Abstract
The reactive zwitterionic intermediates, generated from addition of isoquinoline to alkyl propiolates, react with thiazolidin-2,4-dione to afford 3-[1-(2,4-dioxothiazolidin-3-yl)isoquinolin-2(1H)-yl]acrylates in good yields. Using (Z)-5-arylidenethiazolidine-2,4-diones as the NH-acidic component leads to 3-{1-[(Z)-5-benzylidene-2,4-dioxothiazolidin-3-yl] isoquinolin-2(1H)-yl}acrylates in moderate to good yields in the absence of any catalysts under mild reaction conditions. The reaction between quinoline, ethyl propiolate, and thiazolidin-2,4-dione leads to ethyl 3-[2-(2,4-dioxothiazolidin-3-yl) quinolin-1(2H)-yl]acrylate.
Keywords
Introduction
The prominence of 1,2-dihydroisoquinoline as a basic scaffold in many natural products and biologically active molecules1–3 has promoted considerable efforts towards their synthesis.4–8 Significant efforts continue to be given to the development of novel 1,2-dihydroisoquinoline-based structures and new methods for their construction. For instance, recently it was reported that 1,2-dihydroisoquinoline skeletons could be obtained through the direct addition of various carbon pronucleophiles to ortho-alkynylaryl aldimines catalyzed by a Lewis acid.4,5 This scaffold could also be generated from isoquinolines via a multicomponent reaction.6–12
The rich chemistry that stems from the addition of nucleophiles to activated acetylenic compounds has evoked considerable interest. Aromatic N-heterocycles are known to form zwitterions with activated acetylenes such as dimethyl acetylenedicarboxylate (DMAD). 13 It has been shown that these types of zwitterions can be trapped by a variety of organic acids.14–17 In continuation of current interest in the development of new routes to heterocyclic and carbocyclic systems,18–26 we became interested in developing novel and efficient methods to construct the new 