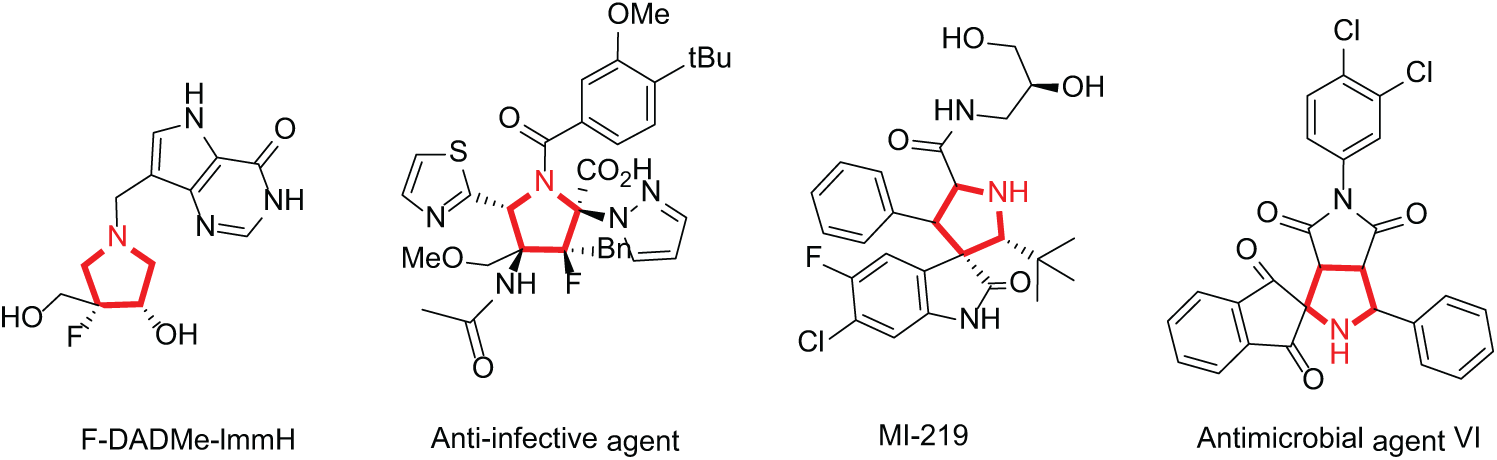
All chemicals and solvents were purchased from commercial vendors (Aldrich or Alfa) and were used as received without further purification. 1H nuclear magnetic resonance (NMR) spectra were recorded on a Bruker AM 500 MHz spectrometer in CDCl3. Chemical shifts are reported in ppm with the solvent resonance as the internal standard (CDCl3, δ 7.26 ppm). 13C NMR spectra were recorded on an NMR instrument operated at 125 MHz with complete proton decoupling. Chemical shifts are reported in ppm with the solvent resonance as the internal standard (CDCl3, δ 77.1 ppm). High-resolution mass spectra (HRMS) were recorded on a Thermo Scientific LTQ Orbitrap XL mass instrument (ESI). Thin-layer chromatography (TLC) was performed on precoated glass-backed plates and visualized with ultraviolet (UV) light at 254 nm. Flash column chromatography was performed on silica gel.
