Abstract
Background:
Cerebral venous thrombosis (CVT) accounts for 0.5–1% of all strokes. The role of endovascular therapy (EVT) in the management of CVT remains controversial and variations in practice patterns are not well known.
Aims:
Here, we present a comprehensive, international characterization of practice patterns and perspectives on the use of EVT for CVT.
Methods:
A comprehensive 42-question survey was distributed to stroke clinicians globally from May to October 2023, asking about practice patterns and perspectives on the use of EVT for CVT.
Results:
The overall response rate was 31% (863 respondents of 2744 invited) across 61 countries. The majority of respondents (74%) supported the use of EVT for CVT in certain clinical situations. Key considerations for decision-making in using EVT favored clinical over radiographic/procedural factors and included worsening level of consciousness (86%) and worsening neurological deficits (76%). In the past 3 years, 56% of respondents used EVT for the treatment of CVT, with most (49.5%) involved in two to five cases. Among interventionalists, significant variability existed in the techniques used for EVT (p < 0.001), with aspiration thrombectomy (56%) and stent retriever (51%) being the most used overall. Regionally, interventionalists from China predominantly used intra-sinus heparin (56%), while this technique was most commonly ranked as “never indicated” throughout the rest of the world (23%). Post-procedure, low molecular weight heparin was the most used anticoagulant (83%), although North American respondents favored unfractionated heparin (37%), while imaging was primarily split between magnetic resonance (71.8%) and computed tomography (65.9%) arteriography or venography.
Conclusion:
Our survey reveals significant heterogeneity in approaches to EVT for CVT, and provides a comprehensive characterization of indications, techniques, and long-term management used by clinicians internationally. This resource will aid in optimizing patient selection and endovascular treatments for future trials.
Introduction
Cerebral venous thrombosis (CVT) is a rare form of stroke that leads to death or dependence in 10–15% of patients.1–3 Due to the rarity of CVT, there is a lack of large, randomized trials to inform management, and existing treatment guidelines are consensus-based, primarily derived from observational studies or small clinical trials with limited statistical power.4–6 Anticoagulation is the recommended first-line standard of care therapy, with endovascular therapy recommended for cases where there has been clinical deterioration despite anticoagulation.5,7
The role of endovascular therapy (EVT) as an adjunctive first-line treatment in the management of CVT remains an area of uncertainty with limited evidence and diverse practices among clinicians. Clinical trials in this area are hindered by undefined eligibility criteria and limited uniformity in approaches to endovascular treatment. The only randomized trial in this area, the Thrombolysis or Anticoagulation for Cerebral Venous Thrombosis (TO-ACT), was stopped early for futility and a propensity score analysis from the large observational Anticoagulation in the Treatment of Cerebral Venous Thrombosis (ACTION-CVT) trial did not demonstrate a survival or functional benefit for EVT in CVT.6,8 However, it is not clear whether EVT may benefit specific patient populations, and whether these populations vary by the technique used. Thus, decision-making around EVT in the treatment of CVT remains case-by-case and current practice patterns are not well known.
To gain a comprehensive understanding of international practices and perspectives on EVT for CVT, we conducted a large, global survey of stroke clinicians across 61 countries aimed at elucidating existing practices in management and influencing factors when considering EVT for CVT. This resource will aid in selecting optimal patient populations, endovascular techniques, and postoperative management for future clinical trials and in clinical practice.
Methods
This survey received ethics approval from our institutional ethics board, the University of British Columbia Clinical Research Ethics Board (approval number H22-02916), and all participants gave informed consent. This report is conducted according to the CROSS guidelines. 9 A survey comprising 42 questions (provided in Supplementary Data) was distributed to stroke neurologists, neurointerventionalists, neurosurgeons, and other relevant clinicians globally through local networks and professional societies, including the Society of Vascular and Interventional Neurology, the German Stroke Trials Network, the Cerebral Venous Thrombosis Consortium, and the Women in Neurointervention WhatsApp group. Survey responses were recorded between May 2023 and October 2023. The time of estimated survey completion was 5–10 min. The survey was sent by electronic mail and answered online using the Qualtrics platform (Qualtrics, Provo, UT), and submission was only possible upon completion of the complete survey. The questionnaire was designed to collect responses on four main categories: (1) respondent demographics and experience with EVT for CVT; (2) clinical, radiographical, and procedural factors considered when assessing a patient for EVT; (3) endovascular techniques used and those never indicated for use in CVT treatment; and (4) use and timing of post-EVT imaging and medical management. Participation was voluntary, and responses were anonymized. Respondents were given the option to enter a random draw for a US$500 gift card to Amazon upon completing the survey. The study complied with local institutional research board regulations and informed consent was implied upon completing the survey. The survey was distributed in English except in China, where participants completed a version translated into Chinese (Mandarin) by a professional medical translator and peer-reviewed by a native speaking co-author in China (Y.C.).
To identify regional differences in survey responses, countries were grouped by continent, specifically North America, Central America, South America, Europe, Asia, Oceania, and Africa. Regions were grouped based on sample size where appropriate. Given the distinct use of a translated survey for distribution in mainland China, we assessed responses from mainland China separately from the rest of Asia. Responses were also grouped by non-interventionalist stroke specialists and interventionalists (including interventional neurologists, interventional radiologists, interventional neuroradiologists, and neurosurgeons). Questions about technical considerations in EVT were directed through branching logic to interventionalists only.
