Abstract
The synthesis and characterization of a novel 6,13-dihydro-6,13-ethanopentacene-15,16-diimine palladium(II) complex are described. The effect of the backbone framework of the α-diimine metal catalyst on the polymerization reaction of norbornene and 1-octene is investigated. Compared to the 9,10-dihydro-9,10-ethanoanthracene-11,12-diimine palladium(II) complex (
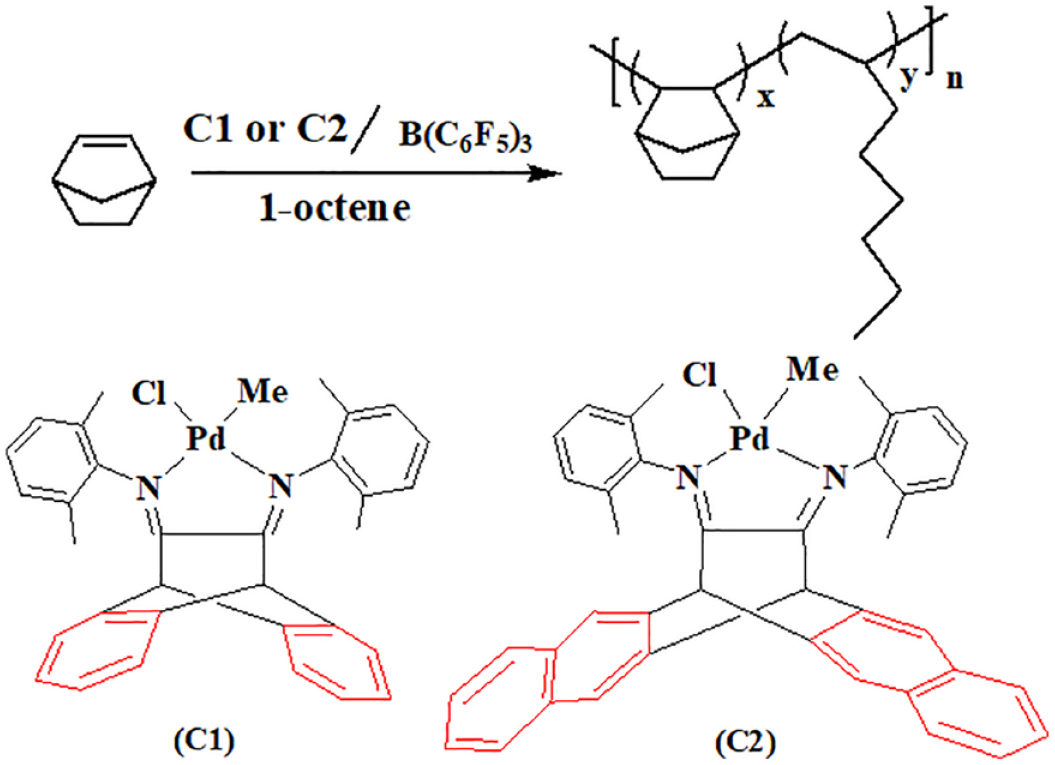
Introduction
Since Brookhart and colleagues discovered aryl-substituted α-diimine nickel and palladium complexes for olefin polymerization,1–3 late-transition-metal catalysts have attracted increasing attention due to their lower oxophilicity and resistance toward polar functionalities.4–13A series of breakthroughs have been made in this field, mainly involving other late transition metal α-diimine catalysts.
