Abstract
A practical approach for the synthesis of cyclic phenyl hexayne is demonstrated through a one-pot deprotection/transformation of magnesium acetylide into a copper acetylide/Sonogashira coupling procedure, followed by dephosphination, Hay coupling, desilylation, and Eglinton coupling. This approach avoids tedious synthetic routes and harsh reaction conditions and affords the product in 41% yield.

A practical approach for the synthesis of cyclic phenyl hexayne from Me3Si-/Ph2P(O)-protected ethyne is demonstrated through a one-pot deprotection/transformation of magnesium acetylide into a copper acetylide/Sonogashira coupling procedure, followed by dephosphination, Hay coupling, desilylation, and Eglinton coupling.
Introduction
Inherently nucleophilic alkynes, important building blocks for preparation of biological active compounds and organic functional compounds, are widely used in organic synthesis.1–11 Phenyl polyynes,
12
especially highly π-conjugated cyclic phenyl polyynes having rigid structures and highly expanded π-systems, can be widely used in organic materials such as organic field-effect transistors (OFETs),
13
organic light-emitting diodes (OLEDs),
14
dye-sensitized solar cells (DSSCs),
15
semiconductors,
16
and liquid crystals.
17
Transition-metal-catalyzed cross-coupling reactions18–23 are efficient approaches for synthesis of polyynes. Haley has synthesized the cyclic phenyl hexayne

Haley’s procedure for the synthesis of cyclic pentayne
Results and discussions
We initiated our studies with the synthesis of expanded Me3Si-/Ph2P(O)-protected ethyne
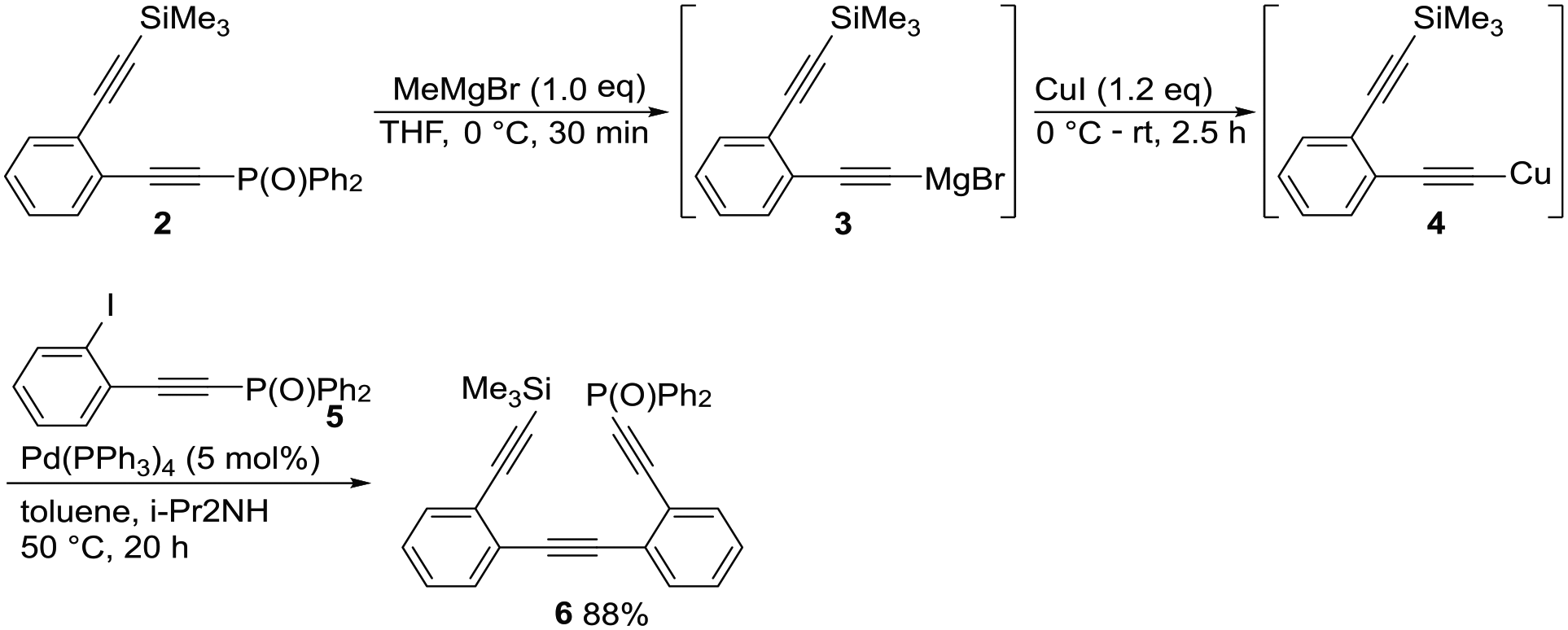
Synthesis of expanded Me3Si-/Ph2P(O)-protected ethyne
Without transformation of the magnesium acetylide

