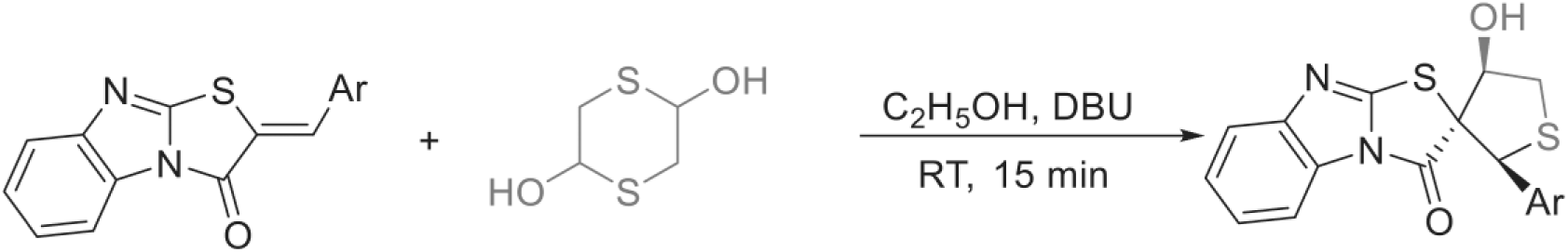
General procedure for the synthesis of (2RS,2′SR,4′SR)-2-aryl-4-hydroxyspiro[benzo[4,5]imidazo[2,1-b][1,3]thiazole-2,3-thiolan]-3-one
A mixture of (Z)-2-arylmethylidene-benzo[4,5]imidazo[2,1-b]thiazol-3(2H)-ones 1 (1 mmol), 1,4-dithiane-2,5-diol (1 mmol) and DBU (50 mol%) in tetrahydrofuran (THF) (5 mL) was stirred at room temperature for 2 h. The solvent was removed, and the product was purified by flash column using petroleum/ether–ethyl acetate mixture (3:1 v/v) as eluent to afford 3.
(2RS,2′SR,4′SR)-4′-hydroxy-2′-phenylspiro[benzo[4,5]imidazo[2,1-b][1,3]thiazole-2,3-thiolan]-3-one (3a): White solid, yield 50%; m.p.: 230–233 °C; 1H NMR (DMSO-d6, 500 MHz): 2.92 (t, J = 10.0 Hz, 1H), 3.40 (dd, J1 = 10.5 Hz, J2 = 7.5 Hz, 1H), 5.06 (dt, J1 = 16.0 Hz, J2 = 7.0 Hz, 1H), 5.45 (s, 1H), 6.57 (d, J = 6.0 Hz, 1H), 7.28–7.33 (m, 3H), 7.34–7.40 (m, 4H), 7.53 (d, J = 8.0 Hz, 1H), 7.85 (d, J = 7.5 Hz, 1H); 13C NMR (DMSO-d6, 125 MHz): 33.30, 51.83, 79.29, 85.01, 112.16, 118.82, 124.14, 126.19, 128.37, 128.73, 128.79, 129.17, 133.93, 148.47, 155.41, 166.63; IR (KBr) v: 1738.4 cm−1; high-resolution electrospray ionization mass spectrometer (ESI-HRMS) calc. for [C18H15N2O2 S2]+ (M + H): 355.0569; found: 355.0569.
(2RS,2′SR,4′SR)-2′-(4-fluorophenyl)-4′-hydroxyspiro[benzo[4,5]imidazo [2,1-b][1,3]thiazole-2,3-thiolan]-3-one (3b): White solid, yield 58%; m.p.: 276–278 °C; 1H NMR (DMSO-d6, 500 MHz): 2.93 (t, J = 10.0 Hz, 1H), 3.41 (dd, J1 = 10.5 Hz, J2 = 7.5 Hz, 1H), 5.05 (dt, J1 = 14.0 Hz, J2 = 7.0 Hz, 1H), 5.46 (s, 1H), 6.58 (d, J = 5.5 Hz, 1H), 7.14 (t, J = 8.5 Hz, 2H), 7.35 (dt, J1 = 20.0 Hz, J2 = 7.5 Hz, 2H), 7.43 (dd, J1 = 8.5 Hz, J2 = 5.5 Hz, 2H), 7.54 (d, J = 8.0 Hz, 1H), 7.85 (d, J = 7.5 Hz, 1H); 13C NMR (DMSO-d6, 125 MHz): 33.44, 51.03, 79.20, 84.99, 112.18, 115.19, 115.37, 118.85, 124.16, 126.22, 129.19, 130.16, 130.18, 130.88, 130.95, 148.49, 155.27, 166.52; IR (KBr) v: 1736.4 cm−1; ESI-HRMS calc. for [C18H14FN2O2 S2]+ (M + H): 373.0475; found: 373.0476.