1,2-dihydroisoquinoline-based structures, with a hope of finding active hits for our particular biological assays. Herein, we report a synthesis of 1,2-dihydroisoquinolines via the three-component reaction of isoquinoline, alkyl propiolates, and CH-acids, under mild reaction conditions.
Results and discussion
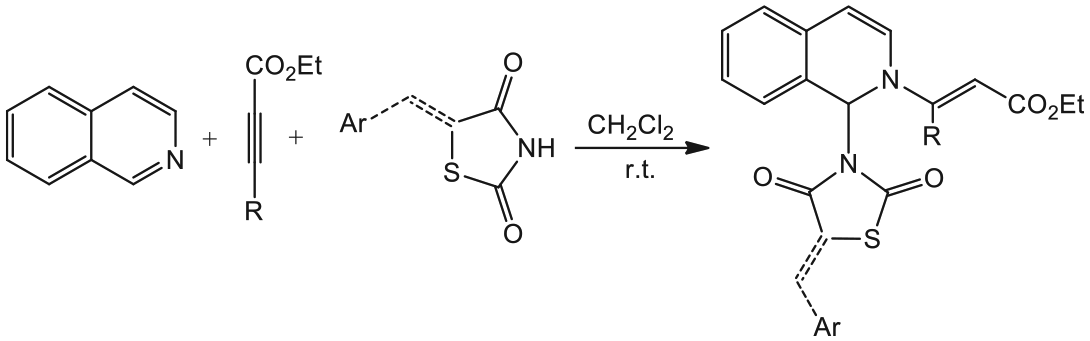
The reaction of isoquinoline (

The products were characterized on the basis of their elemental analyses and their IR, 1H NMR, and 13C NMR spectra. The mass spectra of these compounds exhibited molecular ion peaks at the appropriate m/z values. The 1H NMR spectra of compounds
Using (Z)-5-arylidenethiazolidine-2,4-diones

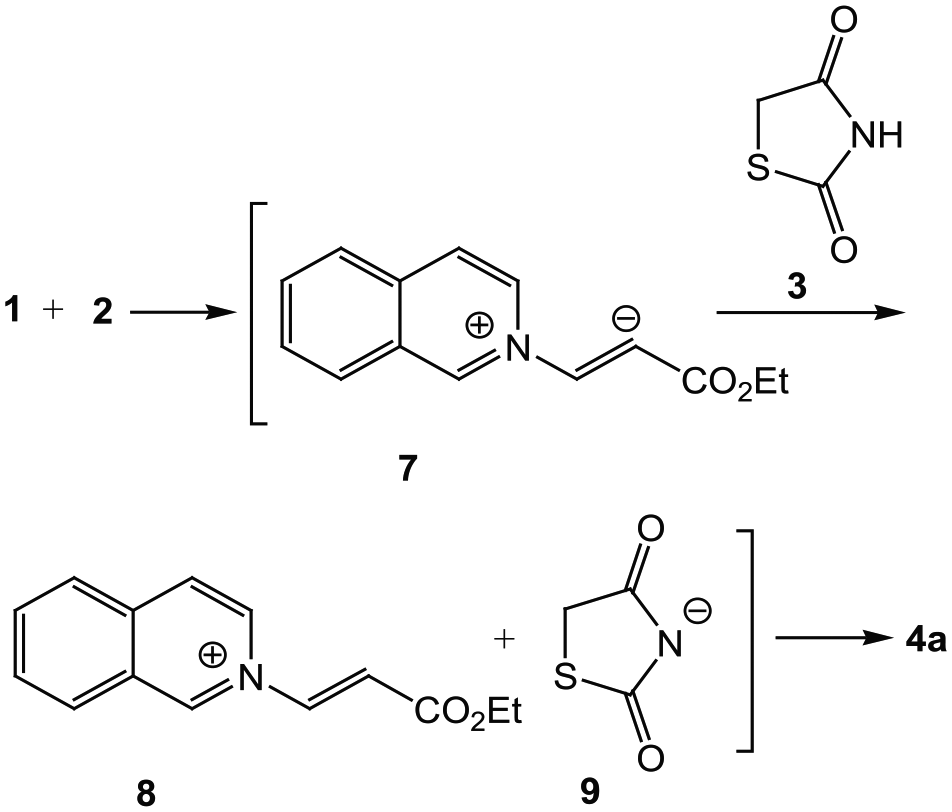
Mechanistically, it is conceivable that the reaction involves initial formation of a 1:1 zwitterionic intermediate
To extend our knowledge of these transformations, we performed the reaction of quinoline

Conclusion
In conclusion, we have developed a convenient route to functionalized [isoquinolin-2(1H)-yl]acrylates from isoquinoline and activated acetylenes in the presence of thiazolidin-2,4-dione or (Z)-5-arylidenethiazolidine-2, 4-diones. Using quinoline produced quinolin-1(2H)-yl]acrylate
Experimental
All of the chemicals used in this study were purchased from Merck and were used without further purification. Melting points were measured on an Electrothermal 9100 apparatus. Elemental analyses for C, H and were performed using a HeraeusCHN-O-Rapid analyzer. UV spectra were recorded on a Shimadzu UV-160A spectrometer. 1H and 13C NMR spectra (CDCl3) were measured with a Bruker DRX-300 Avance spectrometer. IR spectra were recorded on a Shimadzu IR-460 spectrometer. Mass spectra were recorded on a Finnigan-Mat 8430 spectrometer operating at an ionization potential of 70 eV. Chromatography columns were prepared from Aldrich silica gel? (70–230 mesh).