General procedure for the synthesis of 4
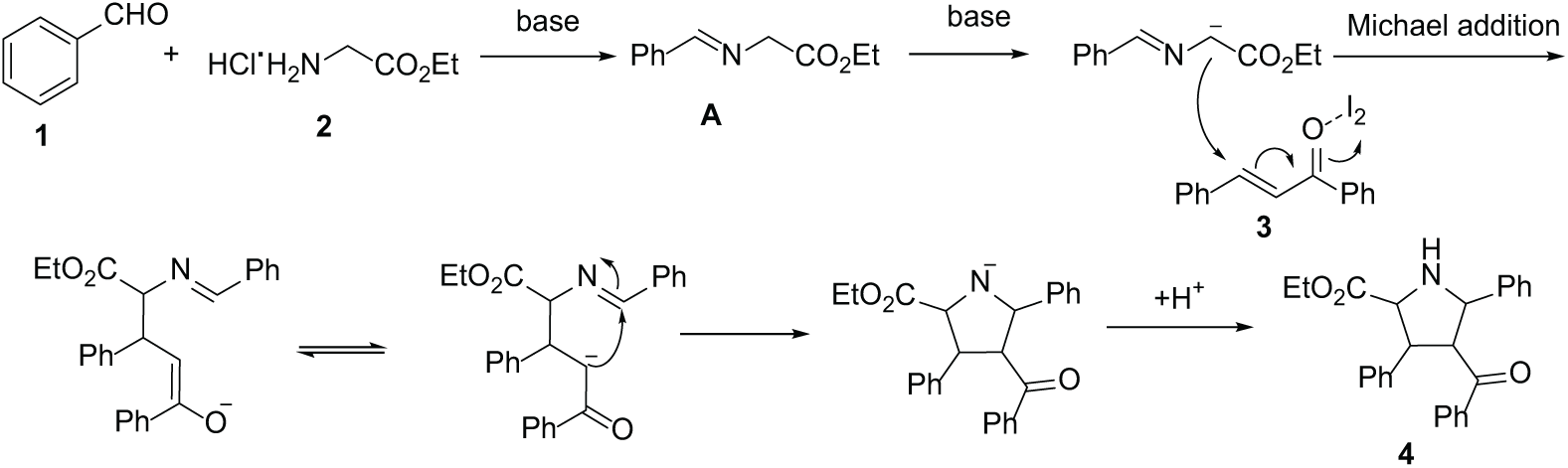
An aromatic aldehyde 1 (0.5 mmol), an amino acid ester 2 (0. 5 mmol), a chalcone 3 (0.25 mmol), K2CO3 (1.5 mmol), I2 (0.2 mmol), and THF (2 mL) were successively added into a closed and dry reaction tube and stirred at 80 °C for 7 h. The reaction was monitored by TLC. After the reaction had finished, the reaction mixture was extracted with ethyl acetate (3 × 10 mL) and washed with water, and the combined organic layers were washed with brine and aqueous Na2S2O3, dried over anhydrous Na2SO4, and concentrated in vacuo. Purification of the crude product by flash chromatography on silica gel (eluent ethyl acetate/petroleum ether = 1/4) gave the desired products 4a–w.
Ethyl 4-benzoyl-3,5-diphenylpyrrolidine-2-carboxylate (4a):
29
White solid (92.9 mg, 93% yield); m.p. 130–132 °C (Lit.
29
126–129 °C); 1H NMR (500 MHz, CDCl3) δ: 7.56 (d, J = 7.2 Hz, 2H), 7.41 (dd, J = 9.3, 7.4 Hz, 3H), 7.35 (m, J = 7.7 Hz, 2H), 7.26 (dd, J = 14.9, 7.3 Hz, 3H), 7.11 (m, 5H), 5.02 (d, J = 8.8 Hz, 1H), 4.60–4.51 (t, J = 8.7 Hz, 1H), 4.28 (dq, J = 10.7, 7.1 Hz, 1H), 4.21–4.16 (m, 1H), 4.13 (dd, J = 7.8 Hz, 2H), 1.19 (t, J = 7.1 Hz, 3H).
Ethyl 4-benzoyl-5-(4-nitrophenyl)-3-phenylpyrrolidine-2-carboxylate (4b): Yellow solid (98.8 mg, 89% yield); m.p. 145–147 °C; 1H NMR (500 MHz, CDCl3) δ: 7.98 (d, J = 8.6 Hz, 2H), 7.60 (d, J = 7.6 Hz, 2H), 7.48 (t, J = 7.3 Hz, 1H), 7.41–7.29 (m, 8H), 7.27 (d, J = 10.4 Hz, 2H), 5.11 (d, J = 8.7 Hz, 1H), 4.64 (t, J = 8.5 Hz, 1H), 4.33–4.24 (m, 1H), 4.18 (ddd, J = 14.3, 12.2, 8.2 Hz, 2H), 4.10 (t, J = 8.7 Hz, 1H), 1.20 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 197.2, 172.2, 148.0, 147.1, 139.1, 137.0, 133.2, 129.0, 128.4, 128.2, 128.0, 127.9, 127.5, 123.9, 67.1, 66.3, 61.5, 61.0, 51.6, 14.2; HRMS (ESI) m/z calcd for C26H25N2O5+ (M + H)+ 445.17580; found: 445.17587.
