Abstract
An efficient method is developed for the synthesis of 4-(3-arylprop-2-ynyloxy)-6-methyl-2-(methylthio)pyrimidines via palladium-catalyzed Sonogashira reactions of 4-methyl-2-(methylthio)-6-(prop-2-yn-1-yloxy)pyrimidine with electron-poor aryl iodides in acetonitrile at room temperature. Excellent yields of the products were obtained in reaction times of 9–11 h.
Introduction
Heterocycles bearing a pyrimidine moiety are reported to show a broad spectrum of pharmacological properties such as antibacterial, 1 anti-HIV, 2 and anticancer. 3 In this regard, antitumor and antiviral are two of the most extensively reported activities of pyrimidine analogs. 4 Moreover, they possess insecticidal and herbicidal activities.5–8
Transition-metal-mediated cross-coupling reactions have proven to be powerful tools for mild and highly efficient carbon–carbon bond formation.9–11 Palladium (Pd)-catalyzed cross-coupling reactions are among the most versatile processes for carbon–carbon bond formation. Among these reactions, the coupling reactions of terminal alkynes with aryl and vinyl halides (Sonogashira reactions) play important roles in modern synthetic chemistry.12,13 This reaction is one of the most widely used C–C bond-forming reactions,14,15 furnishing an impressive route to aryl alkynes, which are interesting intermediates for the preparation of a variety of natural products,16–19 pharmaceuticals, 20 and organic materials.21,22
Accordingly, and based on our progressive endeavors in developing Pd-catalyzed reactions of acetylenes leading to heterocyclic compounds of biological significance via environmentally benign synthetic methodologies,23–27 herein we report the synthesis of new derivatives of 4-(3-arylprop-2-ynyloxy)-6-methyl-2-(methylthio)pyrimidine via the Pd-catalyzed Sonogashira coupling reactions of 4-methyl-2-(methylthio)-6-(prop-2-ynyloxy)pyrimidine with aryl iodides.
Results and discussion
