Abstract
A new series of 4-azasteroidal-17-hydrazone derivatives have been synthesized from androstenedione. Their structures were characterized by analysis and spectroscopic data. The antiproliferative activity of synthesized compounds against three cancer cells (human lung adenocarcinoma, human oesophageal cervical cancer, human gastric adenocarcinoma) and a normal cell line (human gastric mucosal) was investigated. The studies show that the compound bearing a naphthyl group displayed the same antiproliferative activity in vitro against tested cells as

4-azasteroidal -17-hydrazone derivatives have been synthesized. The title compounds were screened for their preliminary anticancer activity against 446 (human lung adenocarcinoma cell line), Eca-109 (human esophageal cervical cancer cell line), AGS (human gastric adenocarcinoma cell line) and a normal cell GES-1(human gastric mucosal).
Introduction
Some steroidal compounds have been used as traditional medicines, such as antibacterial and hormone kind medication. Besides the naturally occurring substances, the majority of steroidal drugs are semi-synthetic compounds.1–4 Modification of steroidal compounds by introduction of heteroatom or replacement of carbon atoms by a heteroatom in steroids often affects the chemical properties of the steroidal molecule and results in alterations of biological activities.5–8 Nitrogen-containing steroid derivatives are common clinical drugs in cancer chemotherapy. It has been reported that 4-azasteroidal analogues may lead to compounds having anticancer activity, cytotoxicity, antibacterial and 5α-reductase inhibitor activity.9–13
Hydrazone compounds and their derivatives are a class of substances displaying antibacterial, antiviral, antitumor or other biological activities.14,15 T Zoltan et al.16–18 have reported some synthetic work of 4-azasteroidal-17-hydrazone. However, the study of hydrazine derivatives with respect to antiproliferative activity has few reports. In this article, novel 4-azasteroidal-17-hydrazone derivatives from the commercially available androstenedione were designed and synthesized. Furthermore, the antiproliferative activity of the compounds against 446 (human lung adenocarcinoma cell line), Eca-109 (human oesophageal cervical cancer cell line), AGS (human gastric adenocarcinoma cell line) and a normal cell line (GES-1) were evaluated in vitro.
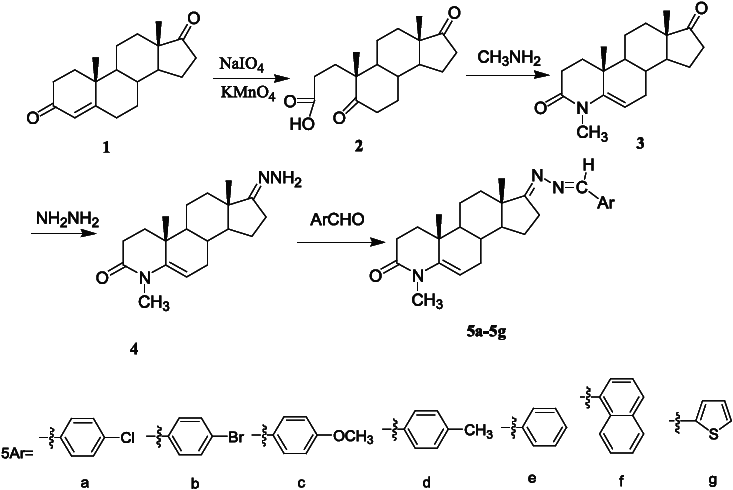
Scheme 1 outlines the synthetic procedures of compounds

