Abstract
A convenient method for the preparation of Coenzyme Q1 from cheap and readily available 3,4,5-trimethoxytoluene is developed. Coenzyme Q1 is synthesized in a moderate yield by a two-step procedure involving the key reaction of an allyl bromide with Coenzyme Q0 through a redox chain reaction. The reaction is efficient and can be used for the synthesis of other Coenzyme Q compounds.

Introduction
Coenzyme Q10 (CoQ10; Figure 1) is an isoprenoid quinone compound 1 and plays a pivotal role in the electron transport chain in respiratory processes. 2 CoQ10 is a natural antioxidant that scavenges free radicals. 3 It is widely used in the treatment of cardiovascular disease and mitochondrial disorders. 4 Coenzyme Q1 (CoQ1; Figure 1) is an important fragment of the Coenzyme Q (CoQ) series which are active in the electron transport and oxidative phosphorylation processes in mitochondria. CoQ1 also acts as a key intermediate in the synthesis of higher CoQ analogues. 5
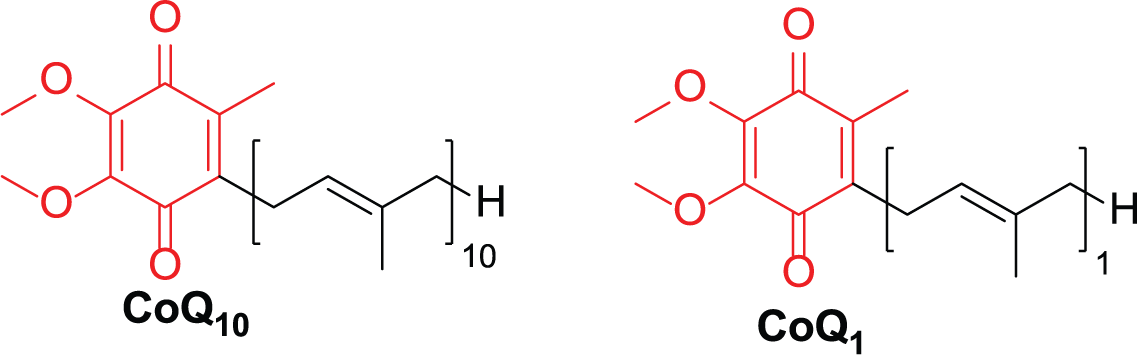
Structures of CoQ10 and CoQ1.
There have been several methods published for the preparation of CoQ1, most of which involve a Lewis acid-catalyzed reaction between an allylic alcohol and hydroquinone, followed by oxidation to the quinones. Hegedus and Waterman 2 and Sato et al. 6 synthesized CoQ1 by reaction of π-allylic nickel complexes with quinones in 26% yield (Scheme 1, eq 1). Yamago et al. 7 reported a radical-mediated synthesis of substituted quinones with organotellurium compounds. 8 However, these reactions were quite sensitive to the reaction conditions, the key reagents π-allylnickel bromide complex and the requisite organotellurium reagent were difficult to prepare. Tabushi et al. 9 reported a β-cyclodextrin-catalyzed allylation–oxidation of hydroquinone to form CoQ1 in 11% yield (Scheme 1, eq 2). Recently, Chen 5 and Borioni et al. 10 started from 3,4,5-tetramethoxytoluene (TMT) to obtain CoQ1 in multiple steps (Scheme 1, eq 3). Unfortunately, all these methods generally gave low yields and complex by-products. Therefore, a general and practical method for efficient CoQ1 synthesis is highly desirable. Here, we report a two-step synthesis of CoQ1 by starting from 3,4,5-trimethoxytoluene with a total yield of 60% (Scheme 1, eq 4).

Various methods for the syntheses of CoQ n : (a) previous work and (b) this work.
Results and discussion
First, a single-step synthesis of CoQ0 is shown in Table 1; this oxidation reaction of TMT is conducted in acetic acid at 50°C in less than 2 h and without using any metal catalyst. This environmentally friendly procedure is based on the oxidant as an oxygen atom donor, and the acidic solvent acetic acid plays an important role in the transformation. The traditional method employing 30% H2O2 as an oxidant gave a yield of 50% (Table 1, entry 1). The use of Na2S2O8 and (NH4)2S2O8 improved the reaction yield (Table 1, entries 3 and 4). The best yield was obtained using K2S2O8 as an oxidant to afford the desired product CoQ0 in 85% yield (Table 1, entry 2). However, when cerium ammonium nitrate (CAN) was used as the oxidant, we did not observe any product CoQ0 (Table 1, entry 5).
