Abstract
Background:
Post-stroke fatigue (PSF) affects around 50% of stroke survivors. Previous systematic reviews of randomized controlled trials found insufficient evidence to guide practice, but most excluded Chinese studies. Furthermore, their searches are now out-of-date.
Aims:
To systematically review and perform a meta-analysis of randomized placebo-controlled trials of pharmacological interventions for treating PSF.
Methods:
We screened Airitri, CNKI, VIP, CINAHL, ClinicalTrials.gov, CENTRAL, Cochrane Stroke Group Trial Register, EMBASE, EU Clinical Trial Register, ISRCTN, MEDLINE, PsycINFO, Wanfang, and WHO ICTRP up to 11 November 2022. Our primary outcome was fatigue severity. We conducted subgroup analysis by drug type and sensitivity analysis after excluding the trials at high risk of bias. Secondary outcomes included mood and quality of life.
Results:
We screened 33,297 citations and identified 10 published completed trials, 6 unpublished completed trials, and 6 ongoing trials. Pharmacological treatments were associated with lower fatigue severity at the end of treatment (10 published completed trials, 600 participants, pooled standardized mean difference (SMD) = −0.80, 95% confidence interval (CI): −1.29 to −0.31; I2 = 86%, p < 0.00001), but not at follow-up (265 participants, pooled SMD = −0.14, 95% CI: −0.38 to 0.10; I2 = 0, p = 0.51). However, these trials were small and had considerable risk of bias. Beneficial effects were seen in trials with low risk of bias on randomization, missing outcome data, and reporting bias. There were insufficient data on secondary outcomes for meta-analysis, but six trials reported improved quality of life.
Conclusion:
There is insufficient evidence to support a particular pharmacological treatment for PSF, thus current clinical guidelines do not require amendment.
Background
More than 15 million people have a stroke each year globally, of whom more than 30% are left with severe long-term disability. 1 Between 25% and 85% of stroke survivors have post-stroke fatigue (PSF), which may persist for many years.2–5 There are important gaps in understanding the etiology of PSF. 6 In 2023, the World Stroke Organization identified the lack of strong recommendations for PSF management. 7 How to manage PSF is a priority for the Europe’s 2018–2030 Stroke Action Plan. 8
Previous systematic reviews of PSF treatment generally sought studies only from English-language databases, though Wu et al., 9 who conducted a Cochrane review in 2014, included Chinese trials. Hinkle et al. 6 included trials from 2000 to 2016, which was updated by Aali et al. 10 to include published trials until January 2020, but these two reviews included only trials published in English. There are two reviews of individual Chinese herbal medicine interventions: Xu et al. 11 reviewed trials of Astragalus membranaceus interventions up to November 2016 and Jin et al. 12 reviewed randomized controlled trials (RCTs) of Buyang Huanwu Tang interventions up to October 2021, but there is no review of all Chinese herbal medicines. Furthermore, these reviews are now out-of-date. Therefore, a comprehensive and up-to-date review is needed to identify all trials (completed and published, completed but not published, and ongoing) of any pharmacological interventions for treating PSF, including trials published in Chinese.
Aims
This systematic review aims to assess “What effect do pharmacological interventions have on post-stroke fatigue and other outcomes in adult stroke survivors with post-stroke fatigue?” (PROSPERO protocol CRD 42022367429, 31 October 2022).
The four objectives were as follows:
To examine the effectiveness of pharmacological treatment in reducing the proportion and/or fatigue severity of stroke survivors with PSF.
To determine the effects of the pharmacological interventions on other outcomes, including quality of life, level of disability and dependency, the ability to return to work, mood, anxiety, apathy, daytime sleepiness, and mortality.
To determine the cost-effectiveness of the pharmacological intervention.
To identify the safety of the pharmacological treatment (i.e. any adverse effect, risk of treatment).
Methods
Search strategy
The following databases were searched from inception till 11 November 2022:
Airitri library;
Chinese National Knowledge Infrastructure (CNKI);
Chinese Scientific Journals Database (VIP);
CINAHL;
ClinicalTrials.gov;
Cochrane Central Register of Controlled Trials (CENTRAL);
Cochrane Stroke Group Trial Register;
EMBASE;
EU Clinical Trial Register;
ISRCTN;
MEDLINE;
PsycINFO;
Wanfang Database;
World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP).
We adopted the MEDLINE search strategy for other databases and translated the search terms for Chinese databases. In addition, we searched ongoing Chinese trials through WHO ICTRP; this platform includes The Chinese Trials Register. We checked the reference lists of all included studies. We did not set language limitations during the literature searches. The search strategies are listed in Appendix A.
Types of studies
We reported the total number of randomized placebo-controlled trials (RCTs) in patients who had PSF at recruitment, where the interventions were used to treat PSF and which measured PSF as an outcome. This included trials that were completed and published, completed but not published, and ongoing.
Types of participants
We included people aged 18 years and older with a clinical diagnosis of stroke, including ischemic, intracerebral, and subarachnoid hemorrhage. We included any method of diagnosis or assessment of PSF.
Types of interventions
We included trials of
A single pharmacological intervention versus placebo;
A combination of two or more pharmacological interventions versus one or more placebo(s).
We excluded trials of
A pharmacological intervention plus usual care versus usual care alone;
Combination of pharmacological and non-pharmacological interventions versus either pharmacological or non-pharmacological interventions alone (due to the possible confounding effect of a non-pharmacological and pharmacological intervention).
Types of outcome measures
Primary outcome: Any type of fatigue measurement at the end of the treatment, including binary (yes/no) or continuous fatigue measurements.
