Abstract
Reactions of 1,4,5-triaryl-2-(4-bromomethyl)phenyl-imidazoles with sodium azide in acetone give the corresponding azidomethyl derivatives, which on 1,3-dipolar cycloaddition with various terminal alkynes in the presence of CuI afford novel 1,2,3-triazole products. On the other hand, treatment of 2,4,5-triaryl-1-(4-hydroxyphenyl)-imidazoles with propargyl chloride in the presence of a base gives the corresponding propargyl ether derivatives, which under CuI-catalyzed 1,3-dipolar cycloaddition with benzyl azide produce 1,2,3-triazole derivatives. All the products are characterized from their spectroscopic data and most are evaluated for fluorescence emission. The optical parameters of the studied products are also reported.
Keywords
Introduction
1,3-Dipolar cycloaddition of azides and alkynes, as one of the most powerful click reactions, offers an efficient methodology for the synthesis of 1,2,3-triazoles.1–3 Several members of these azoles have been widely applied in diverse areas of chemistry such as medicinal and materials hemistry,4–6 and also various streams of applied sciences.7–12 The Cu(I)-catalyzed version of this coupling reaction, which was discovered by the groups of Sharpless and Meldal independently, increases the reaction rate and produces the 1,4-disubstituted triazoles regioselectively.13,14 Continuing efforts and several modifications leading to the production of highly diverse derivatives highlight the special interest in this click reaction.15–17
Polyaromatic molecules with several heterocyclic frameworks and high conjugation have attracted attention due to having high technology applications, especially in biological and optical systems.18–21 Meanwhile, there are many reports on the synthesis of hybrid molecules containing a 1,2,3-triazole moiety with other heterocyclic compounds.22,23 1,2,4,5-Tetraarylimidazole derivatives, which can be prepared by four-component condensation of aromatic aldehydes, benzil, primary amines and ammonium acetate,24,25 have been recently reported by our group26,27 to act as light-emitting backbones. According to our interest in the synthesis of new heterocyclic and polyaromatic systems containing a 1,2,3-triazole ring, 28 herein we report Cu(I)-catalyzed cycloadditions between azidomethyl derivatives of 1,2,4,5-tetraarylimidazoles and terminal alkynes to afford new 1,2,3-triazole structures possessing polyaryl imidazole scaffolds. In