Ethyl 4-benzoyl-3-phenyl-5-(4-(trifluoromethyl)phenyl)pyrrolidine-2-carboxylate (4c): White solid (107.4 mg, 92% yield); m.p. 126–127 °C; 1H NMR (500 MHz, CDCl3) δ: 7.55 (d, J = 7.2 Hz, 2H), 7.45 (t, J = 7.4 Hz, 1H), 7.42–7.38 (m, 2H), 7.38–7.33 (m, 4H), 7.29 (m, 5H), 5.06 (d, J = 8.7 Hz, 1H), 4.59 (t, J = 8.3 Hz, 1H), 4.29 (dq, J = 10.7, 7.1 Hz, 1H), 4.22–4.14 (m, 2H), 4.13–4.09 (m, 1H), 1.20 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 198.2, 172.7, 143.5, 140.2, 137.3, 133.1, 128.8, 128.4, 128.0, 127.8, 127.8, 127.2, 125.0 (q, J = 3.6 Hz), 67.6, 65.8, 61.3, 60.2, 52.4, 14.1; HRMS (ESI) m/z calcd for C27H25F3NO3+ (M + H)+ 468.17810; found: 468.17847.
Ethyl 4-benzoyl-5-(4-fluorophenyl)-3-phenylpyrrolidine-2-carboxylate (4d): White solid (98.1 mg, 94% yield); m.p. 139–141 °C; 1H NMR (500 MHz, CDCl3) δ 7.57 (d, J = 7.3 Hz, 1H), 7.46–7.37 (m, 4H), 7.34 (t, J = 7.6 Hz, 2H), 7.31–7.23 (m, 4H), 7.14–7.08 (m, 2H), 6.79 (t, J = 8.6 Hz, 2H), 5.01 (d, J = 8.9 Hz, 1H), 4.55 (t, J = 8.4 Hz, 1H), 4.27 (dq, J = 10.7, 7.1 Hz, 1H), 4.18 (dq, J = 10.7, 7.1, 1H), 4.12 (dd, J = 14.2, 6.3 Hz, 2H), 1.18 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 198.2, 172.9, 163.0, 161.1, 140.3, 137.3, 135.2 (d, J = 3.2 Hz), 133.0, 129.1 (d, J = 8.2 Hz), 128.7, 128.3, 128.0, 127.8, 127.1, 115.0 (d, J = 21.5 Hz), 67.5, 65.7, 61.2, 60.5, 52.4, 14.1; HRMS (ESI) m/z calcd for C26H25FNO3+ (M + H)+ 418.18130; found: 418.18176.
Ethyl 4-benzoyl-5-(4-bromophenyl)-3-phenylpyrrolidine-2-carboxylate (4e): White solid (112.2 mg, 94% yield); m.p. 157–159 °C; 1H NMR (500 MHz, CDCl3) δ: 7.58 (d, J = 7.2 Hz, 2H), 7.46 (t, J = 7.4 Hz, 1H), 7.40–7.36 (m, 2H), 7.33 (m, 4H), 7.27–7.20 (m, 3H), 7.02 (d, J = 8.4 Hz, 2H), 4.98 (d, J = 8.8 Hz, 1H), 4.55 (t, J = 8.5 Hz, 1H), 4.27 (dq, J = 10.9, 7.1 Hz, 1H), 4.20–4.12 (dd, J = 15.7, 7.2 Hz, 2H), 4.09 (m, 1H), 1.18 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 198.1, 172.8, 140.2, 138.5, 137.3, 133.0, 131.2, 129.1, 128.7, 128.4, 128.0, 127.8, 127.2, 121.5, 67.5, 65.7, 61.3, 60.3, 52.4, 14.1; HRMS (ESI) m/z calcd for C26H25BrNO3+ (M + H)+ 478.10123; found: 478.10150.
Ethyl 4-benzoyl-5-(4-methoxyphenyl)-3-phenylpyrrolidine-2-carboxylate (4f): White solid (77.3 mg, 72% yield); m.p. 134–136 °C; 1H NMR (500 MHz, CDCl3) δ: 7.58 (d, J = 7.2 Hz, 2H), 7.41 (dd, J = 13.8, 7.3 Hz, 3H), 7.33 (t, J = 7.6 Hz, 2H), 7.30–7.22 (m, 3H), 7.05 (d, J = 8.7 Hz, 2H), 6.63 (d, J = 8.7 Hz, 2H), 4.99 (d, J = 8.9 Hz, 1H), 4.53 (t, J = 8.4 Hz, 1H), 4.27 (dq, J = 10.8, 7.1 Hz, 1H), 4.19–4.13 (m, 2H), 4.14–4.08 (m, 1H), 1.17 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 198.4, 173.0, 158.9, 140.5, 137.5, 132.8, 131.5, 128.7, 128.6, 128.3, 128.1, 127.9, 127.0, 113.5, 67.6, 66.0, 61.2, 60.8, 55.1, 52.7, 14.1; HRMS (ESI) m/z calcd for C27H28NO4+ (M + H)+ 430.20128; found: 430.20169.