Secondary outcomes (including, but not limited to):
Health-related quality of life;
Disability;
Level of dependence;
Death;
Return to work;
Mood;
Anxiety;
Apathy;
Daytime sleepiness;
Cost-effectiveness of the interventions;
Adverse effects of the intervention.
Study selection
Citations from database searches were imported into Covidence. S.H.C., A.K., and X.Z. independently screened the titles and abstracts and applied inclusion criteria to the full texts (S.H.C. and X.Z. were responsible for Chinese articles; S.H.C., A.K., and X.Z. were responsible for all other articles). Any disagreements were resolved by discussion with a fourth reviewer (G.M.).
Data extraction
A data extraction form was developed and stored on Covidence. The data extracted included participants, intervention, comparison, outcome, study design, and additional notes (e.g. funding sources and possible conflicts of interest). Independent extraction of English-language articles was done by S.H.C. and A.K., and S.H.C. and X.Z. were responsible for the Chinese studies. Then, reviewers discussed any differences to reach a consensus. For trials that did not report any adverse effects of the treatment, we recorded them as “not reported” rather than assuming no adverse effects. We emailed trials’ investigators to request additional information or data when needed.
Risk of bias and quality assessment
We used the Cochrane risk-of-bias tool 2 for randomized trials (RoB2) to assess the quality of structure in five domains: 13
Randomization process: random allocation methods, concealment, and baseline characteristics that could undermine the randomization process.
Deviations from intended interventions: blinding and analysis appropriateness to estimate intervention effects.
Missing outcome data: outcome data availability and potential bias from missing data.
Outcome measurement: outcome and intervention measurement methods’ consistency across treatment and placebo groups.
Selection of the reported result: adherence to pre-specified outcome measure and analysis plan regardless of the results.
S.H.C. and X.Z. independently evaluated the risk of bias for trials published in Chinese; S.H.C. and A.K. assessed all other trials independently, and then the assessments were checked by G.M. Any disagreements were resolved by discussion. We assessed publication bias using funnel plots (Review Manager 5.4.1).
Data synthesis and analysis
For trials that reported two or more fatigue scales, we used the data from the most widely used scale among all 10 published trials. All trials reported continuous outcomes for our primary outcome of fatigue, so we calculated the standardized mean differences (SMDs) and relevant 95% CIs and performed a random effects meta-analysis. We analyzed the effects of interventions separately at the end of the treatment and at the end of follow-up. There were insufficient data on secondary outcomes to perform a meta-analysis, so we summarized results reported by the trials’ authors.
We explored clinical heterogeneity by type of intervention. We had intended to explore heterogeneity by fatigue duration at recruitment, but insufficient data were available.
Assessment of heterogeneity
Forest plots and chi-square tests with the degree of freedom of k − 1 (k is the number of trials) were used to assess the heterogeneity in intervention effects between trials and pre-specified subgroups, using a p-value of < 0.10 because of the small number of trials. We used I2 to quantify the variability in effect estimates across trials due to heterogeneity.
Sensitivity analysis
We conducted a sensitivity analysis for each domain of the risk of bias (RoB2) to explore methodological heterogeneity. We used Z-test to compare the pooled effect size with the summary effect of all included studies for each sensitivity analysis.
Result
Search results
We retrieved 33,297 citations, obtained full texts for 123 publications, and excluded 96 studies after full-text screening, including 35 RCTs that did not have a placebo. We included a total of 27 publications published in English or Chinese (10 published completed trials enrolling 616 participants,14–23 5 publications of secondary analysis of the MIDAS (Modafinil in Debilitating Fatigue After Stroke) trial,24–28 6 completed but not published trials,29–34 and 6 ongoing trials).35–40 All trials found were written in English or Chinese, except for 7 trials registered on the EU Clinical Trial Register in other languages; we screened and applied our inclusion criteria to these trials because English versions were available in the protocols. The reasons for exclusion and reference are shown in the supplementary information (Appendix D). The Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) diagram is shown in Figure 1.