Treatment of 6-methyl-2-(methylthio)pyrimidine-4(3H)-one (
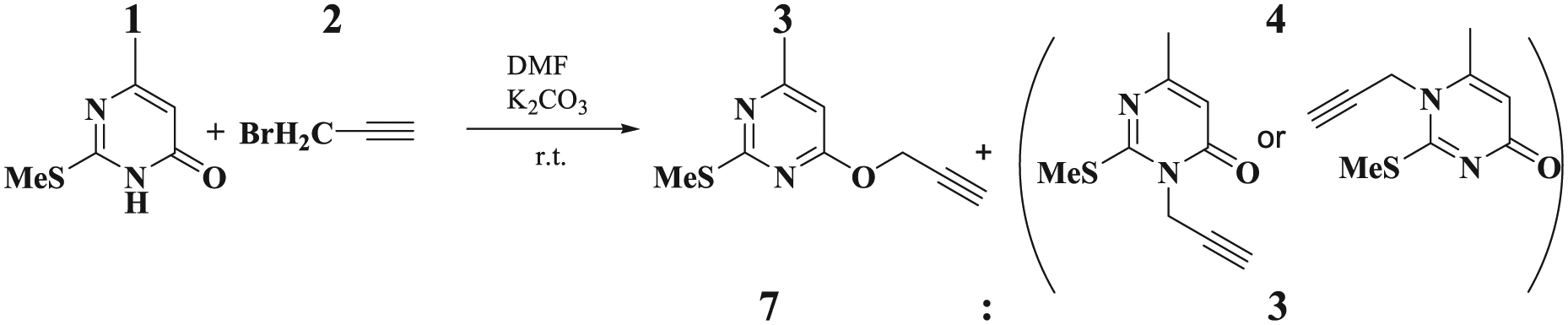
Alkylation of pyrimidine (
Structures (

FTIR spectra for (a) compound (
The 1H NMR spectrum for compound (
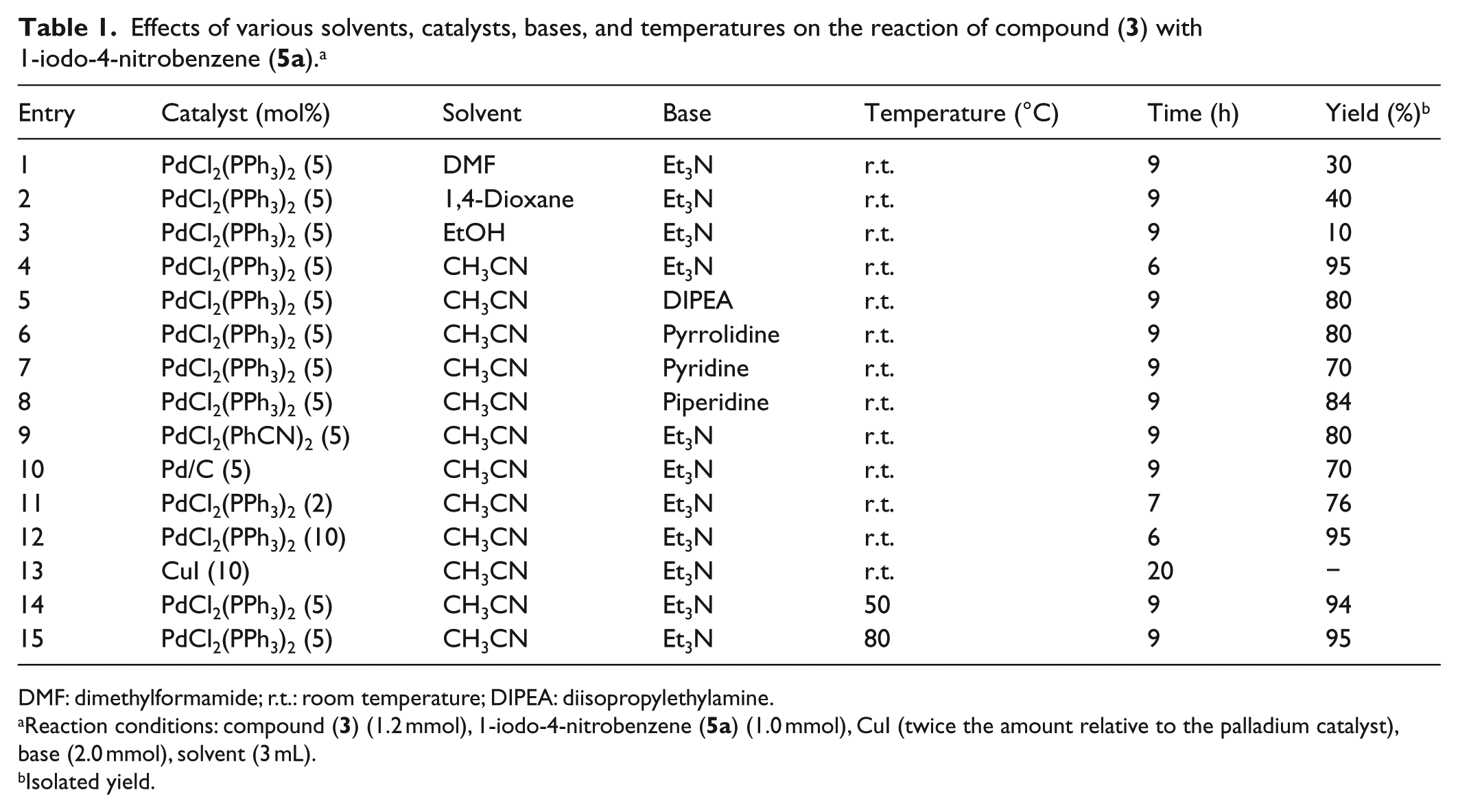
For optimization of the reaction conditions, we chose the reaction of compound (
Effects of various solvents, catalysts, bases, and temperatures on the reaction of compound (
DMF: dimethylformamide; r.t.: room temperature; DIPEA: diisopropylethylamine.
Reaction conditions: compound (
Isolated yield.
The reactions were performed using different solvents such as DMF, CH3CN, EtOH, and 1,4-dioxane in the presence of Et3N as the base at room temperature. As can be seen, the highest reaction yield was obtained when CH3CN was used as the solvent (Table 1, entry 4). Moreover, other parameters such as the base and reaction temperature were also investigated. When organic bases such as Et3N, diisopropylethylamine, pyrrolidine, pyridine, and piperidine were used (Table 1, entries 4–8), the best results were obtained in the case of Et3N (Table 1, entry 4). Also, several catalytic palladium sources such as Pd/C, Pd(PhCN)2Cl2, and PdCl2(PPh3)2 were screened in CH3CN. It was found that PdCl2(PPh3)2 was the best catalyst for this reaction condition (Table 1, entry 4). The conditions with respect to the amount of catalyst were optimized. Decreasing the loading of the catalyst to 2.0 mol% lowered the reaction yield dramatically (Table 1, entry 11). However, increasing the amount of catalyst to 10 mol% only shortened the reaction time and did not have any effect on the reaction yield (Table 2, entry 12). No Sonogashira coupling reaction was observed when CuI alone was used as the catalyst (Table 1, entry 13). Furthermore, the effect of the temperature on the reaction yield was investigated. As shown in Table 1, increasing the temperature did not improve the reaction yield (Table 2, entries 14 and 15).