Categorical data were summarized as counts and percentages, and comparisons were made using the chi-squared test. The Bonferroni correction was used in the form of p-value adjustments for all post hoc analyses. For questions where participants were able to answer more than once, percentages are given relative to the number of respondents, explaining summative percentages above 1.00. To visualize qualitative rankings of factors influencing the use of EVT in patients with CVT, categories (not important, somewhat important and very important) were converted into unweighted, ordered integers (1, 2, and 3, respectively). Sankey diagrams for post-procedural management included only respondents to both modality and timing questions. Statistical significance was set at p ⩽ 0.05. Authors B.A.B. and T.S.F. had full access to all the data in the study and take responsibility for its integrity and the data analysis.
Results
Respondent demographics and experience with EVT for CVT
There were 2744 invited participants. The overall response rate of the survey was 31%, consisting of 863 respondents across 61 countries (Figure 1). Respondent demographics and experience with EVT for CVT are summarized in Tables 1 and 2, respectively. Briefly, the majority of respondents were employed in an academic hospital with a comprehensive stroke center (75.9%), with years of experience varying widely across respondents (Table 1). We received responses from a wide range of relevant specialists, consisting mostly of stroke consultants (60.7%) and neurointerventionalists (34.6%).
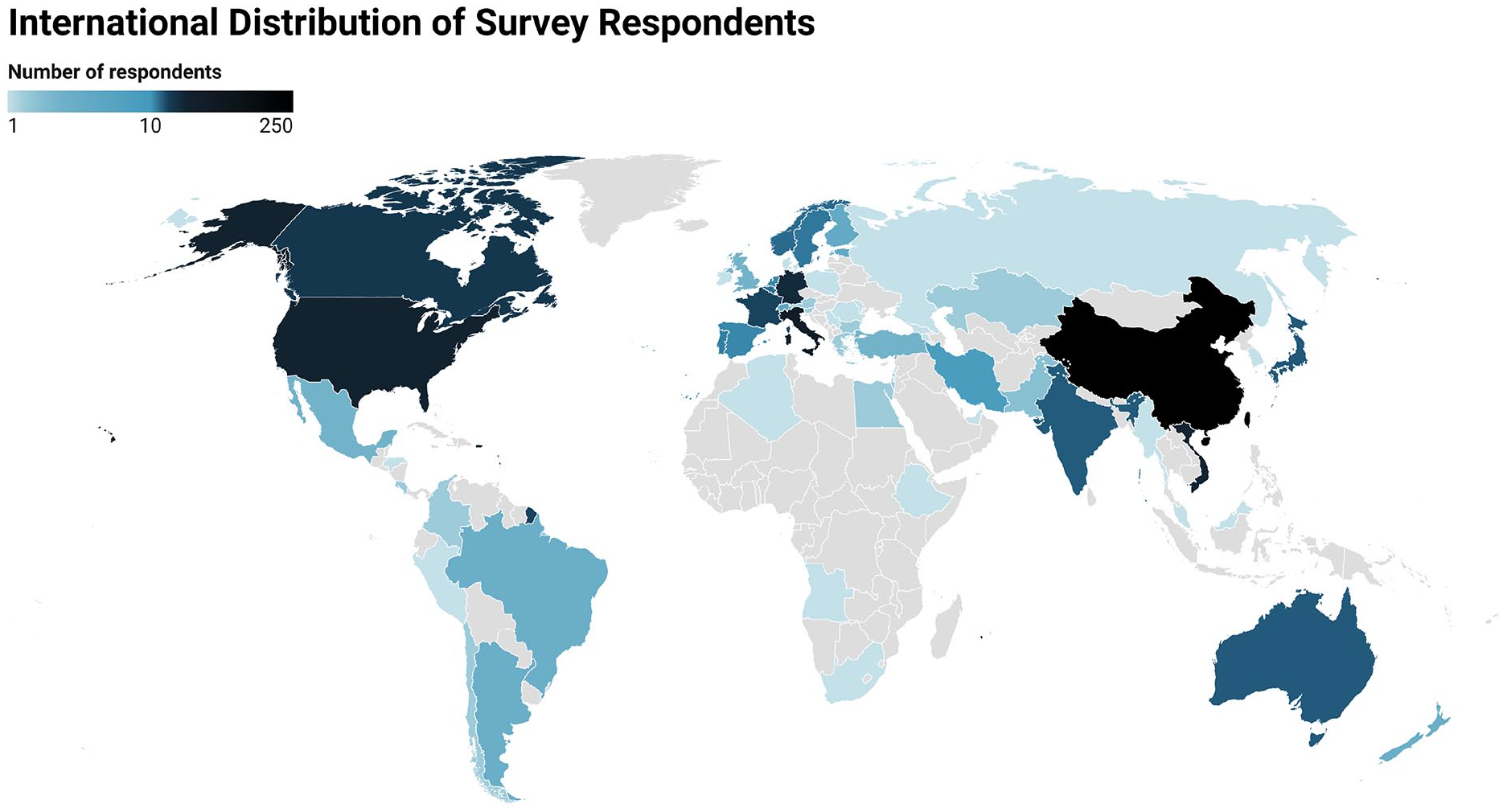
International distribution of the number of survey respondents by country.
Demographics.

Experience with EVT for CVT.