(2RS,2′SR,4′SR)-2′-(4-chlorophenyl)-4′-hydroxyspiro[benzo[4,5]imidazo [2,1-b][1,3]thiazole-2,3-thiolan]-3-one (3c): White solid, yield 48%; m.p.: 284–287 °C; 1H NMR (DMSO-d6, 500 MHz): 2.94 (t, J = 10.0 Hz, 1H), 3.41 (dd, J1 = 10.5 Hz, J2 = 7.5 Hz, 1H), 5.06 (dt, J1 = 14.0 Hz, J2 = 7.0 Hz, 1H), 5.47 (s, 1H), 6.60 (d, J = 6.0 Hz, 1H), 7.32–7.36 (m, 2H), 7.37–7.41 (m, 4H), 7.55 (d, J = 8.0 Hz, 1H), 7.86 (d, J = 7.5 Hz, 1H); 13C NMR (DMSO-d6, 125 MHz): 33.46, 51.05, 79.30, 84.80, 112.21, 118.86, 124.17, 126.23, 128.40, 129.21, 130.62, 133.14, 133.35, 148.50, 155.21, 166.47; IR (KBr) v: 1737.1 cm−1; ESI-HRMS calc. for [C18H14ClN2O2 S2]+ (M + H): 389.0180; found: 389.0184.
(2RS,2′SR,4′SR)-2′-(4-bromophenyl)-4′-hydroxyspiro[benzo[4,5]imidazo [2,1-b][1,3]thiazole-2,3-thiolan]-3-one (3d): White solid, yield 53%; m.p.: 280–281 °C; 1H NMR (DMSO-d6, 500 MHz): 2.94 (t, J = 10.0 Hz, 1H), 3.41 (dd, J1 = 10.5 Hz, J2 = 7.5 Hz, 1H), 5.05 (dd, J1 = 16.0 Hz, J2 = 7.0 Hz, 1H), 5.45 (s, 1H), 6.59 (d, J = 6.0 Hz, 1H), 7.32–7.39 (m, 4H), 7.50 (d, J = 8.5 Hz, 2H), 7.55 (d, J = 8.0 Hz, 1H), 7.85 (d, J = 7.5 Hz, 1H); 13C NMR (DMSO-d6, 125 MHz): 33.46, 51.11, 79.33, 84.72, 112.21, 118.87, 121.98, 124.18, 126.24, 129.22, 130.91, 131.33, 133.59, 148.50, 155.20, 166.46; IR (KBr) v: 1736.7 cm−1; ESI-HRMS calc. for [C18H14BrN2O2 S2]+ (M + H): 432.9675; found: 432.9680.
(2RS,2′SR,4′SR)-4′-hydroxy-2′-(4-methoxyphenyl)spiro[benzo[4,5]imidazo [2,1-b][1,3]thiazole-2,3-thiolan]-3-one (3e): White solid, yield 52%; m.p.: 254–256 °C; 1H NMR (DMSO-d6, 500 MHz): 2.90 (t, J = 10.0 Hz, 1H), 3.38 (dd, J1 = 10.5 Hz, J2 = 7.5 Hz, 1H), 3.65 (s, 3H), 5.04 (dt, J1 = 13.5 Hz, J2 = 7.5 Hz, 1H), 5.38 (s, 1H), 6.54 (d, J = 6.0 Hz, 1H), 6.84 (d, J = 8.5 Hz, 2H), 7.30–7.32 (m, 2H), 7.33–7.38 (m, 2H), 7.54 (d, J = 8.0 Hz, 1H), 7.84 (d, J = 7.5 Hz, 1H); 13C NMR (DMSO-d6, 125 MHz): 33.30, 51.35, 54.97, 79.19, 85.32, 112.15, 113.68, 118.81, 124.12, 125.45, 126.19, 129.18, 130.05, 148.49, 159.37, 166.67; IR (KBr) v: 1736.4 cm−1; ESI-HRMS calc. for [C19H17N2O3 S2]+ (M + H): 385.0675; found: 385.0679.