Synthesis of 4-(3-arylprop-2-ynyloxy)-6-methyl-2-(methylthio)pyrimidines (

Reaction conditions: compound (
Isolated yield.
Using the optimized reaction conditions (Table 1, entry 4), the scope of the reaction was explored with various electron-poor aryl iodides (
Mechanistic details for the formation of 4-(3-arylprop-2-ynyloxy)-6-methyl-2-(methylthio)pyrimidines (

A plausible mechanism for formation of 4-(3-arylprop-2-ynyloxy)-6-methyl-2-(methylthio)pyrimidines (
Conclusion
We have described an efficient method for the synthesis of 4-(3-arylprop-2-ynyloxy)-6-methyl-2-(methylthio)pyrimidines catalyzed by Pd–Cu through Sonogashira coupling reactions of 4-methyl-2-(methylthio)-6-(prop-2-yn-1-yloxy)pyrimidine (
Experiment
General
The reagents and solvents used were supplied from Merck, Fluka, or Aldrich. Melting points were determined using a brand electro-thermal C14500 apparatus. The reaction progress and the purity of compounds were monitored using thin-layer chromatography (TLC) using silica gel plates (Merck 60 F250). All known compounds were identified by comparing their melting points and 1H NMR data with those of authentic samples. The 1H NMR (300 MHz) and 13C NMR (75 MHz) spectra were recorded on a Bruker Avance DPX-250 FT-NMR spectrometer. The chemical shifts are given as δ values against tetramethylsilane as the internal standard, and the J values are given in Hertz. Microanalysis was performed on a PerkinElmer 240-B microanalyzer.
Synthesis of 4-methyl-2-(methylthio)-6-(prop-2-yn-1-yloxy)pyrimidine (3) and N1- or N3-propynylated pyrimidine (4 )
Propargyl bromide (
Compound (
Compound (
Synthesis of 4-(3-arylprop-2-ynyloxy)-6-methyl-2-(methylthio)pyrimidines (6a–g )
A mixture of aryl iodide (
4-Methyl-2-(methylthio)-6-{[3-(4-nitrophenyl)prop-2-yn-1-yl]oxy}pyrimidine (
4-Methyl-2-(methylthio)-6-{[3-(2-nitrophenyl)prop-2-yn-1-yl]oxy}pyrimidine (
4-Methyl-2-(methylthio)-6-{[3-(3-nitrophenyl)prop-2-yn-1-yl]oxy}pyrimidine (
4-{[3-(4-Chloro-3-nitrophenyl)prop-2-yn-1-yl]oxy}-6-methyl-2-(methylthio)pyrimidine (
4-{[3-(2-Chloro-4-nitrophenyl)prop-2-yn-1-yl]oxy}-6-methyl-2-(methylthio)pyrimidine (
4-{[3-(4-Chloro-2-nitrophenyl)prop-2-yn-1-yl]oxy}-6-methyl-2-(methylthio)pyrimidine (
4-{[3-(2-Methyl-4-nitrophenyl)prop-2-yn-1-yl]oxy}-6-methyl-2-(methylthio)pyrimidine (
Supplemental Material
supplementary_information – Supplemental material for Sonogashira coupling reactions: Synthesis of 4-substituted-6-methyl-2-(methylthio)pyrimidines catalyzed by Pd–Cu
Supplemental material, supplementary_information for Sonogashira coupling reactions: Synthesis of 4-substituted-6-methyl-2-(methylthio)pyrimidines catalyzed by Pd–Cu by Fatemeh Rezaeimanesh, Mohammad Bakherad and Hossein Nasr-Isfahani in Journal of Chemical Research
Footnotes
Acknowledgements
The authors gratefully acknowledge the support of the Research Council of the Shahrood University of Technology.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Research Council of the Shahrood University of Technology.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