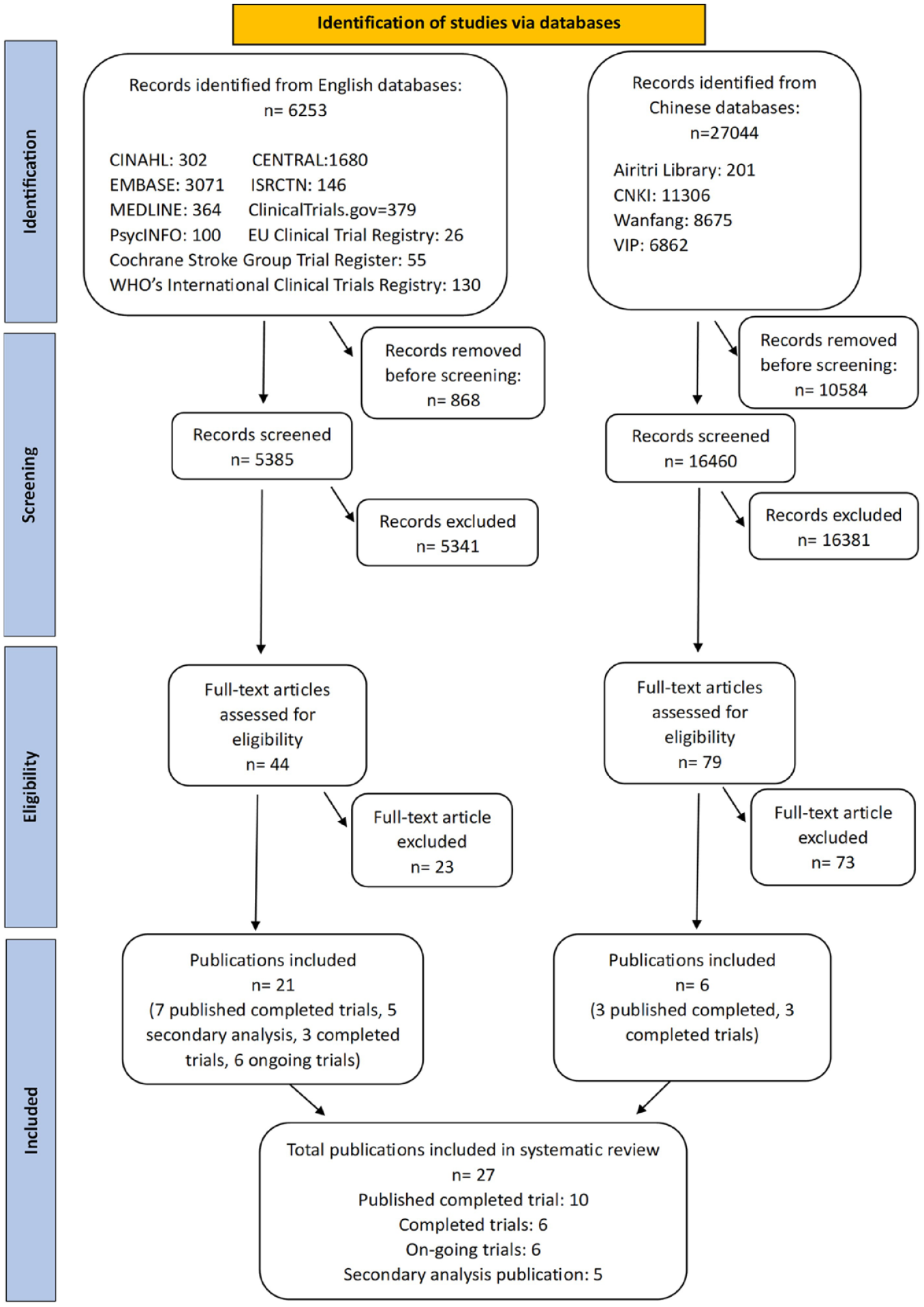
PRISMA flow diagram showing the selection of trials.
Study characteristics
Of 10 included published RCTs (n = 616 at randomization; n = 600 analyzed),14–23 3 were cross-over RCTs, so we used data just from the first phase of treatment,14,16,18 and 7 were parallel RCTs.15,17,19–23 All trials measured fatigue immediately at the end of treatment using continuous outcome measures, most commonly the fatigue severity scale (FSS) (Table 1). The duration of treatment ranged from 28 days to 3 months. Five trials measured fatigue longer term with the duration of follow-up ranging from 8 weeks to 12 months. Further details, including the characteristics of the trials and the primary and secondary outcomes, are in the supplementary information (Appendix B).
Participant characteristics and study design of the included trials.
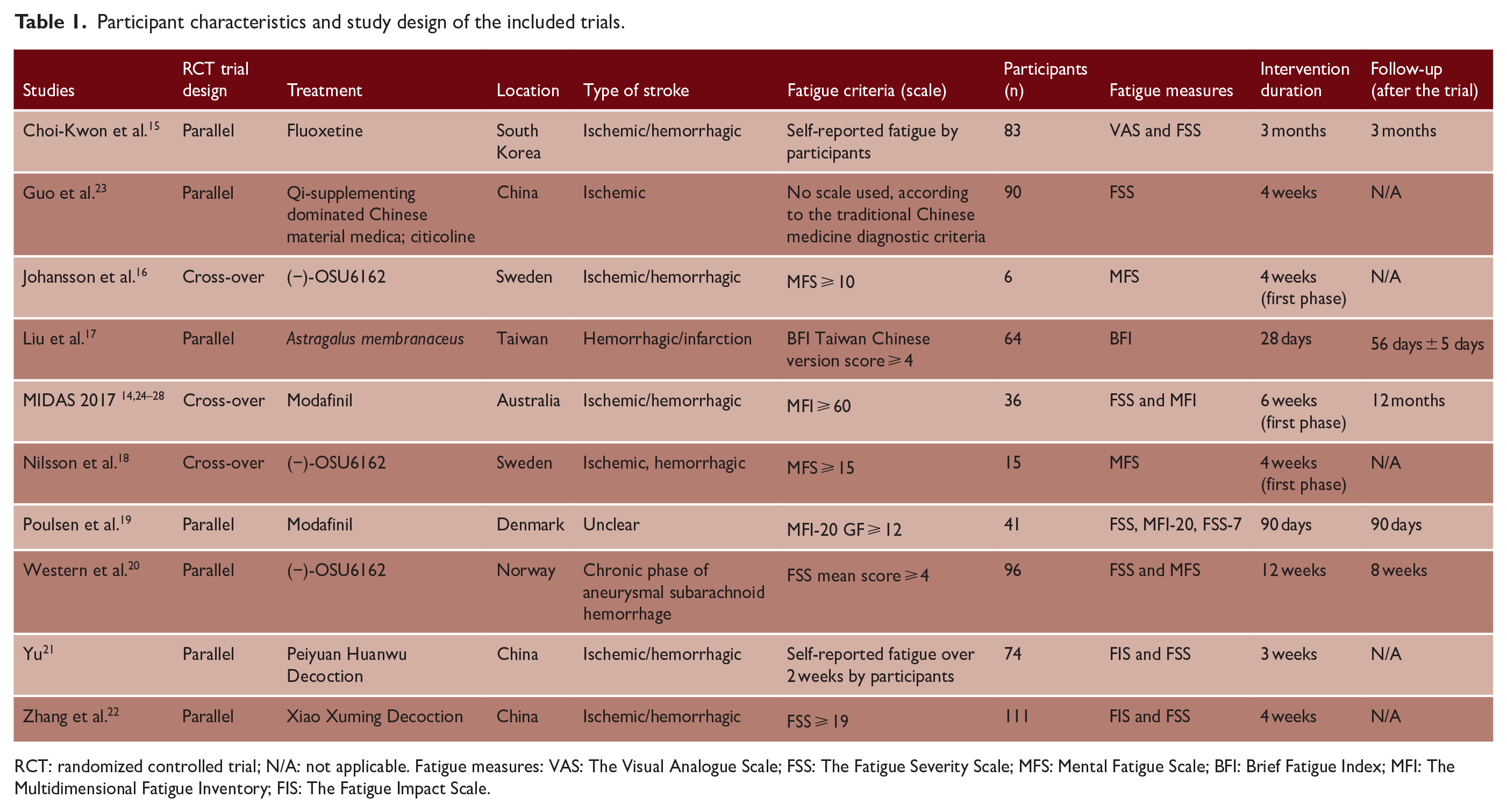
RCT: randomized controlled trial; N/A: not applicable. Fatigue measures: VAS: The Visual Analogue Scale; FSS: The Fatigue Severity Scale; MFS: Mental Fatigue Scale; BFI: Brief Fatigue Index; MFI: The Multidimensional Fatigue Inventory; FIS: The Fatigue Impact Scale.