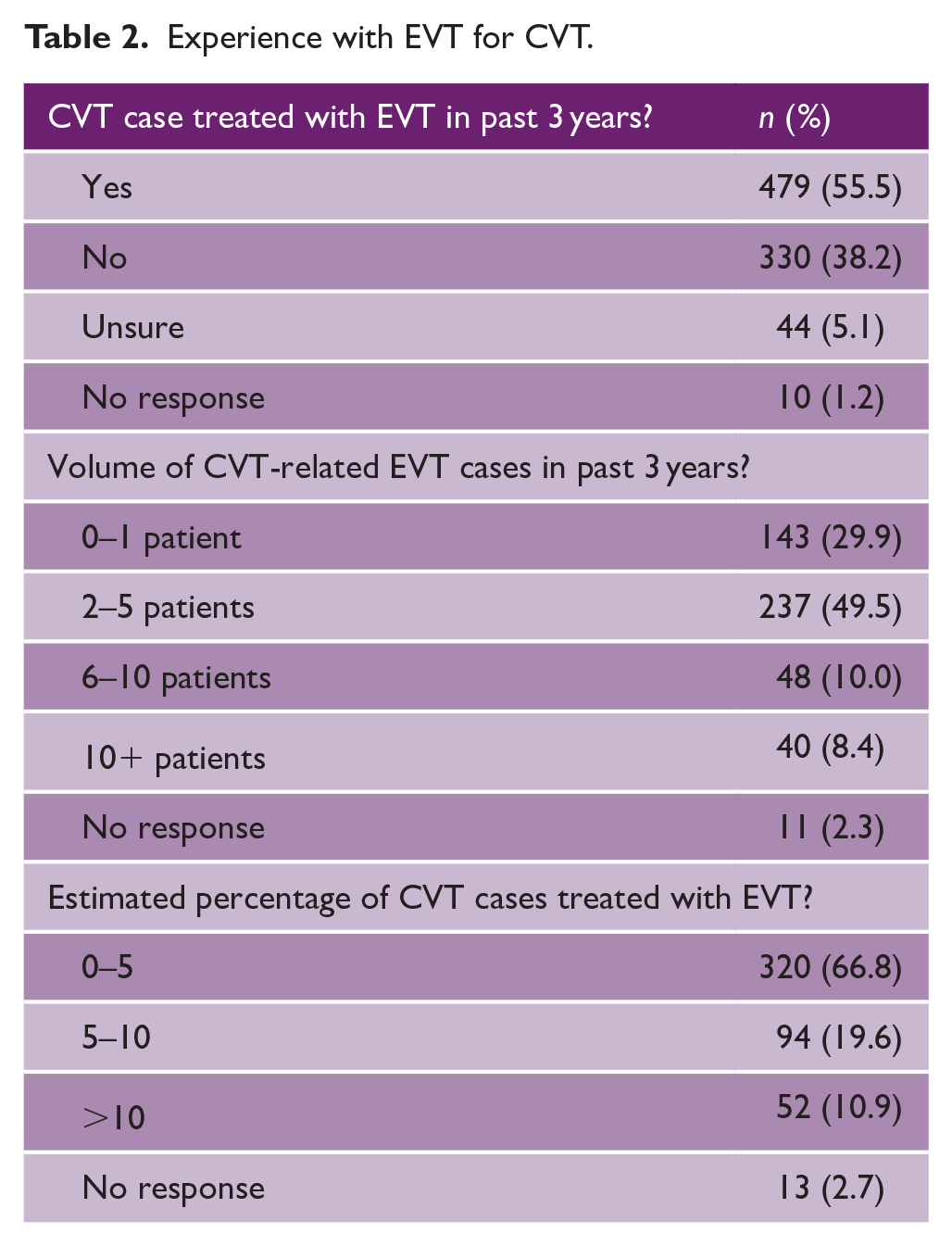
The majority of respondents had treated at least one case of CVT using EVT within the past 3 years (55.5%), and of those who had, most had done so in two to five patients (49.5%; Table 2). In addition, most of these respondents (66.8%) estimated that EVT was used in less than 5% of CVT cases (Table 2). The majority of respondents (74%) favored the use of EVT over standard medical treatment for patients with CVT in certain situations.
Factors influencing the decision to use EVT for CVT
Overall, clinical factors were ranked as more important than radiographic and procedural factors by respondents when considering the use of EVT for CVT treatment, although this was not a direct comparison made in the survey. In terms of clinical factors, worsening level of consciousness (LOC), worsening clinical deficits besides LOC, and having a trial of anticoagulation prior to EVT were ranked as the most important factors (ranked as “very important” by 86%, 76%, and 74% of respondents, respectively; Figure 2). As for radiographic and procedural factors, the location and burden of the thrombus, the probability of recanalization, and the interventionalist’s experience with EVT for CVT ranked as most important in this regard (Figure 2). The responses of interventionalists and non-interventionalists were similar with respect to the importance of clinical, radiographic, and procedural factors in the decision-making process (Figure S1).
