Abstract
A series of 5-methyl-4-methylene-7-methylthio-2-arylmino-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile derivatives were synthesized via tandem aza-Wittig and annulation reactions with {N-[3-acetyl-5-cyano-2-methyl-6-(methylthio)pyridin-4-yl]imino}triphenylphosphorane and aryl isocyanate in dry dichloromethane. Their structures were clearly confirmed by infrared, 1H NMR, 13C NMR, high-resolution mass spectrometry, and X-ray single-crystal diffraction. All newly synthesized compounds were screened for herbicidal activities against monocotyledonous and dicotyledonous plants. The results indicated that the target compound 2-[(4-methoxyphenyl)amino]-5-methyl-4-methylene-7-(methylthio)-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile showed 100% inhibition rate to grain sorghum (monocotyledonous) at the concentration of 100 mg L−1.

Introduction
As one of the nitrogen- and oxygen-containing fused heterocycles, pyridooxazine derivatives occupy an important place in medicinal chemistry owing to their notable pharmacological and biological activities such as thermochromic agents, 1 antinociceptives, 2 antivirals, 3 antibacterials, 4 antimalarials, 5 anticancer agents, 6 tissue factor inhibitors, 7 and scavenger agents of carbohydrates. 8 In addition, the pyridooxazine derivative pazufloxacin mesilate (PZFX) is a novel quinolone antimicrobial agent for injection. 9 In recent years, the synthesis and biological activity of these derivatives have become a hot topic in biological and pharmacological studies.
Among the pyridooxazine derivatives, most of the synthetic studies are focused on pyrido[2,3-b][1,4]oxazines,10,11 pyrido[2,1-b][1,3]oxazines, 12 pyrido[3,2-b][1,4] oxazines,13,14 pyrido[4,3-b][1,4]oxazines, 15 and pyrido[3,2-b][1,2]oxazines. 16 However, several reports are available on the synthesis of pyrido[4,3-d][1,3]oxazine derivatives. Hansen synthesized a series of pyrido[4,3-d][1,3]oxazines by the reaction of 4-amino-3-picolinic acid with substituted benzoyl chloride. 17 Raslanc and colleagues 18 reported a new preparative approach, utilizing tandem aza-Wittig and annulation reactions with the corresponding iminophosphoranes, aromatic isocyanate, and acid chloride to give two stable pyrido[4,3-d][1,3]oxazine compounds. Therefore, to discover novel pyrido[4,3-d][1,3]oxazine compounds with excellent biological activity is of greater theoretical and application value.
The aza-Wittig reaction has been improved to be a versatile and powerful technique for synthesizing five- to eight-membered N-heterocycles, including some active natural compounds.19,20 Some N-containing compounds, which are not easily prepared by other methods, have been synthesized via the tandem aza-Wittig reactions of functionalized iminophosphoranes under mild conditions. 21 The tandem aza-Wittig reactions of functionalized iminophosphoranes were also applied in the synthesis of many fused N-heterocycles such as pyrazolopyrimidine, 22 triazolopyrimidine, 23 thienopyrimidine, 24 pyridopyrimidine, 25 imidazolopyrimidine, 26 and furopyrimidine derivatives. 27 Recently, we have become interested in the synthesis of new bioactive fused heterocycles, such as pyrido[4,3-d][1,3]oxazines from iminophosphoranes, with the aim of evaluating their herbicidal activities. In this paper, an effective method for the preparation of a novel series of pyrido[4,3-d][1,3]oxazine derivatives from the {N-[3-acetyl-5-cyano-2-methyl-6-(methylthio)pyridin-4-yl]imino}triphenylphosphorane is described. The subsequent herbicidal testing indicated that many target compounds possessed herbicidal activity against the root and stalk of radish (Raphanus sativus), grain sorghum (Sorghum bicolor), cucumber (Cucumis sativus), purple medic (Medicago sativa), rape (Brassica napus), and barnyard grass (Echinochloa crusgalli).