Ethyl 4-benzoyl-3-phenyl-5-(p-tolyl)pyrrolidine-2-carboxylate (4g): White solid (97.1 mg, 94% yield); m.p. 160–162 °C; 1H NMR (500 MHz, CDCl3) δ: 7.58 (d, J = 7.2 Hz, 2H), 7.41 (dd, J = 14.2, 7.3 Hz, 3H), 7.34 (t, J = 7.6 Hz, 2H), 7.30–7.23 (m, 3H), 7.02 (d, J = 8.1 Hz, 2H), 6.91 (d, J = 7.9 Hz, 2H), 5.00 (d, J = 8.8 Hz, 1H), 4.55 (t, J = 8.4 Hz, 1H), 4.28 (dq, J = 10.7, 7.1 Hz, 1H), 4.28–4.25 (m, 1H), 4.17–4.11 (m, 2H), 2.20 (s, 3H), 1.18 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 198.5, 172.9, 140.6, 137.5, 137.2, 136.3, 132.7, 128.8, 128.7, 128.2, 128.1, 127.9, 127.3, 127.0, 67.7, 66.4, 61.2, 60.8, 52.8, 20.9, 14.1; HRMS (ESI) m/z calcd for C27H28NO3+ (M + H)+ 414.20637; found: 414.20679.
Ethyl 4-benzoyl-5-(naphthalen-1-yl)-3-phenylpyrrolidine-2-carboxylate (4h): White solid (100.1 mg, 89% yield); m.p. 160–162 °C; 1H NMR (500 MHz, CDCl3) δ: 7.98 (d, J = 8.5 Hz, 1H), 7.61 (d, J = 8.0 Hz, 1H), 7.54–7.40 (m, 7H), 7.37–7.29 (m, 3H), 7.12 (d, J = 7.4 Hz, 2H), 7.04 (t, J = 7.4 Hz, 1H), 6.80 (t, J = 7.8 Hz, 2H), 5.75 (d, J = 7.7 Hz, 1H), 4.70 (dd, J = 7.7, 4.5 Hz, 1H), 4.37 (dq, J = 10.7, 7.1 Hz, 1H), 4.31–4.23 (m, 2H), 4.17 (dd, J = 7.7, 4.5 Hz, 1H), 1.30 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 200.7, 172.4, 142.6, 136.8, 133.3, 133.0, 132.0, 130.8, 129.0, 128.8, 127.9, 127.7, 127.3, 127.3, 127.1, 125.9, 125.2, 125.2, 123.8, 122.5, 68.4, 62.9, 61.2, 59.7, 54.2, 14.2; HRMS (ESI) m/z calcd for C30H28NO3+ (M + H)+ 450.20637; found: 450.20651.
Ethyl 4-benzoyl-3-phenyl-5-(thiophen-2-yl)pyrrolidine-2-carboxylate (4i): White solid (80.9 mg, 80% yield); m.p. 160–162 °C; 1H NMR (500 MHz, CDCl3) δ 7.72 (d, J = 7.5 Hz, 2H), 7.48 (t, J = 7.4 Hz, 1H), 7.40 (d, J = 7.4 Hz, 2H), 7.34 (dd, J = 17.2, 7.9 Hz, 4H), 7.24 (m, 1H), 7.05 (d, J = 4.5 Hz, 1H), 6.77–6.69 (m, 2H), 5.30 (d, J = 8.3 Hz, 1H), 4.57 (t, J = 8.2 Hz, 1H), 4.27 (dq, J = 10.8, 7.1 Hz, 1H), 4.19–4.14 (m, 2H), 4.13 (dd, J = 8.1, 5.7 Hz, 1H), 1.19 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 197.4, 172.8 8, 143.2, 140.2, 137.3, 133.0, 128.7, 128.4, 128.1, 127.9, 127.1, 126.6, 125.5, 124.7, 67.2, 61.7, 61.2, 60.3, 51.6, 14.1; HRMS (ESI) m/z calcd for C24H24NO3S+ (M + H)+ 406.14714; found: 406.14761.