addition, catalytic cyclizations of propargyloxy derivatives of the prepared tetraarylimidazoles with benzyl azide to diversify the products are also reported. All of the synthesized products are evaluated for their absorption and fluorescence emission properties.
Results and discussion
The starting compounds
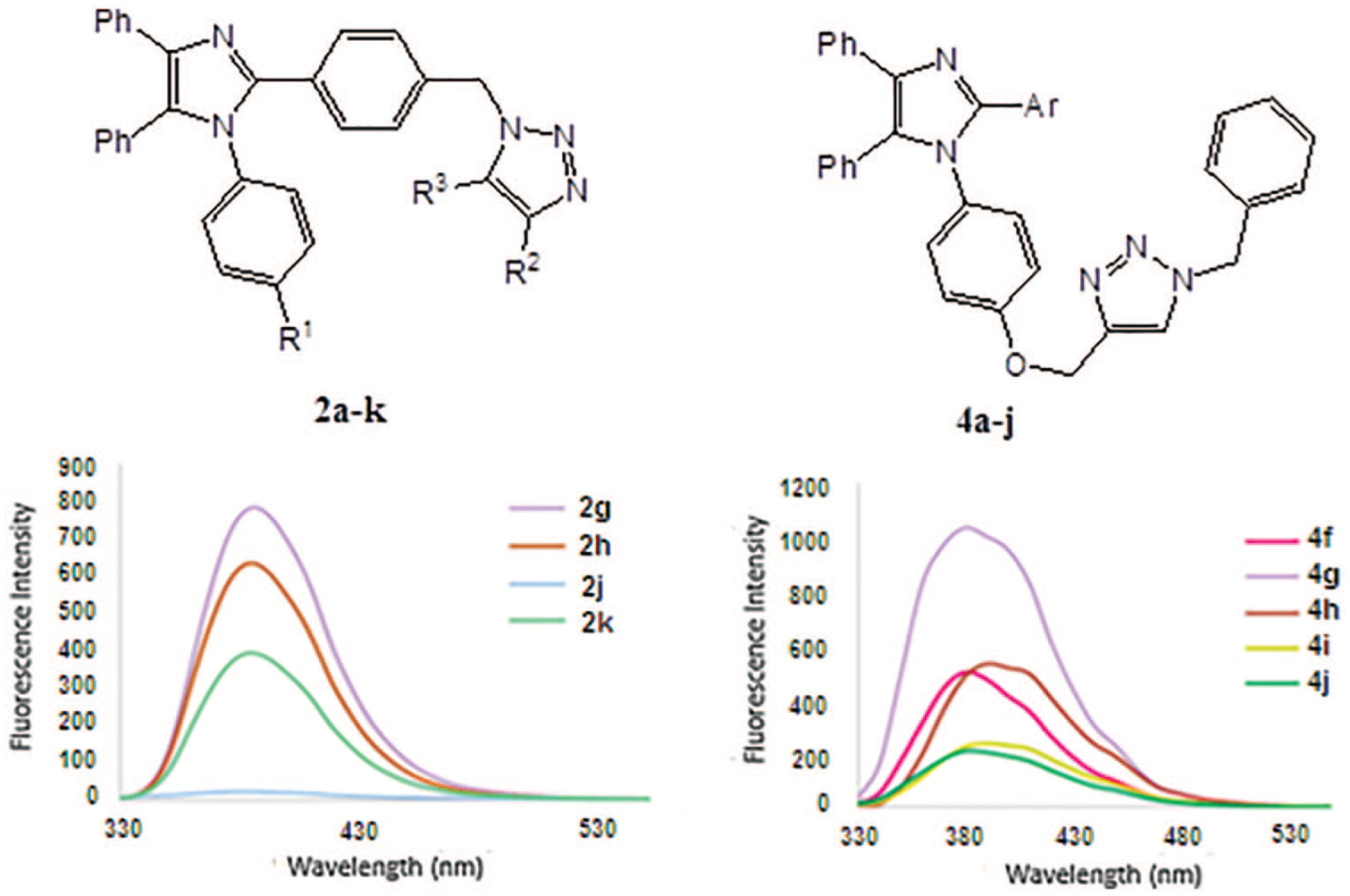
The synthesis of 1,2,3-triazole derivatives
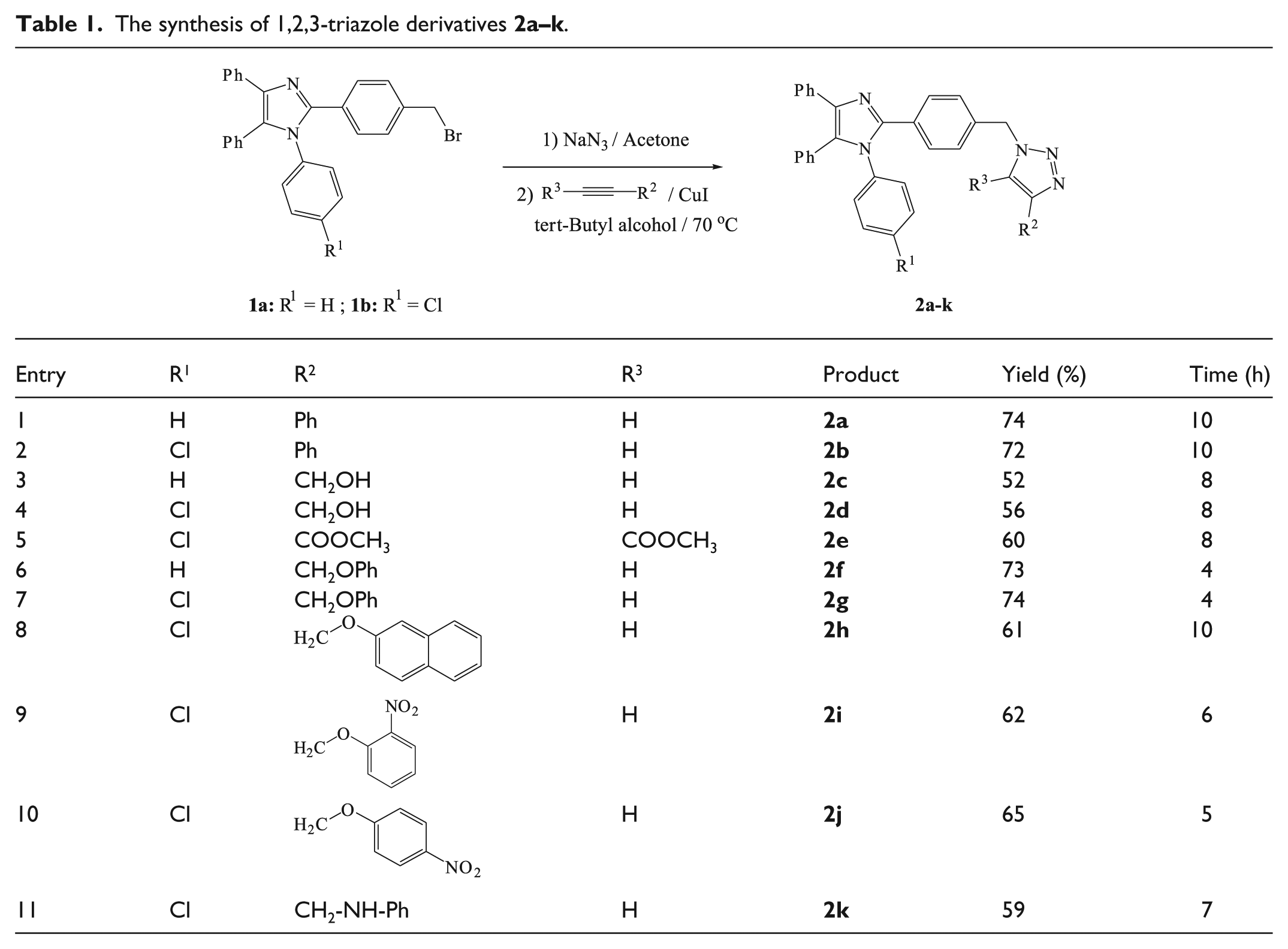
To study the optical properties of the synthesized compounds, we recorded the UV-Vis and fluorescence spectra of some chosen products in dilute (10-6 M) ethanolic solutions. Two absorbtion peaks appeared for each of the studied materials, one in the range of 223–234 nm and the other in the range of 284–296 nm (Table 2). Calculated molar extinction coefficients (εmax) for the more intense peaks (223–234 nm) indicate strong π–π* transitions. The fluorescence emission spectra of the samples and the related descriptions are presented in Figure 1 and Table 2, respectively.

The fluorescence spectra of dilute solutions (10-6 M) of selected products
Fluorescence and UV-Vis parameters of selected products
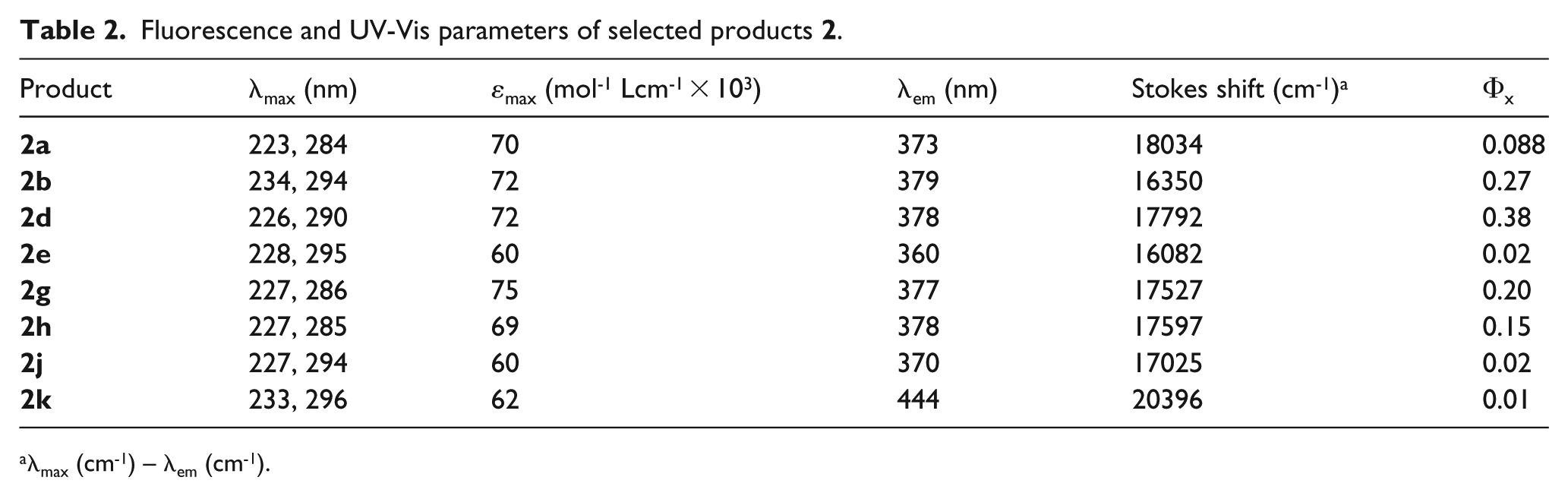
λmax (cm-1) – λem (cm-1).
