Abstract
A decade on from the first positive thrombectomy trials, hyperacute therapies for ischemic stroke continue to rapidly advance. Effective treatments remain limited to reperfusion, although several cytoprotective approaches continue to be investigated. Intravenous fibrinolytics are now demonstrated to be beneficial up to 24 h in patients selected using perfusion imaging, but their role in patients with non-disabling symptoms appears very limited. Tenecteplase is superior to alteplase in meta-analysis of the latest trials, and adjuvant thrombolytics are an area of active investigation. Endovascular thrombectomy is beneficial in a wide range of anterior and posterior circulation large vessel occlusions up to 24 h after onset with the more distal occlusions, mild presentations, and >24 h window being the main frontiers to be tested in ongoing trials. Imaging parameters are prognostic but appear not to modify the relative treatment benefit of thrombectomy versus standard medical care. Therefore, deciding who not to treat with thrombectomy is a key clinical challenge that requires careful but rapid integration of clinical, imaging, and patient preference considerations. Systems of care to accelerate delivery of these highly effective therapies will maximize benefits for the greatest number of patients with stroke.
Keywords
Introduction
The field of stroke medicine has had some notable landmarks, perhaps most notably the 1995 publication of the first positive thrombolytic trial 1 and the 2015 publication of multiple positive thrombectomy trials,2–6 with transformative impact in reducing disability for patients with ischemic stroke. As we reach the 10th anniversary of the presentation of MR CLEAN 2 at the World Stroke Congress in Budapest 2014, it is timely to reflect on the advances in hyperacute stroke therapies and the frontiers we continue to investigate. This issue of the International Journal of Stroke includes a number of topical articles relevant to ischemic stroke acute therapy.
Intravenous fibrinolytics
Tenecteplase
Alteplase within 0–4.5 h is well established 7 with extension to 9-h post-onset or the midpoint of sleep based on perfusion mismatch imaging recommended in several regions.8–10 Tenecteplase has now been compared with alteplase in more patients than alteplase was compared with placebo,11–21 and superiority for the outcome of modified Rankin scale 0–1 (no disability) at 90 days demonstrated in study-level meta-analysis (Figure 1). A large scale shift to tenecteplase use has already occurred in North America and tenecteplase is now licensed for stroke in Europe, with the European Stroke Organization guidelines recommending tenecteplase use. 22 It is, therefore, likely that the rest of the world will also switch to tenecteplase once the supply shortage outside North America eases. The absolute benefit over alteplase in the general thrombolytic-eligible population at a modified Rankin scale 0–1 threshold is a clinically meaningful ~3% (number needed to treat to achieve no disability of ~33). The benefit may be greater in patients with large vessel occlusion.14,22 Just as importantly, the simplicity of bolus administration (compared to 60-min alteplase infusion) ensures the full dose is given and facilitates transport between or within hospitals for thrombectomy when required.