(2RS,2′SR,4′SR)-4′-hydroxy-2′-(4-(methylthio)phenyl)spiro[benzo[4,5]imidazo [2,1-b][1,3]thiazole-2,3-thiolan]-3-one (3f): White solid, yield 46%; m.p.: 248–250 °C; 1H NMR (DMSO-d6, 500 MHz): 2.38 (s, 3H), 2.92 (t, J = 10.0 Hz, 1H), 3.39 (dd, J1 = 10.5 Hz, J2 = 7.5 Hz, 1H), 5.05 (dt, J1 = 16.0 Hz, J2 = 7.0 Hz, 1H), 5.41 (s, 1H), 6.57 (d, J = 6.0 Hz, 1H), 7.16 (d, J = 8.0 Hz, 2H), 7.29–7.33 (m, 2H), 7.34–7.39 (m, 2H), 7.54 (d, J = 8.0 Hz, 1H), 7.85 (d, J = 7.5 Hz, 1H); 13C NMR (DMSO-d6, 125 MHz): 14.04, 33.32, 51.36, 79.30, 85.06, 112.19, 118.85, 124.16, 125.23, 126.22, 129.20, 129.28, 130.13, 139.15, 148.49, 155.41, 166.60; IR (KBr) v: 1743.6 cm−1; ESI-HRMS calc. for [C19H17N2O2 S3]+ (M + H): 401.0447; found: 401.0446.
(2RS,2′SR,4′SR)-4′-hydroxy-2′-(p-tolyl)spiro[benzo[4,5]imidazo [2,1-b][1,3]thiazole-2,3-thiolan]-3-one (3g): White solid, yield 61%; m.p.: 262–264 °C; 1H NMR (DMSO-d6, 500 MHz): 2.18 (s, 3H), 2.91 (t, J = 10.0 Hz, 1H), 3.39 (dd, J1 = 10.5 Hz, J2 = 7.5 Hz, 1H), 5.05 (dt, J1 = 13.5 Hz, J2 = 7.5 Hz, 1H), 5.40 (s, 1H), 5.56 (d, J = 5.5 Hz, 1H), 7.08 (d, J = 8.0 Hz, 2H), 7.27–7.29 (m, 2H), 7.30–7.39 (m, 2H), 7.53 (d, J = 8.0 Hz, 1H), 7.85 (d, J = 7.5 Hz, 1H); 13C NMR (DMSO-d6, 125 MHz): 20.55, 33.26, 51.61, 79.31, 85.14, 112.15, 118.80, 124.12, 126.18, 128.62, 128.94, 129.19, 130.85, 138.20, 148.49, 155.51, 166.68; IR (KBr) v: 1736.1 cm−1; ESI-HRMS calc. for [C19H17N2O2 S2]+ (M + H): 369.0726; found: 369.0724.
(2RS,2′SR,4′SR)-4′-hydroxy-2′-(3-methoxyphenyl)spiro[benzo [4,5]imidazo[2,1-b][1,3]thiazole-2,3-thiolan]-3-one (3h): White solid, yield 56%; m.p.: 196–198 °C; 1H NMR (DMSO-d6, 500 MHz): 2.92 (t, J = 10.0 Hz, 1H), 3.39 (dd, J1 = 10.5 Hz, J2 = 7.5 Hz, 1H), 3.64 (s, 3H), 5.06 (dd, J1 = 15.5 Hz, J2 = 7.0 Hz, 1H), 5.43 (s, 1H), 6.57 (d, J = 6.0 Hz, 1H), 6.82 (d, J = 8.5 Hz, 1H), 6.90-6.95 (m, 2H), 7.20 (t, J = 8.0 Hz, 1H), 7.35 (dt, J1 = 19.5 Hz, J2 = 7.0 Hz, 2H), 7.54 (d, J = 8.0 Hz, 1H), 7.86 (d, J = 7.5 Hz, 1H); 13C NMR (DMSO-d6, 125 MHz): 33.28, 51.70, 54.95, 79.24, 84.90, 112.13, 113.89, 114.50, 118.83, 120.75, 124.16, 126.21, 129.20, 129.53, 135.55, 148.50, 155.49, 158.88, 166.71; IR (KBr) v: 1737.2 cm−1; ESI-HRMS calc. for [C19H17N2O3 S2]+ (M + H): 385.0675; found: 385.0678.
