Abstract
A novel, reactive, yellow fluorescent organosilicon dye, N-propyl-(diethoxy)methyl-silane-4-dimethylamino-naphthalimide, is designed and synthesized and is used to fabricate a covalently yellow fluorescent silicone oil by polycondensation of hydroxy-terminated polydimethylsiloxane. The chemical structure and optical properties of the N-propyl-(diethoxy)methyl-silane-4-dimethylamino-naphthalimide and covalently yellow fluorescent silicone oil are characterized by 1H nuclear magnetic resonance, 13C nuclear magnetic resonance, mass spectrometry, Fourier transform infrared, UV–Vis, and fluorescence spectra. The results indicate that the fluorescence quantum yields of N-propyl-(diethoxy)methyl-silane-4-dimethylamino-naphthalimide and covalently yellow fluorescent silicone oil are 2.61% and 3.5%, respectively. The λmax values of their dichloromethane solutions are 416 nm, and their λex and λem are 430 and 509 nm, respectively. Furthermore, covalently yellow fluorescent silicone rubbers are prepared using tetraethoxysilane as a cross-linker, and some of their properties are investigated. Thermogravimetric analysis and dynamic mechanical analysis show that heat resistance and tan δ of the covalently yellow fluorescent silicone rubber are improved in comparison with silicone rubbers without N-propyl-(diethoxy)methyl-silane-4-dimethylamino-naphthalimide moieties. Solvent extraction experiments indicate that the solvent resistance of the covalently yellow fluorescent silicone rubber is much better than that of noncovalently yellow fluorescent silicone rubber.

Introduction
With their excellent characteristics of temperature resistance, permeability, and biocompatibility, 1 silicone materials have been widely applied in various fields like biomedicines,2–4 semiconductors, 5 three-dimensional (3D) printing, and so on. 6 There is currently a great demand for colorized organosilicon products. However, due to the poor compatibility between pigments/dyes and polysiloxanes, 7 problems such as precipitation of pigments during storage, uneven color distribution on usage, poor color fastness to solvent resistance, and strong bio-toxicity often occur, which have limited the applications of such products.
It has been demonstrated that the light fastness and solvent fastness of covalently colored polymer latex films were much better than those of noncovalently colored films.8–10 Similar investigations have also been focused on the covalent bonding of the chromophore to silicone, and some progress has been made. For example, Meng et al.11–13 combined a series of fluorescence dyes with organoalkoxysilanes through covalent bonds and then connected the resulting fluorescent organoalkoxysilanes to mesoporous silica via sol–gel reaction to produce chemosensors, which inherited the good properties of mesoporous silica, that is, biocompatibility and thermal stability. Oehrlein 14 chemically bonded anthraquinone dyes with siloxy-containing molecules, followed by reaction with silsesquioxane, to prepare colorized silsesquioxane, which has been used as bodily-contact colorant. Yan and colleagues15,16 used terthiophene and perylenediimide derivatives to covalently bond with small silicone molecules containing hydroxy or alkoxy groups and then copolymerized to fabricate colored alternating copolysiloxane. Banning 17 prepared colorized polydimethylsiloxane (PDMS) by reaction of amino-containing PDMS with reactive dyes, figuring out the compatibility issue between PDMS and dyes. Both Xu and Wang covalently bonded chromophores with small silicone molecules in order to prepare colorized silicone resins.18,19 It is known that alkoxysilanes with two or three alkoxy groups are the basic molecule to obtain the polysiloxanes. As described above, many researchers focus on the colored trialkoxysilanes and use them as cross-linker to prepare colored silicone rubbers and silicone resins. It is noted that those colored trialkoxysilanes are solid, and a certain amount of solvent is needed in the cross-linking process, which will greatly affect the properties of the resulting colored silicone rubber because of the poor solubility between solvent and reinforcing fillers. On the other hand, colored silicone oils cannot be obtained from this kind of colored organosilicon. Thus, colored dialkoxysilanes which have two alkoxy groups are demanded to solve the above problems and improve the properties of the colored silicone materials. Because of its unique photophysical properties and stable chemical structure, naphthalimide has been widely used in various fields.20,21 In this paper, the naphthalimide group was selected as a chromophore to fabricate covalently bound colored silicones. For this purpose, a yellow fluorescent organosilicon dye with a chromophore and a diethoxysilane group, N-propyl-(diethoxy)methyl-silane-4-dimethylamino-naphthalimide (PDSNA), was first prepared via a two-step organic reaction, and then, it was used as a chain extender to react with hydroxy-terminated polydimethylsiloxane (PDMS-OH) to fabricate the covalently yellow fluorescent silicone oil (CYF-SO). The structures and optical properties of the PDSNA and CYF-SO were characterized. Afterwards, using CYF-SO, PDMS-OH, and tetraethoxysilane (TEOS) as raw materials, covalently yellow fluorescent silicone rubbers (CYF-SRs) were prepared, and their thermal and solvent resistance properties were investigated.
Results and discussion
Synthesis and characterization of the reactive silicone dye
Design of the yellow fluorescent silicone dye
PDSNA, a reactive silicone dye containing a yellow fluorescent chromophore and a diethoxysilane group, was designed and synthesized. This can then be used as a chain extender to react with PDMS-OH to produce CYF-SO, and then further to prepare CYF-SRs. As a comparison, N-butyl-4-dimethylamino-naphthalimide (BDNA), an unreactive yellow fluorescent dye with the same chromophore and without a diethoxysilane group, was also fabricated. Since BDNA could not react with the silanol group, noncovalently yellow fluorescent silicone rubber (NCYF-SR) was prepared by using the same amount of BDNA to replace PDSNA. The chemical structure of the silicone dyes was confirmed by 1H nuclear magnetic resonance (NMR), 13C NMR, Fourier transform infrared (FTIR), and mass spectrometry (MS).
UV–Vis absorption spectra
The UV–Vis absorption spectra of PDSNA and DMANA (4-dimethylamino-naphthalimide) in dichloromethane (DCM) are given in Figure 1. Both spectra show three absorption bands: two located in the UV region and one located in the visible region, and the corresponding data are given in Table 1.