The catalytic features of the late-transition-metal α-diimine catalysts rely on the electronic or steric influence of the ligand used.14–18 However, the current reported catalysts have some defects, such as poor thermal stability and short catalytic lives, which limit their applications in industrial polymerization. As far as we know, bulky substituents can hinder the rotation of aniline moieties and protect the metal center effectively, which leads to the enhancement of the thermal stability of the catalyst. α-Diimine nickel catalysts with a bulky camphyl backbone were synthesized by Wu and colleagues,19,20 which showed good thermal stability toward olefin polymerizations and enabled special branched polyolefins to be obtained. Sun and colleagues21–24 have reported a series of Ni(II) complexes bearing unsymmetrical α-diimine ligands that exhibit high activity and thermal stability in olefin polymerization. Chen and colleagues 25 showed that a naphthyl-α-diimine nickel complex bearing chiral bulky sec-phenethyl groups at the o-naphthyl position exhibited good catalytic activity and resulted in branched polymers (42–88/1000C) with high molecular weights (Mn: (4.3–15.2) × 104 g·mol−1) and narrow molecular weight distributions (MWDs; Mw/Mn = 1.13−1.29, room temperature (RT)). From the aforementioned results, the incorporation of bulky axial substituents in the α-diimine ligand is crucial for the enhancement of the thermal stability of the catalyst.