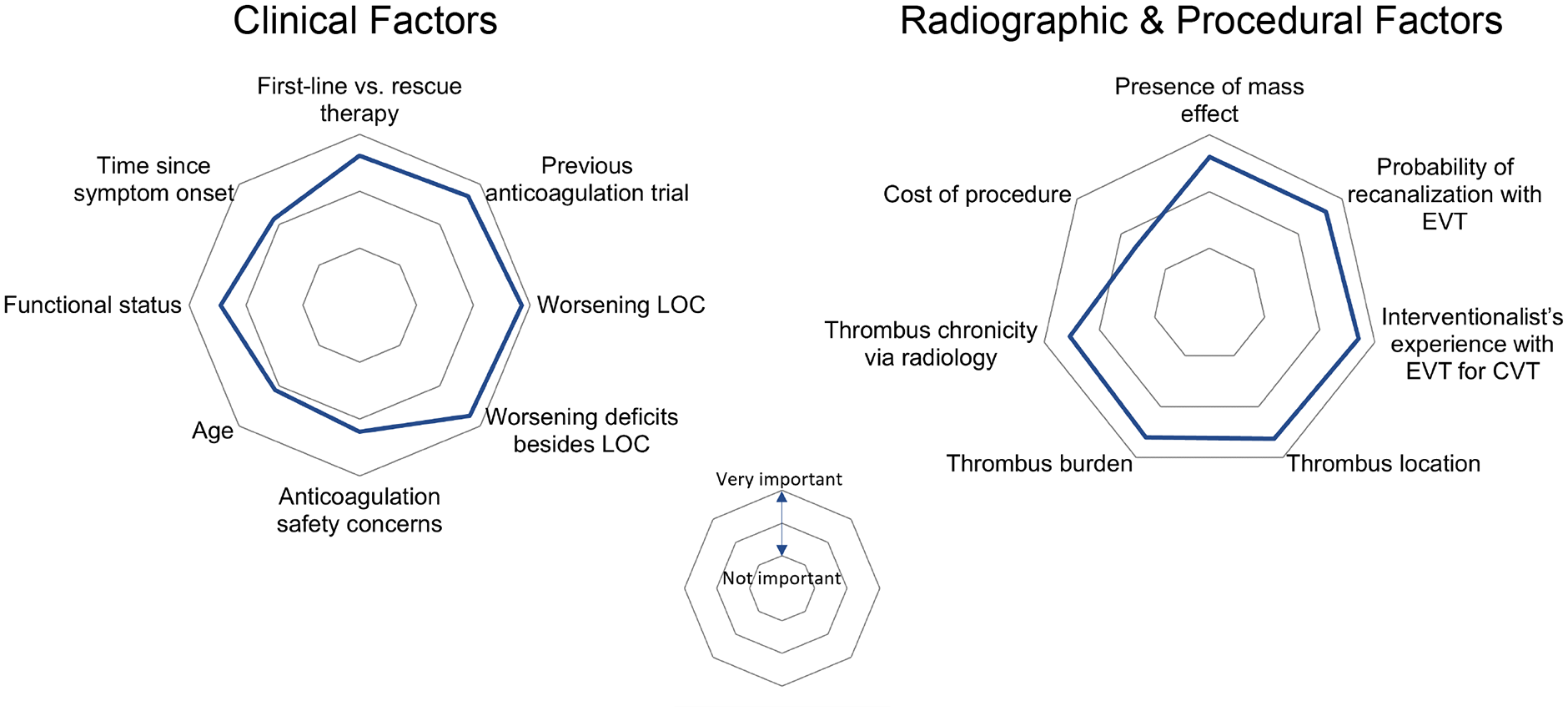
Importance of clinical and radiographical/procedural factors when considering the use of EVT for CVT treatment.
Use of endovascular techniques for CVT
Regarding access, almost all interventionalists (98%) believed the dural sinuses were amenable to endovascular intervention, while a minority believed the deep cerebral veins to be amenable to CVT (39%). There was substantial heterogeneity among interventionalists regarding preferred endovascular techniques (Figure 3), with mechanical thrombectomy with aspiration (56.2%) and stent retriever (50.5%) being the most utilized overall, followed by direct thrombolysis with tissue plasminogen activator (33.4%), direct intra-sinus administration of heparin (32.8%), and balloon angioplasty (23.4%). In addition to overall heterogeneity in techniques used, a chi-squared test comparing responses by region showed significant geographical variations (p < 0.001). Differing from responses from other countries, interventionalist respondents from China instead used direct intra-sinus administration of heparin most commonly (56.3%). In contrast, this technique was ranked highest as “never indicated” by respondents outside of China (23%; Figure 3).
