,5-dihydro-1,3,4-oxadiazol-2-yl)methyl]phthalazin-1(2H)-one (17 )
A mixture of the acetic acid hydrazide
2-[(1,3,4-Oxadiazol-2-yl)methyl]-4-(4-isopropylphenyl)phthalazin-1(2H)-one (18 )
A mixture of acetic acid hydrazide
4-(4-Isopropylphenyl)-2-[(5-methyl-1,3,4-oxadiazol-2-yl)methyl]phthalazin-1(2H)-one (19 )
A mixture of acetic acid hydrazide
2-{2-[4-(4-Isopropylphenyl)-1-oxophthalazin-2(1H)-yl]acetyl}-N-phenylhydrazine-1-carbothioamide (20 )
A mixture of acetic acid hydrazide
2-[2-(3,5-Dimethyl-1H-pyrazol-1-yl)-2-oxoethyl]-4-(4-isopropylphenyl)phthalazin-1(2H)-one (21 )
A mixture of acetic acid hydrazide
2-[2-(3,5-Diamino-1H-pyrazol-1-yl)-2-oxoethyl]-4-(4-isopropylphenyl)phthalazin-1(2H)-one (22 )
A mixture of acetic acid hydrazide
1-{2-[4-(4-Isopropylphenyl)-1-oxophthalazin-2(1H)-yl]acetyl}pyrazolidine-3,5-dione (23 )
A mixture of acetic acid hydrazide
2-[2-(5-Amino-2,3-dihydro-1H-pyrazol-1-yl)-2-oxoethyl]-4-(4-isopropylphenyl)phthalazin-1(2H)-one (24 )
A mixture of acetic acid hydrazide
N-(1,3-Dioxoisoindolin-2-yl)-2-[4-(4-isopropylphenyl)-1-oxophthalazin-2(1H)-yl]acetamide (25 )
A mixture of acetic acid hydrazide
Antimicrobial activity assay
The in vitro antimicrobial activity of the newly synthesized compounds was measured against specific microbial strains using the disk diffusion technique in which sterile Whatman No. 5 filter paper (11 mm diameter) disks were utilized. 38 Ethanol was selected to dissolve the tested compounds then the 11-mm disks were loaded with 10 mg mL−1 of the test compound under investigation (50 µL). Subsequently, the disks were left with caution under hot air streams to reach complete dryness.
Test plates were made ready via pouring freshly prepared Muller–Hinton agar medium (10 mL) seeded with selected microbes. The disks were placed on the surface of agar plates then incubation for 1 h at 5 °C took place to secure good diffusion. Thereafter, all the plates were incubated at 37 °C for 24 h. Finally, the outgrowth of the microorganisms was recorded and the average IZ were measured in millimeters and used as criteria for the antimicrobial activity. Importantly, the average growth of microbes was calculated after the plates were done in triplicate. The observed clear zone size is directly proportional to the inhibitory effect of the investigated compound. In each experiment, a negative control was incorporated by using a pure solvent disk. For comparison, a disk with Amoxicillin (standard drug) was screened for antimicrobial activity using the same conditions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to extend their profound appreciation to Fayoum University for the financial support.