Results and discussion
Synthesis and spectroscopic characterization
The synthetic route of the novel pyrido[4,3-d][1,3]oxazine derivatives is illustrated in Scheme 1. 2-methyl-3-acetyl-4-amino-5-cyan-6-methylthio-pyridine
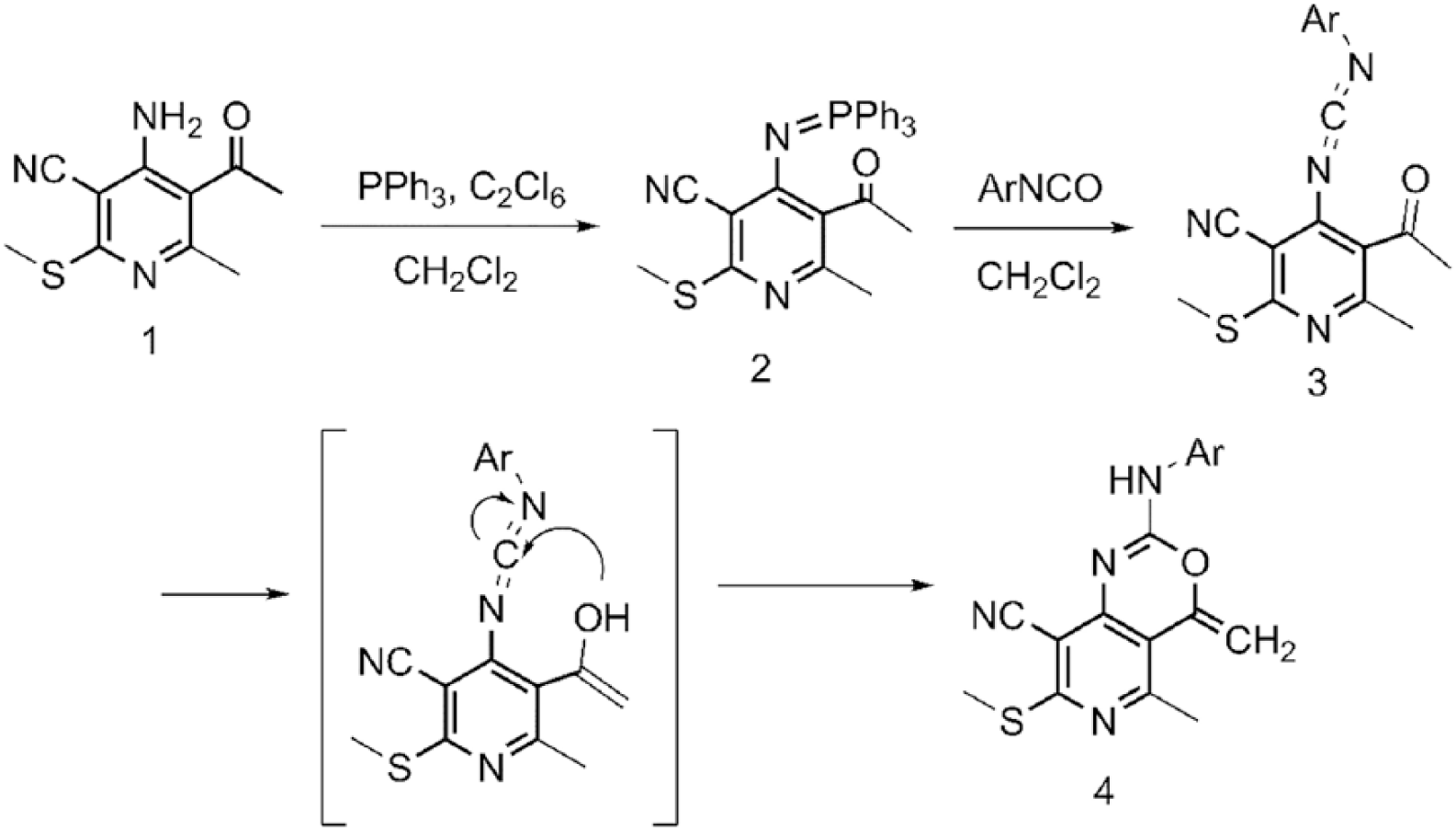
The synthetic route of the compound
Structures of products

The title compounds

ORTEP diagram of
X-ray crystallography analysis information for 4g.