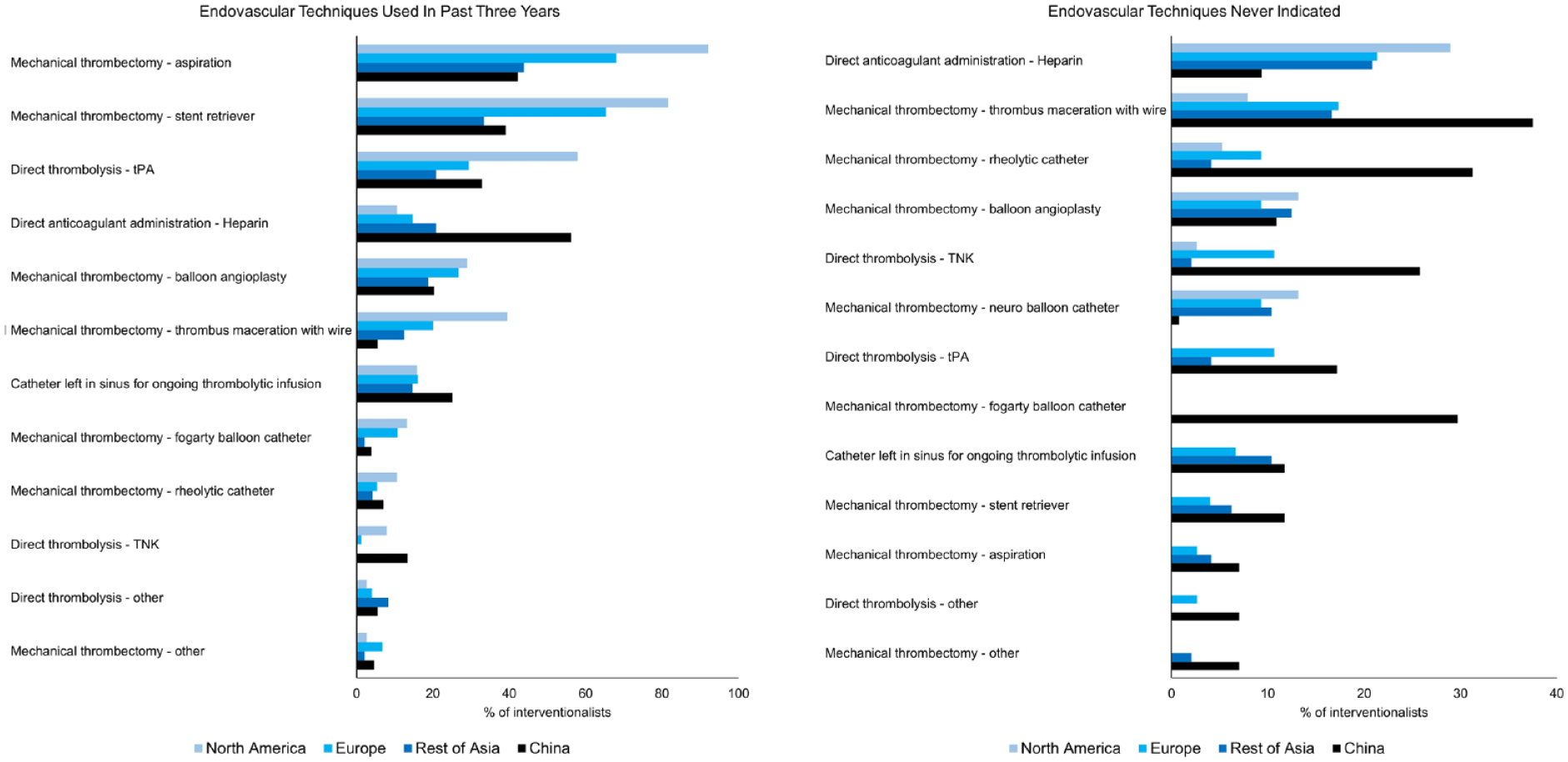
Interventionalist’s use and perceptions of endovascular techniques for the treatment of CVT, stratified by geographical region.
Post-procedural imaging and medical management
Preferred post-procedural imaging was largely distributed between magnetic resonance arteriography or venography (MRA/MRV; 71.8%) and computed tomography arteriography or venography (CTA/CTV; 65.9%); digital subtraction angiography (DSA) was used less frequently (18.2%; Figure 4). Respondents from Asia in particular showed a slight preference for MRA/MRV over CTA/CTV (Figure S2). Timing of post-procedural imaging was also not consistent, with clinicians most frequently selecting an imaging timepoint of 24 h (69.2%).
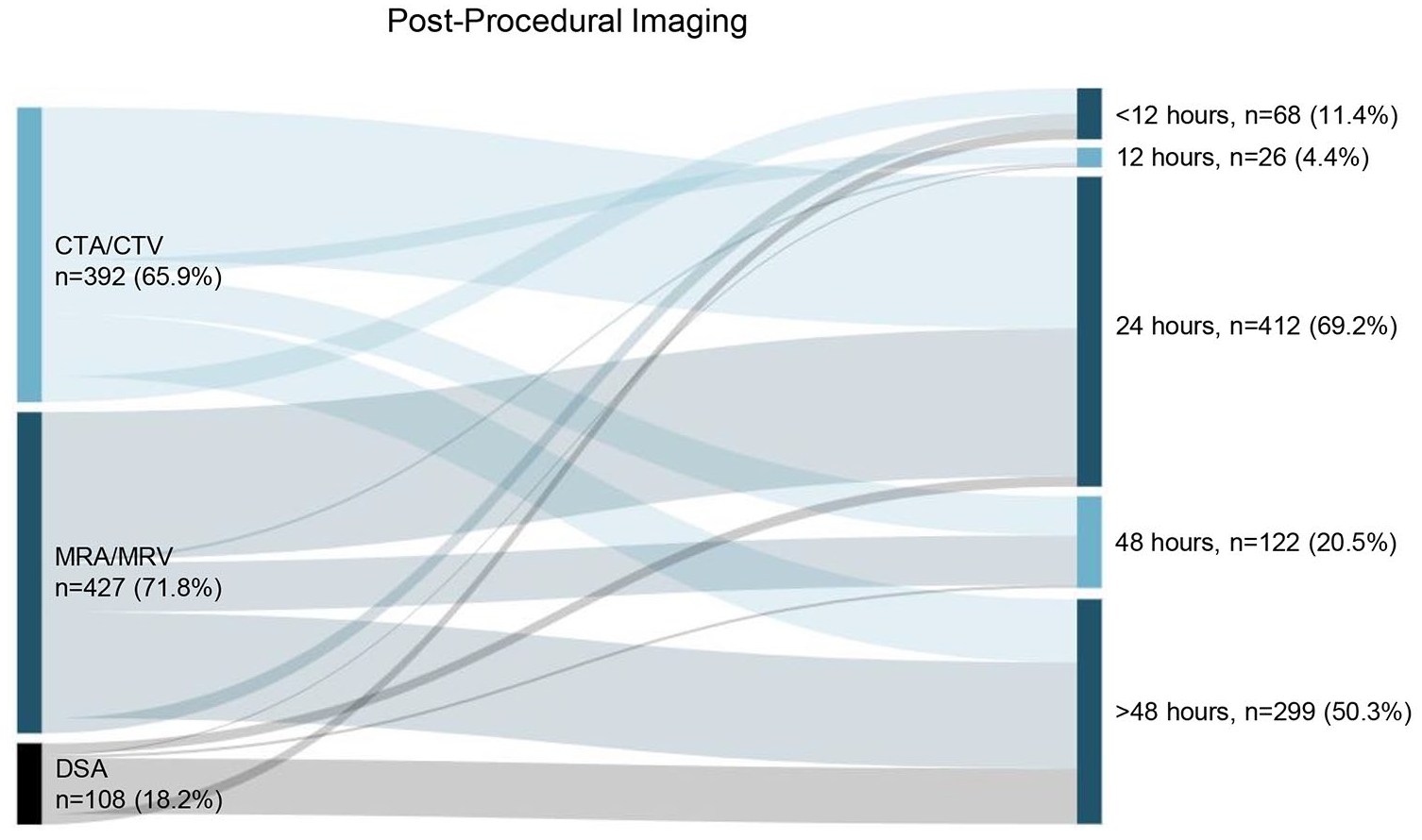
Post-procedural imaging modalities and timing following EVT for CVT. Percentages totaled over 100% as respondents were allowed to select multiple answer options. CTA/CTV—computed tomography angiography/venography; MRA/MRV—magnetic resonance angiography/venography; DSA—digital subtraction angiography.