All 10 published completed trials are included in the meta-analysis. One trial included two intervention groups versus a placebo control group, one with Chinese herbal medicine (referring to Guo 2012a in the meta-analysis) and another using citicoline (Guo 2012b). 23 Therefore, 11 trial arms were included in the meta-analysis (and we ensured that we did not “double-count” the placebo arm).
Ongoing and completed trials
Six randomized placebo-controlled trials have been completed (386 participants), but not yet published,29–34 including three trials on Buyang Huanwu decoction (also known as Peiyuan huanwu decoction),30,33,34 two on OSU6162,16,29 and one on Perispinal Etanercept. 31 A further six ongoing randomized placebo-controlled trials were identified, including four Chinese herbal medicine trials (proposed sample size 112, 180, 400, and 432), a modafinil (proposed sample size 300), and a trial of BP1.3656 (histamine H3 receptor antagonist) intervention (proposed sample size 54).35–40 The supplementary information shows each completed and ongoing trial’s characteristics (Appendix C).
Risk of bias in included studies
We summarized the risk of bias in the 10 included trials (Figures 2 and 3). For cross-over trials, we assessed the risk of bias based on the result of fatigue measurement from the first period of the trial.14,16,18
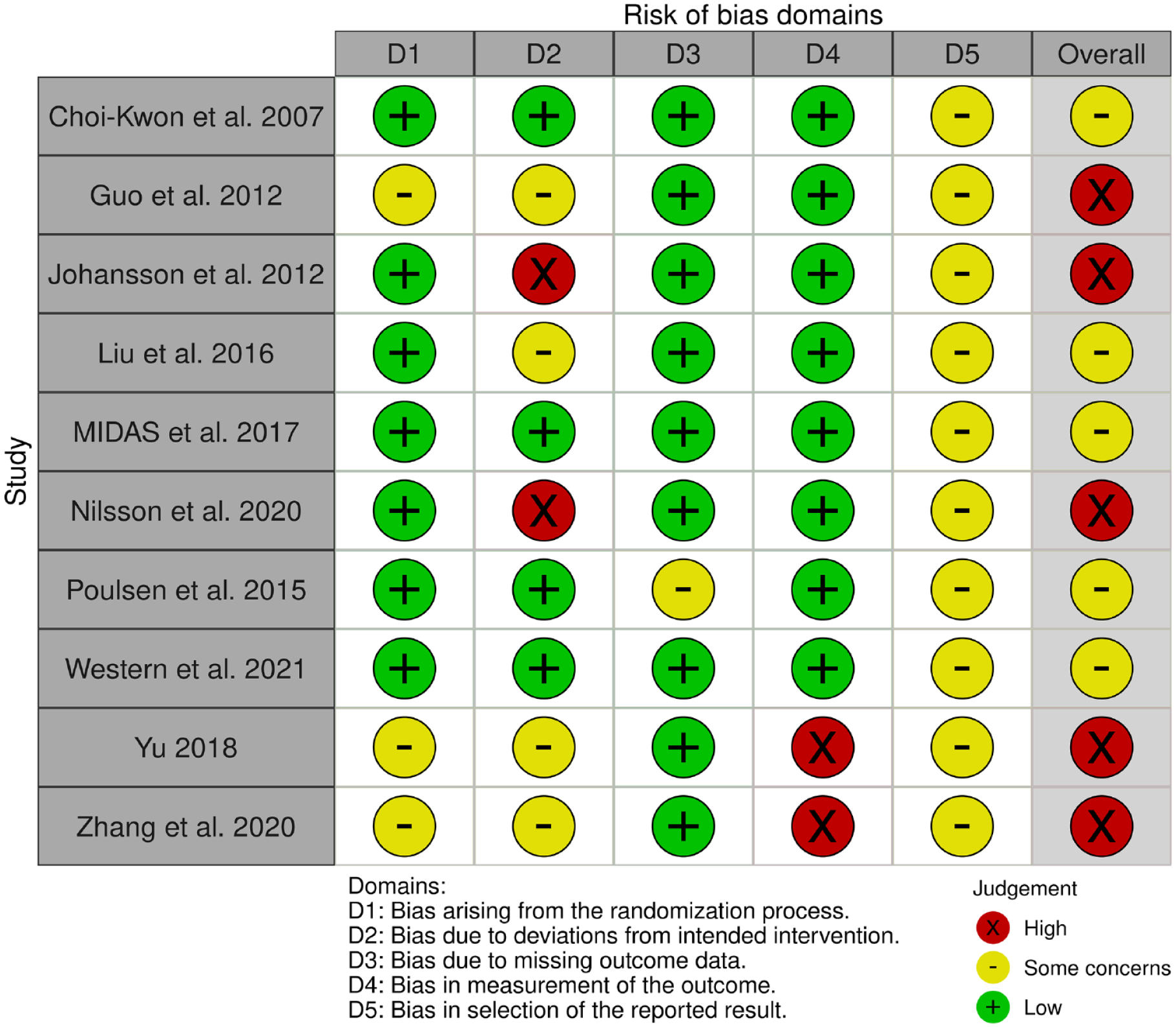
Risk of bias summary: review authors’ judgments on each risk of bias domain for each included study.
