Abstract
Treatment of a series of aromatic hydrocarbons and O-carbethoxy phenol substrates with a mixture of chlorosulfonic acid and dimethoxymethane in CH2Cl2 catalyzed by zinc iodide affords the corresponding chloromethyl derivatives in good to excellent yields.
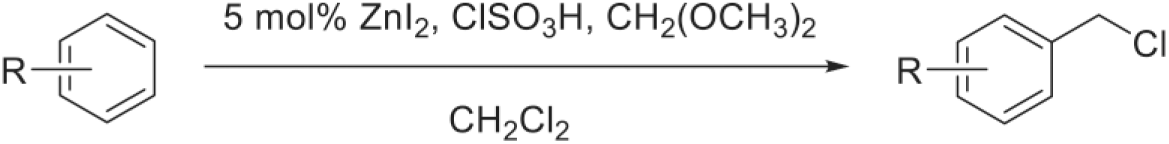
Introduction
Chloromethyl substituted aromatic compounds are promising key intermediates because of their easy transformation to a variety of fine or special chemicals, polymers and pharmaceuticals. The chloromethylation of aromatic compounds has been well documented in the literature.1–8 The oldest method for the synthesis of this class of compounds involves the chloromethylation of aromatic hydrocarbons with hydrochloric acid and either trioxane or paraformaldehyde in the absence of any catalyst;6–8 however, the reaction rate is very low and unsuitable for practical chemical processes. Lewis acids such as zinc chloride, stannic chloride, aluminium chloride, and boron trifluoride are excellent catalysts for this reaction.9–11 Among these acids, zinc chloride is an effective catalyst in hydrochloric acid solution. However, a stoichiometric amount of catalyst to substrate is required, making the work-up procedure tedious. These catalysts, in general, suffer from the inherent problems of corrosiveness, high susceptibility to water, environmental hazards, waste control after the reaction, etc.12–14 Rare-earth metal triflates were recently reported as active catalysts for this reaction, but these catalysts are expensive and not suitable for industrial use.15,16 The use of ionic liquids17–21 and surfactant micelles22,23 as catalysts for the chloromethylation of aromatic hydrocarbons has also been reported. However, the ionic liquids are inevitably associated with one or more disadvantages, such as high cost or difficulty of synthesis. Therefore, it is important to replace these highly corrosive, hazardous and polluting acid catalysts with environmentally friendly catalysts which are active under mild conditions.
Here we wish to report a simple and effective chloromethylation of aromatic compounds with dimethoxymethane and chlorosulfonic acid catalyzed by ZnI2 in CH2Cl2 under mild conditions (Scheme 1).

ZnI2-catalyzed chloromethylation of aromatic compounds.
Results and discussion
We initiated our studies by carrying out the chloromethylation of benzene with dimethoxymethane and chlorosulfonic acid in the presence of 5 mol% of ZnI2 in CH2Cl2 at 5−10°C. After stirring for 0.5 h, the desired benzyl chloride was afforded in 76% yield. In order to find the scope of this reaction, several aromatic hydrocarbons were examined in the presence of 5 mol% of ZnI2. The results are summarized in Table 1. As shown in Table 1, the reaction proceeded smoothly at 5−10°C in a short time and provided good yields (Table 1, entries 1–6). The p-substituted benzyl chloride was exclusively afforded from the various aromatic hydrocarbons screened. Moreover, the chloromethylation of biphenyl provided the mono-chloromethylated product in good yield (Table 1, entry 2). In addition, we have found that the chloromethylation of 3,5-dimethylphenol could not be carried out under similar conditions due to the coordination of its hydroxyl group with ZnI2, which led to deactivation of the substrate. When the hydroxyl group of 3,5-dimethylphenol was protected by reaction with ethyl chloroformate, the chloromethylation could be carried out smoothly and provided the desired product in nearly quantitative yield (Table 1, entry 6). The chloromethylation of various substituted O-carbethoxy phenol derivatives proceeded in good to excellent yields in a short time (Table 1, entries 6–10). Unfortunately, we found that the chloromethylation of some O-carbethoxy phenol derivatives did not occur even if the reaction time was prolonged and the reaction temperature increased. These were O-carbethoxy phenol derivatives bearing an electron-withdrawing group (i.e. Cl, NO2) which were inert under similar conditions. The chloromethylation of O-carbethoxy-1-naphthol mainly provided O-carbethoxy-8-chloromethyl-1-naphthol (
Chloromethylation of aromatic compounds catalyzed by ZnI2 a .

Reaction conditions: The reaction mixture of aromatic compounds (26 mmol), chlorosulfonic acid (31 mmol) and dimethoxymethane (31 mmol) was prepared in CH2Cl2 (30 mL) at −10°C in the presence of 5 mol% of ZnI2, followed by stirring at 5−10°C for t h.
All products were identified by their 1H and 13C NMR spectra.
Yields of products isolated by column chromatography.