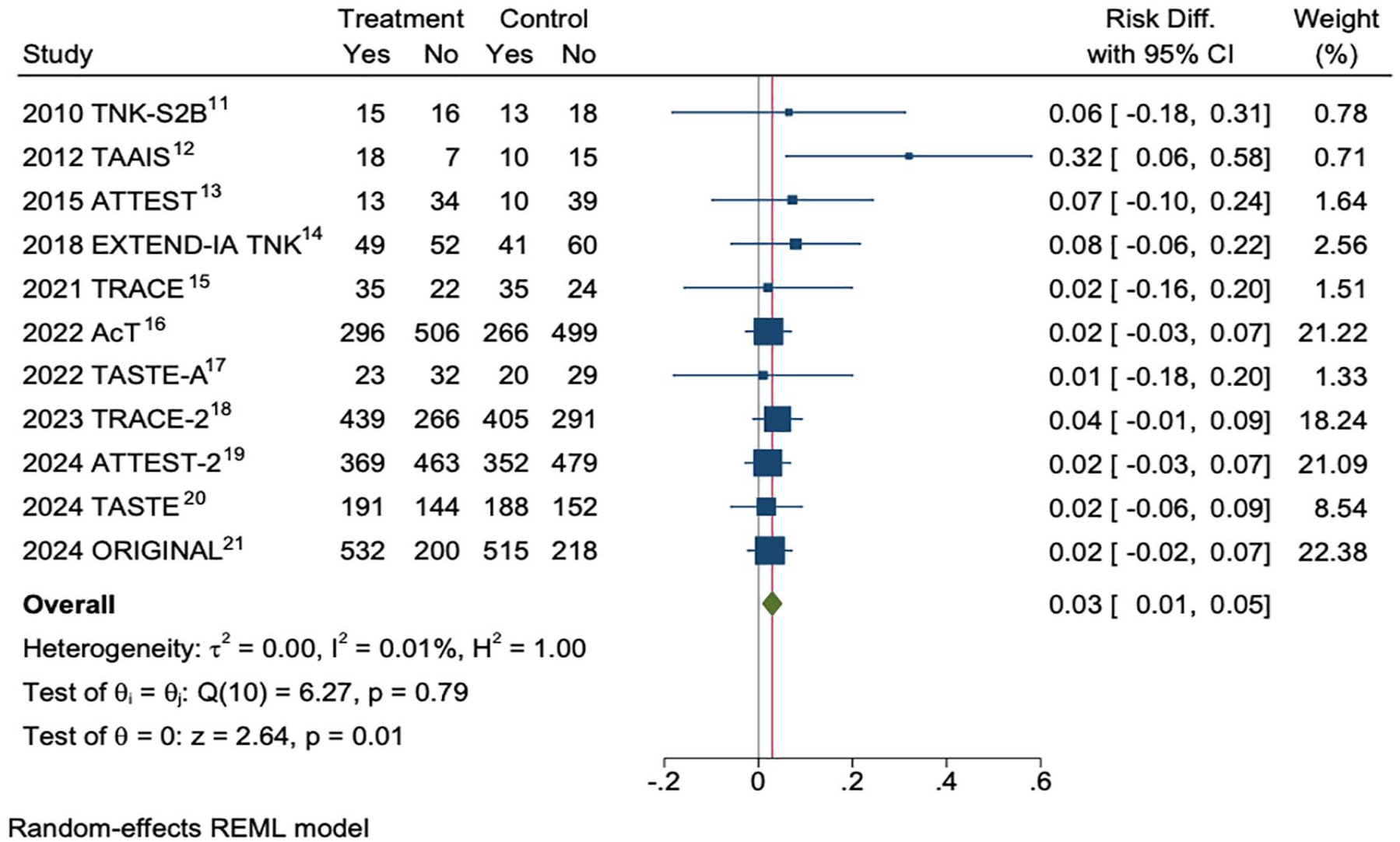
Meta-analysis of trials comparing tenecteplase 0.25 mg/kg versus alteplase 0–4.5 h after stroke onset for the outcome of modified Rankin Scale 0–1 (no disability) at 90 days.
Extended time window
The latest data using tenecteplase have extended the time window to 24 h using perfusion imaging selection. The TRACE-III trial randomized patients with large vessel occlusion who could not access thrombectomy to tenecteplase versus standard care and demonstrated an absolute benefit in mRS 0–1 of ~10%, 23 similar to alteplase 0–3 h in the broad lytic-eligible population 7 or 4.5–9 h in the perfusion mismatch population. 8 The TRACE-III results directly apply to patients who cannot access thrombectomy (which is the majority globally). However, even in developed countries, most patients first present to a non-thrombectomy capable hospital and require inter-hospital transport, with no guarantee of still being eligible for thrombectomy on arrival. Dedicated trials for this scenario are ongoing (NCT04454788) but TRACE-III provides a rationale for giving tenecteplase while arranging transfer for possible thrombectomy. There is residual uncertainty in patients able to immediately access thrombectomy, given the neutral results of the TIMELESS trial. 24 However, there were no safety concerns in TIMELESS, and the median 15 min between tenecteplase and arterial puncture is shorter than could be achieved in routine practice due to trial-related consent, randomization, and clinical trial pharmacy procedures. It was also shorter than the median lytic to puncture time of 25 min in the pooled trials of direct thrombectomy. 25 This allowed little opportunity for the lytic to have an effect on pre-thrombectomy reperfusion. As suggested by the increase in post-thrombectomy reperfusion when thrombolytic was given in the pooled direct thrombectomy trials, 25 there may also be a facilitatory effect of active thrombolytic on thrombectomy success. However, proving this would require a large sample size to establish what is probably a <5% effect size on the dichotomized modified Rankin scale.
Mild stroke
Mild stroke continues to present decision-making challenges but recent trials have clarified that there is little benefit (and some risk) of fibrinolytics in patients without disabling symptoms.26,27 The TEMPO-2 trial, 28 the protocol for which appears in this issue of International Journal of Stroke, 29 selected a subgroup of patients with vessel occlusion or perfusion lesion. These patients had a high rate of deterioration in prior studies30,31 but the phase 3 trial was negative. The risk of deterioration was unexpectedly low at 8%, there was no hint of benefit in functional outcome (despite increased recanalization with tenecteplase), and there was an increase in symptomatic hemorrhage and death. However, most deaths were late and appeared unrelated (e.g. cancer) so that may have been a chance finding. Nonetheless, the TEMPO-2 results clearly did not show any benefit of tenecteplase in these patients, suggesting that the most appropriate management is rapid loading with dual antiplatelets and close observation for any deterioration.