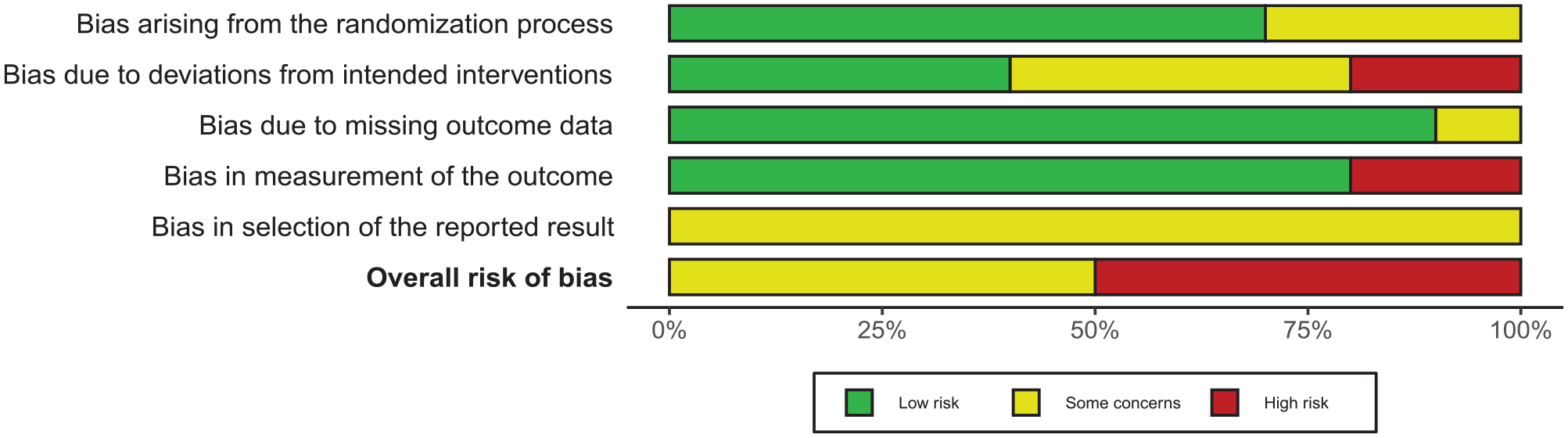
Risk of bias graph: each risk of bias domain presented as percentages across all included studies.
Effect of intervention on fatigue
Fatigue at the end of the treatment
We performed a meta-analysis using SMD with 95% CI. One trial reported median and interquartile range (IQR) in the measure of fatigue at the end of the treatment, 19 so the results were converted into an estimated mean and standard deviation (SD) using the Quantile Estimation method, 41 aided by a statistician, Dr Niall Anderson. Of 10 trials (600 participants),14–23 fatigue severity was lower in the intervention group (pooled SMD = −0.80, 95% CI: −1.29 to −0.31) at the end of the treatment, with significant heterogeneity (I2 = 86%, p < 0.00001) (Figure 4).