There are a large number of papers on α-diimine nickel and palladium complexes containing bulky substituents at the ortho-position of the aniline rings. However, very little work has been performed on studies of the backbone steric effects of α-diimine metal complexes for olefin polymerization. Recently, we successfully explored a type of thermostable and highly active 9,10-dihydro-9,10-ethanoanthracene-11,12-diimine Pd(II) catalysts with three-dimensional geometry and large steric hindrance on the backbone toward norbornene (NB) polymerization.
26
In this context, we became very interested in the influence of the backbone steric effects of such α-diimine palladium complexes on olefin polymerization. Herein, we report the application of a 6,13-dihydro-6,13-ethanopentacene-15,16-diimine palladium(II) complex (
Results and discussion
Synthesis of the polycyclic aromatic palladium complexes
6,13-Dihydro-6,13-ethanopentacene-15,16-diimine formed the corresponding palladium complex (
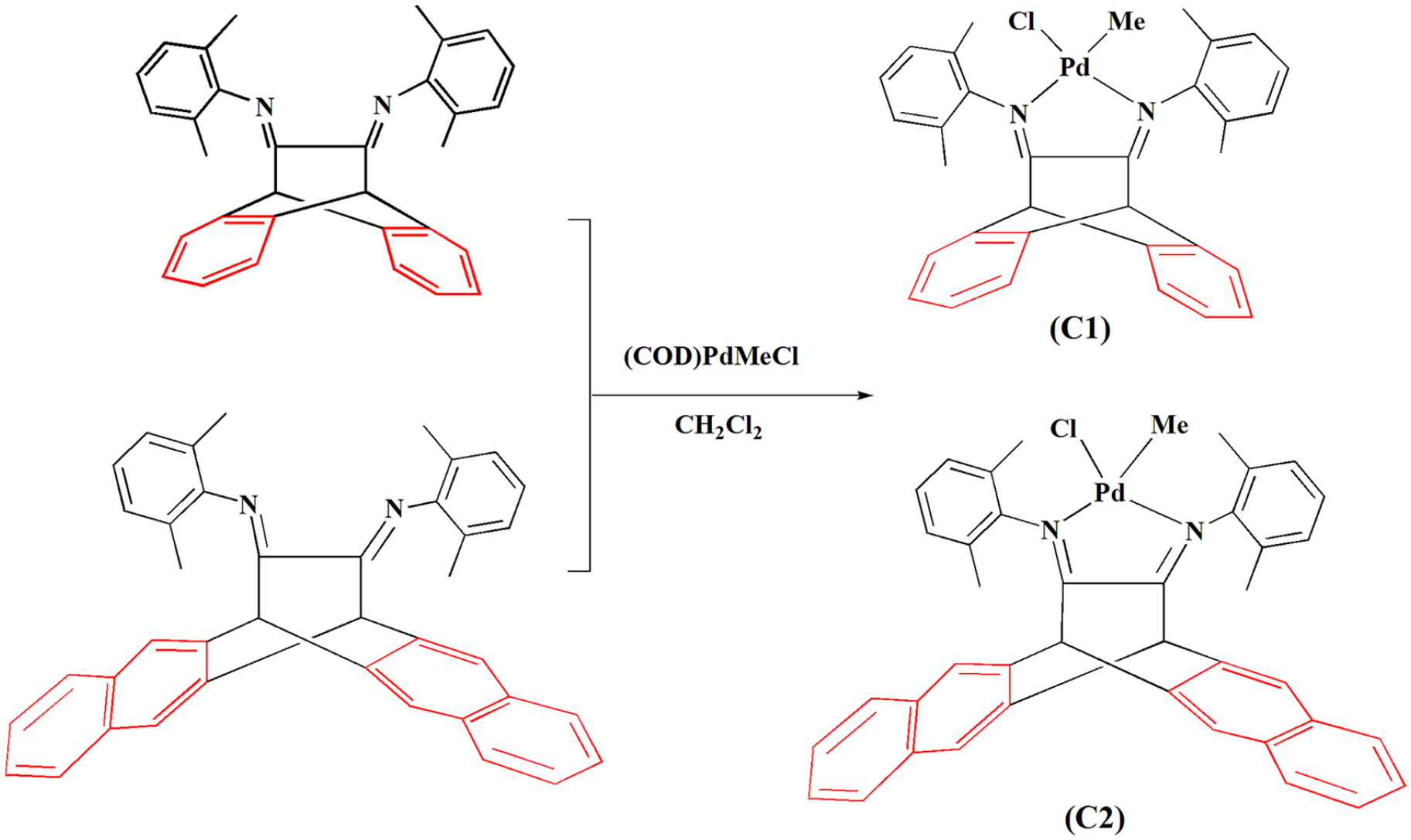
Synthesis of the polycyclic aromatic palladium complexes.
Copolymerization of NB with 1-octene