UV–Vis absorption spectra of PDSNA and DMANA in DCM (25 mg L−1).
Photophysical properties of DMANA, PDSNA, and CYF-SO.

QY: quantum yield; DMANA: 4-dimethylamino-naphthalimide; PDSNA: N-propyl-(diethoxy)methyl-silane-4-dimethylamino-naphthalimide; CYF-SO: covalently yellow fluorescent silicone oil; λex: excitation peak wavelength; λem: emission peak wavelength.
Fluorescent spectra
The fluorescent spectrum of PDSNA in DCM (5 mg L−1) is presented in Figure 2(a). Its excitation peak wavelength (λex) and emission peak wavelength (λem) were, respectively, at 430 and 509 nm, and Stokes shift was 79 nm. Compared with DMANA, its λex hypochromatic shift was 3 nm and λem hypochromatic shift was 6 nm. As indicated in Figure 2(b), there was a linear relationship between the PDSNA concentration and its fluorescent intensity, which was the basis of the quantitative analysis of PDSNA in our following work.

(a) Fluorescent emission spectrum of PDSNA in DCM (5 mg L−1) and (b) relationship between the fluorescence intensity and concentration of PDSNA in DCM.
The fluorescence quantum yield (QYs) represents the ratio of the number of photons emitted to the number absorbed, which is regarded as an imperative parameter for fluorescent substance characterization. The QYs of PDSNA was 2.61%, which was higher than that of DMANA (QYs = 1.20%). The reason was that when the imide group with strong electrophilic effect was connected with a propyl group as an electron-donating group, the internal charge transfer would be enhanced, resulting in intensifying of the fluorescent characteristics. 20
Synthesis and characterization of the CYF-SO
As a chain extender of PDMS-OH, PDSNA can react with the HO-Si group to link the yellow fluorescent chromophore to PDMS. By changing the dosage of PDSNA and the viscosity of PDMS-OH, two CYF-SO series were prepared, and their spectral characteristics including infrared (IR), UV–Vis, and fluorescence were examined.
FTIR analysis
Samples of CYF-SO (2) and CYF-SO (b) were selected to study the FTIR analysis, and as control experiments, the FTIR spectra of PDMS-OH and PDSNA were also recorded. As shown in Figure 3, the absorptions at 1702 and 1662 cm−1 were due to stretching vibrations for C=O, while those at 1613, 1589, and 1570 cm−1 were characteristic absorptions for naphthalene and benzene. The above-mentioned absorptions appeared in the samples of CYF-SO (2) and CYF-SO (b) but were not traceable in the spectrum of PDMS. Combined with the subsequent test on solvent resistance against NCYF-SR, it could be demonstrated that PDSNA has been covalently bonded to PDMS, that is, CYF-SO was prepared successfully.