Central retinal artery occlusion
Another unanswered question is the role of intravenous fibrinolytics for central retinal artery occlusion. Prior trials have not demonstrated benefit with alteplase but may not have enrolled patients sufficiently early to salvage retina. The protocol for the REVISION trial in this issue of the International Journal of Stroke 32 details the planned trial, which enrolls within 4.5 h of onset based on meta-analysis of the existing data. 33
Adjuvant thrombolytics
Despite improvements with tenecteplase, the majority of patients with large vessel occlusion still require thrombectomy and patients with distal internal carotid and proximal M1 middle cerebral artery occlusions rarely reperfuse rapidly with fibrinolytics. 34 This has sparked interest in adjuvant agents that target different components of thrombus. A dense shell containing platelets, von Willebrand factor, and DNA has been recently recognized to encapsulate thrombi and likely increases resistance to fibrinolytics but may respond to adjuvant combinations. 35 The MOST trial studied argatroban and eptifibatide in combination with fibrinolytics but the study was terminated early for futility. 36 Adding aspirin or heparin increased the risk of bleeding.37,38 Novel antiplatelets, von Willebrand factor antagonists, DNase, alpha-2-antiplasmin inhibitors, and other agents are all in trials (Table 1) with the aim of improving intravenous thrombolytic efficacy without excessive bleeding risk. The stagnant flow immediately proximal to the thrombus reduces access to endogenous and exogenous thrombolytic substances and trials are testing whether nanoparticles can overcome this barrier to pharmacological therapies. 39
Key clinical questions in hyperacute stroke therapy.
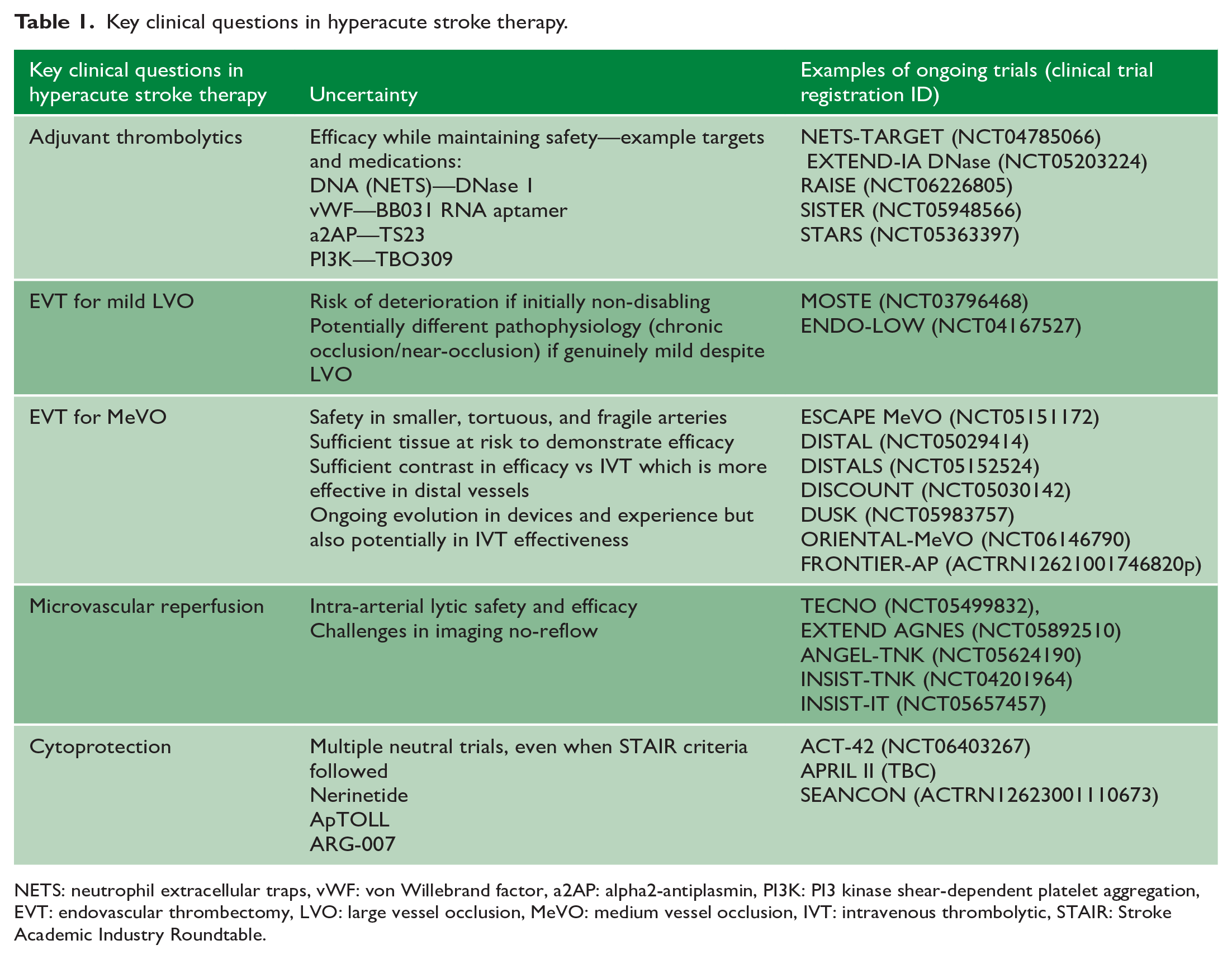
NETS: neutrophil extracellular traps, vWF: von Willebrand factor, a2AP: alpha2-antiplasmin, PI3K: PI3 kinase shear-dependent platelet aggregation, EVT: endovascular thrombectomy, LVO: large vessel occlusion, MeVO: medium vessel occlusion, IVT: intravenous thrombolytic, STAIR: Stroke Academic Industry Roundtable.
