Abstract
A new method for the synthesis of indolin-2-ones has been realized by an I2-promoted oxidative reaction from 1,2,3,3-tetramethyl-3H-indolium iodides. This transformation proceeded smoothly under metal-free and peroxide-free conditions in a cascade manner.
Introduction
Indolin-2-one rings have been recognized as crucial motifs in diverse natural products and pharmaceutically relevant entities.1–3 Several drug discovery programs in industry and academia are anchored around the indolin-2-one scaffold, and drugs such as Sunitinib and Nintedanib are in clinical use for targeted anticancer therapies.4,5 A number of methods for the synthesis of indolin-2-one have been reported. Classical synthetic methods include the derivatization of other heterocycles (such as Wolff–Kishner reduction of isatin and oxidation of indole), Friedel–Crafts cyclizations of α-haloacetanilides, and variations in the Fischer indole synthesis.6–8 Cyclizations of 2-haloacryloylanilide derivatives by a variety of radical initiators have also been used to prepare indolin-2-ones.9–12 In recent years, metal-mediated activations/cyclizations on precrafted advanced precursors have been a commonly pursued approach (Scheme 1). Notable examples are Cu-catalyzed intermolecular C–H cyclization (Scheme 1(a)), 13 Pd-catalyzed insertion of isocyanide (Scheme 1(b)), 14 ruthenium-catalyzed intramolecular alkene hydroarylation (Scheme 1(c)), 15 and Fe-mediated hydrometallation-cyclization (Scheme 1(d)). 16 In addition, Ni-catalyzed aromatic C–H alkylation (Scheme 1(e)) and Ir-catalyzed radical cyclization under visible light (Scheme 1(f)) have also been used to construct indolin-2-one rings.17,18

Representative recent approaches for accessing indolin-2-ones.
Recently, due to the need for “greener chemistry,” chemists are beginning to develop transformations under metal-free conditions. Iodine, a cheap, nontoxic, and abundant halogen, is attracting more and more attention, especially in oxidative coupling reactions to form new carbon–carbon (C–C) or carbon–heteroatom (C–X) bonds.19,20 An in situ iodination usually occurs in iodine-mediated oxidative couplings, followed by in situ oxidation to generate an electrophilic aldehyde group.21–23 1,2,3,3-Tetramethyl-3H-indolium iodide, a well-known synthetic agent of many fluorescent dyes, is considered to contain an activated methyl group which can react with an aldehyde or carbonyl group.24,25 In our previous research, the activated C-2 methyl group reacted with molecular iodine to form an aldehyde via Kornblum oxidation in the presence of dimethyl sulfoxide (DMSO). 26 Based on these results, we report an I2-promoted one-pot method for the synthesis of indolin-2-ones under metal-free conditions.
Results and discussion
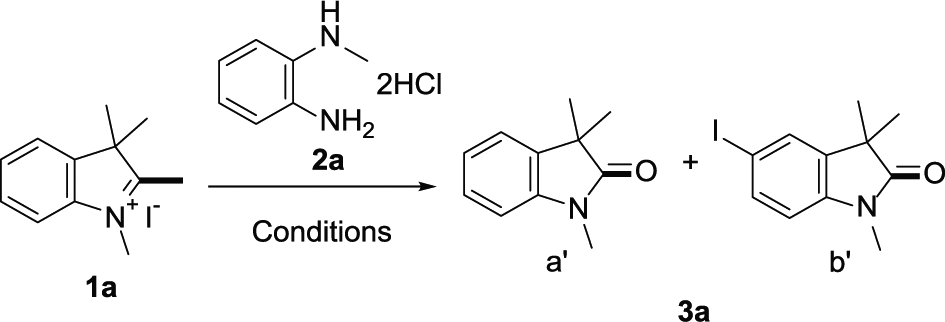
The reaction is demonstrated by using 1,2,3,3-tetramethyl-3H-indol-1-ium iodide (
Optimization of the reaction conditions a .