FTIR spectra of the PDMS, PDSNA, CYF-SO (2), and CYF-SO (b).
UV–Vis absorption analysis
The UV–Vis absorption spectra of CYF-SO prepared via different methods are shown in Figure 4. Compared with PDSNA, the position of the visible region absorption peak λmax (416 nm) and its peak pattern showed no evident change, indicating that the chemical structure of the chromophoric group did not change during polycondensation; thus, the synthesized silicone oil possessed the same color as PDSNA. In addition, the absorbance of the CYF-SO increased gradually with the increasing dosage of PDSNA as shown in Figure 4(a), while for the silicone oils prepared with different viscosities of PDMS-OH and an equal amount of PDSNA, the absorbances were almost the same as shown in Figure 4(b).

UV–Vis absorption spectra of CYF-SO in DCM (25 mg L−1): (a) with different amounts of PDSNA and (b) with different viscosities of PDMS-OH.
Fluorescent spectrum analysis
The fluorescence emission spectra of CYF-SO in DCM (0.25 g L−1, λem = 509 nm) are shown in Figure 5. It can be observed that the peak pattern of the fluorescent emission spectra is similar to that of PDSNA (Figure 2), and the λem at 506 nm exhibited a slight 3- to 5-nm hyperchromic shift to PDSNA, indicating that the naphthalimide fluorophore kept its own fluorescence property after being covalently bonded onto the PDMS chain. The fluorescence intensity of the CYF-SO (DCM solution) at the λem increased with the increasing amount of PDSNA (shown in Figure 5(a)), indicating that the brightness of the fluorescence could be controlled by adjusting the PDSNA dosage. On the other hand, with the viscosity of PDMS-OH increasing, the fluorescence intensity of the samples had no significant variation (shown in Figure 5(b)), which proved that the viscosity of PDMS-OH did not affect the fluorescent properties.

Fluorescence emission spectra of the CYF-SO in DCM (0.25 g L−1): (a) with different amounts of PDSNA and (b) with different viscosities of PDMS-OH.
As shown in Table 1, the QYs was also different between the final CYF-SO product and its precursor PDSNA. With the increasing dosage of PDSNA, the fluorescence intensity of CYF-SO increased gradually, and their QYs was all higher than that of PDSNA. A possible reason for the remarkable improvement of QYs after covalent bonding with polysiloxane chains was that the chromophoric group was distributed over the polysiloxane chains evenly, thus reducing the odds of making them to collide and quench.9,21
CYF-SRs and their properties
In order to understand the influence of PDSNA on the thermal properties of CYF-SRs, CYF-SR was fabricated using 1.0 wt% of PDSNA, and common silicone rubber (SR) without using PDSNA was prepared according to the traditional preparation method.
Thermogravimetric analysis test
Thermogravimetric analysis (TGA) is an important method to test the thermal stability of materials. The results of TGA for the silicone rubbers prepared with different formulations are given in Figure 6 and Table 2. The TInitial of CYF-SRs was 10 °C higher than that of SR, and the T50% loss of CYF-SRs was 17 °C higher than that of SR, meaning that the PDSNA can improve the heat resistance of the rubbers.

TGA thermogram of the CYF-SR and SR.
TGA mass loss data of CYF-SR and SR.

TGA: thermogravimetric analysis; SR: silicone rubber; CYF-SR: covalently yellow fluorescent silicone rubber.
Dynamic mechanical analysis test
Dynamic mechanical analysis (DMA) is a technique used to measure the glass transition temperature and loss factor. As indicated in Figure 7 and Table 3, compared with SR, the tan δ value of the CYF-SR increased greatly, while Tg had no obvious change, which indicated that the naphthalimide unit could increase the friction with the molecular chain and consume energy, which made the tan δ value higher.