Ethyl 4-(4-nitrobenzoyl)-3,5-diphenylpyrrolidine-2-carboxylate (4j): Yellow solid (52.3 mg, 47% yield); m.p. 157–159 °C; 1H NMR (500 MHz, CDCl3) δ 8.06 (d, J = 7.0 Hz, 2H), 7.65–7.59 (m, 2H), 7.43–7.39 (m, 2H), 7.37–7.35 (m, 2H), 7.28 (d, J = 8.6 Hz, 1H), 7.13–7.05 (m, 5H), 5.04 (d, J = 8.9 Hz, 1H), 4.51 (t, J = 8.6 Hz, 1H), 4.33–4.24 (m, 1H), 4.21–4.17 (m, 1H), 4.17–4.12 (m, 2H), 1.19 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 197.6, 172.7, 147.5, 147.1, 139.8, 137.0, 133.5, 128.8, 128.6, 128.4, 128.0, 127.8, 127.4, 123.2, 67.5, 65.3, 61.4, 59.9, 52.1, 14.1; HRMS (ESI) m/z calcd for C26H25N2O5+ (M + H)+ 445.17580; found: 445.17587.
Ethyl 4-(4-bromobenzoyl)-3,5-diphenylpyrrolidine-2-carboxylate (4k): Creamy-white (76.7 mg, 64% yield); m.p. 112–116 °C; 1H NMR (500 MHz, CDCl3) δ: 7.41–7.37 (m, 6H), 7.35 (m, 2H), 7.30–7.23 (m, 1H), 7.16–7.06 (m, 5H), 5.00 (d, J = 8.8 Hz, 1H), 4.46 (dq, J = 8.7, 7.7 Hz, 1H), 4.28 (dq, J = 8.7, 7.7 Hz, 1H), 4.21–4.15 (m, 1H), 4.11 (dd, J = 17.3, 9.7 Hz, 2H), 1.19 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 197.7, 172.7, 140.6, 138.9, 136.2, 131.5, 129.5, 128.8, 128.3, 127.9, 127.8, 127.4, 127.2, 67.7, 66.7, 61.3, 60.8, 52.9, 14.1; HRMS (ESI) m/z calcd for C26H25BrNO3+ (M + H)+ 478.10123; found: 478.10175.
Ethyl 4-(4-methylbenzoyl)-3,5-diphenylpyrrolidine-2-carboxylate (4l): White solid (86.9 mg, 84% yield); m.p. 125–127 °C; 1H NMR (500 MHz, CDCl3) δ: 7.49 (d, J = 8.2 Hz, 2H), 7.40 (d, J = 7.4 Hz, 2H), 7.34 (t, J = 7.6 Hz, 2H), 7.24 (t, J = 7.3 Hz, 1H), 7.17–7.05 (m, 7H), 5.01 (d, J = 8.7 Hz, 1H), 4.54 (t, J = 8.4 Hz, 1H), 4.28 (dq, J = 10.7, 7.1 Hz, 1H), 4.20–4.14 (m, 2H), 4.14–4.08 (m, 1H), 2.32 (s, 3H), 1.19 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 198.7, 172.9, 139.2, 137.6, 137.5, 136.7, 128.2, 128.1, 128.1, 127.7, 127.6, 127.4, 67.9, 66.7, 61.2, 60.7, 52.5, 21.1, 14.2; HRMS (ESI) m/z calcd for C27H28NO3+ (M + H)+ 414.20637; found: 414.20679.
Ethyl 4-(4-methoxybenzoyl)-3,5-diphenylpyrrolidine-2-carboxylate (4m): White solid (86.9 mg, 81% yield); m.p. 122–124 °C; 1H NMR (500 MHz, CDCl3) δ: 7.58 (d, J = 8.9 Hz, 2H), 7.41–7.37 (m, 2H), 7.34 (d, J = 10.4, 4.9 Hz, 2H), 7.26–7.22 (m, 1H), 7.18–7.06 (m, 5H), 6.75 (dd, J = 9.4, 2.4 Hz, 2H), 4.99 (d, J = 8.7 Hz, 1H), 4.50 (t, J = 8.3 Hz, 1H), 4.28 (dq, J = 7.1, 11.7 Hz, 1H), 4.17 (dq, J = 7.1, 11.7 Hz, 1H), 4.15 (dd, J = 9.3 Hz, 8.7 Hz, 2H), 4.10 (t, J = 8.0 Hz, 1H), 3.81 (s, 3H), 1.19 (t, J = 8.0 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 196.8, 172.9, 163.3, 140.8, 139.4, 130.7, 130.4, 128.7, 128.1, 127.9, 127.6, 127.4, 127.0, 113.4, 67.8, 66.8, 61.2, 60.3, 55.4, 52.9, 14.2; HRMS (ESI) m/z calcd for C27H28NO4+ (M + H)+ 430.20128; found: 430.20175.