DMSO: dimethyl sulfoxide; HPLC: high-performance liquid chromatography.
Reaction conditions:
Ratio of a′ to b′ in the isolated product is determined by HPLC.
Isolated yield of
Methanesulfonic acid (2.0 mmol) was added.
H2O (1 mL) was added.
3-Å molecular sieves were added.
Pyridine (0.1 mL) was added.
Ratio of a′ to b′ in the reaction solution was determined by HPLC.
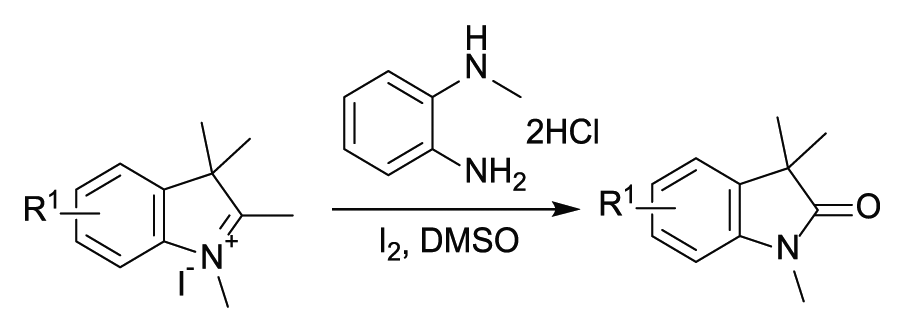
With optimized conditions in hand, the generality and scope of the reaction were investigated (Table 2). 1,2,3,3-Tetramethyl-3H-indol-1-ium iodides
Substrate scope a .

DMSO: dimethyl sulfoxide; NMR: nuclear magnetic resonance.
Reaction was performed with
Yields of isolated products.
The ratio was determined by 1H NMR spectroscopy.
To gain more information about the reaction mechanism, a model reaction of
With the aforementioned results and previous reports in mind,
26
a possible mechanism is proposed in Scheme 2 using 1,2,3,3,5-pentamethyl-3H-indol-1-ium iodide

A plausible reaction mechanism.
In conclusion, a molecular I2 promoted C–C bond cleavage has been developed to construct indolin-2-ones from simple and readily available indolinium iodides. In addition, the reaction performs well in the absence of any metal or peroxide. Further studies to elucidate a detailed mechanism and further explorations of this I2/DMSO-promoted oxidative reaction are currently underway in our laboratory.
Experimental analysis
All reagents and solvents were obtained from commercial suppliers and were used without further purification. The method of preparing starting compounds
General procedure for the synthesis of indolin-2-ones 3
A mixture of quaternary salt
1,3,3-Trimethylindolin-2-one and 7-iodo-1,3,3-trimethylindolin-2-one (mixture obtained from entry 7 in Table 1,
1,3,3,5-Tetramethylindolin-2-one (
5-Chloro-1,3,3-trimethylindolin-2-one (
5-Methoxy-1,3,3-trimethylindolin-2-one (
5-Bromo-1,3,3-trimethylindolin-2-one (
1,3,3-Trimethyl-5-(trifluoromethyl)indolin-2-one (
5,7-Dichloro-1,3,3-trimethylindolin-2-one (
1,3,3,5,7-Pentamethylindolin-2-one (
1,1,3-Trimethyl-1,3-dihydro-2H-benzo[e]indol-2-one (
7-Bromo-1,3,3-trimethylindolin-2-one (
1,3,3,7-Tetramethylindolin-2-one and 5-Iodo-1,3,3,7-tetramethylindolin-2-one (mixture,
1-Ethyl-3,3-dimethylindolin-2-one and 1-Ethyl-5-iodo-3,3-dimethylindolin-2-one (mixture,
1′-Methylspiro[cyclohexane-1,3′-indolin]-2′-one and 5′-Iodo-1′-methylspiro [cyclohexane-1,3′-indolin]-2′-one (mixture,
Supplemental Material
Supporting_Information_for_An_iodine-promoted_one-pot_and_metal-free_access_to_indolin-2-ones – Supplemental material for An iodine-promoted one-pot and metal-free access to indolin-2-ones
Supplemental material, Supporting_Information_for_An_iodine-promoted_one-pot_and_metal-free_access_to_indolin-2-ones for An iodine-promoted one-pot and metal-free access to indolin-2-ones by Yong Zhang, Xi Zong and Min Ji in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant No. 81671745).
Supplemental material
Supplemental material for this article, which contains the NMR spectra and preliminary study of reaction mechanism, is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.