DMA curves of CYF-SR and SR.
DMA results on CYF-SR and SR.

DMA: dynamic mechanical analysis; CYF-SR: covalently yellow fluorescent silicone rubber.
Solvent resistance
To clarify the color fastness of the CYF-SR to solvent resistance, an NCYF-SR with the same amount of the dye was prepared as a control experiment. Since DCM could not dissolve the CYF-SR, but did dissolve BDNA and PDSNA perfectly, it was used as the solvent in Soxhlet extraction experiments. UV–Vis absorption spectrometry was used to measure the λmax absorbance of PDSNA and BDNA with different concentrations, and results are listed in Table 4. The smaller the extraction rate (Re) value, the better color fastness to solvent resistance.
Extraction results for NCYF-SR and CYF-SR with DCM.

CYF-SR: covalently yellow fluorescent silicone rubber; NCYF-SR: noncovalently yellow fluorescent silicone rubber; DCM: dichloromethane.
It was evident that the dye of NCYF-SR was extracted easily, within 1 h, and the Re reached up to 77.2%, which was much higher than CYF-SRs. As the extraction time increased, the Re value of both rubbers increased. After 5 h, the Re of the CYF-SR was 7.0%, while the Re of NCYF-SR reached 85%, indicating that the solvent resistance of the CYF-SR was much better than that of the NCYF-SR.
Conclusion
The synthesized reactive dye PDSNA, an organosilicon monomer containing a yellow fluorescent chromogenic group and a diethoxy group, could be used as a chain extender to prepare CYF-SO and CYF-SR. The λmax value of PDSNA and CYF-SO was 416 nm in the UV–Vis absorption spectrum. The QYS of PDSNA and CYF-SO were 2.61% and 3.5%, respectively. Compared with common silicone rubber without color, CYF-SRs had certain benefits with respect to both heat resistance and loss factor in the low temperature zone. Compared with the NCYF-SR, the CYF-SR turned out to be a significant improvement regarding color fastness to solvent resistance.
Experimental
Materials
4-Bromo-1,8-naphthalic anhydride and 3-(diethoxy(methyl)silyl)propan-1-amine were purchased from Adamas Reagent Co., Ltd. (China) PDMS-OH (molecular weight 4200, viscosity 30 cP) was purchased from Alfa Aesar Co., Ltd. (China) PDMS-OH with different viscosities (500, 2000, 5000, and 8000 cP) were purchased from Jinan Xingfei Long Chemical Co., Ltd. TEOS (China), ditin butyl dilaurate, and 3-dimethylaminopropionitrile were purchased from Aladdin Industrial Co. (Shanghai, China). DCM and ethanol were obtained from Shanghai Titan Scientific Co., Ltd, (China) and isopentanol was purchased from Modern Oriental (Beijing) Technology Development Co., Ltd.
Characterization
1H NMR and 13C NMR spectra were obtained on a JOEL-ECS 400 spectrometer (JEOL, Japan) with tetramethylsilane as the internal standard and using CDCl3 as the solvent. MS spectra were obtained on a liquid chromatograph-mass spectrometer (LCMS)-ion trap (IT)/ time-of-flight (TOF) spectrometer (Shimadzu, Japan). FTIR spectra were recorded on a Nicolet 560 FTIR spectrometer (Thermo Fisher Scientific, USA). UV–Vis absorption spectra were obtained on a T6 UV–Vis spectrometer (Purkinje General, China http://www.pgeneral.com/). Fluorescent spectra were recorded on an RF-5301PC luminescence spectrometer (Shimadzu, Japan).
TGAs were measured on a TGA Q5000 (TA, USA), where the N2 flow rate was 50 mL min−1, and the heating rate was 10 K min−1. TInitial is the initial thermal decomposition temperature, and T10% loss is the temperature with mass loss up to 10%, where T50% loss and T90% loss share the same meaning. DMAs were recorded on a DMA Q800 (TA, USA) in stretch mode, where test frequency was 1 Hz and the heating rate was 5 K min−1.
Measurement of the fluorescent quantum yields (QY) was accomplished using Rhodamine 6G (QY 95%) as reference. 22 At sample’s excitation peak wavelength (λex), measure the UV–Vis absorbances and the area fluorescence intensities of the sample and the reference in ethanol, respectively. Then, the values of QYs were calculated with the following equation
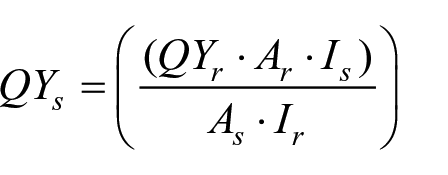
where QYr is the QY of the Rhodamine 6G reference, that is, 95%; As and Ar represent the absorbances at the excitation wavelength of the sample and the reference, respectively; and Is and Ir are the area fluorescence intensities of the sample and the reference, respectively.
Solvent fastness was tested using a Soxhlet apparatus as follows: CYF-SR and NCYF-SR were both extracted with DCM for 5 h. The extraction rate (Re) was calculated as follows