General procedure for the preparation of compounds 4, 6 and 11
To a stirred solution of thiazolidin-2,4-dione
(2E)-Ethyl 3-[1-(2,4-dioxothiazolidin-3-yl)isoquinolin-2(1H)-yl]acrylate (4a)
Yield 0.3 g (87%), yellow crystals, mp 70–72 °C. IR (KBr, CHCl3): 1687 (C=O), 1748 (C=O) cm−1. 1H NMR (300 MHz, CDCl3): δ = 1.30 (3H, t, 3J 7.1 Hz, Me), 4.19 (2H, q, 3J 7.1 Hz, CH2O), 4.84 (1H, d, 2J 3.7 Hz, CH2S), 5.40 (1H, d, 3J 13.6 Hz, CH), 5.83 (1H, d, 3J 7.5 Hz, CH), 5.95 (1H, d, 2J 3.7 Hz, CH2S), 6.38 (1H, d, 3J 7.5 Hz, CH), 7.05 (1H, d, 3J 7.6 Hz, CH), 7.06 (1H, s, CH), 7.14 (1H, d, 3J 7.6 Hz, CH), 7.19 (1H, t, 3J 7.5 Hz, CH), 7.32 (1H, t, 3J 7.5 Hz, CH), 7.48 (1H, d, 3J 13.6 Hz, CH). 13C NMR (75 MHz, CDCl3) δ = 14.9 (Me), 35.9 (CH2S), 55.0 (CH), 60.0 (CH2O), 92.5 (CH), 107.3 (CH), 125.4 (CH), 126.0 (CH), 126.6 (CH), 127.8 (C), 128.0 (C), 129.4 (CH), 132.2 (CH), 146.3 (CH), 168.7, 172.2, 173.7, (3C=O). MS (EI, 70 eV): m/z (%) = 344 (M+, 5), 299 (25), 271 (40), 129 (85), 128 (27), 115 (100), 99 (80), 73 (43). Anal. calcd for C17H16N2O4S: C, 59.29; H, 4.68; N, 8.13. Found: C, 59.52; H, 4.61; N, 8.18%.
(2E)-Ethyl 3-[1-(2,4-dioxothiazolidin-3-yl)isoquinolin-2(1H)-yl]-3-phenylacrylate (4b)
Yield 0.4 g (85%), yellow crystals, mp 71–73 °C. IR (KBr, CHCl3): 1691 (C=O), 1739 (C=O) cm−1. 1H NMR (300 MHz, CDCl3): δ = 1.05 (3H, t, 3J 7.1 Hz, Me), 3.96 (2H, q, 3J 7.1 Hz, CH2O), 4.87 (1H, d, 2J 3.1 Hz, CH2S), 4.65 (1H, d, 2J 3.1 Hz, CH2S), 5.77 (1H, d, 3J 7.7 Hz, CH), 6.07 (1H, d, 3J 7.9 Hz, CH), 7.08 (1H, s, CH), 7.09 (1H, d, 3J 7.5 Hz, CH), 7.12 (1H, d, 3J 7.5 Hz, CH), 7.23 (1H, t, 3J 7.6 Hz, CH), 7.24 (1H, s, CH), 7.34 (1H, t, 3J 7.6 Hz, CH), 7.45–7.48 (5H, m, CH). 13C NMR (75 MHz, CDCl3) δ = 14.5 (Me), 35.9 (CH2S), 54.9 (CH), 60.0 (CH2O), 94.5 (CH), 109.3 (CH), 125.4 (CH), 126.0 (CH), 126.6 (CH), 127.8 (C), 128.0 (C), 129.0 (2CH), 129.2 (2CH), 129.5 (CH), 129.8 (CH), 132.0 (CH), 134.6 (C), 157.0 (C), 167.5, 171.2, 172.7, (3C=O). MS (EI, 70 eV): m/z (%) = 420 (M+, 8), 375 (31), 347 (42), 175 (85), 129 (87), 128 (29), 115 (100), 73 (42). Anal. calcd for C23H20N2O4S: C, 65.70; H, 4.79; N, 6.66. Found: C, 65.94; H, 4.73; N, 6.71%.