Single-step synthesis of CoQ0.
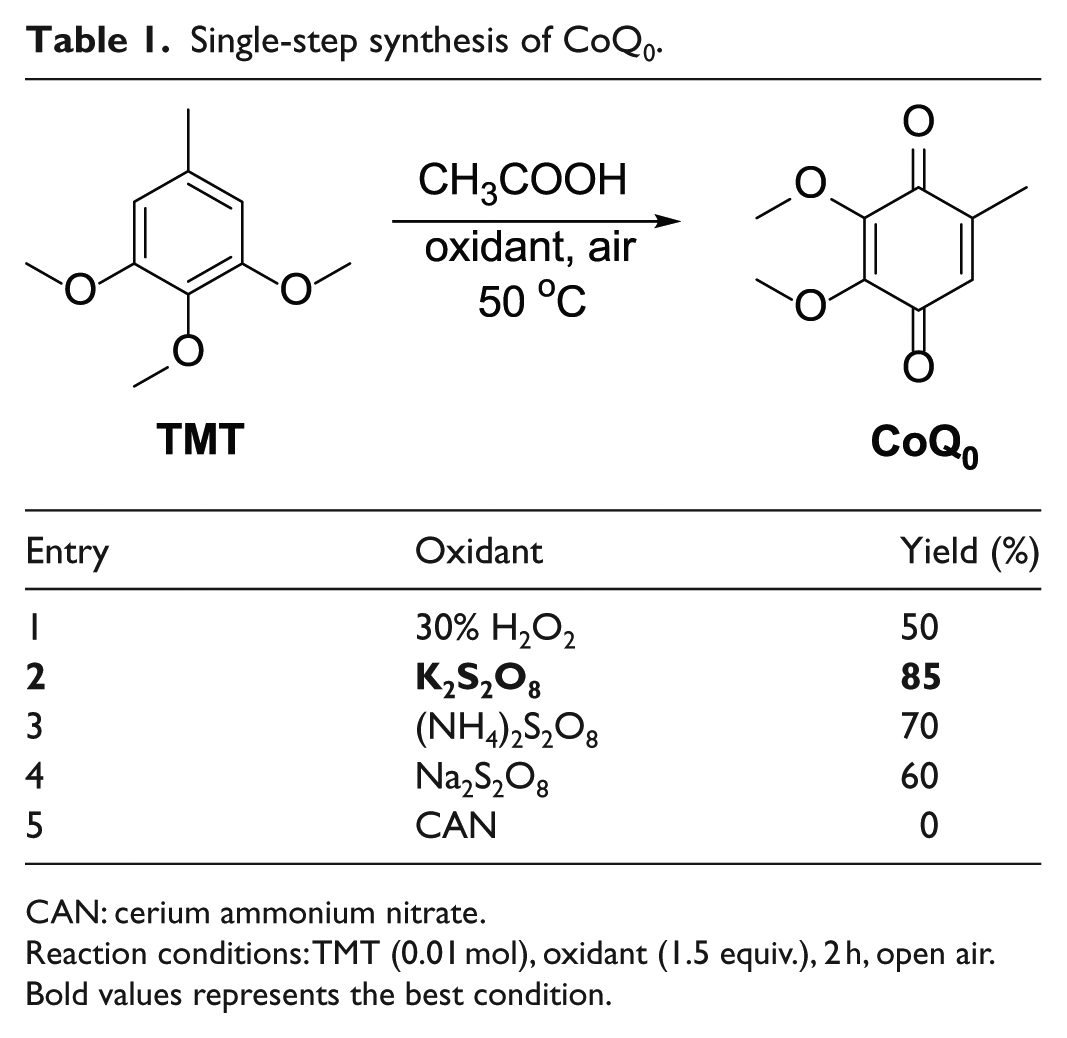
CAN: cerium ammonium nitrate.
Reaction conditions: TMT (0.01 mol), oxidant (1.5 equiv.), 2 h, open air.
Bold values represents the best condition.