where m1 and m denote the extracted fluorescent dye amount and the fluorescent dye amount used in the method, respectively. The relation between absorbance (y) and concentration (x) was fitted accordingly. The absorbance at the λmax was measured. Both m1 and m were determined by UV–Vis analysis.
Synthesis of the reactive dye and the unreactive dye
The reactive yellow fluorescent dye, PDSNA, and the unreactive yellow fluorescent dye, BDNA, were prepared. The synthetic route is given in Scheme 1.
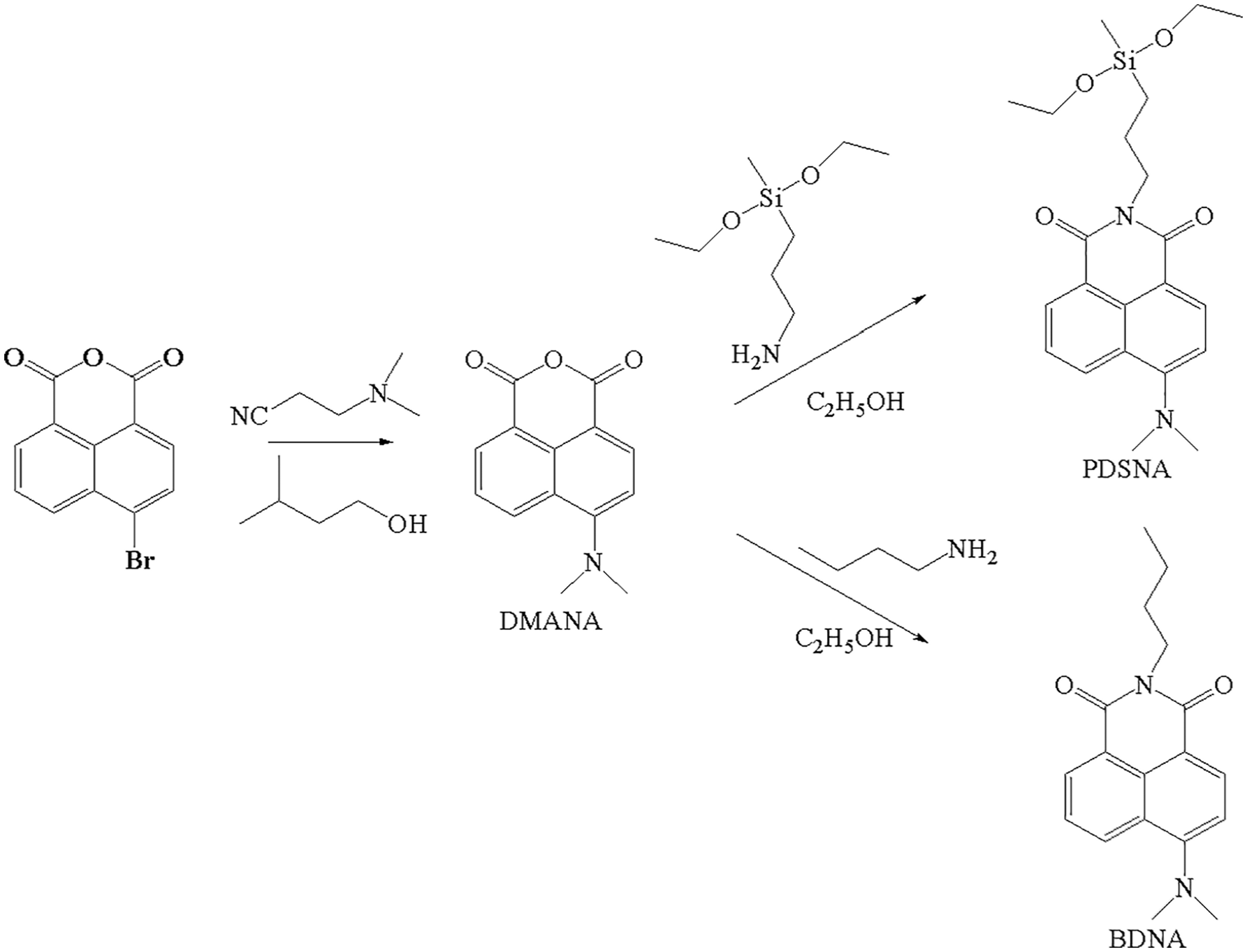
Synthesis of the reactive yellow fluorescent dye PDSNA and the unreactive yellow fluorescent dye BDNA.
Synthesis of DMANA
Compound DMANA was prepared according to reported procedures. 23 1H NMR (400 MHz, CDCl3): δ 8.54 (d, J = 7.3 Hz, 1H), 8.49 (d, J = 8.6 Hz, 1H), 8.43 (d, J = 8.3 Hz, 1H), 7.67 (t, J = 7.9 Hz, 1H), 7.11 (d, J = 8.2 Hz, 1H), 3.18 (s, 6H) (Supplemental material, Figure S1); 13C NMR (101 MHz, CDCl3): δ 161.69, 160.74, 157.70, 134.99, 133.25, 132.94, 132.87, 125.15, 124.90, 119.28, 113.37, 109.73, 44.73 (Supplemental material, Figure S2); FTIR (KBr, cm−1): 2958, 2871 (CH3), 2925, 2854 (CH2), 2797 (N-CH3), 1683, 1640 (C=O stretch), 1584, 1521, 1495 (NA and benzene ring) (Supplemental material, Figure S3); HRMS (ESI): m/z 242.0812 [M + H]+ (Supplemental material, Figure S4).
Synthesis of PDSNA
In a 100-mL three-neck flask, 3-(diethoxy-methyl-silanyl)-propylamine (2.30 g, 12 mmol), DMANA (2.41 g, 10 mmol), and ethanol (50 mL) were added, and the mixture was stirred at reflux under N2 for 6 h. Upon reaction completion, the solvent was removed by rotary evaporation, and the yellow solid was purified by silica gel column chromatography using a mixture of DCM/ethyl acetate (40:1) as eluent to give the product (3.11 g, 75% yield). 