(2E)-Ethyl 3-{1-[(Z)-5-benzylidene-2,4-dioxothiazolidin-3-yl]isoquinolin-2(1H)-yl}acrylate (6a)
Yield 0.32 g (75%), yellow crystals, mp 73–75 °C. IR (KBr, CHCl3): 1676 (C=O), 1735 (C=O) cm−1. 1H NMR (300 MHz, CDCl3): δ = 1.35 (3H, t, 3J 7.1 Hz, Me), 4.30 (2H, q, 3J 7.1 Hz, CH2O), 6.12 (1H, d, 3J 9.1 Hz, CH), 6.65 (1H, d, 3J 9.1 Hz, CH), 7.06 (1H, d, 3J 14.7 Hz, CH), 7.20 (1H, s, CH), 7.45–7.55 (9H, m, CH), 7.93 (1H, d, 3J 14.7 Hz, CH), 7.96 (1H, s, CH). 13C NMR (75 MHz, CDCl3) δ = 14.3 (Me), 56.3 (CH), 62.1 (CH2O), 94.5 (CH), 108.7 (CH), 121.4 (C), 125.3 (CH), 125.9 (CH), 126.4 (CH), 127.4 (C), 128.8 (C), 129.0 (2CH), 129.1 (2CH), 129.4 (CH), 129.8 (CH), 132.0 (CH), 134.5 (C), 135.5 (CH), 146.7 (CH), 167.9, 171.3, 172.9, (3C=O). MS (EI, 70 eV): m/z (%) = 432 (M+, 7), 387 (30), 359 (44), 342 (35), 204 (100), 129 (86), 99 (81), 90 (24), 73 (46). Anal. calcd for C24H20N2O4S: C, 66.65; H, 4.66; N, 6.48. Found: 66.68; H, 4.63; N, 6.50%.
(E)-Ethyl 3-{1-[(Z)-5-(3-methylbenzylidene)-2,4-dioxothiazolidin-3-yl]isoquinolin-2(1H)-yl}acrylate (6b)
Yield 0.31 g (71%), yellow crystals, mp 84–86 °C. IR (KBr, CHCl3): 1682 (C=O), 1748 (C=O) cm−1. 1H NMR (300 MHz, CDCl3): δ = 1.33 (3H, t, 3J 7.1 Hz, Me), 2.43 (3H, s, Me), 4.31 (2H, q, 3J 7.1 Hz, CH2O), 6.11 (1H, d, 3J 9.1 Hz, CH), 6.63 (1H, d, 3J 9.1 Hz, CH), 7.06 (1H, d, 3J 14.7 Hz, CH), 7.22 (1H, s, CH), 7.27–7.45 (8H, m, CH), 7.93 (1H, d, 3J 14.7 Hz, CH), 7.95 (1H, s, CH). (75 MHz, CDCl3) δ = 14.4 (Me), 21.8 (Me), 57.1 (CH), 62.3 (CH2O), 96.4 (CH), 108.9 (CH), 121.4 (C), 125.5 (CH), 125.8 (CH), 126.6 (CH), 127.5 (C), 128.7 (C), 129.0 (2CH), 129.2 (2CH), 129.8 (CH), 132.0 (CH), 134.5 (C), 135.5 (CH), 136.3 (C), 147.0 (CH), 167.5, 170.9, 171.8, (3C=O). MS (EI, 70 eV): m/z (%) = 446 (M+, 5), 341 (12), 268 (16), 197 (16), 170 (100), 148 (36), 129 (27), 97 (36), 57 (54), 43 (45). Anal. calcd for C25H22N2O4S: C, 67.25; H, 4.97; N, 6.27. Found: C, 67.00; H, 4.93; N, 6.31%.