Endovascular thrombectomy
Endovascular thrombectomy (EVT) is now established to reduce disability for patients with anterior circulation large vessel occlusion of the internal carotid, M1 middle cerebral and basilar arteries, up to 24 h after stroke onset.40,41 Patients with tandem cervical occlusion with intracranial large vessel occlusion also clearly benefit. Meta-analysis of 0–6 h trials also demonstrated benefit of EVT in patients with proximal or dominant M2 occlusions. 42
The initial extension from 6–24 h in the anterior circulation using perfusion 43 or clinical-core 44 mismatch criteria appears to have now been replaced by broader eligibility with clear benefit in patients with Alberta Stroke Program Earlt CT Score (ASPECTS) 3–5,45–49 and even ASPECTS 0–2 in the LASTE trial that included patients within 7 h of onset. 49 Using perfusion imaging, there was no upper limit in core volume for benefit of EVT versus medical management evident in the SELECT-2 trial using ordinal analysis of modified Rankin scale (mRS). 50 However, absolute rates of functional independence (mRS 0–2) and independent ambulation (mRS 0–3) dropped substantially with increased volumes of ischemic core. Improvements in outcome ceased to be statistically significant >120 mL in the RESCUE Japan LIMIT trial, based on modeled confidence intervals. However, point estimates remained favorable beyond that point and the 120 mL threshold may simply reflect low patient numbers and, therefore, wide confidence intervals rather than physiology. 51
Large core: to treat or not to treat?
Elderly patients were under-represented or actively excluded from the randomized trials, and patients also had to have good premorbid function. These factors make decisions in clinical practice more challenging, as the likelihood of the patient achieving what they may regard as an acceptable outcome needs to be rapidly estimated when deciding whether to offer thrombectomy, based on an extrapolation of trial data. Potential meaningful recovery should not be narrowly defined as mRS 0–2. Particularly in the large core population, an mRS of 3, and for some patients even mRS 4, would be preferable to a high probability of mRS 5–6 with medical therapy. 52 Some argue that all patients with large vessel occlusion should be treated intensively, and then, decisions can be made about goals of care in the coming days. Such goals of care decisions need to bear in mind the increase in early neurological deterioration with thrombectomy observed in SELECT-2. 46 Patients with large core frequently have a complicated early course and rarely exhibit the early major recovery that is common in patients with smaller ischemic core. Therefore, avoidance of the self-fulfilling prophecy of early limitations on care is advisable if the initial decision to pursue thrombectomy has been made. The volume and location of ischemic core are strongly prognostic and can be considered in conjunction with patient frailty and expected time to reperfusion to further refine predicted outcome. 46 There are some patients in whom the combination of extensive ischemic injury, premorbid frailty, co-morbidity, or known wishes means that the likelihood of an outcome acceptable to the patient is so low as to be essentially futile. Thrombectomy is resource-intensive, and, in such cases, a decision can be made in the emergency department to take a conservative approach.