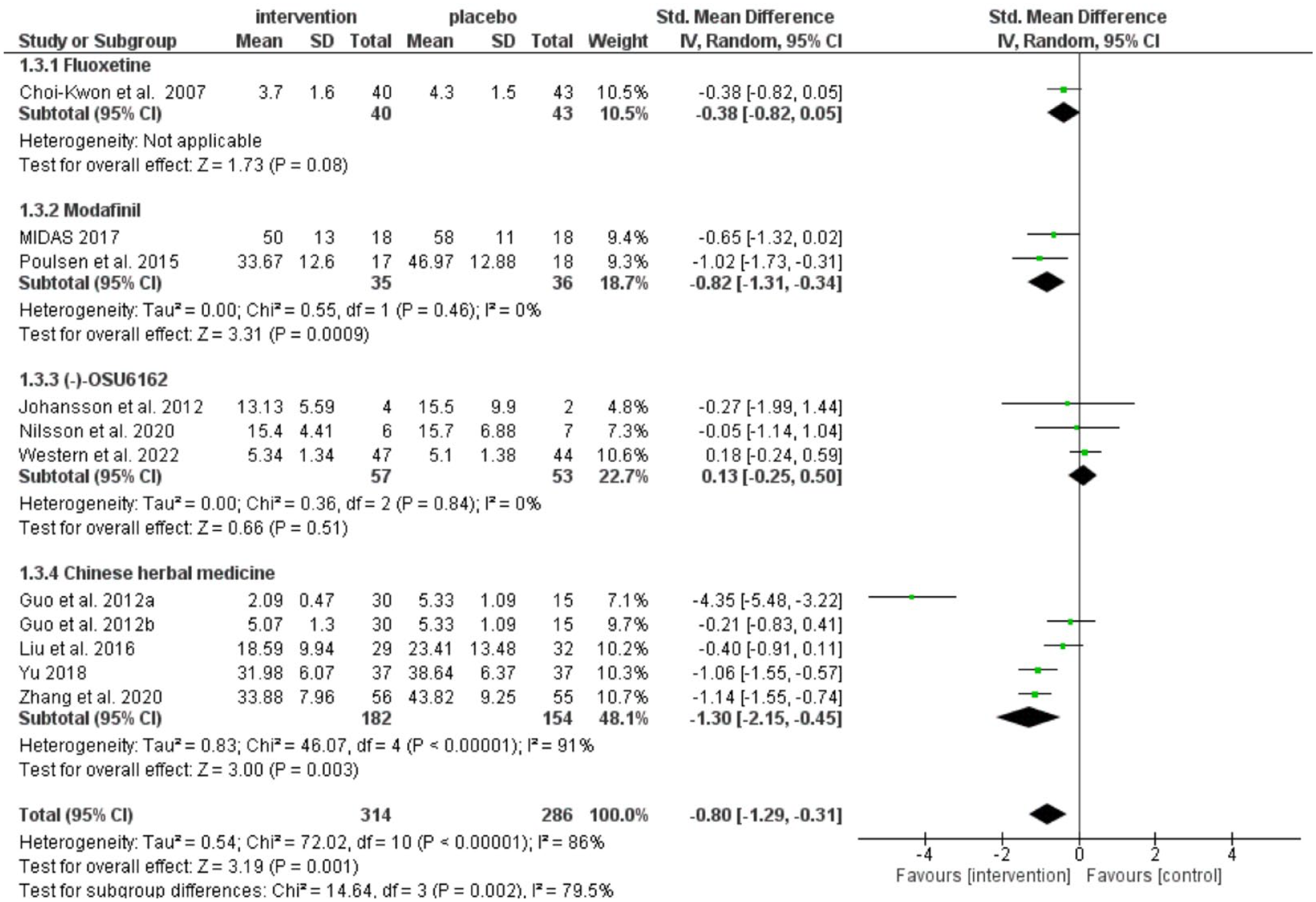
Treatment effects on fatigue at the end of the treatment.
Fatigue at follow-up
Five trials measured fatigue at follow-up (283 participants),14,15,17,19,20 of which data from four trials (265 participants) could be included in the meta-analysis.15,17,19,20 There was no difference in fatigue severity between groups at follow-up (pooled SMD = −0.14, 95% CI: −0.38 to 0.10), with no significant heterogeneity (I2 = 0, p = 0.51) (Figure 5).
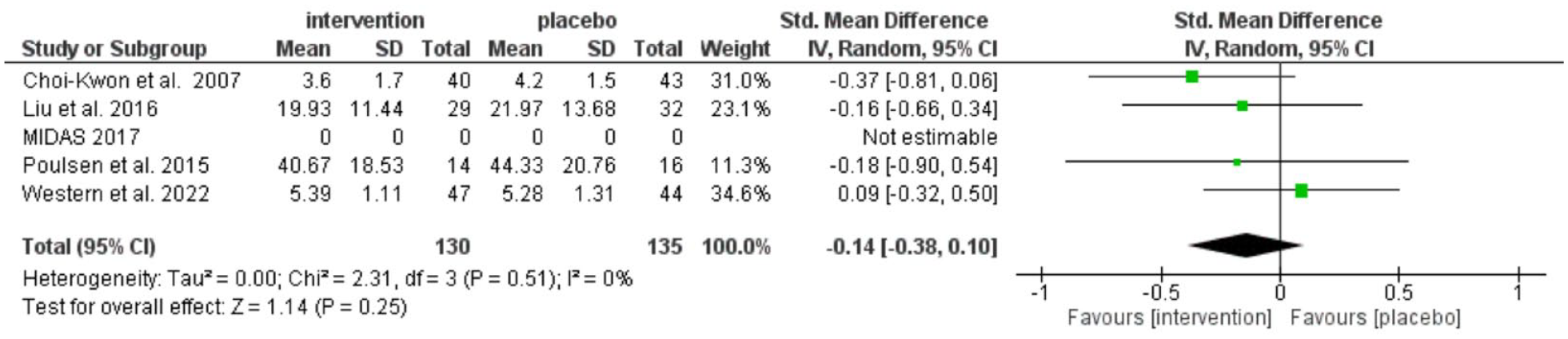
Treatment effects on fatigue of the included trials at follow-up.
Subgroup analysis
We categorized the interventions into four groups: fluoxetine, modafinil, (−)-OSU6162, and Chinese herbal medicines (Figure 4). Fatigue severity was lower in modafinil (pooled SMD = −0.82, 95% CI: −1.31 to −0.34) and Chinese herbal medicine group (pooled SMD = −1.30, 95% CI: −2.15 to −0.45), but not in fluoxetine (pooled SMD = −0.38, 95% CI: −0.82 to 0.05) and (−)-OSU6162 treatments (pooled SMD = 0.13, 95% CI: −0.25 to 0.50). However, the two modafinil trials were both small. There was considerable heterogeneity in the Chinese herbal medicine subgroup (I2 = 91%, p = 0.003). There was only one fluoxetine trial included for analysis. We had intended to perform a subgroup analysis based on the fatigue duration at recruitment, but no trial reported these data.