1H NMR (400 MHz, CDCl3): δ 8.55 (d, J = 7.3 Hz, 1H), 8.46 (d, J = 8.2 Hz, 1H), 8.42 (d, J = 9.0 Hz, 1H), 7.64 (t, J = 8.0 Hz, 1H), 7.10 (d, J = 8.2 Hz, 1H), 4.14 (t, J = 7.8 Hz, 2H), 3.74 (q, J = 7.0 Hz, 4H), 3.09 (s, 6H), 1.78 (dt, J = 16.0, 8.1 Hz, 2H), 1.19 (t, J = 7.0 Hz, 6H), 0.73 (t, J = 7.2 Hz, 2H), 0.11 (s, 3H) (Supplemental material, Figure S5); 13C NMR (101 MHz, CDCl3): δ 164.50, 163.97, 156.86, 132.55, 131.09, 130.91, 130.17, 125.22, 124.84, 123.07, 114.97, 113.26, 58.15, 44.78, 42.77, 21.66, 18.45, 11.33, −4.86 (Supplemental material, Figure S6); FTIR (KBr, cm−1): 2969, 2876 (CH3), 2795 (N-CH3), 1693, 1654 (C=O stretch), 1587, 1516, 1484 (NA and benzene ring), 1102, 1074 (Si-OC2H5) (Supplemental material, Figure S7); HRMS (ESI): m/z 437.1862 [M + Na]+ (Supplemental material, Figure S8)
Synthesis of BDNA
To replace 3-(diethoxy-methyl-silanyl)-propylamine with N-butylamine (2.19 g, 30 mmol), DMANA (2.41 g, 10 mmol) and ethanol (50 mL) were added in a 100-mL round-bottom flask. And the mixture was stirred at reflux for 6 h. Upon reaction completion, the solvent was removed by rotary evaporation, and the yellow solid was washed with deionized water three times. Yellow solid product was given (2.90 g, 99% yield). 1H NMR (400 MHz, CDCl3): δ 8.54 (d, J = 7.2 Hz, 1H), 8.44 (d, J = 8.2 Hz, 1H), 8.40 (d, J = 8.5 Hz, 1H), 7.63 (t, J = 7.9 Hz, 1H), 7.09 (d, J = 8.4 Hz, 1H), 4.14 (t, J = 7.7 Hz, 2H), 3.07 (s, 6H), 1.69 (p, J = 8.2 Hz, 2H), 1.42 (h, J = 7.5 Hz, 2H), 0.95 (t, J = 7.2 Hz, 3H) (Supplemental material, Figure S9); 13C NMR (101 MHz, CDCl3): δ 164.69, 164.16, 156.94, 132.63, 131.13, 131.02, 130.27, 125.37, 124.97, 123.23, 115.21, 113.41, 44.87, 40.11, 30.36, 20.50, 13.97 (Supplemental material, Figure S10); FTIR (KBr, cm−1): 2955, 2868 (CH3), 2797 (N-CH3), 1770, 1723 (C=O stretch), 1582, 1522, 1491 (NA and benzene ring) (Supplemental material, Figure S11); HRMS (ESI): m/z 297.1593 [M + H]+ (Supplemental material, Figure S12).
Preparation of CYF-SO