Ethyl 4-benzoyl-3-(4-nitrophenyl)-5-phenylpyrrolidine-2-carboxylate (4n): Yellow solid (51.2 mg, 46% yield); m.p. 122–124 °C; 1H NMR (500 MHz, CDCl3) δ: 8.20 (d, J = 8.7 Hz, 2H), 7.58 (dd, J = 11.2, 8.3 Hz, 4H), 7.49–7.40 (m, 1H), 7.35–7.26 (m, 2H), 7.15–7.03 (m, 5H), 5.06 (d, J = 9.1 Hz, 1H), 4.56 (t, J = 9.2 Hz, 1H), 4.30–4.23 (m, 2H), 4.22–4.18 (m, 1H), 4.18–4.12 (m, 1H), 1.17 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 197.6, 172.7, 147.5, 147.1, 139.8, 137.0, 133.5, 128.8, 128.6, 128.4, 128.0, 127.8, 127.4, 123.2, 67.5, 65.3, 61.4, 59.9, 52.1, 14.1; HRMS (ESI) m/z calcd for C26H25N2O5+ (M + H)+ 445.17580; found: 445.17587.
Ethyl 4-benzoyl-3-(4-bromophenyl)-5-phenylpyrrolidine-2-carboxylate (4o): White solid (59.9 mg, 50% yield); m.p. 132–134 °C; 1H NMR (500 MHz, CDCl3) δ: 7.55 (d, J = 7.2 Hz, 2H), 7.47–7.45 (m, 2H), 7.42 (dd, J = 4.9, 3.7 Hz, 1H), 7.30–7.27 (m, 4H), 7.11–7.07 (m, 5H), 5.00 (d, J = 9.0 Hz, 1H), 4.49 (t, J = 8.7 Hz, 1H), 4.27 (dq, J = 14.3, 7.1 Hz, 1H), 4.17 (dd, J = 17.9, 7.1 Hz,1H), 4.10 (m, 2H), 1.20 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 197.9, 172.7, 139.5, 139.3, 137.3, 132.9, 131.8, 129.7, 128.3, 128.2, 128.0, 127.7, 127.5, 120.9, 67.4, 66.4, 61.3, 60.8, 51.8, 14.2; HRMS (ESI) m/z calcd for C26H25BrNO3+ (M + H)+ 478.10123; found: 478.10129.
Ethyl 4-benzoyl-3-(4-methoxyphenyl)-5-phenylpyrrolidine-2-carboxylate (4p): White solid (89.2 mg, 83% yield); m.p. 98–99 °C; 1H NMR (500 MHz, CDCl3) δ: 7.56 (d, J = 7.2 Hz, 2H), 7.42 (t, J = 7.4 Hz, 1H), 7.32 (d, J = 8.7 Hz, 2H), 7.30–7.25 (m, 2H), 7.15–7.05 (m, 5H), 6.88 (d, J = 8.7 Hz, 2H), 5.00 (d, J = 8.8 Hz, 1H), 4.54–4.48 (t, J = 8.6 Hz 1H), 4.28 (dq, J = 10.7, 7.1 Hz, 1H), 4.17 (dq, J = 10.7, 7.1 Hz, 1H), 4.08 (dd, J = 17.4, 9.4 Hz, 2H), 3.80 (s, 3H), 1.21 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 198.5, 173.0, 158.6, 139.4, 137.5, 132.8, 132.5, 128.8, 128.2, 128.1, 128.0, 127.6, 127.4, 114.1, 67.7, 66.5, 61.2, 60.8, 55.2, 52.1, 14.2; HRMS (ESI) m/z calcd for C27H28NO4+ (M + H)+ 430.20128; found: 430.20151.