Herbicidal activity
The target compounds
Conclusion
In summary, we designed and synthesized 11 pyrido[4,3-d][1,3]oxazine derivatives via tandem aza-Wittig reaction. The preliminary bioassay of these title compounds was studied. Based on the herbicidal testing, we found that some target compounds possessed excellent herbicidal activity against the root and stalk of radish, grain sorghum, cucumber, purple medic, rape, and barnyard grass. The study in this work demonstrated an effective method to synthesize fused heterocycles containing pyrido[4,3-d][1,3]oxazines with good herbicidal activity.
Experimental analysis
General experimental methods
All commercial reagents were purchased from J&K Chemical Co. Et3N and dichloromethane (DCM) were dried according to standard methods and distilled before use. Melting points were determined on an electrothermal melting-point apparatus and are uncorrected. A Nicolet 6700 spectrometer was used to record IR spectra (KBr pellets). 1H NMR, 13C NMR, and 19F NMR spectra were recorded on a Bruker 400 spectrometer with CDCl3 or dimethyl sulfoxide-d6 (DMSO-d6) as the solvent. The HRMS analysis was acquired on a micro-TOF II instrument. The X-ray single-crystal diffraction was performed with a SuperNova E G8910B Instrument.
Synthesis of {N-[3-Acetyl-5-cyano-2-methyl-6-(methylthio)pyridin-4-yl]imino}triphenylphosphorane 2
General procedure for compound 4
Intermediate
5-methyl-4-methylene-7-(methylthio)-2-(phenylamino)-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile (
5-methyl-4-methylene-7-methylthio-2-(p-tolylamino)-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile (
2-[(3,4-dichlorophenyl)amino]-5-methyl-4-methylene-7-(methylthio)-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile (
2-[(4-chlorophenyl)amino]-5-methyl-4-methylene-7-(methylthio)-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile (
5-methyl-4-methylene-7-(methylthio)-2-{[2-(trifluoromethyl)phenyl]amino}-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile (
2-[(3-chlorophenyl)amino]-5-methyl-4-methylene-7-(methylthio)-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile (
2-[(4-methoxyphenyl)amino]-5-methyl-4-methylene-7-(methylthio)-4H-pyrido [4,3-d][1,3]oxazine-8-carbonitrile (
2-(benzylamino)-5-methyl-4-methylene-7-(methylthio)-4H-pyrido[4,3d][1,3] oxazine-8-carbonitrile (
2-[(3,5-dimethylphenyl)amino]-5-methyl-4-methylene-7-(methylthio)-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile (
5-methyl-4-methylene-7-(methylthio)-2-{[4-(trifluoromethyl)phenyl]amino}-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile (
2-[(4-fluorophenyl)amino]-5-methyl-4-methylene-7-(methylthio)-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile (
Supplemental Material
Supporting_Information – Supplemental material for Design, synthesis, and herbicidal activity of novel 2-(arylamino)-5-methyl-4-methylene-7-(methylthio)-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile derivatives
Supplemental material, Supporting_Information for Design, synthesis, and herbicidal activity of novel 2-(arylamino)-5-methyl-4-methylene-7-(methylthio)-4H-pyrido[4,3-d][1,3]oxazine-8-carbonitrile derivatives by Yi Sun, Wenfei Huang, Zhiqiang Li, Tao Wang and Jin Luo in Journal of Chemical Research
Footnotes
Acknowledgements
The authors are grateful to the National Natural Science Foundation of China and the Natural Science Foundation of Jiangxi Province in China for their financial support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was supported by the National Natural Science Foundation of China (grant nos. 21562026 and 21762025) and the Natural Science Foundation of Jiangxi Province in China (grant no. 20161BAB213072).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