Publication bias
There were insufficient data points to interpret the Funnel plot (Figure 6).
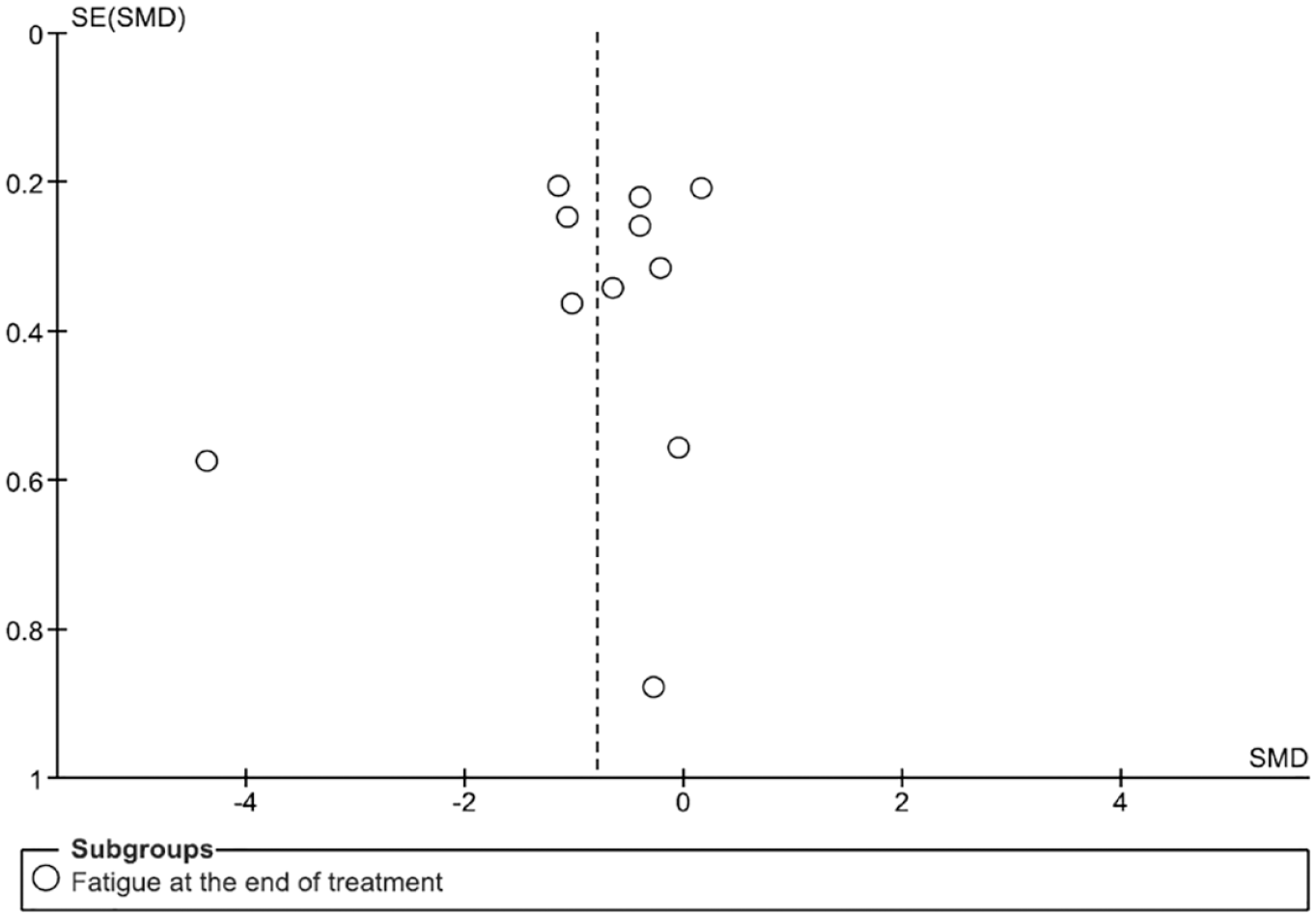
Funnel plot of the meta-analysis of published trials.
Sensitivity analysis
For each domain, only trials with a low risk of bias for that domain were included for analysis (Appendix E).
Domain 1: randomization process. Seven trials were at low risk of bias and were included in the analysis (325 participants).14–20 There was a significant effect on PSF (pooled SMD = −0.35, 95% CI: −0.67 to −0.03), with no statistically significant heterogeneity (I2 = 43%, p-value = 0.10).
Domain 2: deviation from intended interventions. Four trials were at low risk of bias (246 participants).14,15,19,20 There was no significant effect on treating PSF (pooled SMD = −0.41, 95% CI: −0.91 to 0.08), with possible substantial heterogeneity (I2 = 71%, p-value = 0.02).
Domain 3: missing outcome data. Nine trials were at low risk of bias and were included in the analysis (565 participants).14–18,20–23 There was a significant effect on PSF (pooled SMD = −0.78, 95% CI: −1.31 to −0.25), with substantial heterogeneity (I2 = 87, p-value < 0.00001).
Domain 4: measurement of outcome. Eight trials were at low risk of bias and were included in the analysis (415 participants).14–20,23 There was a significant effect on PSF (pooled SMD = −0.73, 95% CI: −1.33 to −0.13), with substantial heterogeneity (I2 = 86%, p-value < 0.00001).