Inspired by Xu and Li’s 11 work on the alkylation of p-quinones by a redox chain reaction, we tried to synthesize CoQ1 by the allylation of CoQ0 with 1-bromo-3-methyl-2-butene 1; the results are shown in Table 2. Diethyl 1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate (Hantzsch ester) was selected as an initiator which can accelerate the alkylation according to the literature, 12 and the reaction works with many Lewis acids as catalysts in dichloromethane as the solvent at room temperature (r.t.). Typical Lewis acid catalysts were screened in the reaction; however, AlCl3, ZnCl2, and FeCl3 did not catalyze the reaction (Table 2, entries 1–3). The solvents used for this reaction was crucial, acetone, tetrahydrofuran (THF), CH3CN, or toluene as the solvent led to low yields of CoQ1 (Table 2, entries 5–8). On the basis of these screening studies, the optimum conditions employed BF3·Et2O as the catalyst and dichloromethane as the solvent.
Redox chain reaction for the synthesis of CoQ1.

CoQ1: coenzyme Q1; N.R.: no reaction; THF: tetrahydrofuran; r.t.: room temperature.
Reaction conditions: CoQ0 (0.01 mol), compound
Bold values represents the best condition.
Conclusion
In summary, we have developed a convenient synthetic protocol for the preparation of CoQ1 from cheap and readily available 3,4,5-trimethoxytoluene (TMT) in two steps. The overall yield of CoQ1 is 60%. The intermediate CoQ0 was also obtained in 85% yield in the first step. The second redox chain reaction between allyl bromide and CoQ0 provided a one-step procedure for the direct introduction of allyl groups on quinones in good yield. The reaction is efficient, clean, and the workup is easy. This method may find use for the synthesis of other CoQ compounds.
Experimental section
All reactions were monitored by thin-layer chromatography (TLC; SiO2, petroleum ether (PE; b.p. 50°C–70°C)/ethyl acetate (EtOAc), 5:1). Melting points were measured on BUCHI Melting Point M-565. Nuclear magnetic resonance (NMR) and mass spectra were recorded on a Bruker Avance III-HD 400 NMR and a TripleTOF mass spectrometer, respectively. Potassium persulfate, ammonium persulfate, Hantzsch ester, and BF3·Et2O were purchased from Adamas, P. R. of China, and used without further purification.
General method for the preparation of 2,3-dimethoxy-5-methyl-1,4-benzoquinone (CoQ0)
3,4,5-Trimethoxytoluene (1.82 g, 10 mmol) was dissolved in a mixture of acetic acid (10 mL) and catalytic H2SO4 (0.01 mL), then a solution of the oxidant (15 mmol) was added dropwise over 10 min. The mixture was stirred and heated at 50°C for 1 h. After cooling, the reaction mixture was extracted with CH2Cl2 (3 × 10 mL). The combined organic phases were washed with H2O and NaHCO3, then dried over anhydrous Na2SO4 and evaporated under reduced pressure. The residue was purified by silica gel column chromatography (PE/EtOAc, 5:1) to give CoQ0.
CoQ0, red-colored needles, m.p. 55°C–58°C (Lit. 13 57°C–59°C).
1H NMR (400 MHz, CDCl3): δ = 6.44 (q, J = 1.7 Hz, 1H), 4.02 (s, 3H, OCH3), 4.00 (s, 3H, OCH3), 2.04 (d, J = 1.6 Hz, 3H, CH3).
13C NMR (101 MHz, CDCl3): δ = 184.4 (C
MS (ESI): m/z = 205 [M + Na]+.
The spectroscopic data are in accord with the literature. 13
General method for the preparation of 2,3-dimethoxy-5-methyl-6-(3-methyl-2-butenyl)-1,4-benzoquinone (CoQ1)
1-Bromo-3-methyl-2-butene (
1H NMR (400 MHz, CDCl3): δ = 4.32 (t, 1H, J = 7.0 Hz, C=CH), 3.96 (s, 3H, CH3O), 3.94 (s, 3H, CH3O), 3.12 (d, 2H, J = 7.0 Hz, CH2), 2.14 (s, 3H, CH3), 1.75 (s, 3H, CH3), 1.65 (s, 3H, CH3).
13C NMR (101 MHz, CDCl3): δ = 180.0 (C
MS (ESI): m/z = 251 [M + H]+.
The spectroscopic data are in accord with the literature. 5
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (Nos 31600740 and 81803353), the Natural Science Foundation of Jiangsu Province (BK20160443), the Six Talent Peaks Project in Jiangsu Province (SWYY-094), the Jiangsu Provincial Key Laboratory for Bioresources of Saline Soils (Nos JKLBS2016013 and JKLBS2017010), and the College Students Practice Innovation Training Program of Yancheng Teachers University (Provincial key projects).


