Abstract
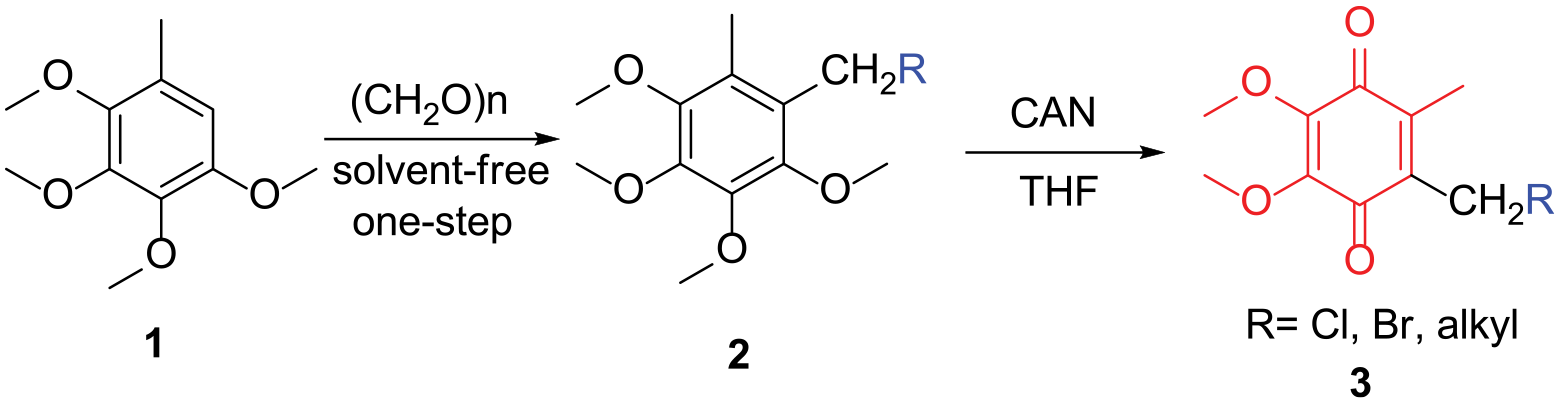
A series of 1,4-benzoquinones was prepared in a high yield by a two-step reaction starting from 2,3,4,5-tetramethoxytoluene by a Blanc reaction and subsequent oxidation.
Introduction
Coenzyme Q10 (CoQ10, Figure 1) is a 1,4-benzoquinone molecule with a side chain of 10 isoprenyl units, occurs naturally in human and most bacteria and acts as an electron carrier in mitochondrial respiratory chains.
1
CoQ10 is widely used in the treatment of mitochondrial disorders and neurodegenerative movement disorders,
2
such as Parkinson’s disease, Alzheimer’s disease, and Friedreich’s ataxia.
3
Synthetic CoQ analogues (

CoQ10 and compounds
Among the published syntheses of CoQ10, Lipshutz et al.
5
and Negishi et al.
6
developed efficient procedures based on metal-catalyzed cross-coupling between a chloromethyl-CoQ0 (compound
We now present an efficient and reproducible synthesis of compounds
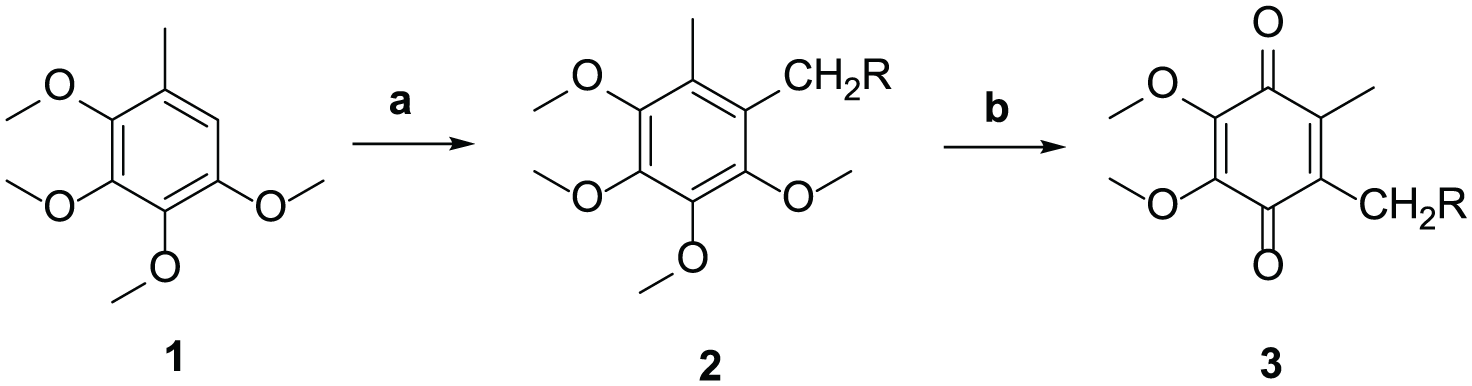
Reagents and conditions: (a) paraformaldehyde and 37% HCl, 47% HBr, morpholine, or piperidine (35 °C) and (b) CAN and THF/H2O (25 °C).
Results and discussion
As shown in Table 1, a two-step method for the synthesis of 1,4-benzoquinones is described. In the first step, Blanc reaction of compound
Two-step synthesis of 1,4-benzoquinones (

Conclusion
In summary, a two-step route for the synthesis of 1,4-benzoquinones
Experimental
All reactions were monitored by thin layer chromatography (TLC; SiO2, petroleum ether (boiling range in 60–90oC)/EtOAc 5:1); nuclear magnetic resonance (NMR) and mass spectra were recorded on a Bruker AVANCE III-HD 400 NMR spectrometer and a TripleTOF mass spectrometer, respectively. Elemental analyses were performed on a Perkin-Elmer PE2400 Elemental Analyzer. All reagents, for example, paraformaldehyde and CAN, were purchased from Adamas, P.R. China, and used without further purification. 2,3,4,5-Tetramethoxytoluene
General procedure for compounds 2a–2d
Different solvents (37% HCl, 47% HBr, morpholine, and piperidine) were added dropwise at room temperature to stirred mixtures of compound
Compound
Compound
Compound
Anal. calcd for C16H25NO5: C, 61.72; H, 8.09; N, 4.50; O, 25.69; found: C, 61.70; H, 8.10; N, 4.52; O, 25.71%.
Compound
Anal. calcd for C17H27NO4: C, 65.99; H, 8.80; N, 4.53; O, 20.68; found: C, 66.00; H, 8.81; N, 4.50; O, 20.69%.
General procedure for compounds 3a-3d
A solution of CAN (0.002 mol) in water (5 mL) was added dropwise to a solution of each compound
Compound
Compound
Compound
Compound
Footnotes
Author contributions
Yong-Fu Qiu, Bin Lu, and Yi-Yu Yan contributed equally to this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (Nos. 31600740 and 81803353), the Natural Science Foundation of Jiangsu Province (BK20160443), the Six Talent Peaks Project in Jiangsu Province (SWYY-094), the Jiangsu Provincial Key Laboratory for Bioresources of Saline Soils (Nos. JKLBS2016013 and JKLBS2017010) and the College students practice innovation training program of Yancheng Teachers University (Provincial key projects).