(E)-Ethyl 3-{1-[(Z)-5-(4-methylbenzylidene)-2,4-dioxothiazolidin-3-yl]isoquinolin-2(1H)-yl}acrylate (6c)
Yield 0.36 g (85%), yellow crystals, mp 85–87 °C. IR (KBr, CHCl3): 1673 (C=O), 1738 (C=O) cm−1. 1H NMR (300 MHz, CDCl3): δ = 1.38 (3H, t, 3J 7.1 Hz, Me), 2.43 (3H, s, Me), 4.27 (2H, q, 3J 7.1 Hz, CH2O), 6.03 (1H, d, 3J 14.7 Hz, CH), 6.60 (1H, d, 3J 7.5 Hz, CH), 7.30 (1H, s, CH), 7.31 (2H, d, 3J 7.5 Hz, CH), 7.41 (2H, d, 3J 7.5 Hz, CH), 7.55 (1H, t, 3J 7.9 Hz, CH), 7.70 (1H, t, 3J 7.5 Hz, CH), 7.85 (1H, s, CH), 8.18–8.21 (2H, m, CH), 8.45 (1H, d, 3J 7.5 Hz, CH), 8.74 (1H, d, 3J 14.7 Hz, CH). 13C NMR (75 MHz, CDCl3) δ = 14.3 (Me), 22.0 (Me), 58.3 (CH), 62.2 (CH2O), 96.5 (CH), 108.8 (CH), 121.4 (C), 125.5 (CH), 125.8 (CH), 126.7 (CH), 127.6 (C), 128.9 (C), 129.0 (2CH), 129.2 (2CH), 129.8 (CH), 132.7 (CH), 134.7 (C), 135.6 (CH), 136.5 (C), 146.5 (CH), 168.9, 172.5, 173.3, (3C=O). MS (EI, 70 eV): m/z (%) = 446 (M+, 5), 341 (12), 268 (16), 197 (16), 170 (100), 148 (36), 129 (27), 97 (36), 57 (54), 43 (45). Anal. calcd for C25H22N2O4S: C, 67.25; H, 4.97; N, 6.27. Found: C, 67.03; H, 4.93; N, 6.31%.
(2E)-Ethyl 3-{1-[(Z)-5-(4-nitrobenzylidene)-2,4-dioxothiazolidin-3-yl]isoquinolin-2(1H)-yl}acrylate (6d)
Yield 0.32 g (72%), yellow crystals, mp 96–98 °C. IR (KBr, CHCl3): 1671 (C=O), 1737 (C=O) cm−1. 1H NMR (300 MHz, CDCl3): δ = 1.38 (3H, t, 3J 7.1 Hz, Me), 4.43 (2H, q, 3J 7.1 Hz, CH2O), 6.96 (1H, d, 3J 7.2 Hz, CH), 7.05 (1H, d, 3J 14.7 Hz, CH), 7.31 (1H, s, CH), 7.51 (1H, t, 3J 7.5 Hz, CH), 7.57–7.70 (3H, m, CH), 7.94 (1H, d, 3J 14.7 Hz, CH), 7.74 (1H, s, CH), 8.05 (2H, d, 3J 8.3 Hz, 2CH), 8.37 (2H, d, 3J 8.8 Hz, 2CH), 8.68 (1H, d, 3J 7.2 Hz, CH). 13C NMR (75 MHz, CDCl3) δ = 14.3 (Me), 58.3 (CH), 62.2 (CH2O), 96.5 (CH), 108.8 (CH), 121.4 (C), 125.5 (CH), 125.8 (CH), 126.8 (CH), 127.7 (C), 128.9 (C), 129.0 (2CH), 129.5 (2CH), 129.9 (CH), 132.7 (CH), 134.7 (C), 139.7 (CH), 144.5 (C), 146.5 (CH), 168.9, 172.5, 173.3, (3C=O). MS (EI, 70 eV): m/z (%) = 477 (M+, 6), 432 (28), 404 (28), 342 (35), 249 (100), 135 (29), 129 (83), 99 (89), 73 (39). Anal. calcd for C24H19N3O6S: C, 60.37; H, 4.01; N, 8.80. Found: C, 60.31; H, 4.09; N 8.75%.