For immediate post-procedural medical management, low molecular weight heparin (LMWH; 82.5%) was preferred in contrast to unfractionated heparin (37.4%; Figure 5). North American respondents in particular favored unfractionated heparin over LMWH (Figure S3). Preferred duration of initial post-procedural anticoagulation ranged from 0 to more than 10 days. Choice of subsequent oral anticoagulation was also split, with 70.8% of respondents preferring direct oral anticoagulants (DOACs) and 55.2% vitamin K antagonists (Figure 5). The preferred duration for long-term anticoagulation was most commonly 6–12 months (67.2%).
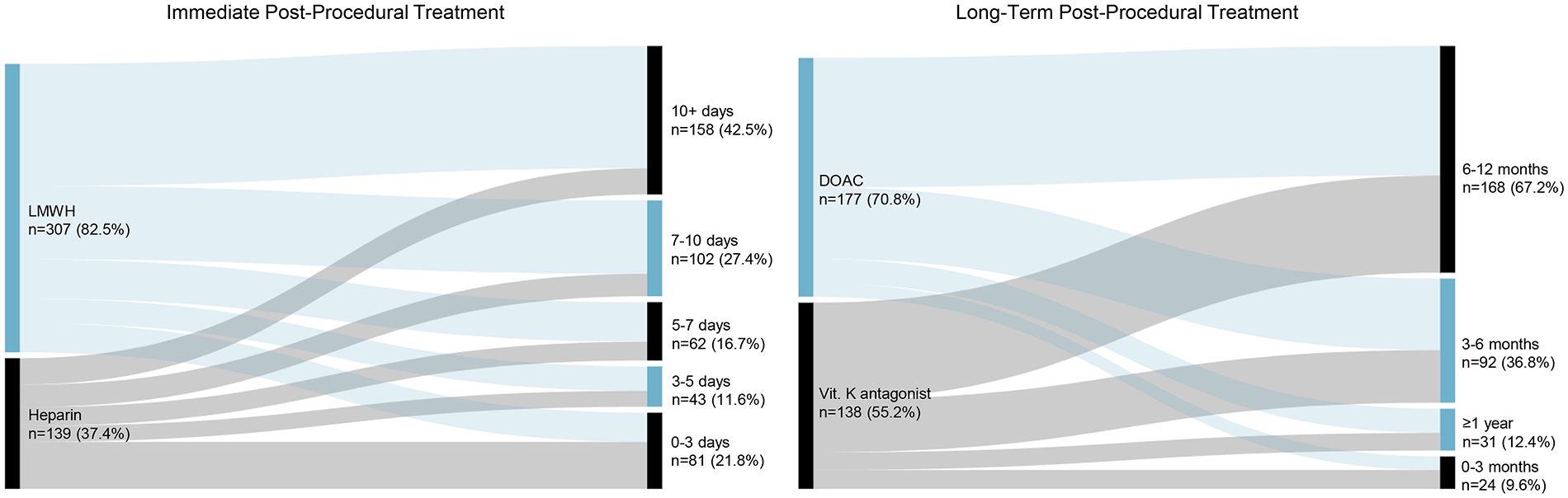
Post-procedural medical management following EVT for CVT. Short-term bridging therapy and long-term therapy were assessed. Percentages totaled over 100% as respondents were allowed to select multiple answer options. LMWH—low molecular weight heparin; DOAC—direct oral anticoagulant.
