Abstract
Background:
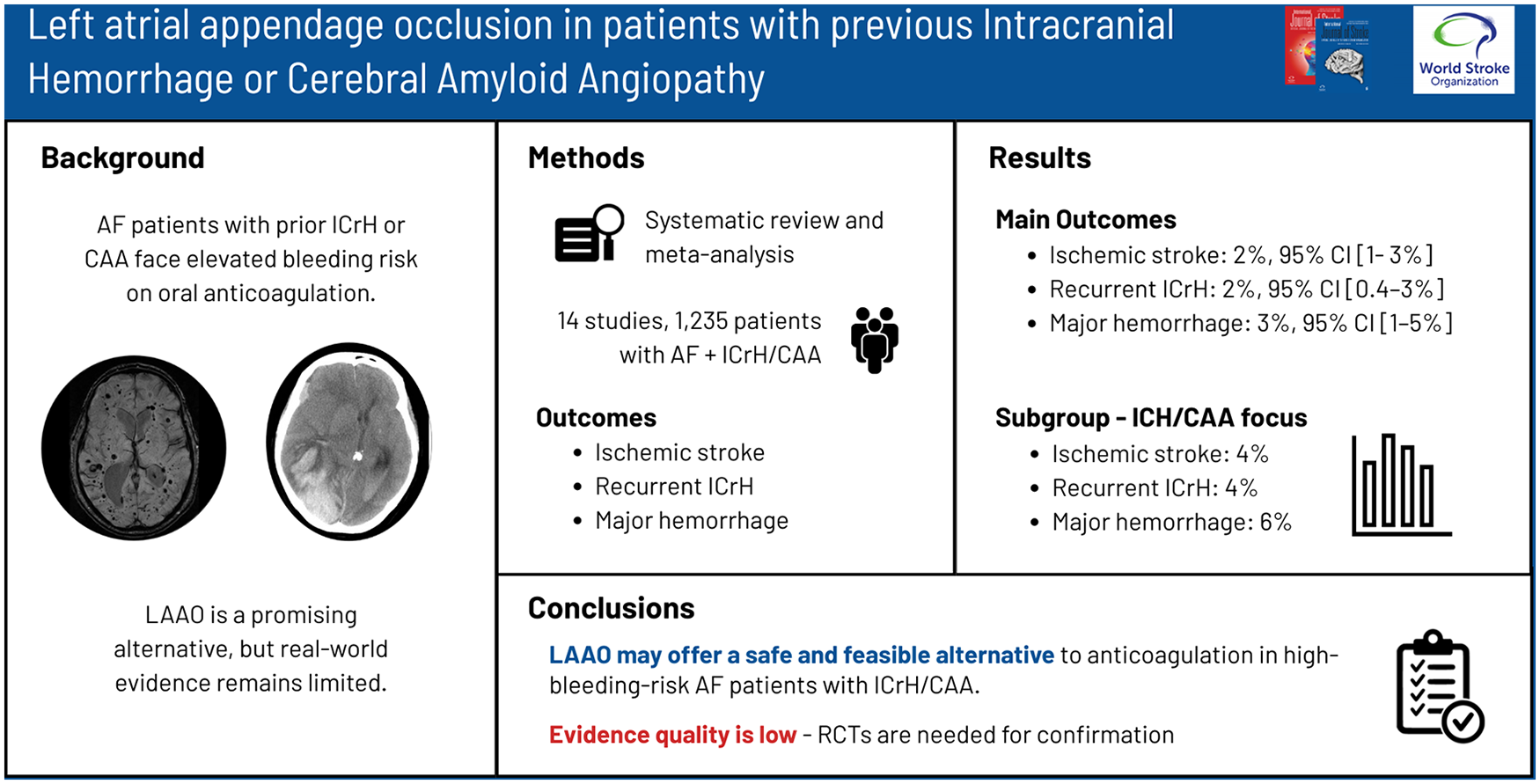
Patients with atrial fibrillation (AF) on oral anticoagulation (OAC) who have a history of intracranial hemorrhage (ICrH) or cerebral amyloid angiopathy (CAA) have an elevated risk of ICrH recurrence. Left atrial appendage occlusion (LAAO) has emerged as a potential alternative to OAC for stroke prevention in high-bleeding-risk AF patients. Small observational studies suggest that LAAO may be safe and feasible in patients with ICrH, when using single or dual antiplatelet therapy or short-term OAC post-procedure, though data remain limited. This systematic review and meta-analysis aims to consolidate evidence on the safety and efficacy of LAAO in patients with prior ICrH or CAA.
Methods:
We conducted a systematic review and meta-analysis examining the safety and efficacy of LAAO in patients with non-valvular AF and prior ICrH and/or CAA. PubMed/MEDLINE and EMBASE (via Scopus) databases were systematically searched from inception until 29 February 2024. Eligible studies included randomized control trials, observational studies, and case series (⩾10 participants) reporting stroke events in patients with AF and previous history of ICrH and/or CAA undergoing LAAO. Pooled incidence rates (IRs) with corresponding 95% confidence intervals (CIs) were calculated for primary outcomes (post-procedural ischemic stroke and recurrent ICrH) and secondary outcomes.
Results:
Fourteen studies including 1235 patients met inclusion criteria. The pooled average follow-up period was 17.1 months. The pooled IRs for ischemic stroke, recurrent ICrH, and major hemorrhage were 2% (95% CI: 1–3%, I2 = 2%), 2% (95% CI: 0.4–3%, I2 = 45%) and 3% (95% CI: 1–5%, I2 = 54%), respectively. In prespecified subgroup analyses of studies focusing on patients with intraparenchymal hemorrhage and/or CAA, pooled IRs for ischemic stroke, recurrent ICrH, and major hemorrhage IR of 4% (95% CI: 1–8%), 4% (95% CI: 0.4–10%) and 6% (95% CI: 3–12%), respectively.
Conclusions:
LAAO may be a safe and effective treatment for selected AF patients with a history of ICrH and/or CAA, but the quality of evidence is poor. Future randomized controlled trials are essential to validate LAAO’s efficacy and long-term safety.
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