Areas of uncertainty for thrombectomy benefit
Key current frontiers in EVT are mild severity and distal occlusion site. Patients with mild severity (NIHSS <6) were under-represented or excluded from the pivotal EVT trials. To have such mild symptoms despite large vessel occlusion, these patients must have excellent collaterals and, potentially, some will have chronic occlusions or chronic near-occlusion with acute thrombosis that may respond differently to EVT. The neutral data with thrombolytics and surprisingly low rate of deterioration in TEMPO-2, 28 despite vessel occlusion, make the results of ongoing trials (MOSTE NCT03796468 and ENDO-LOW NCT04167527) even more keenly anticipated. In basilar artery occlusion, there is uncertain benefit of EVT when NIHSS is <10. 41 Unfortunately, the NIHSS is less well correlated with subsequent disability in posterior circulation stroke. In particular, dysphagia and truncal ataxia are not scored but should probably be considered when assessing potential benefit of reperfusion therapy. 53
Distal medium vessel occlusions (Figure 2) are an active area of investigation with at least seven randomized trials ongoing. The evidence for EVT in meta-analysis extends to proximal or dominant M2 middle cerebral artery occlusions. 42 Beyond that, increased arterial tortuosity, reduced luminal diameter, and wall thickness increase the risk of vascular injury with EVT. The territory at risk is reduced and the relative effectiveness of thrombolytics increased so randomized trials are crucial and the first reports are expected in 2025. Perfusion mismatch has been suggested to modulate the benefit of distal thrombectomy 54 but is not required for selection in the current randomized trials. The risk–benefit balance is likely to see-saw with progressive improvements in both mechanical devices and proceduralist experience versus improvements in intravenous thrombolytic strategies, which may make this an active field of enquiry for some time to come.
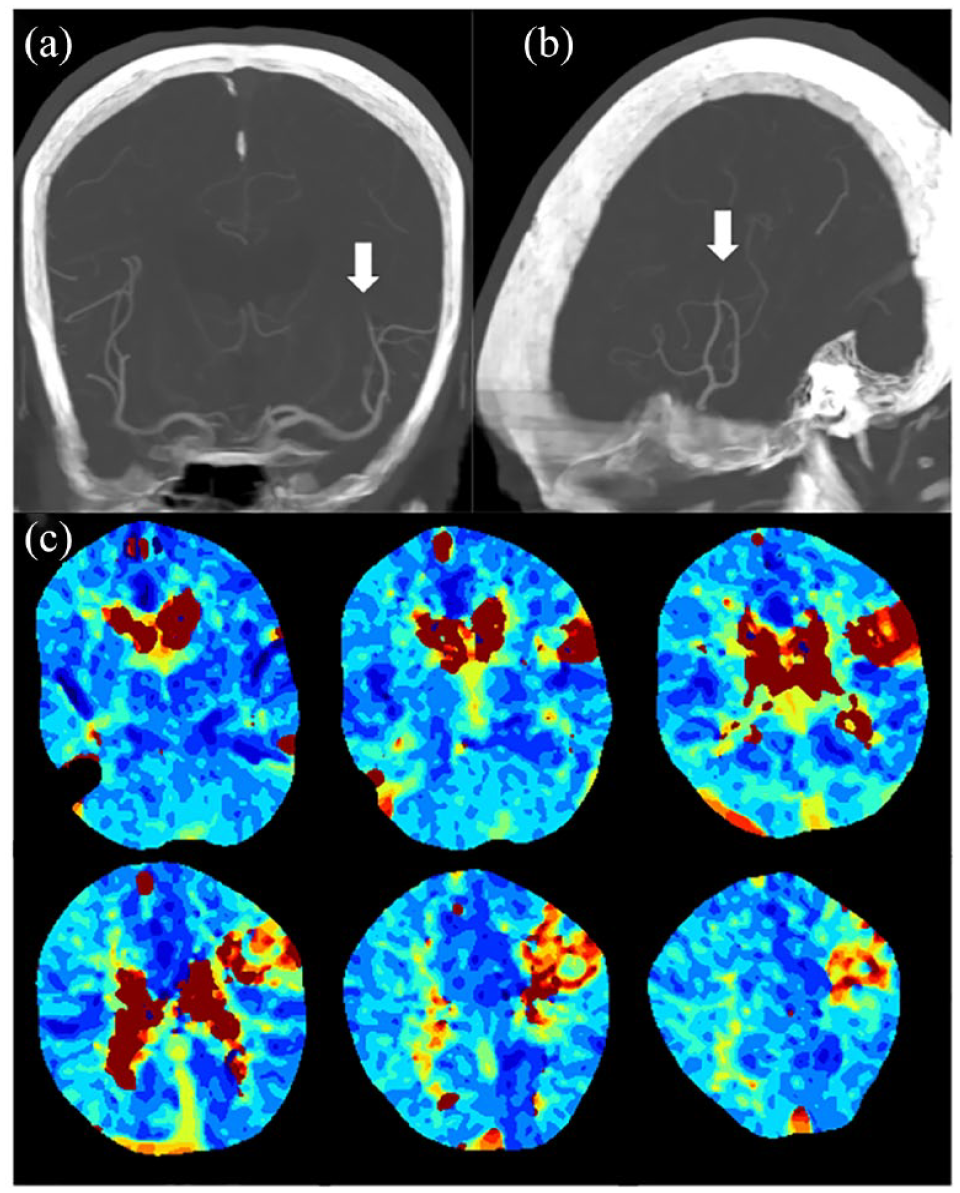
Perfusion imaging assists in finding the site of occlusion on computed tomography (CT) angiography in a patient with distal vessel occlusion and indicates the spatial location and extent of tissue at risk: (a) coronal and (b) sagittal maximum intensity projections of CT angiogram (arrows = distal left middle cerebral artery occlusion); (c) time to maximum (Tmax) map demonstrating the critically hypoperfused region.