In a 100-mL flask, a pre-determined dosage of PDSNA, 10.00 g of PDMS-OH, 50 mL of DCM, and two drops of dibutyltin dilaurate were added, and the mixture was stirred at reflux under N2 for 6 h. Upon reaction completion, the solvent was removed by rotary evaporation and a yellow oily liquid was obtained.
Two series of CYF-SO were synthesized: Fixed viscosity of PDMS-OH at 30 cP and changing dosage of PDSNA at 0.25, 0.50, 0.75, and 1.00 g to obtain the first series CYF-SO (1)–(4) with different dye concentrations. Fixed dosage of PDSNA at 0.10 g and changing viscosity of PDMS-OH at 500, 2000, 5000, and 8000 cP to prepare the second series CYF-SO (a)–(d) with different PDMS-OH viscosities.
Preparation of CYF-SRs
To prepare CYF-SRs, 2.08 g of PDMS-OH (viscosity 30 cP) and 2.08 g of CYF-SO (2) were added to 7.92 g of PDMS-OH (viscosity 2000 cP) separately, followed by the addition of 0.10 g of dibutyltin dilaurate and 0.20 g of TEOS, and the mixture was stirred and put into the mold at 60 °C for 12 h. The mold was kept in room temperature for another 12 h. By demolding, SR and CYF-SR can be obtained.
As a control experiment, NCYF-SR was prepared by replacing PDSNA with the same number of moles of BDNA, using CYF-SR preparation formulation.
Supplemental Material
Electronic_Supplementary_Information – Supplemental material for Synthesis and characterization of a novel, reactive, yellow fluorescent organosilicon dye and its polysiloxanes
Supplemental material, Electronic_Supplementary_Information for Synthesis and characterization of a novel, reactive, yellow fluorescent organosilicon dye and its polysiloxanes by Bo Ning, Jun-Wen Cao, Xiao-Teng Zhou, He Qin, Ling-Xiao Li and Cheng-You Kan in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Key Research and Development Program of China (2017YFD0200704).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
