Abstract
The binuclear silver(I) complex [Ag2(η2-SPPh3)2(SPPh3)2](BF4)2 has been synthesized. Crystal analysis shows the coordination sphere around Ag(I) can be described as a three-coordinated slightly distorted planar triangular configuration with both monodentate and monoatomic bidentate bridging coordination modes for the SPPh3 ligand. The fluorescence properties of free SPPh3 and the Ag(I) complex have been studied in the solid state. The Ag(I) complex shows quasi-reversible Ag+/Ag pairs in its electrochemistry and has significant antioxidant activity against hydroxyl radicals.

Introduction
Transition metal complexes have attracted considerable attention because of their potential applications as functional materials as well as their structural diversity.1–3 For the synthesis of coordination compounds, d10 metals are widely used because their flexible coordination sphere allows the generation of different kinds of structures with ligands containing nitrogen, sulfur or oxygen.4–8 Ag(I) is among the most labile metal ion due to its d10 electronic configuration with versatile coordination number varying from 2 to 8.9–11 The Ag(I) ion demonstrates linear, trigonal, square planar, square-pyramidal, trigonal-bipyramidal, tetrahedral, and octahedral coordination geometries with high affinity to both nitrogen and sulfur atoms.12–14 Moreover, This ion is apt to form short Ag-Ag contacts as well as ligand unsupported interaction, which has been proved to be two of the most important factors contributing to the formation of such complexes and their special properties.15–16 Furthermore, silver complexes show potential applications in photoluminescence, molecular recognition, and as antioxidant, antibacterial, and anticancer agents.17–19
Sulfur is a soft base that easily forms stable compounds with a transition metal such as silver that is a soft acid. 20 Triphenylphosphine sulphide and its derivatives are important chemical reagents and ligands for homogeneous catalysts, and have broad application prospects in flame retardant, antistatic and anti-corrosion agents.21,22 Therefore, research work on sulfur-containing metal complexes not only has important theoretical significance for enriching the structural chemistry of these compounds, but also has potential application value in many fields of biomedicine and materials science.23–25 In this work, we report the synthesis, structure, fluorescence, electrochemical properties and antioxidant activity of a binuclear Ag(I) complex containing triphenylphosphine sulphide ligand.
Results and discussion
Characterisation of the complexes
The Ag(I) complex prepared in this study is soluble in polar aprotic solvents such as dimethylformamide (DMF) and dimethyl sulfoxide (DMSO), and slightly soluble in acetonitrile, but insoluble in water. Elemental analysis indicates that the composition of complex is [Ag2(η2-SPPh3)2(SPPh3)2](BF4)2 (
Crystal structure of complex 1
An ORTEP diagram of

Molecular structure and atom numberings of
Selected bond lengths (Å) and bond angles (°) of

Symmetry transformations used to generate equivalent atoms: #1: -x + 1,-y,-z + 1.

A view of the packing in
Fluorescence properties
The d
10
metal coordination complexes have attractive fluorescent properties, which make them eligible candidates for chemical sensors or optical devices.
33
Hence, the fluorescent emission spectra of SPPh3 and

The solid-state fluorescent emission spectra of the SPPh3 ligand and
Electrochemical studies
The electrochemical properties of SPPh3 and

Cyclic voltammogram of
Electrochemical data and electrochemical potentials and energy of
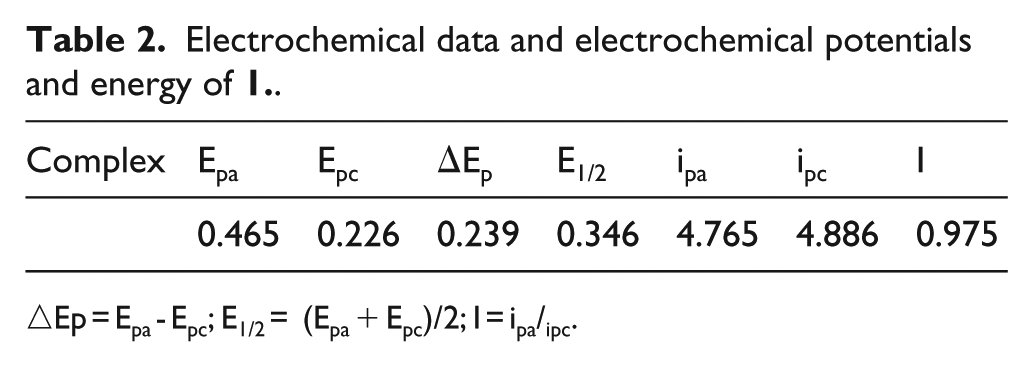
△Ep = Epa - Epc; E1/2 = (Epa + Epc)/2; I = ipa/ipc.
Antioxidant properties
On the basis of relevant reports in the literature, some silver complexes may display good biological activity, such as antioxidant activity.
41
Therefore, we also investigated whether complex