Ethyl 4-benzoyl-3-(3-methoxyphenyl)-5-phenylpyrrolidine-2-carboxylate (4q): White solid (90.2 mg, 84% yield); m.p. 104–106 °C; 1H NMR (500 MHz, CDCl3) δ: 7.57 (t, J = 9.2 Hz, 2H), 7.44–7.36 (m, 2H), 7.34 (t, J = 7.6 Hz, 1H), 7.30–7.22 (m, 2H), 7.19–7.05 (m, 5H), 7.03–6.85 (m, 1H), 6.75 (d, J = 8.9 Hz, 1H), 5.00 (d, J = 8.7, 3.8 Hz, 1H), 4.52 (dt, J = 23.8, 8.2 Hz, 1H), 4.32–4.23 (m, 1H), 4.16 (dd, J = 12.1, 8.2 Hz, 2H), 4.10 (dd, J = 17.5, 9.3 Hz, 1H), 3.81 (s, 3H), 1.20 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 198.6, 172.9, 159.8, 142.4, 139.1, 137.5, 132.8, 129.7, 128.2, 128.1, 128.1, 127.6, 127.4, 120.1, 113.9, 112.2, 77.3, 77.1, 76.8, 67.7, 66.7, 61.2, 60.6, 55.2, 52.9, 14.2; HRMS (ESI) m/z calcd for C27H28NO4+ (M + H)+ 430.20128; found: 430.20151.
Ethyl 4-benzoyl-5-phenyl-3-(p-tolyl)pyrrolidine-2-carboxylate (4r): White solid (93.4 mg, 90% yield); m.p. 149–150 °C; 1H NMR (500 MHz, CDCl3) δ: 7.55 (d, J = 7.2 Hz, 2H), 7.41 (t, J = 7.4 Hz, 1H), 7.30–7.24 (m, 4H), 7.18–7.06 (m, 7H), 5.00 (d, J = 8.7 Hz, 1H), 4.55–4.51 (t, J = 8.4 Hz, 1H), 4.33–4.25 (m, 1H), 4.18 (dq, J = 10.7, 7.1 Hz, 1H), 4.09 (dd, J = 18.6, 10.8 Hz, 2H), 2.33 (s, 3H), 1.21 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 198.7, 173.0, 139.3, 137.6, 137.5, 136.7, 128.2, 128.1, 128.1, 127.7, 127.6, 127.4, 67.9, 66.7, 61.2, 60.7, 52.5, 21.1, 14.2; HRMS (ESI) m/z calcd for C27H28NO3+ (M + H)+ 414.20637; found: 430.20654.
Ethyl 4-(2-hydroxybenzoyl)-3,5-diphenylpyrrolidine-2-carboxylate (4s): White solid (53.2 mg, 51% yield); m.p. 160–162 °C; 1H NMR (500 MHz, CDCl3) δ: 11.79 (s, 1H), 7.61 (d, J = 6.8 Hz, 1H), 7.36 (qd, J = 8.3, 1.3 Hz, 6H), 7.21–7.16 (m, 2H), 7.16–7.08 (m, 3H), 6.79 (dd, J = 18.2, 8.2 Hz, 2H), 5.06 (d, J = 8.8 Hz, 1H), 4.59 (t, J = 8.5 Hz, 1H), 4.28 (dq, J = 10.7, 7.2 Hz, 1H), 4.18 (dd, J = 10.1, 3.6 Hz, 2H), 4.16–4.11 (m, 1H), 1.18 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 203.8, 172.8, 162.3, 140.1, 138.9, 136.2, 129.9, 128.8, 128.2, 127.9, 127.8, 127.2, 127.1, 120.0, 118.6, 118.2, 67.6, 67.0, 61.3, 60.1, 52.1, 14.1; HRMS (ESI) m/z calcd for C26H26NO4+ (M + H)+ 416.18563; found: 416.18570.