To investigate the influence of the backbone framework of the catalysts on the polymerization activity, the copolymerization of NB with 1-octene using different co-monomers and monomer feed ratios in the presence of complex
Copolymerization of NB with 1-octene catalyzed by polycyclic aromatic palladium complexes/B(C6F5)3 systems. a .
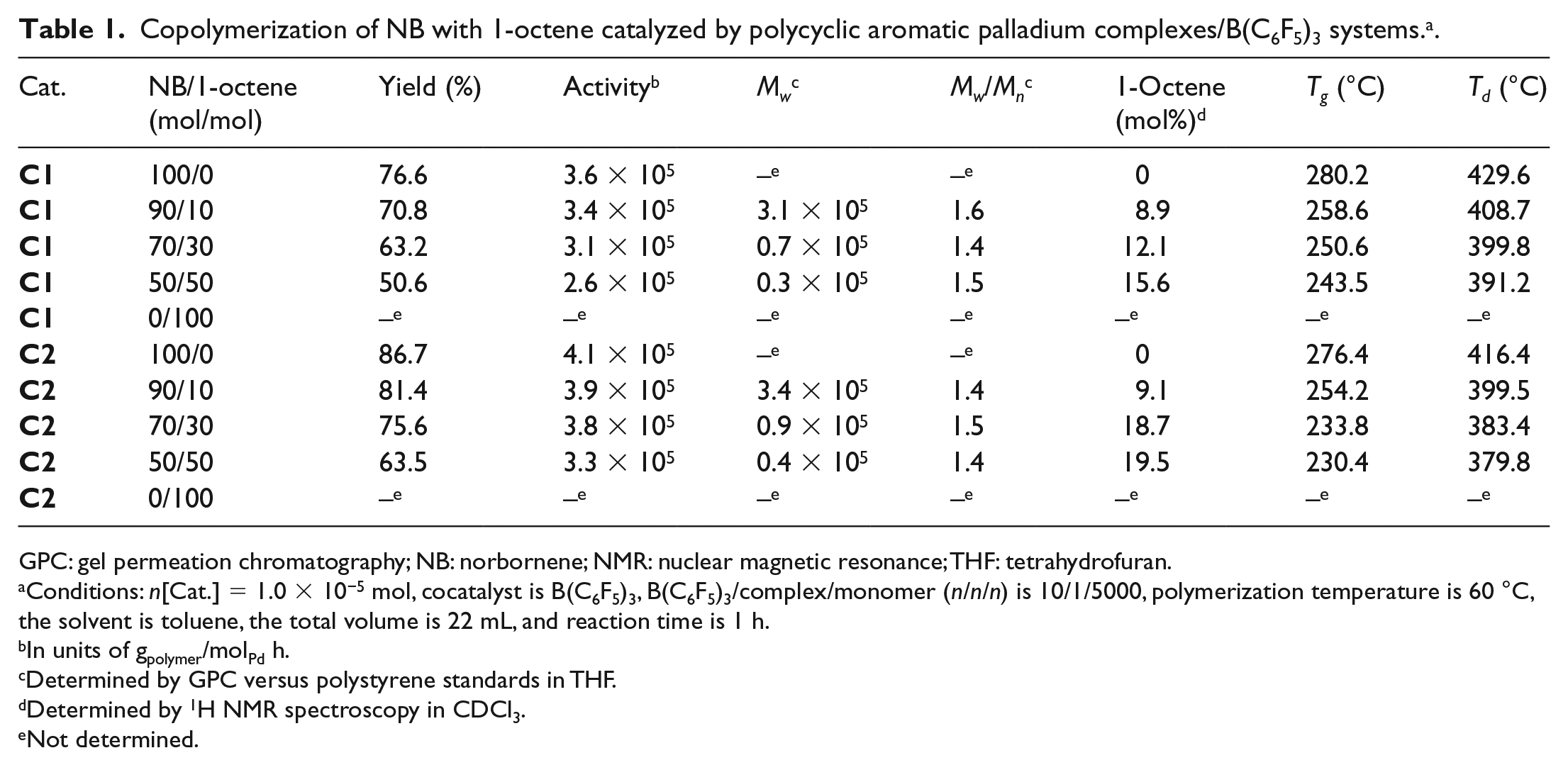
GPC: gel permeation chromatography; NB: norbornene; NMR: nuclear magnetic resonance; THF: tetrahydrofuran.
Conditions: n[Cat.] = 1.0 × 10−5 mol, cocatalyst is B(C6F5)3, B(C6F5)3/complex/monomer (n/n/n) is 10/1/5000, polymerization temperature is 60 °C, the solvent is toluene, the total volume is 22 mL, and reaction time is 1 h.
In units of gpolymer/molPd h.
Determined by GPC versus polystyrene standards in THF.
Determined by 1H NMR spectroscopy in CDCl3.
Not determined.
Besides the activity, the backbone framework of catalyst also affected the incorporation rate of the monomer as well as the yield. As shown in Table 1, the 1-octene content in the copolymers was 8.9%–15.6% for
All the copolymers of NB and 1-octene were soluble in common organic solvents (such as CHCl3, CH2Cl2, cyclohexane, and tetrahydrofuran (THF)) as well as chlorobenzene at ambient temperature, which exhibited better solubility than polynorbornene (PNB).