Microvascular pathophysiology and cytoprotection
Despite effective reperfusion, the majority of patients with large vessel occlusion do not regain independence. 40 In part, this may reflect irreversible injury prior to reperfusion. However, increasingly, it seems that what we have previously regarded as irreversibly injured could perhaps be at least partially salvageable, particularly with novel approaches to treat microvascular occlusions. 55 Temporary reversal in diffusion abnormalities after reperfusion generally returns with current therapies but may represent an opportunity for novel interventions. 56 Preclinical data indicate capillary sludging with debris and neutrophils with pericyte contraction, despite macrovascular reperfusion, and may be amenable to treatment. 57
Future developments in cytoprotection may modify the time is brain paradigm and several agents remain in trials. However, the recent neutral ESCAPE-NEXT trial (NCT04462536) gives pause for thought, after promising preclinical and early phase human data, positive pre-specified subgroup analysis, and biological plausibility of deleterious interaction with plasmin in alteplase-treated patients in the previous phase 3 trial. 58 The potential signal of benefit when nerinetide was administered prehospital (NCT02315443) will be tested in an early time window trial. Other agents including ApTOLL with positive phase 2 data (NCT04734548) are proceeding to phase 3.
Systems of care
Reperfusion therapies are highly time sensitive.7,59 Hence, optimizing systems to deliver treatment faster is one of the most impactful strategies to reduce stroke-related disability. Fibrinolytics are more effective when given earlier and data indicate superiority of bridging with alteplase (and tenecteplase) prior to thrombectomy versus direct thrombectomy, at least within 2 h 20 min of stroke onset, based on modeled confidence intervals. 60 Again, there is no evidence of inferiority or harm as soon as the >2 h 20 min threshold is crossed and the precise threshold likely reflects study power and modeling as much as physiology. As a result, the ideal models deliver thrombolytic as early as possible and then proceed to EVT in the subgroup with suitable vessel occlusion as quickly as possible. Mobile stroke units are ideally suited to this paradigm with rapid thrombolytic administration, potentially within 1 h of onset, and definitive triage of thrombectomy-eligible patients to an appropriate hospital.61,62 Current mobile stroke units are resource-intensive, but future technological advances may allow more routine remote clinical assessment, and perhaps even imaging assessment in standard ambulances.
Role of imaging
Imaging is a key to stroke diagnosis and therapeutic decision-making and, therefore, a critical component of the stroke system of care, and also a substantial time investment.
Whatever imaging protocol is utilized should be acquired in a single sitting (i.e. not a non-contrast CT followed by CT angiogram at a separate later session). Tenecteplase has the advantage of being able to be easily administered in the scanner room between sequences if necessary. Magnetic resonance imaging (MRI) is used as the primary modality in some regions but there needs to be immediate scanner access and strategies to manage unstable patients or those with known metallic implants or unknown implant status.
The initial imaging should be made available to all teams involved (including those receiving the patient for EVT when inter-hospital transfer is required) to avoid repeat imaging whenever possible. Studies have indicated that collaterals are usually stable within at least 3 h 63 and with recent large core trial data, the question has to be asked, “Would repeating the CT change the decision to perform EVT?” If the answer is no then there seems little justification to delay reperfusion to re-image. With improving flat panel CT acquisitions, some questions such as the presence of hemorrhagic transformation can be addressed in the angiography suite. Direct to angiography suite approaches are being trialed but data so far predominantly relate to patients transferred from another hospital (i.e. no repeat imaging) rather than patients direct from the field (no imaging prior to angiography), and the latter subgroup did now show benefit. 64
Although recent large core EVT trials have reduced reliance on perfusion imaging for EVT selection 6–24 h, 50 IV thrombolytic treatment beyond 4.5 h is still reliant on perfusion imaging.8,23 Prior data have not demonstrated benefit of alteplase >4.5 h in patients selected based on non-contrast CT. 7 Late window thrombolytic treatment, therefore, relies on establishing a target for therapy (a significant perfusion mismatch estimating the ischemic penumbra) and an absence of severe or extensive CT hypodensity (to reduce risk of symptomatic intracerebral hemorrhage, Figure 3 65 ). Patients without target mismatch had no signal of benefit and potential harm in meta-analysis 8 and patients with severe CT hypodensity contributed a majority of symptomatic ICH in TRACE-III. 23 While the subgroup of patients with vessel occlusion and no major non-contrast CT changes are likely to have a perfusion mismatch profile, that “simple” approach risks missing patients with a treatable target or delaying treatment while carefully scrutinizing a CT angiogram for distal vessel occlusion that would have been instantly visible on a perfusion map (Figure 2). The reliance on minimal hypodensity on CT is also less generalizable and objective and will potentially exclude patients who would have met perfusion-based criteria.