Synthesis of expanded Me3Si-/Ph2P(O)-protected ethyne
With triyne
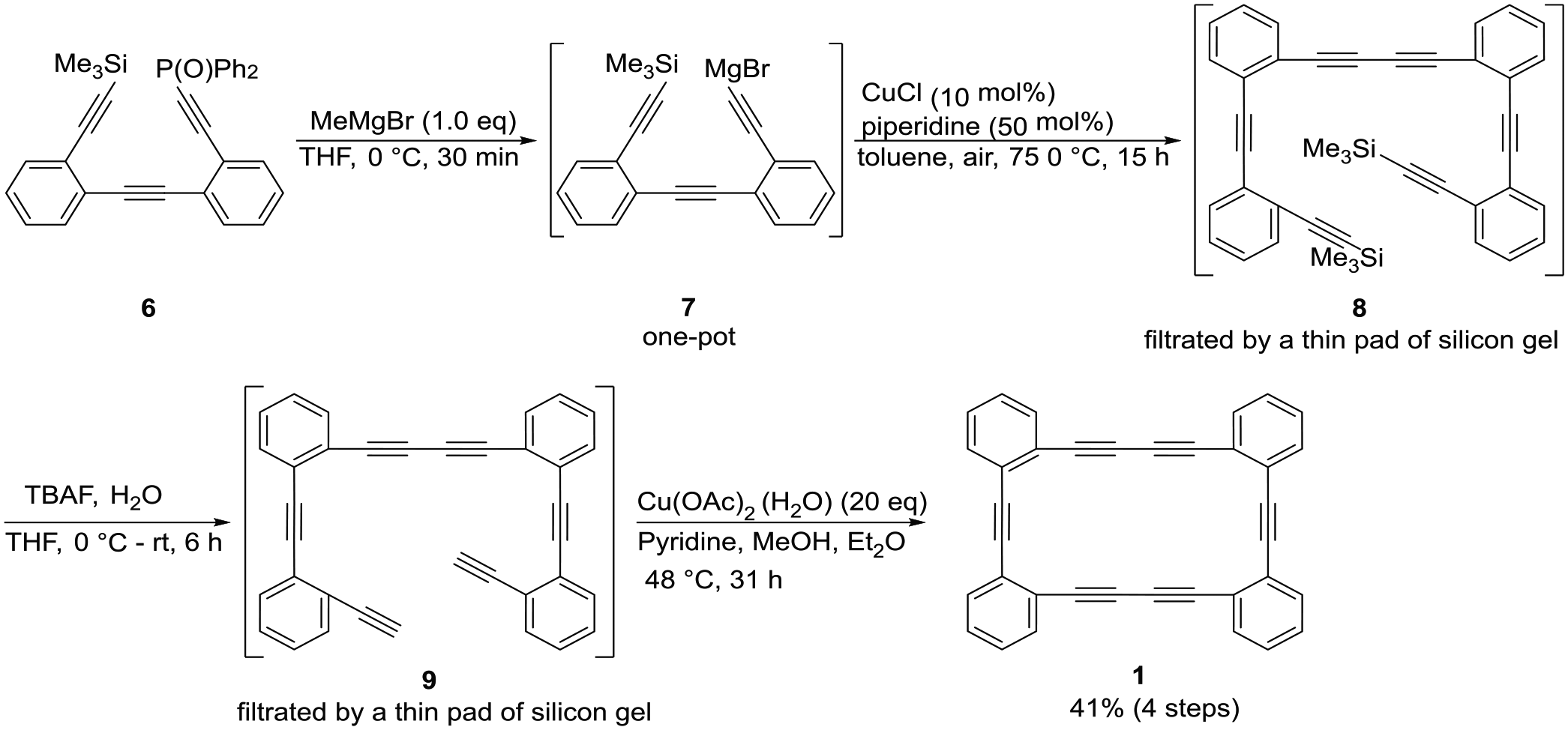
Synthesis of cyclic phenyl hexayne
Conclusion
In conclusion, we have developed an efficient procedure for the synthesis of cyclic phenyl hexayne from Me3Si-/Ph2P(O)-protected ethynes through a one-pot deprotection/transformation of magnesium acetylide into copper acetylide/Sonogashira coupling procedure followed by dephosphination, Hay coupling, desilylation, and Eglinton coupling. Our procedure demonstrates some advantages over the previous route, such as a short sequence, mild reaction conditions, and a high total yield.
Experiment
Dry solvents, reagents, and catalysts were purchased as analytical grade and used without further purification. Starting materials
Synthesis of 6
To a tetrahydrofuran (THF) solution (10.0 mL) of
Synthesis of cyclic phenyl hexayne 1
To a THF solution (5 mL) of
Supplemental Material
supplementary_material – Supplemental material for Synthesis of cyclic phenyl hexayne from Me3Si-/Ph2P(O)-protected ethynes
Supplemental material, supplementary_material for Synthesis of cyclic phenyl hexayne from Me3Si-/Ph2P(O)-protected ethynes by Li Wu, Li-fen Peng, Zhi-fang Hu, Hong Wang, Zi-long Tang, Yin-chun Jiao and Xin-hua Xu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the National Natural Science Foundation of China (Nos. 21802040 and 21877034), the Natural Science Fund Youth Project of Hunan Province (No. 2018JJ3145), the General Project of Hunan Education Department (No. 17C0629), and the Open Foundation of Key Laboratory of Theoretical Organic Chemistry and Functional Molecule of Ministry of Education, Hunan University of Science and Technology (No. E21843).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