Synthesis of compounds
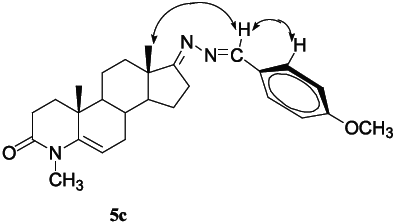
To further investigate the configuration of the 17-hydrazone derivatives,
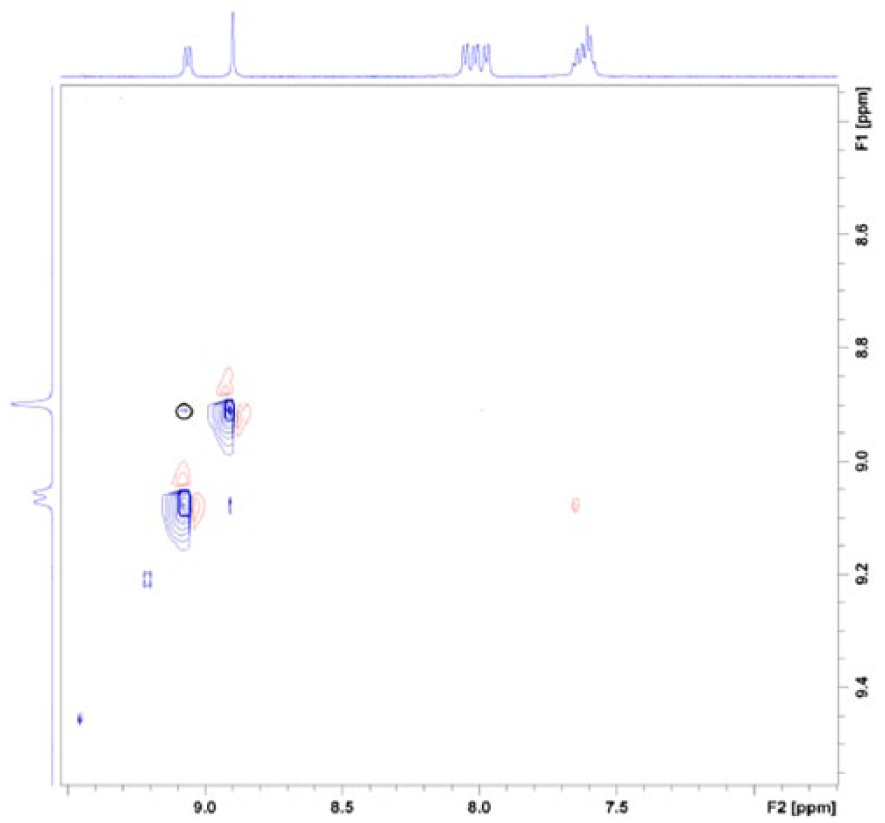
NOESY spectrum of (a)
Partially enlarged NOESY spectrum of
Example of NOE relationships of compound
To evaluate the antiproliferative activity of the compounds, we determined their IC50 values on 446 (human lung adenocarcinoma cell line), Eca-109 (human oesophageal cervical cancer cell line) and AGS (human gastric adenocarcinoma cell line) cancer cells and one normal cell line (human gastric mucosal cell GES-1) using the MTS (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetazolium) assay. MTS is a compound that can be taken up by viable cells and reduced by a mitochondrial dehydrogenase forming a formazan product in living cells. The absorbance of the formazan product at 492 nm is in linear proportion to cell numbers. The results are summarized as IC50 values in μmol L−1 in Table 1.
In vitro a antiproliferative activities (IC50 in μmol L−1) of 4-azasteroidal-17-hydrazone derivatives.

ND: not determined.
Data represent the mean values of three independent determinations.
From Table 1, it is clear that some of the compounds show moderate to good cytotoxicity against 446, Eca-109 and AGS cancer cell lines and very weak toxicity towards the GES-1 normal cell line. In general, all the compounds were more active against the oesophageal cervical cancer cell line (Eca-109) than the other two cell lines. Compound
The information obtained from the studies is valuable for the design of novel steroidal chemotherapeutic drugs. Our findings provide new evidence showing the relationship between the chemical structure and biological function.
Experimental
All chemicals and solvents were of analytical grade and solvents were purified by general methods before being used. Melting points (m.p.) were determined on an X4 apparatus and were uncorrected. Infrared (IR) spectra were determined with KBr pellets on an FTS-135 spectrophotometer. 1H and 13C nuclear magnetic resonance (NMR) spectra were recorded on a Bruker 500 NMR spectrometer (Bruker Instruments Inc., Billerica, MA, USA) at either 500 MHz (1H NMR) or 125 MHz (13C NMR). Electrospray ionization mass spectrometry (ESI-MS) was performed on an LCQ Advantage MAX spectrometer. Elementary analyses were performed by a vario ELsuperuser.
5,17-Dioxo-A-nor-3,5-secoandrostan-3-oic acid (2 )
A mixture of androstenedione
4-Methyl-4-aza-5-androstene-3,17-dione (3 )
A mixture of
3-Oxo-4-methyl-4-aza-5-androstene-17-hydrazone (4 )
Compound
General procedure for the synthesis of compounds (5a–g )
To a stirred solution of compound
Biological activity
Cell viability assay
The anticancer potency of test compounds was measured using the MTS assay. The cells (1 × 104) were seeded in 96-well plates and cultured for 24 h, followed by treatment with different concentrations of the test compound for a range of durations for the different experiments. A volume 20 μL of MTS solution was added to each well and incubated for 3 h at 37 °C, following which the supernatant was removed and 150 μL DMSO was added. The absorbance was recorded using a microplate reader (Titertek Multiskan; Flow Laboratories, North Ryde, Austria) at a wavelength of 492 nm. All experiments were performed six times. The cell growth inhibition rate was calculated using the following formula: 1 − ODexperiment/ODcontrol. The IC50 values were calculated as the concentration of drug yielding 50% cell survival.21,22
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of P.R. China (Grant Nos 81772550 and 81502032), the Programme of Science and Technology of Hebei (17211412) and the Project of Hebei University of Science and Technology (Grant No. 1182120).
