Abstract
Convenient syntheses of quinazoline, benzothiazole, thiadiazole, imidazole, and thiourea derivatives starting from 3-(thiophen-2-yl)prop-2-enoyl isothiocyanate are described. The structures of the synthesized compounds are confirmed from their microanalytical and spectral data. Some of the products are examined for their antibacterial activity against Gram-positive and Gram-negative bacteria and fungi.
Keywords
Introduction
Extensive studies on the chemistry of aroyl isothiocyanates have established the value of these reagents as starting materials for the syntheses of a wide variety of heterocyclic compounds and thiourea derivatives.1–11 Highly reactive α,β-unsaturated acyl isothiocyanates can be useful intermediates in organic synthesis, because their multiple bonds can participate in cyclization reactions. 12 In addition, intramolecular cyclization of α,β-unsaturated acylthioureas, formed in the reaction of α,β-unsaturated acyl isothiocyanates and amines, have been reported.13–16 In this investigation, we utilized a heterocyclic α,β-unsaturated acyl isothiocyanate in order to synthesize several heterocyclic compounds expected to demonstrate antibacterial activity against Gram-positive and Gram-negative bacteria and fungi.
Results and discussion
In the current study, transformations of 3-(thiophen-2-yl)prop-2-enoyl isothiocyanate (
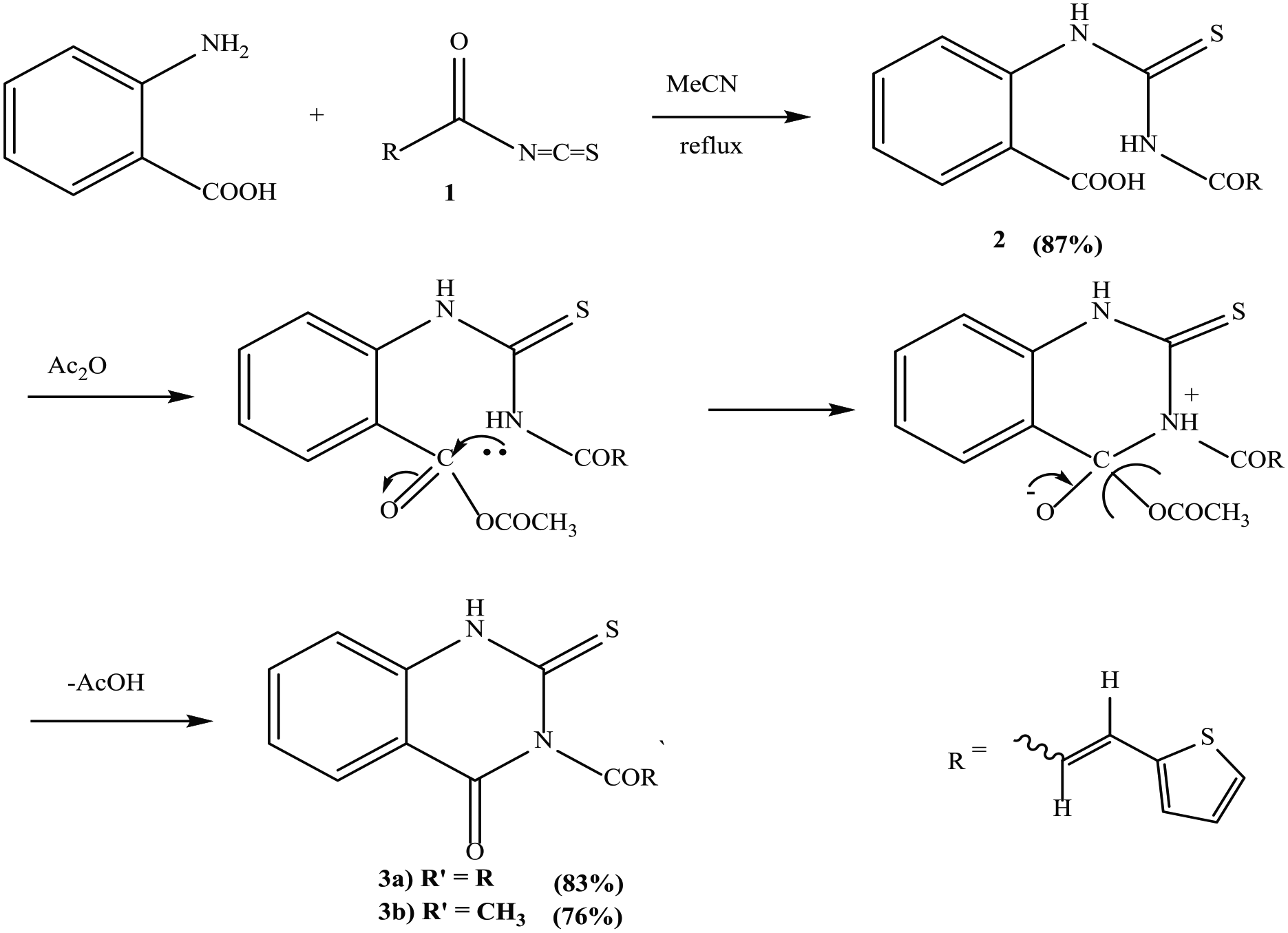
Synthesis of quinazolines
Treatment of isothiocyanate

Reactions of isothiocyanate
The interaction of isothiocyanate
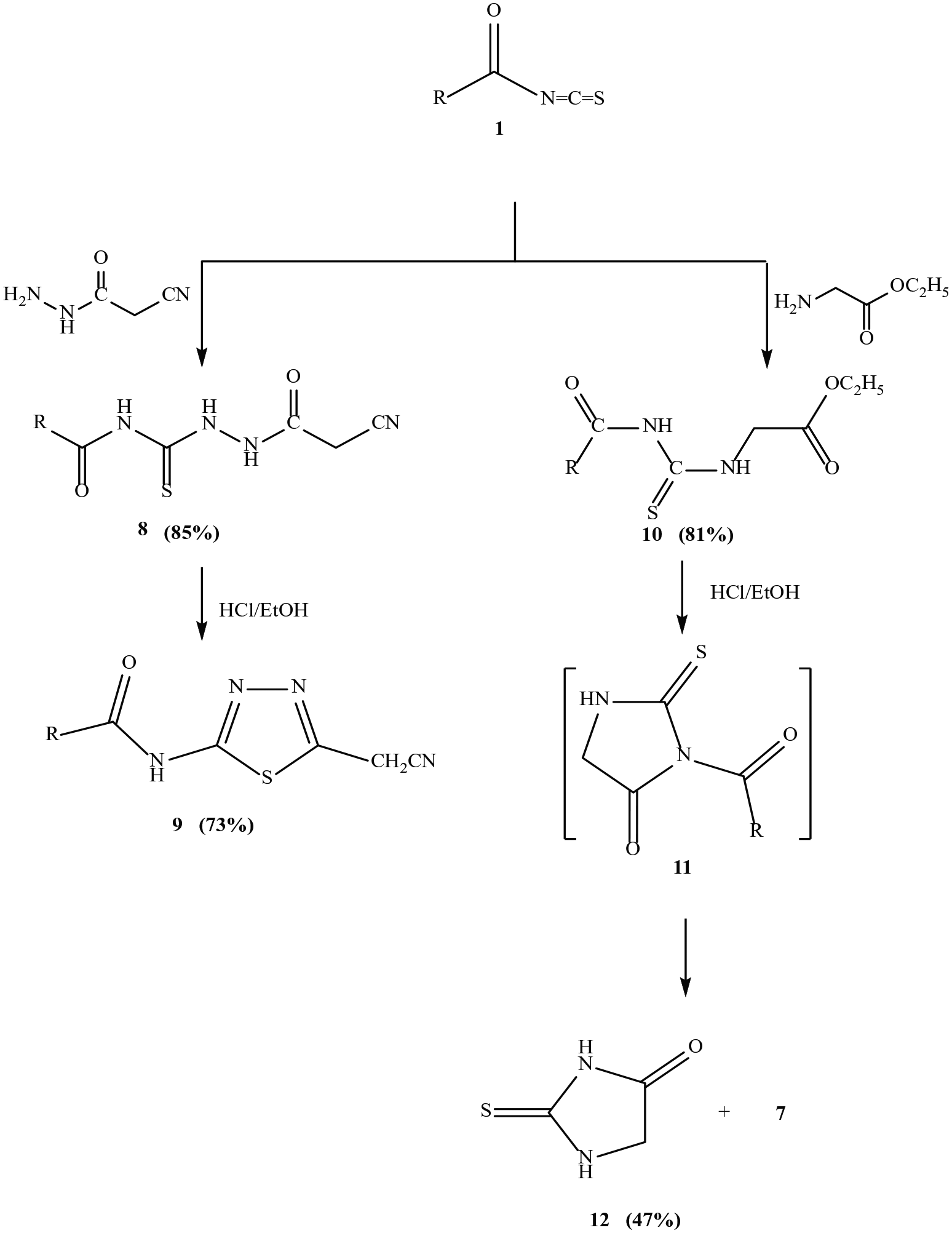
Reactions of isothiocyanate
Refluxing a solution of the isothiocyanate

Formation of compounds
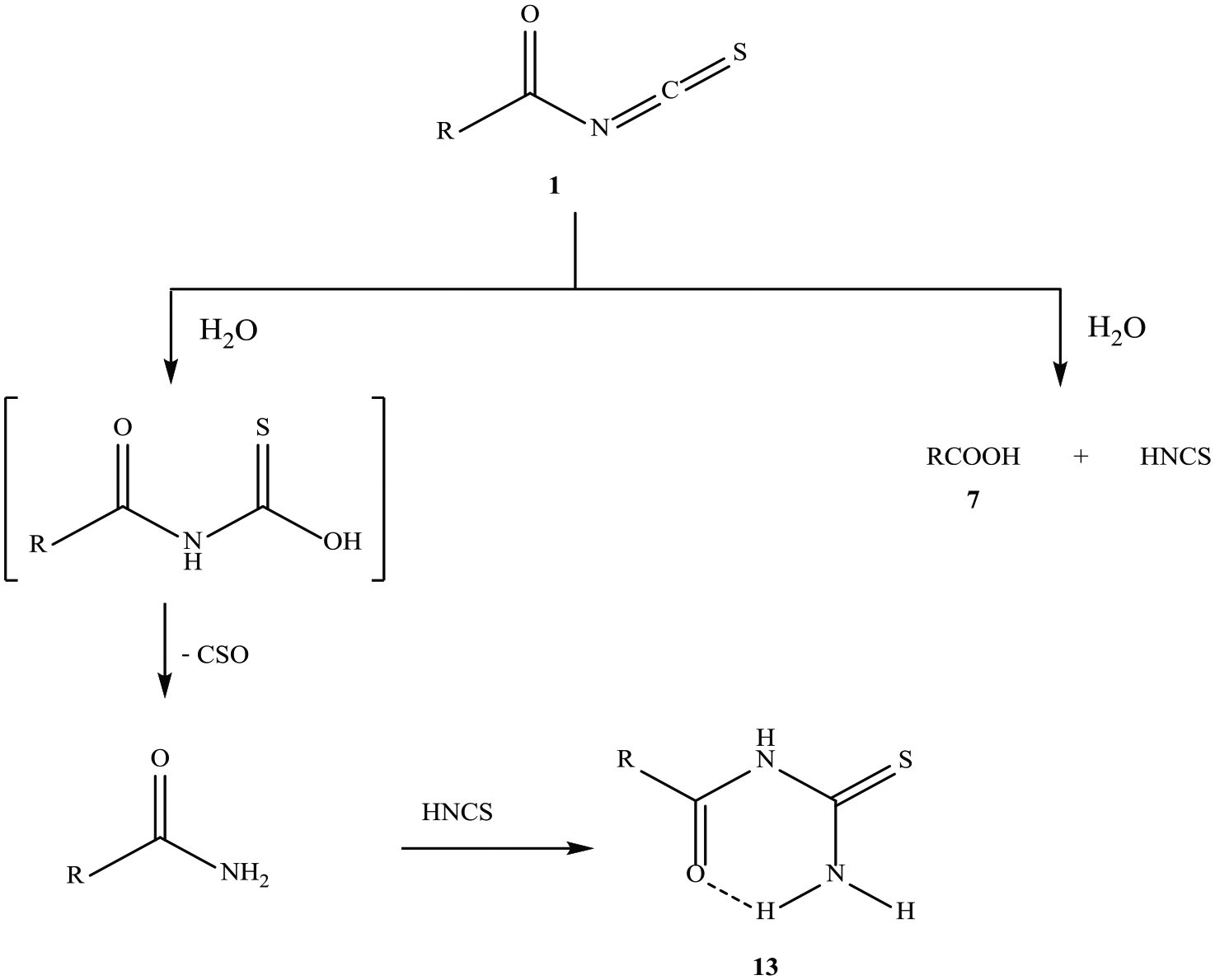
The mechanism of formation of compound
The mechanism of formation of compound
Antimicrobial activity
The prepared heterocyclic compounds
Antimicrobial Activity Data.