As seen, compound
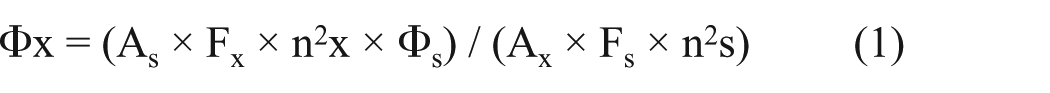
According to the equation, x is the symbol of the samples and s is the symbol of the standard, where Φ is the fluorescence quantum yield, F is the area under the fluorescence emission curves and A shows the absorbance at the excitation wavelength (270 nm), and nx and ns are the refractive indexes of ethanol and water (1.36 and 1.33, respectively).
The Stokes shifts of the synthesized compounds, calculated from the difference between the emission wavelength λem and the absorption wavelength λmax, are relatively good, indicating low overlap between their absorption and emission spectra.
We then prepared the 1-(4-hydroxyphenyl)-2,4,5-triarylimidazoles
The synthesis of 1,2,3-triazole derivatives
The optical behavior of the products obtained was also evaluated in dilute (10-6 M) ethanolic solutions. Figure 2 shows the fluorescence spectra of the samples
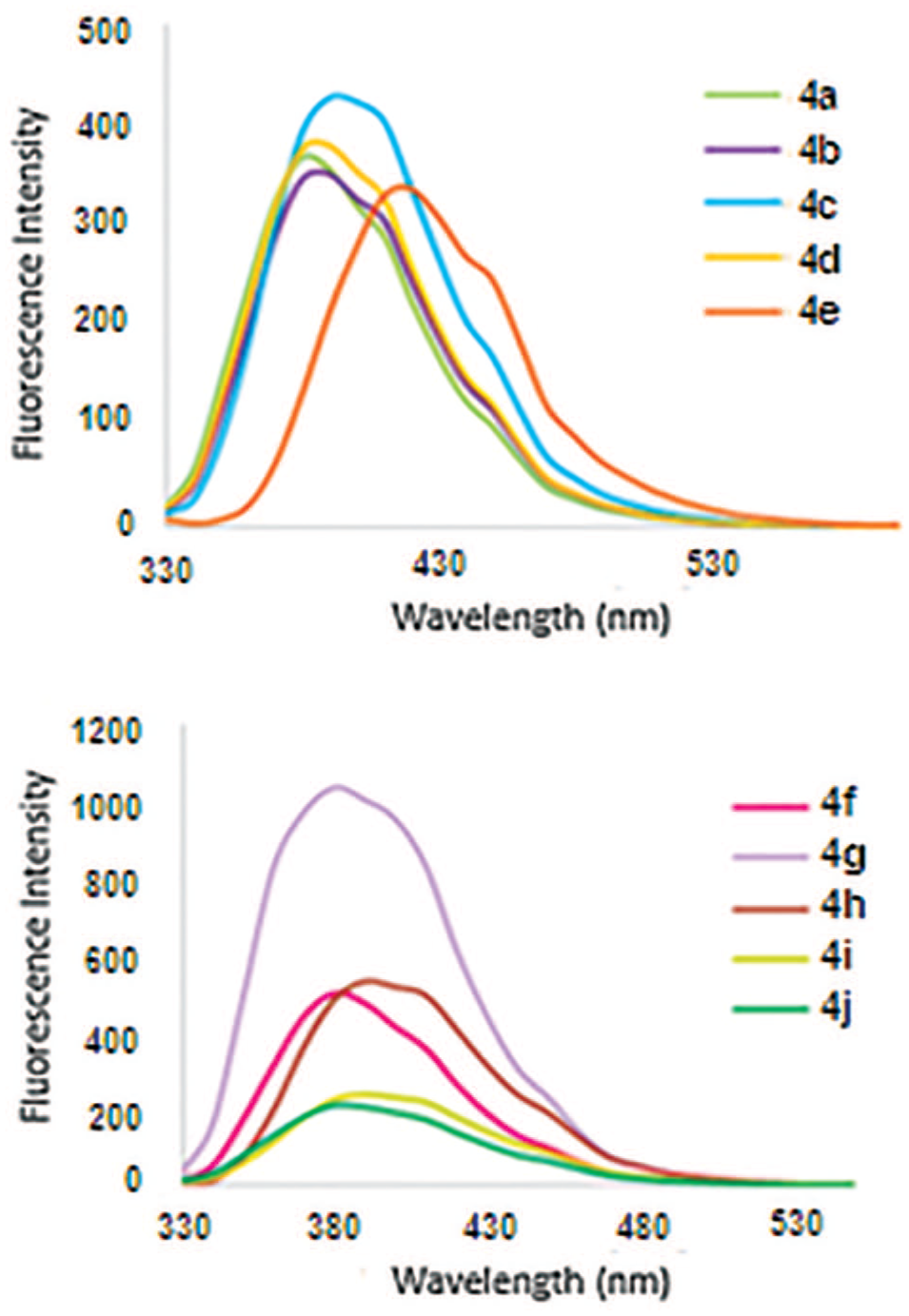
The fluorescence spectra of dilute solutions (10-6 M) of products
Fluorescent and UV–Vis parameters of products

A single product was obtained from each of the above Cu(I) catalysed cycloaddition reactions and was identified by analogy with the literature as the 1,4-disubstituted triazole rather than the 1,5-disubstituted isomer.
34
The triazole-H signal in the 1H NMR spectra of the products
Conclusion
In conclusion, we have reported the synthesis and optical properties of some novel tetraaryl imidazole derivatives possessing a 1,2,3-triazole ring. These products were prepared by CuI-catalyzed 1,3-dipolar cycloadditions of either azidomethyl derivatives of tetraaryl imidazoles with various commercial and prepared terminal acetylenes, or propargyloxy derivatives of tetraaryl imidazoles with benzyl azide. All the products were characterized from their spectroscopic data (supplemental material) and most of them were evaluated in view of their optical properties. Fluorescence spectra as well as the optical parameters (λmax, εmax, λem, quantum yields of emission and Stokes shifts) of the products have been reported. The highest values of emission quantum yields in two series of the products were obtained for compounds