(2E)-Ethyl 3-{1-[(Z)-2,4-dioxo-5-((thiophen-2-yl)methylene)thiazolidin-3-yl]isoquinolin-2(1H) yl}acrylate (6e)
Yield 0.28 g (70%), yellow crystals, mp 73–75 °C. IR (KBr, CHCl3): 1689 (C=O), 1735 (C=O) cm−1. 1H NMR (300 MHz, CDCl3): δ = 1.36 (3H, t, 3J 7.1 Hz, Me), 4.29 (2H, q, 3J 7.1 Hz, CH2O), 6.12 (1H, d, 3J 9.1 Hz, CH), 6.63 (1H, d, 3J 9.1 Hz, CH), 7.05 (1H, d, 3J 14.7 Hz, CH), 7.29 (1H, s, CH), 7.45–7.55 (7H, m, CH), 7.92 (1H, d, 3J 14.7 Hz, CH), 7.96 (1H, s, CH). 13C NMR (75 MHz, CDCl3) δ = 14.3 (Me), 56.3 (CH), 62.1 (CH2O), 94.5 (CH), 108.7 (CH), 121.4 (C), 125.3 (CH), 125.9 (CH), 126.4 (CH), 127.4 (C), 128.8 (C), 129.0 (CH), 129.1 (CH), 129.4 (CH), 130.5 (CH), 132.9 (CH), 134.3 (CH), 137.5 (C), 148.5 (CH), 167.9, 171.3, 172.9, (3C=O). MS (EI, 70 eV): m/z (%) = 438 (M+, 8), 393 (29), 365 (42), 342 (34), 209 (100), 129 (89), 99 (88), 96 (31), 73 (38). Anal. calcd for C22H18N2O4S2: 60.26; H, 4.14; N, 6.39. Found: C, 60.33; H, 4.07; N, 6.41%.
(2E)-Ethyl 3-[2-(2,4-dioxothiazolidin-3-yl)quinolin-1(2H)-yl]acrylate (11)
Yield 0.22 g (65%), yellow crystals, mp 86–88 °C. IR (KBr, CHCl3): 1685 (C=O), 1736 (C=O) cm−1. 1H NMR (300 MHz, CDCl3): δ = 1.33 (3H, t, 3J 7.1 Hz, Me), 4.22 (2H, q, 3J 7.1 Hz, CH2O), 4.61 (1H, d, 2J 3.7 Hz, CH2S), 5.30 (1H, d, 3J 13.6 Hz, CH), 5.50 (1H, d, 2J 3.7 Hz, CH2S), 5.99 (1H, dd, 3J 9.9 Hz, 3J 4.3 Hz, CH), 6.80 (1H, d, 3J 4.3 Hz, CH), 6.89 (1H, d, 3J 9.8 Hz, CH), 7.05 (1H, d, 3J 7.6 Hz, CH), 7.14 (1H, d, 3J 7.6 Hz, CH), 7.19 (1H, t, 3J 7.5 Hz, CH), 7.32 (1H, t, 3J 7.5 Hz, CH), 7.48 (1H, d, 3J 13.6 Hz, CH). 13C NMR (75 MHz, CDCl3) δ = 14.9 (Me), 35.9 (CH2S), 55.0 (CH), 60.0 (CH2O), 92.5 (CH), 107.3 (CH), 122.4 (CH), 123.0 (CH), 124.6 (CH), 129.3 (C), 129.1 (C), 130.1 (CH), 135.7 (CH), 149.3 (CH), 168.7, 172.2, 173.7, (3C=O). MS (EI, 70 eV): m/z (%) = 344 (M+, 5), 299 (25), 271 (40), 129 (85), 128 (27), 115 (100), 99 (80), 73 (43). Anal. calcd for C17H16N2O4S: C, 59.29; H, 4.68; N, 8.13. Found: C, 59.32; H, 4.61; N 8.05%.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