Graphical Representation of Antimicrobial Activity.
Experimental analysis
Melting points were measured on an electrothermal melting point apparatus and are uncorrected. The elemental analyses were recorded on a Perkin-Elemer 2400 CHN elemental analyzer (USA). The IR spectra were recorded on a FTIR Maltson (infinity series) spectrophotometer LABX (Canada) as KBr disks. The 1H NMR spectra were measured using a Varian Gemini 300-MHz spectrometer LABX (Canada), with chemical shifts (δ) expressed in ppm downfield from tetramethylsilane (TMS) as the internal standard, in DMSO-d6. Mass spectra were determined on a Shimadzu GC-MSQP 1000 EX instrument (Japan) operating at 70 eV. Thin-layer chromatography (TLC) was run using TLC aluminum sheets coated with silica gel F254 (Merck, England) to monitor the progress of all reactions and the homogeneity of the synthesized compounds. 3-(Thiophen-2-yl)prop-2-enoyl isothiocyanate (
Reactions of isothiocyanate (1 ) with the different nucleophiles; general procedure
To a solution of isothiocyanate
(E, Z)-2-(3-(3-(thiophen-2-yl)acryloyl)thioureido)benzoic acid (
(E)-N-(3,1-benzothiazol-2-yl)-3-(thiophen-2-yl)acrylamide (
(E)-1-(2-hydroxyphenyl)-3-(3-(thiophen-2-yl)acryloyl)thiourea (
(E)-1-(2-aminophenyl)-3-(3-(thiophen-2-yl)acryloyl)thiourea (
(E)-1-(2-cyanoacetyl)-4-(3-(thiophen-2-yl)acryloyl)thiosemicarbazide (
(E)-ethyl 2-(3-(3-(thiophen-2-yl)acryloyl)thioureido)acetate (
(E/Z)-1-(3-(thiophen-2-yl)acryloyl)thiourea (
(E)-1-(piperidin-1-yl)-3-(thiophen-2-yl)prop-2-en-1-one (
Formation of compounds 3a and 3b
A solution of compound
(E)-3-(3-(thiophen-2-yl)acryloyl)-2-thioxo-2,3-dihydroquinazolin-4(1H)-one (
3-acetyl-2-thioxo-2,3-dihydroquinazolin-4(1H)-one (
Formation of compounds 6 and 7
A solution of compound
2-amino-3,1-benzothiazole (
(E)-3-(thiophen-2-yl)acrylic acid (
Formation of compounds 9 and 12
A solution of compound
(E)-N-(5-(cyanomethyl)-1,3,4-thiadiazol-2-yl)-3-(thiophen-2-yl)acrylamide (
2-thioxoimidazolidin-4-one (
Microbacterial activity
The newly synthesized heterocyclic compounds listed in Table 1 were tested for their antibacterial activity against Gram-positive bacteria (Bacillus cereus and Staphylococcus aureus), Gram-negative bacteria (Escherichia coli and Pseudomonas aeruginosa), and fungi (Candida albicans and Aspergillus fumigatus) at a concentration of 100 μg/mL in dimethyl sulfoxide (DMSO). Nutrient agar and potato dextrose agars were used to culture the bacteria and fungi, respectively. The plates were inculcated by the bacteria or fungi and incubated for 24 h at 37 °C for bacteria and for 72 h at 28 °C for fungi, and then the inhibition zones of microbial growth surrounding the filter paper disk (5 mm) were measured in millimeters.
Conclusion
The synthetic utilization of α,β-unsaturated acyl isothiocyanate is based on the different reactivities of both centers toward nucleophiles. It is observed from all the reactions mentioned that the nucleophilic attack proceeds at the isothiocyanato group rather than at the α,β-unsaturated system, which reflects the higher reactivity of the former. Compounds
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