Experimental
Melting points were measured on an Electrothermal MEL-TEMP apparatus (model 1202D). FTIR spectra were obtained with a Bruker Tensor 27 spectrometer; υ in cm-1. 1H NMR and 13C NMR spectra were recorded with a Bruker Spectrospin Avance 400 spectrometer operating at 400 and 100 MHz, respectively; chemical shifts are given in parts per million (ppm, δ) relative to tetramethylsilane or solvent peaks as internal standards [δ: CDCl3: 7.26 ppm (1H), 76 ppm (13C); DMSO-d6: 2.50 ppm (1H), 39.5 ppm (13C)]. Elemental analyses were measured using a Vario EL ΙΙΙ apparatus (Elementar Co.). UV-Vis spectra were recorded on an Analytic Jena Specorel 250 spectrometer. Preparative thin-layer chromatography (PLC) was performed with prepared glass-backed plates (20 × 20 cm2, 500 μ) using silica gel (Merck Kieselgel 60 PF254 + 366). Fluorescence emission spectra were acquired on a Jasco FP-750 spectrofluorimeter.
Synthesis of products 2a–k : general procedure
A mixture of bromide
4-phenyl-1-[4-(1,4,5-triphenyl-1H-imidazol-2-yl)benzyl]-1H-1,2,3-triazole (
Synthesis of products 4a–j : general procedure
A mixture of phenol
1-Benzyl-4-{[4-(2,4,5-triphenyl-1H-imidazol-1-yl)phenoxy]methyl}-1H-1,2,3-triazole (
Supplemental Material
Supporting_information_PDF – Supplemental material for Synthesis and optical properties of novel 1,2,3-triazole derivatives possessing highly substituted imidazoles
Supplemental material, Supporting_information_PDF for Synthesis and optical properties of novel 1,2,3-triazole derivatives possessing highly substituted imidazoles by Zarrin Ghasemi, Arezoo Mirzaie, Roqhayeh Arabzadeh, Zahra Fathi and Ameneh Abolghassemi Fakhree in Journal of Chemical Research
Footnotes
Acknowledgements
The research affairs of the University of Tabriz are gratefully appreciated.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the Iran National Science Foundation (INSF) for financial support.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