Characterization of the copolymers
The obtained copolymers were characterized by gel permeation chromatography (GPC) and the results are shown in Table 1. The molecular weights of the obtained copolymers were up to 105, and the MWDs of the obtained polymers were all relatively narrow (MWD < 1.6). The molecular weights of the copolymers prepared using the
1H and 13C NMR spectra of the copolymers
The structures of the poly(NB-co-1-octene) polymers obtained using the

The 1H NMR spectra of the poly(NB-co-1-octene) polymers with: (a) 8.9%, (b) 12.1%, (c) 15.6%, (d) 9.1%, (e) 18.7%, and (f) 19.5% of 1-octene. Spectra (a)–(c) are obtained using
From the above, it can be seen that the 1-octene content in the copolymers obtained using
The 13C NMR spectra of poly(NB-co-1-octene) containing 19.5% of 1-octene obtained by using the

The 13C NMR spectra of poly(NB-co-1-octene) obtained using
Fourier-transform infrared spectroscopy spectra of the copolymers
The Fourier-transform infrared spectroscopy (FTIR) spectra of poly(NB-co-1-octene) obtained by the

The FTIR curves of poly(NB-co-1-octene)s with: (a) 8.9%, (b) 12.1%, (c) 15.6%, (d) 9.1%, (e) 18.7%, and (f) 19.5% of 1-octene. Spectra (a)–(c) are obtained using
Wide-angle X-ray diffraction analyses of the copolymers
Partial information on the conformation of the copolymers prepared using the

WXRD curves of poly(NB-co-1-octene)s with: (a) 8.9%, (b) 12.1%, (c) 15.6%, (d) 9.1%, (e) 18.7%, and (f) 19.5% of 1-octene. Spectra (a)–(c) are obtained using
TGA and DSC analyses of the copolymers
The decomposition temperatures (Tds) of the copolymers with different 1-octene contents prepared using the
As shown in Table 1, the glass transition temperatures (Tgs) of the copolymers occur at high temperatures. The Tg values of the copolymers decreased as the 1-octene molar ratio increased in the copolymers, and the Tgs of the polymers obtained with the
Conclusion
A novel 6,13-dihydro-6,13-ethanopentacene-15,16-diimine palladium(II) complex (
Experimental
Materials
All manipulations of air-sensitive and water-sensitive compounds were performed under an inert atmosphere. Solvents were purified using standard procedures. Toluene was dried over sodium/benzophenone and distilled under nitrogen before use. Tris(pentafluorophenyl) borane (B(C6F5)3, 97 %) was purchased from J&K (Tianjin, China). NB (98%) was purchased from Sigma-Aldrich (Shanghai, China) and purified by drying over sodium and distilling at 106 °C under N2, and used as a 0.4 g/mL (4.25 mol/L) solution in toluene. 1-Octene was purchased from Sigma-Aldrich (Shanghai, China) and purified by washing twice with aqueous sodium hydroxide (5.0 wt%) and twice with water to remove inhibitors. This was followed by drying over anhydrous CaCl2 and distillation over CaH2 under an argon atmosphere at reduced pressure.
Characterization
The NMR spectra of the copolymers were recorded on a Bruker ARX 600 NMR spectrometer at ambient temperature, with CDCl3 as the solvent. GPC was carried out on a Breeze Waters system using polystyrenes as the standard and THF as the eluent at a flow rate of 1.0 mL/min. The infrared (IR) spectra were recorded on a Shimadzu IR Prestige-21 FTIR spectrophotometer. Thermal gravimetric analysis (TGA) measurements were performed on a Perkin-Elmer TGA 7 instrument from RT to 600 °C at a rate of 20 °C/min under a nitrogen atmosphere. The differential scanning calorimetry (DSC) measurements were obtained on a Shimadzu DSC-60 with a heating/cooling rate of 10 °C/min under a nitrogen atmosphere. The WXRD curves were recorded on a Bruker D8 Focus X-ray diffractometer, operating at 40 kV and 40 mA with a copper target (λ = 1.54 Å) and at a scanning rate of 2°/min.
Synthesis of the 9,10-dihydro-9,10-ethanoanthracene-11,12-diimine methyl palladium chloride complex (
Synthesis of the 6,13-dihydro-6,13-ethanopentacene-15,16-diimine methyl palladium chloride complex (
Polymerization
All procedures were carried out under a nitrogen atmosphere. A typical copolymerization procedure was as follows: a toluene solution of NB and 1-octene was added via a syringe into a 100 mL two-necked round-bottom flask containing a magnetic stir bar. The toluene solution of palladium complex was then added followed by toluene solution of B(C6F5)3. The total volume was kept constant at 22 mL. The polymerization reaction was performed for 1 h at 60 °C. The reaction was terminated by addition of acidic ethanol (90:10, ethanol/HCl). The resulting precipitated polymers were collected by filtration and washed with ethanol several times and then dried under vacuum at 60 °C.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Nos 21664014 and 21764016) and the Natural Science Foundation of Jiangxi Province (No. 20181BAB203002).