Discussion
The management of CVT presents a significant challenge due to its rarity and the absence of well-established treatment guidelines. Only one randomized trial has assessed the efficacy of EVT compared with the standard medical management. The TO-ACT trial, published in 2019, included patients with characteristics associated with adverse outcomes, including ICH at baseline, deep venous involvement, decreased level of consciousness, or “mental status disorder.” Participants were randomized to EVT without prespecified techniques versus best medical therapy and the trial stopped for futility after 67 patients of a target of 164 were recruited, failing to demonstrate a benefit for the primary outcome of modified Rankin scale (mRS) 0–1 at 12 months. 8 When meta-analyzed with the multinational, observational ACTION-CVT cohort study, there remained no consistent signal of benefit for EVT in the treatment of CVT. 6 Subsequent observational studies have not demonstrated significant benefit of using EVT, but also have not identified safety concerns with EVT used as an adjunct therapy.6,10,11 In addition, full recanalization was found to be associated with improved outcomes, although sample sizes were small. The clinical landscape of EVT for CVT is reminiscent of early attempts at EVT for acute ischemic stroke, perhaps failing to show efficacy due to a lack of optimized patient selection criteria or technique use. In addition, the interpretation of outcomes from the existing observational literature is substantially influenced by selection bias. The high mortality rates in CVT patients treated with EVT arises from the fact that only the most severely affected patients undergo EVT, because it is most commonly used as a rescue therapy.6,12 While not well understood, determining the impact of EVT on the treatment of CVT will be crucial in progressing patient outcomes.
This survey represents the largest, most comprehensive characterization of EVT use in CVT to date, reflecting the experience of 863 stroke clinicians across 61 countries. Our findings confirm that despite the paucity of literature to date supporting EVT, it continues to be used for therapy in certain cases of CVT. However, techniques and post-procedural management are varied, and substantial uncertainty remains around the characteristics that might make a patient with CVT a “good” candidate for EVT. The survey uncovered substantial heterogeneity in the techniques employed for EVT, with mechanical thrombectomy with aspiration, mechanical thrombectomy with stent retriever, direct thrombolysis with tissue plasminogen activator, direct administration of heparin, and balloon angioplasty being the most commonly used. This diversity in approach reflects the lack of evidence-based guidelines in this area stemming from the absence of robust trials assessing the superiority of one technique over another. Instead, the most commonly used techniques reflect those used for the treatment of acute ischemic stroke, 13 suggesting clinicians’ choice of technique may be influenced by their familiarity with those techniques and comfort in using those associated devices. Regional variations were also observed, most strikingly with China favoring direct anticoagulation with heparin, a technique ranked most commonly as “never indicated” in other parts of the world. There have been few studies comparing EVT techniques for CVT and currently there is insufficient evidence to suggest which endovascular approach and device is optimal.14,15 Overall, our findings emphasize a need for contemporary clinical trials to guide clinical decision-making and establish evidence-based standardized practices. By providing a comprehensive characterization of indications, techniques, and postoperative management used by clinicians internationally, this resource will aid in optimizing patient selection and endovascular treatments for future trials and clinical decision-making.
This study is subject to limitations common to physician surveys, including a response rate of 31% and the possibility of responder bias and centrality bias. The survey may have been completed preferentially by respondents favoring EVT in CVT, and thus our results may differ from CVT care in general. Given that the survey was not translated to languages other than Chinese, it is possible that there may have been differential interpretation of survey questions or selection bias for English-speaking clinicians. Furthermore, outside of China, only respondents that comprehended academic-level English were able to complete the survey. We also cannot definitively exclude the possibility of duplicate responses, although IP addresses were reviewed to ensure no duplicates were present. Importantly, the wide scope of our survey limited our exploration of specific factors influencing decision-making regarding the use of EVT for CVT, which is a needed area for future research, and would help to further improve patient selection in subsequent clinical trials. Regardless, this work provides a baseline for understanding current international practice patterns for the treatment of a rare disease based on hundreds of responses from expert clinicians.
Conclusion
This international survey highlights the considerable heterogeneity in the approaches to EVT for CVT among stroke clinicians globally. The lack of standardized practices in patient selection, procedural techniques, and post-procedural management emphasizes a persisting need for contemporary high-quality clinical evidence to guide practice, and this characterization of these factors will act as a resource to guide future clinical investigations. Future trials should not only assess the efficacy and safety of different EVT approaches, but also provide guidance on patient selection criteria and post-procedural care. Establishing a global consensus on EVT protocols for CVT based on the availability of local devices will be crucial in improving patient outcomes and fostering a more evidence-based approach to the management of this challenging condition.