A plot of scavenging percentage (%) versus concentration of complex
Conclusion
In this work, a new binuclear silver(I) complex
Experimental
Materials and methods
All chemicals and solvents were reagent grade and used without further purification. [Ag(CH3CN)4BF4], CH2Cl2 and hexane are all commercially produced. The triphenylphosphine sulphide (SPPh3) ligand was synthesized according to the reported method. 46 Na2HPO4–NaH2PO4 buffer and ethylenediaminetetraacetic acid (EDTA)-Fe(II) solution were prepared using twice-distilled water. Electronic spectra were taken on a Lab-Tech UV Bluestar spectrophotometer. Absorbance was measured with a Spectrumlab 722 sp spectrophotometer at room temperature. The C, H and N elemental analyses were performed using a Carlo Erba 1106 elemental analyzer. The IR spectra were recorded in the 4000–400 cm-1 region with a Nicolet FT-VERTEX 70 spectrometer using KBr pellets. Electrochemical measurements were performed on a LK2005A electrochemical analyser under N2 at 283 K. The fluorescence spectra were obtained with an F97 Pro fluorescence spectra fluorophotometer.
Synthesis of [Ag2(η2-SPPh3)2(SPPh3)2](BF4)2 (1 )
A mixture of [Ag(CH3CN)4](BF4) (0.0372 g, 0.1 mmol) and SPPh3 (0.0441 g, 0.15 mmol) in CH2Cl2 solution (5 mL) was stirred at ambient temperature for 2 h to yield a colourless solution. The solution was filtered and hexane slowly diffused into the solution. Colourless crystals suitable for X-ray diffraction studies were obtained after 5 days. Yield: 63%; Anal. calcd for C72 H60Ag2B2F8P4S4: C, 55.20; H, 3.86; found: C, 54.93; H, 3.81%. Selected IR data (KBr ν/cm-1). 1593(w), 1463(m), 1437(s), 1310(m), 1101(s), 1057(s), 987(m), 749(m), 695(s), 596(m), 515(s). UV–Vis (in DMF), λmax (nm): 268.