To examine the halide anion effect, reactions using ZnCl2 and ZnBr2 were compared under similar reaction conditions (5 mol % of catalyst) in the chloromethylation of 3,5-dimethyl O-carbethoxy phenol derivatives with dimethoxymethane and chlorosulfonic acid. ZnCl2 and ZnBr2 were less effective in terms of substrate conversion and provided only moderate yields of
A plausible mechanism for this catalysis is shown as Scheme 2. Treatment of dimethoxymethane with chlorosulfonic acid in situ produces methyl chloromethyl ether. The formation of the chloromethyl cation ([ClCH2]+) is then promoted by ZnI2. Electrophilic substitution of the aromatic compound gives the desired chloromethylated derivative.
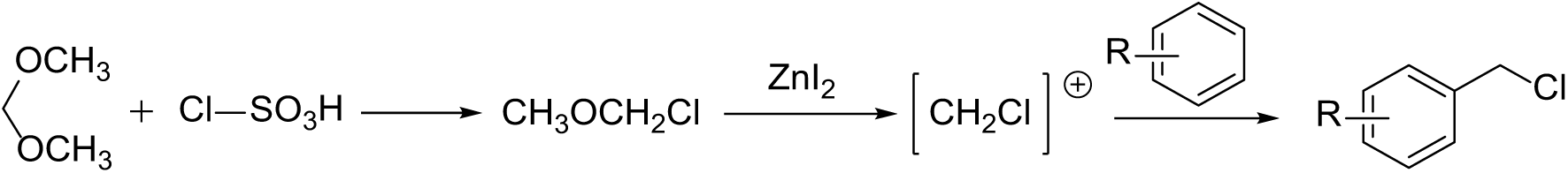
Plausible mechanism of chloromethylation of aromatic derivatives catalyzed by ZnI2.
In conclusion, we have demonstrated the excellent reactivity of ZnI2 as catalyst in the chloromethylation of aromatic compounds with dimethoxymethane and chlorosulfonic acid. This catalytic reaction enables the chloromethylation to be carried out in CH2Cl2 at room temperature in good to excellent yields. This method has some advantages such as simple operation, mild conditions, and easy product isolation. This chloromethylation method has shown great prospects for industrial application. Further aspects of the catalysis and the application to organic synthesis and practical pharmaceutical processes are under investigation.
Experimental
For product purification by flash column chromatography, silica gel (200~300 mesh) and light petroleum ether (boiling range 60–90°C) were used. NMR spectra were taken on a Bruker AM-500 spectrometer with TMS as an internal standard and CDCl3 as solvent. Reaction monitoring was accomplished by thin-layer chromatography (TLC) on silica gel Polygram SILG/UV 254 plates. HRMS were determined on a Waters
Chloromethylation of aromatic compounds catalyzed by zinc iodide; general procedure
A flask was charged with 5 mol% of ZnI2 (1.3 mmol), chlorosulfonic acid (31 mmol) and CH2Cl2 (30 mL), followed by dropwise addition of dimethoxymethane (31 mmol) at −10°C. After stirring the reaction mixture at −10°C for 30 min, the aromatic compound (26 mmol) was slowly added. The resulting mixture was then stirred at 5–10°C for the time recorded in Table 1. The reaction was monitored by TLC analysis. After completion, the reaction was quenched by addition of water (10 mL) in an ice bath. After extraction with CH2Cl2 (3 x 20 mL), the organic phase was washed with 5% sodium carbonate solution (2 x 10 mL), water (2 x 10 mL) and brine (2 x 20 mL), then evaporated to dryness under reduced pressure. The residue was purified by flash column chromatography on a silica gel using petroleum ether (boiling range: 60–90°C) and ethyl acetate as eluents to give the desired product.
Spectroscopic data for the products (Table 1, entries 1–12)
4-(Chloromethyl)-1,1’-biphenyl (
1-(Chloromethyl)-4-ethylbenzene (
1-(Chloromethyl)-4-isopropylbenzene (
1-(tert-Butyl)-4-(chloromethyl)benzene (
O-Carbethoxy-3,5-dimethyl-4-chloromethylphenol (
O-Carbethoxy-2,6-dimethyl-3-chloromethylphenol (
O-Carbethoxy-2,3-dimethyl-4-chloromethylphenol (
O-Carbethoxy-2,4-dimethyl-5-chloromethylphenol (
O-Carbethoxy-2-methyl-5-chloromethylphenol (
O-Carbethoxy-8-chloromethyl-1-naphthol (
Diethyl [methylenebis(naphthalene-8,1-diyl)] bis(carbonate) (
1-Chloro-8-(chloromethyl)naphthalene (
Bis(8-chloronaphthalen-1-yl)methane (
Supplemental Material
JCR1805724_ESI_copyrdited – Supplemental material for An efficient and convenient chloromethylation of some aromatic compounds catalyzed by zinc iodide
Supplemental material, JCR1805724_ESI_copyrdited for An efficient and convenient chloromethylation of some aromatic compounds catalyzed by zinc iodide by Jian Tang, Hongtao Liu, Jing Zhou and Xingxian Zhang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article, which contains the 1H and 13C NMR data, is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
