Abstract
Background:
Vertebral artery origin stenosis (VAOS) is a common cause of posterior circulation ischemic events, and endovascular treatment serves as an alternative treatment. However, conventional endovascular treatment methods are related to high risk of restenosis. It is unclear whether the drug-coated balloon (DCB) can reduce restenosis risk of VAOS.
Methods:
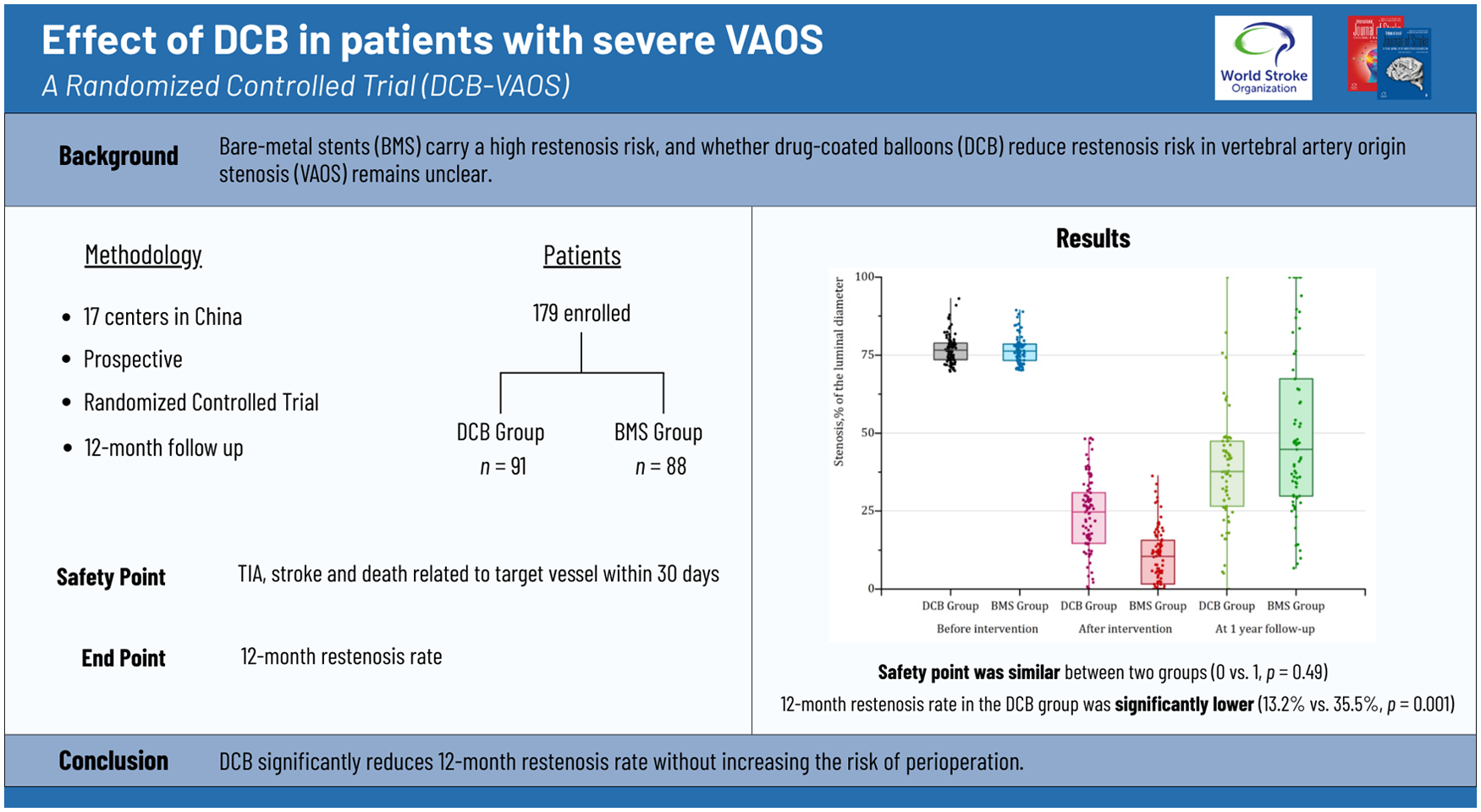
This was a prospective, multicenter, randomized trial conducted from 6 January 2020 to 1 October 2023 in China. Symptomatic patients with severe VAOS were randomly allocated in a 1:1 ratio to undergo either DCB or bare-metal stent (BMS) and followed up for 12 months. The primary safety endpoint was the incidence of transient ischemic attack, stroke, or death related to target vessel within 30 days post-procedure. The primary efficacy endpoint was the rate of 12-month restenosis.
Results:
A total of 179 patients were enrolled with 91 in the DCB group and 88 in the BMS group. No significant difference was observed in the rates of transient ischemic attack, stroke, or death related to target vessel within 30 days between the DCB and BMS groups (0 (0.0%) vs. 1 (1.1%); P = 0.49). The 12-month restenosis rate was significantly lower in the DCB group compared to the BMS group (10/76 (13.2%) vs. 27/76 (35.5%); risk ratio = 0.37; 95% confidence interval = 0.19 to 0.71; P = 0.001).
Conclusion:
This trial demonstrated that DCB may reduce restenosis risk in symptomatic patients with severe VAOS compared to BMS.
Registration:
URL: https://clinicaltrials.gov (unique identifier: NCT03910166).
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
