Abstract
Background:
An in-hospital double-dose influenza vaccination strategy’s effect on preventing major adverse cardiovascular events (MACE) in patients with previous stroke is still uncertain. This study is a prespecified analysis of the vaccine against influenza to avoid cardiovascular events after the Acute Coronary Syndrome (VIP-ACS) trial.
Methods:
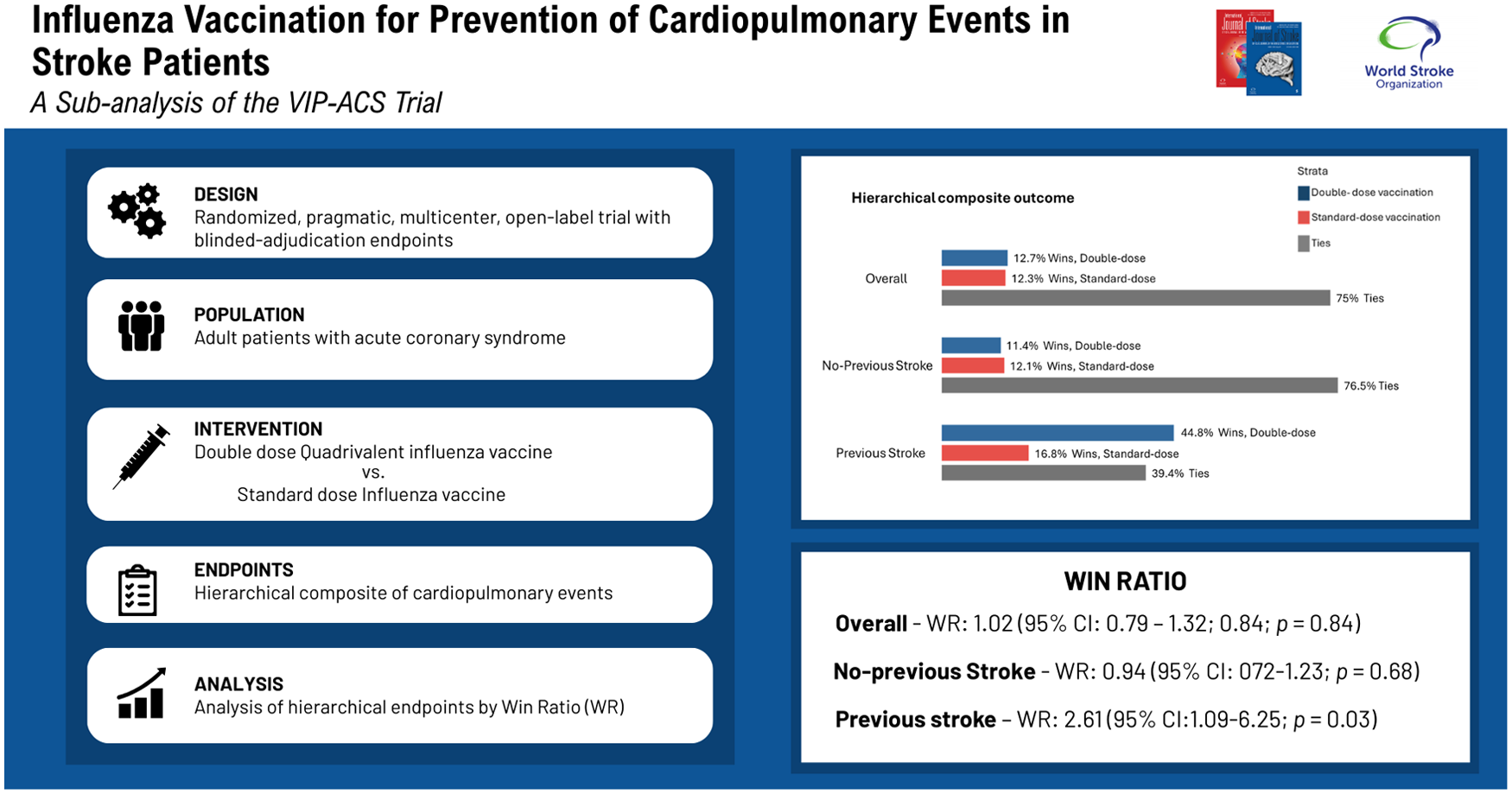
The VIP-ACS trial was a randomized, pragmatic, multicenter, open-label trial with blinded-adjudication endpoints. Adult patients with acute coronary syndrome (ACS) ⩽ seven days of hospitalization were randomized to an in-hospital double-dose quadrivalent inactivated influenza vaccine or a standard-dose vaccine at 30 days post-randomization. The primary endpoint was a hierarchical composite of all-cause death, myocardial infarction (MI), stroke, unstable angina, hospitalization for heart failure, urgent coronary revascularization, and hospitalization for respiratory causes, analyzed by the win ratio (WR) method. The secondary endpoint was a hierarchical composite consisting of CV death, MI and stroke (MACE). Patients were followed for 12 months each influenza season.
Results:
The trial enrolled 1801 patients (31% female). A total of 67 patients had a history of stroke. There were no significant differences between groups on the primary hierarchical endpoint: 11.4% wins (862 patients) in the double-dose vaccine group versus 12.1% wins (872 patients) in the standard-dose vaccination group (WR: 0.94; 95% CI: 0.72–1.24; p = 0.69) without a history of stroke. However, in-hospital double-dose vaccination favored individuals (34 patients) with previous stroke (WR: 2.62; 95% CI: 1.10–6.25; p = 0.03; 43.9% wins vs 16.8% wins). Results were consistent for hierarchical MACE (WR: 3.01; 95% CI: 1.15–7.88; p = 0.02; 41.3% wins vs 13.7% wins) in favor of in-hospital double-dose vaccination (34 patients).
Conclusions:
After an ACS, in-hospital double-dose influenza vaccination prevents hospitalizations and death compared with standard-dose vaccination at 12 months in individuals with previous strokes.
ClinicalTrials.gov number:
NCT04001504
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