(2RS,2′SR,4′SR)-2′-(3,5-dimethoxyphenyl)-4′-hydroxyspiro[benzo[4,5]imidazo [2,1-b][1,3]thiazole-2,3-thiolan]-3-one (3i): White solid, yield 57%; m.p.: 190–191 °C; 1H NMR (DMSO-d6, 500 MHz): 2.90 (t, J = 10.0 Hz, 1H), 3.38 (dd, J1 = 9.5 Hz, J2 = 8.5 Hz, 1H), 3.61 (s, 6H), 5.04 (dd, J1 = 14.0 Hz, J2 = 7.0 Hz, 1H), 5.37 (s, 1H), 6.38 (s, 1H), 6.48 (s, 2H), 6.56 (d, J = 5.5 Hz, 1H), 7.36 (dt, J1 = 20.0 Hz, J2 = 7.5 Hz, 2H), 7.55 (d, J = 8.0 Hz, 1H), 7.87 (d, J = 7.5 Hz, 1H); 13C NMR (DMSO-d6, 125 MHz): 33.26, 51.80, 55.09, 79.14, 84.77, 99.96, 106.70, 112.08, 118.85, 124.19, 126.23, 129.22, 136.35, 148.52, 155.58, 160.12, 166.80; IR (KBr) v: 1745.9 cm−1; ESI-HRMS calc. for [C20H19N2O4 S2]+ (M + H): 415.0781; found: 415.0782.
(2RS,2′SR,4′SR)-4′-hydroxy-2′-(3,4,5-trimethoxyphenyl)spiro[benzo[4,5] imidazo[2,1-b][1,3]thiazole-2,3-thiolan]-3-one (3j): White solid, yield 52%; m.p.: 211–213 °C; 1H NMR (DMSO-d6, 500 MHz): 2.91 (t, J = 10.0 Hz, 1H), 3.39 (dd, J1 = 10.5 Hz, J2 = 7.5 Hz, 1H), 3.54 (s, 3H), 3.63 (s, 6H), 5.03 (dt, J1 = 14.0 Hz, J2 = 7.0 Hz, 1H), 5.38 (s, 1H), 6.56 (d, J = 6.0 Hz, 1H), 6.63 (s, 2H), 7.35 (dt, J1 = 19.5 Hz, J2 = 7.5 Hz, 2H), 7.54 (d, J = 7.5 Hz, 1H), 7.86 (d, J = 7.5 Hz, 1H); 13C NMR (DMSO-d6, 125 MHz): 33.40, 52.24, 55.74, 59.91, 78.85, 85.02, 105.83, 112.00, 118.85, 124.17, 126.21, 129.23, 129.50, 137.48, 148.50, 152.38, 155.56, 166.82; IR (KBr) v: 1736.3 cm−1; ESI-HRMS calc. for [C21H21N2O5 S2]+ (M + H): 445.0886; found: 445.0888.
(2RS,2′SR,4′SR)-2′-(4-(tert-butyl)phenyl)-4′-hydroxyspiro[benzo[4,5]imidazo [2,1-b][1,3]thiazole-2,3-thiolan]-3-one (3k): White solid, yield 50%; m.p.: 225–227 °C; 1H NMR (DMSO-d6, 500 MHz): 1.16 (s, 9H), 2.93 (t, J = 10.0 Hz, 1H), 3.39 (dd, J1 = 10.5 Hz, J2 = 7.5 Hz, 1H), 5.05 (dd, J1 = 16.0 Hz, J2 = 7.5 Hz, 1H), 5.42 (s, 1H), 6.56 (d, J = 6.0 Hz, 1H), 7.31–7.33 (m, 4H), 7.34–7.37 (m, 2H), 7.53 (d, J = 7.5 Hz, 1H), 7.85 (d, J = 7.5 Hz, 1H); 13C NMR (DMSO-d6, 125 MHz): 30.86, 33.21, 34.22, 51.22, 79.65, 84.87, 112.21, 118.81, 124.11, 125.21, 126.15, 128.58, 129.24, 131.02, 148.47, 151.14, 155.54, 166.74; IR (KBr) v: 1740.6 cm−1; ESI-HRMS calc. for [C22H23N2O2 S2]+ (M + H): 411.1195; found: 411.1199.