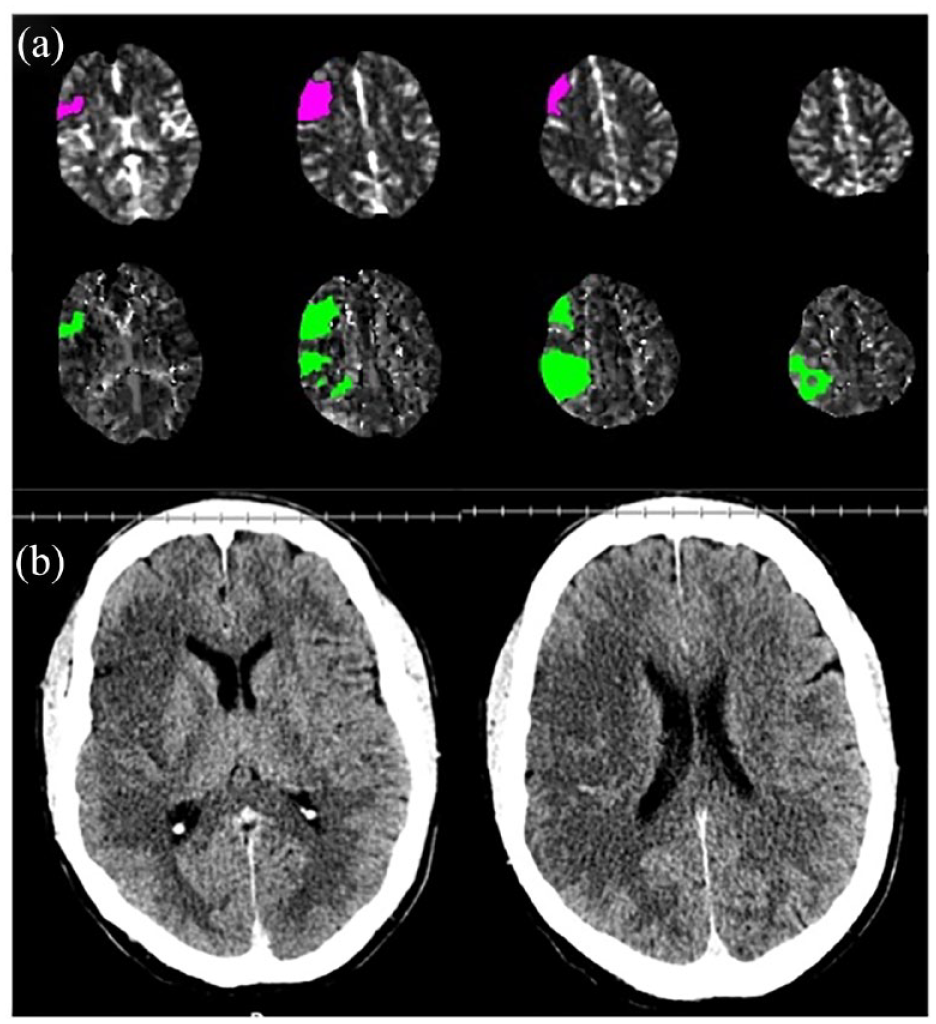
(a) Automated CT perfusion processing suggests perfusion mismatch: Ischemic Core 13 mL, Tmax > 6 s 39 mL = mismatch volume 26 mL, mismatch ratio 3.0. However, (b) unequivocal non-contrast CT hypodensity extends beyond the current perfusion lesion and cerebral blood flow within residual parietal hypoperfusion is no longer below the 30% threshold for ischemic core, despite clear CT hypodensity in that region. Manually outlined CT hypodensity volume was 105 mL. Partial reperfusion and collateral improvement increase in prevalence as time passes and can create a false appearance of perfusion mismatch. Hence, CT perfusion needs to be interpreted in conjunction with non-contrast CT. This patient does not have a true perfusion mismatch and would not have met eligibility for the EXTEND trial of thrombolytic >4.5 h. 65
There are other potential benefits of perfusion imaging in patients with suspected stroke (Table 2) and, when performed routinely to maintain familiarity and high data quality, the additional time taken to acquire this information is minimal and can be performed in parallel with other management processes. Perfusion imaging can improve diagnosis and reduce missed treatment opportunities. This is particularly true at primary stroke centers and is feasible based on Australian experience.66,67 These centers without on-site thrombectomy access also likely benefit the most from the 4.5–24 h thrombolytic time window extension. 23 Information on the location and extent of ischemic core from perfusion imaging is strongly prognostic. 52 This is particularly relevant in patients with co-morbidity or frailty who were not included in randomized trials, or to guide discussion in patients with a strong preference to avoid disability. 50 While core volume may not dissuade the team from pursuing EVT, the information provides useful framing of potential outcomes with family and guides preparation for likely complications such as malignant edema. Perfusion imaging can also identify patients for trials of new therapies to advance care which are likely to be best targeted to those patients who are less likely to have excellent outcome with reperfusion alone.