Ethyl 4-(2-naphthoyl)-3,5-diphenylpyrrolidine-2-carboxylate (4t): White solid (91.1 mg, 81% yield); m.p. 118–120 °C; 1H NMR (500 MHz, CDCl3) δ 8.14 (s, 1H), 7.86 (d, J = 8.0 Hz, 1H), 7.79 (d, J = 8.0 Hz, 1H), 7.69 (d, J = 8.7 Hz, 1H), 7.62–7.49 (m, 3H), 7.47–7.42 (m, 2H), 7.36 (t, J = 7.7 Hz, 2H), 7.27–7.24 (m, 1H), 7.17–7.13 (m, 2H), 7.08–6.99 (m, 3H), 5.11 (d, J = 8.8 Hz, 1H), 4.75–4.70 (t, J = 8.5 Hz, 1H), 4.30 (dq, J = 10.8, 7.1 Hz, 1H), 4.20 (dd, J = 15.1, 6.5 Hz, 2H), 4.18–4.11 (m, 1H), 1.20 (t, J = 7.1 Hz, 1H); 13C NMR (125 MHz, CDCl3) δ: 198.2, 172.9, 140.6, 139.3, 135.3, 134.8, 132.2, 129.8, 129.4, 128.7, 128.4, 128.1, 127.9, 127.6, 127.6, 127.3, 127.1, 123.8, 67.7, 66.7, 61.2, 60.7, 52.8, 14.1; HRMS (ESI) m/z calcd for C30H28NO3+ (M + H)+ 450.20637; found: 450.20651.
Ethyl 3,5-diphenyl-4-(thiophene-2-carbonyl)pyrrolidine-2-carboxylate (4u): White solid (76.1 mg, 75% yield); m.p. 128–130 °C; 1H NMR (500 MHz, CDCl3) δ: 7.47 (d, J = 3.8 Hz, 1H), 7.41 (dd, J = 3.8, 1.1 Hz, 1H), 7.40–7.33 (m, 4H), 7.27 (ddd, J = 7.1, 5.8, 1.5 Hz, 2H), 7.21 (dd, J = 8.2, 1.2 Hz, 2H), 7.19–7.09 (m, 3H), 6.95 (dd, J = 4.9, 3.9 Hz, 1H), 5.00 (d, J = 8.5 Hz, 1H), 4.34–4.30 (m, 1H), 4.30–4.25 (m, 1H), 4.22–4.12 (m, 2H), 4.11–4.07 (m, 1H), 1.21 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 191.1, 172.7, 145.0, 140.8, 138.7, 133.8, 131.9, 128.7, 128.2, 127. 8, 127.7, 127.7, 127.3, 127.1, 67.8, 67.1, 62.1, 61.2, 52.8, 14.1; HRMS (ESI) m/z calcd for C24H24NO3S+ (M + H)+ 406.14714; found: 406.14755.
Methyl 4-benzoyl-3,5-diphenylpyrrolidine-2-carboxylate (4v):
30
White solid (86.9 mg, 90% yield); m.p. 159–161 °C (Lit.
30
150–154°C); 1H NMR (500 MHz, CDCl3) δ: 7.55 (d, J = 7.4 Hz, 2H), 7.44–7.38 (m, 3H), 7.36 (t, J = 7.6 Hz, 2H), 7.25 (d, J = 8.0 Hz, 3H), 7.17–7.03 (m, 5H), 5.01 (d, J = 8.7 Hz, 1H), 4.59–4.42 (t, J = 8.4 Hz, 1H), 4.20 (d, J = 8.9 Hz, 1H), 4.16–4.12 (t, J = 7.2 Hz, 1H), 3.77 (s, 1H).
Ethyl 4-benzoyl-2-methyl-3,5-diphenylpyrrolidine-2-carboxylate (4w): Light yellow solid (60.1 mg, 58% yield); m.p. 143–145 °C; 1H NMR (500 MHz, CDCl3) δ: 7.61 (d, J = 7.2 Hz, 2H), 7.46–7.39 (m, 2H), 7.30 (dt, J = 8.0, 4.2 Hz, 5H), 7.26–7.20 (m, 1H), 7.19–7.12 (m, 2H), 7.12–7.06 (m, 3H), 5.05 (d, J = 9.5 Hz, 1H), 4.94–4.88 (m, 1H), 1.32 (s, 3H), 1.32 (t, J = 7.1 Hz, 3H); 13C NMR (125 MHz, CDCl3) δ: 197.7, 175.2, 139.6, 137.7, 137.6, 132.7, 128.7, 128.3, 128.2, 128.1, 128.0, 127.7, 127.6, 127.2, 68.5, 64.1, 61.6, 56.8, 55.0, 21.4, 14.2; HRMS (ESI) m/z calcd for C27H28NO3+ (M + H)+ 414.20637; found: 414.20648.