Supplemental Material
sj-pdf-1-wso-10.1177_17474930241304206 – Supplemental material for International practice patterns and perspectives on endovascular therapy for the treatment of cerebral venous thrombosis
Supplemental material, sj-pdf-1-wso-10.1177_17474930241304206 for International practice patterns and perspectives on endovascular therapy for the treatment of cerebral venous thrombosis by Benjamin A Brakel, Alexander D Rebchuk, Johanna Ospel, Yimin Chen, Manraj KS Heran, Mayank Goyal, Michael D Hill, Zhongrong Miao, Xiaochuan Huo, Simona Sacco, Shadi Yaghi, Ton Duy Mai, Götz Thomalla, Grégoire Boulouis, Hiroshi Yamagami, Wei Hu, Simon Nagel, Volker Puetz, Espen Saxhaug Kristoffersen, Jelle Demeestere, Zhongming Qiu, Mohamad Abdalkader, Sami Al Kasab, James E Siegler, Daniel Strbian, Urs Fischer, Jonathan Coutinho, Anita Munckhof, Diana Aguiar de Sousa, Bruce CV Campbell, Jean Raymond, Xunming Ji, Gustavo Saposnik, Thanh N Nguyen and Thalia S Field in International Journal of Stroke
Supplemental Material
sj-pdf-2-wso-10.1177_17474930241304206 – Supplemental material for International practice patterns and perspectives on endovascular therapy for the treatment of cerebral venous thrombosis
Supplemental material, sj-pdf-2-wso-10.1177_17474930241304206 for International practice patterns and perspectives on endovascular therapy for the treatment of cerebral venous thrombosis by Benjamin A Brakel, Alexander D Rebchuk, Johanna Ospel, Yimin Chen, Manraj KS Heran, Mayank Goyal, Michael D Hill, Zhongrong Miao, Xiaochuan Huo, Simona Sacco, Shadi Yaghi, Ton Duy Mai, Götz Thomalla, Grégoire Boulouis, Hiroshi Yamagami, Wei Hu, Simon Nagel, Volker Puetz, Espen Saxhaug Kristoffersen, Jelle Demeestere, Zhongming Qiu, Mohamad Abdalkader, Sami Al Kasab, James E Siegler, Daniel Strbian, Urs Fischer, Jonathan Coutinho, Anita Munckhof, Diana Aguiar de Sousa, Bruce CV Campbell, Jean Raymond, Xunming Ji, Gustavo Saposnik, Thanh N Nguyen and Thalia S Field in International Journal of Stroke
Supplemental Material
sj-pdf-3-wso-10.1177_17474930241304206 – Supplemental material for International practice patterns and perspectives on endovascular therapy for the treatment of cerebral venous thrombosis
Supplemental material, sj-pdf-3-wso-10.1177_17474930241304206 for International practice patterns and perspectives on endovascular therapy for the treatment of cerebral venous thrombosis by Benjamin A Brakel, Alexander D Rebchuk, Johanna Ospel, Yimin Chen, Manraj KS Heran, Mayank Goyal, Michael D Hill, Zhongrong Miao, Xiaochuan Huo, Simona Sacco, Shadi Yaghi, Ton Duy Mai, Götz Thomalla, Grégoire Boulouis, Hiroshi Yamagami, Wei Hu, Simon Nagel, Volker Puetz, Espen Saxhaug Kristoffersen, Jelle Demeestere, Zhongming Qiu, Mohamad Abdalkader, Sami Al Kasab, James E Siegler, Daniel Strbian, Urs Fischer, Jonathan Coutinho, Anita Munckhof, Diana Aguiar de Sousa, Bruce CV Campbell, Jean Raymond, Xunming Ji, Gustavo Saposnik, Thanh N Nguyen and Thalia S Field in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.T. reports funding from the European Commission (European Union’s Horizon 2020 research and innovation program), the German Research Foundation, and the German Innovations Fund; and personal consulting fees or honoraria for lectures from Acandis, Alexion, Amarin, AstraZeneca, Bayer, Boehringer Ingelheim, BristolMyersSquibb/Pfizer, Daiichi Sankyo, Stryker, all outside the submitted work. J.E.S. has served as a consultant for AstraZeneca, and has received funding from the National Institutes of Health (R61NS135583), Viz.ai, Philips, and Medtronic. U.F. reports research support of the Swiss National Science Foundation and the Swiss Heart Foundation; PI of the ELAN trial, Co-PI of the DISTAL, TECNO, SWIFT DIRECT, SWITCH, ELAPSE and ICARUS trial; Steering Committee Member of the DO_IT trial; research grants from Medtronic (BEYOND SWIFT, SWIFT DIRECT), from Stryker, Rapid medical, Penumbra, Medtronic and Phenox (DISTAL), and from Boehringer Ingelheim (TECNO), whereas all fees were paid to the institution; consultancies for Medtronic, Stryker, and CSL Behring (fees paid to institution); participation in an advisory board for AstraZeneca (former Alexion/Portola), Boehringer Ingelheim, Biogen, AbbVie and Acthera (fees paid to institution); member of a clinical event committee (CEC) of the COATING study (Phenox) and member of the data and safety monitoring committee (DSMB) of the TITAN, LATE_MT and IN EXTREMIS trials; President of the Swiss Neurological Society, and President-elect of the European Stroke Organisation. J.C. has received research support paid to his institution from Boehringer Ingelheim, Bayer, and AstraZeneca, and is co-founder and shareholder of TrianecT. T.N.N. discloses being Associate Editor of Stroke, and on the Advisory Board of Aruna Bio, Brainomix. T.S.F. discloses being on the Advisory Board for Bayer, Novartis, AstraZeneca, an expert witness testimony, and is on the board of DESTINE Health.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by an Innovative Clinical Trials Planning Grant from the Canadian Institute of Health Research. T.S.F. is supported by a Sauder Family/Heart and Stroke Professorship of Stroke Research.
Data availability
Survey questions are included in the Supplementary Data. Response data are available upon request to the corresponding author, T.S.F. (
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