Benefits of perfusion imaging.
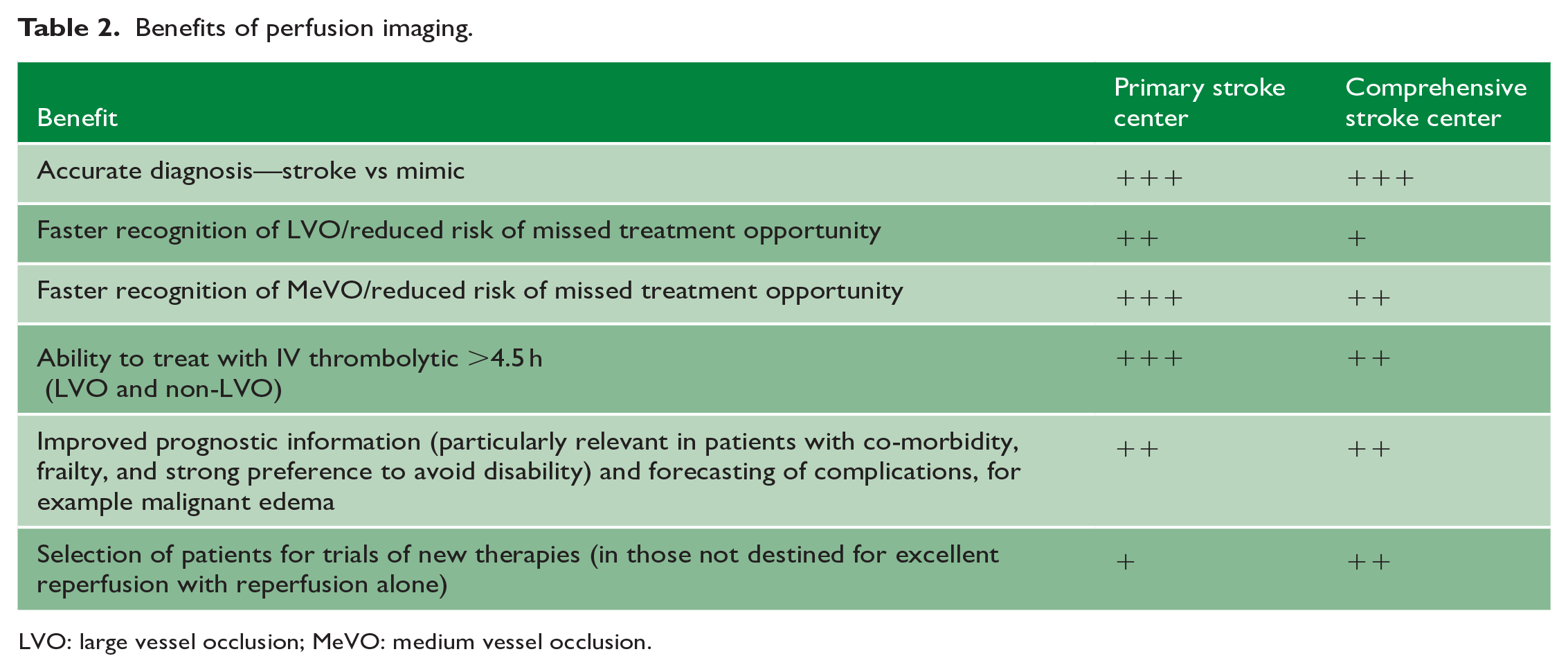
LVO: large vessel occlusion; MeVO: medium vessel occlusion.
Conclusion
Hyperacute stroke therapies are advancing rapidly and, currently, remain reperfusion-focused. Delivering proven therapies faster, while concurrently investigating potentially more effective strategies in trials, is both exciting and challenging as we push the frontiers of stroke medicine.
Footnotes
Acknowledgements
The authors thank Prof Leonid Churilov, Professor of Biostatistics, University of Melbourne, Australia, for assistance with the meta-analysis figure.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