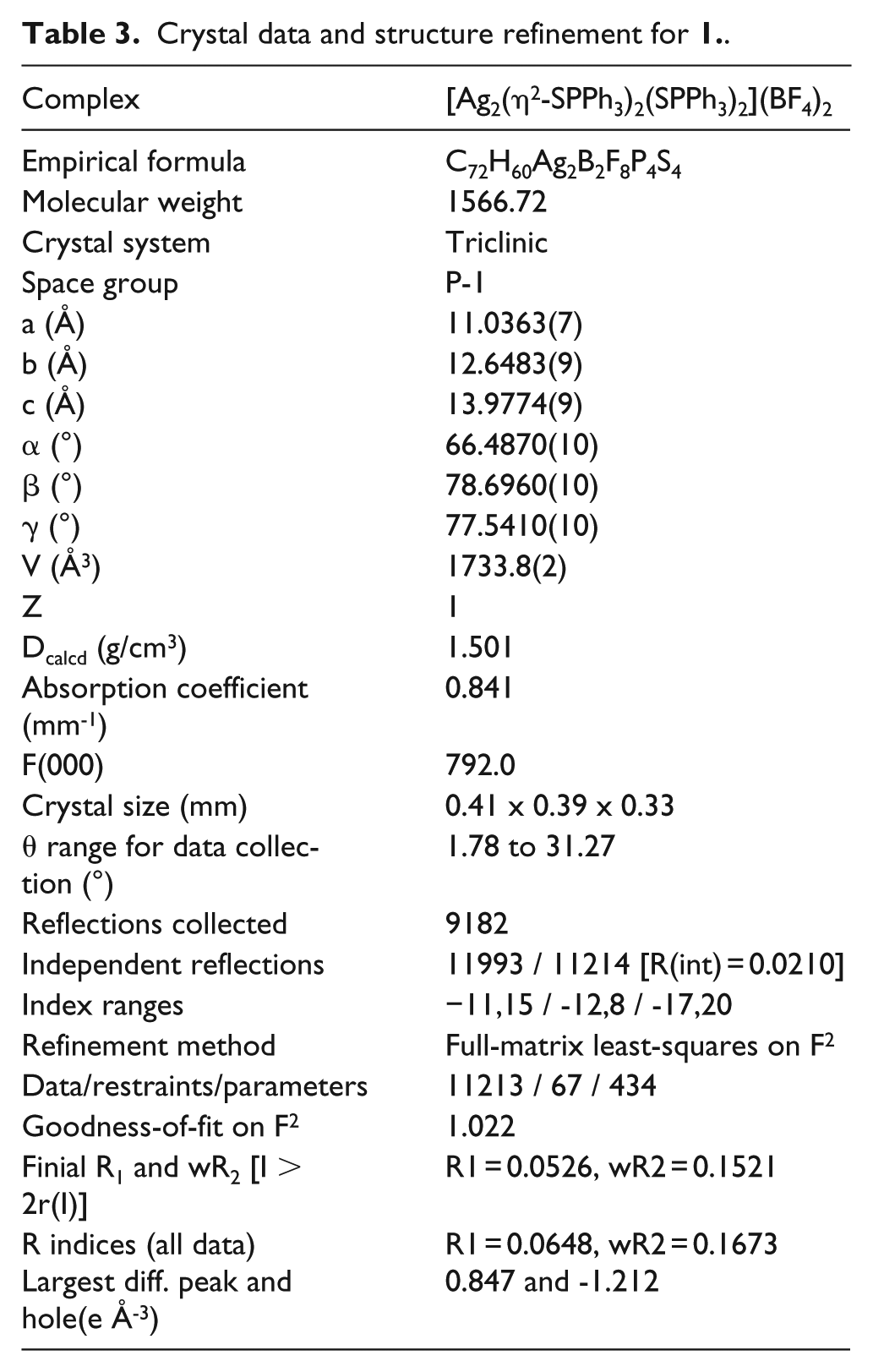
X-ray crystallography
A suitable single crystal of
Crystal data and structure refinement for
Hydroxyl radical scavenging activity
Hydroxyl radicals were generated in aqueous media through the Fenton-type reaction.51,52 The reaction mixture (3 mL) contained 1 mL of 0.1 mmol aqueous safranin, 1 mL of 1.0 mmol aqueous EDTA-Fe(II), 1 mL of 3% aqueous H2O2, and a series of quantitative micro-additions of solutions of the test compound. A sample without the test compound was used as the control. The reaction mixtures were incubated at 37°C for 30 min in a water bath. The absorbance was then measured at 520 nm.53–55 The scavenging effect for OH• was calculated from the following expression: Scavenging ratio (%) = ((Asample - Ar) / (Ao - Ar)) × 100%, where Asample is the absorbance of the sample in the presence of the test compound, Ar is the absorbance of the blank sample in the absence of the test compound, and Ao is the absorbance in the absence of the test compound, EDTA-Fe(II) and H2O2.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Young Scholars Science Foundation of Lanzhou Jiaotong University (2018004), Foundation of A Hundred Youth Talents Training Programme of Lanzhou Jiaotong University (152022) and Natural Science Foundation of Gansu Province (17JR5RA090).
Supplemental material
Crystallographic data (excluding structure factors) for the structures reported in this paper have been deposited with the Cambridge Crystallographic Data Centre with reference numbers CCDC 1879118. Copies of the data can be obtained, free of charge, on application to the CCDC, 12 Union Road, Cambridge CB2 1EZ, UK. Tel.:+44 01223 762 910; fax: +44 01223 336 033; e-mail: ![]() .
.