Domain 5: selection of the reported result (reporting bias). All 10 included trials had some or a high risk of bias regarding the selection of the reported results; hence, no trial was included for this sensitivity analysis.
Secondary outcome
There was insufficient data on secondary outcomes to perform a meta-analysis, so we narratively reviewed these data.
No trial reported death, dependency, return to work, apathy, and daytime sleepiness. Only the MIDAS trial assessed the cost-effectiveness of the modafinil treatment and suggested that modafinil is cost-effective in treating fatigue, with an average cost of AUD 3.60 per day. 24
Six trials assessed health-related quality of life.14,17,19,20,22,23 Two modafinil trials reported improvement in Stroke Specific Quality of Life Scale (SSQOL) score: improvement in the domains of self-reported energy, mobility, social roles, vision, and thinking were observed after 6 weeks of modafinil treatment in MIDAS trial; 14 another trial conducted by Poulsen et al. 19 reported significant improvement in the upper extremity function, work and productivity, and languages after 90 days of modafinil treatment (p < 0.05), with the improvement in languages and thinking persisted at follow-up (0.01 < p < 0.05). Three trials on Chinese herbal medicine assessed and reported improvement in overall quality of life: Guo et al. 23 reported participants in the Qi-supplementing dominated Chinese material medica group scored higher in the SSQOL (p < 0.01) than those receiving citicoline (p < 0.01) or placebo (p < 0.01) after 4 weeks; Zhang et al. 22 reported improvement in SSQOL in patients received 4 weeks of Xiao Xuming Decoction (p < 0.05); Liu et al. 17 reported improvement in European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30) on role functioning, cognitive functioning, and global QOL (p < 0.05), and SF-36 physical and social functioning (p < 0.05) after-physical and social functioning (p < 0.05) after 28 days treatment with Astragalus membranaceus, and social functioning was significantly improved at 56 days follow-up (p < 0.05). For (−)-OSU6162, Western et al. 20 reported that 36-Item Short Form Health Survey (SF-36) scores in vitality were improved in both the treatment and placebo groups (p < 0.05).
Five trials assessed disability at the end of the intervention.16–19,21 No significant effect on the Frenchay Activity Index (FAI) scores was observed in one of the (−)-OSU6162 trials, 16 but the other (−)-OSU6162 trial did find an improvement (p = 0.0097). 18 One modafinil trial by Poulsen et al. 19 reported no improvement in the Modified Barthel-100 Index (MBI) score, modified Rankin Scale (mRS) in physical impairment, and Scandinavian Stroke Scale (SSS) in neurological disability at the end of treatment or at 90 days follow-up. Two trials of Chinese herbal medicine reported an improvement in MBI score, including a trial of 3 weeks of treatment of Peiyuan Huanwu Decoction (p < 0.05) 21 and a trial of Astragalus membranaceus after 28 days of treatment and at follow-up (p < 0.05). 17
Six trials assessed anxiety.14–16,18–20 Johansson et al. 16 and Nilsson et al. 18 both reported an improvement in Comprehensive Psychopathological Rating Scale (CPRS) scores at the end of the (−)-OSU6162 treatment, whereas Western et al. 20 reported that Beck Depression Inventory (BDI-II) scores improved significantly in both (−)-OSU6162 and placebo groups, and Beck Anxiety Inventory (BAI) score improved only in the placebo group after 12 weeks of treatment. Poulsen et al. 19 reported no effect on the Major Depression Inventory (MDI) score from modafinil intervention. In the MIDAS trial, modafinil had no significant impact on anxiety, mood, and overall Depression, Anixety and Stress Scale (DASS) scores (p = 0.7688). 14 Choi-Kwon et al. 15 reported that fluoxetine significantly improved emotional incontinence and anger proneness (p < 0.05), but not depression (p = 0.05).
Only six trials reported adverse events,14,16–20 of which four assessed adverse events by interviewing or reviewing patients’ records during the interventions.14,18–20 The assessment methods were unclear in two trials.16,17 All reported mild adverse reactions in both the placebo and intervention groups, and no serious adverse events were observed.
Discussion
Summary of main results
This systematic review and meta-analysis is the most up-to-date synthesis of all placebo-controlled RCTs of pharmacological interventions to treat PSF. We synthesized 10 completed RCTs recruiting 616 participants. Interventions included fluoxetine, modafinil, (−)-OSU6162, and Chinese herbal medicines. Although meta-analysis suggested a beneficial effect with lower severity of fatigue of the pharmacological interventions at the end of the treatment (pooled SMD = −0.80, 95% CI: −1.29 to −0.31), there was a substantial risk of bias. Of the five trials (n = 283) that measured fatigue at follow-up, the treatments had no benefit (pooled SMD = −0.14, 95% CI: −0.38 to 0.10). There was insufficient data to perform a meta-analysis on the quality of life and other secondary outcomes, but some individual trials reported some benefits.
Limitations of the included studies
First, the sample size of each included trial was small (ranging from 6 to 111 participants), and there was significant heterogeneity between trials in fatigue criteria at recruitment, intervention types, and outcome measures for fatigue. Moreover, the rationale of primary outcome measure scale chosen was not described, limiting our understanding of which aspect of the fatigue the trialists were attempting to study. Even among the Chinese herbal medicine trials, there was considerable heterogeneity, which could be explained by the formulation of the Chinese herbal medicine, because the decoctions used in each trial had a different composition. Second, there is a lack of evidence and understanding of which ingredient(s) or components are associated with any observed clinical benefits or adverse effects, as decoctions often contain a complex composition and lack of standardization of the quantity of each ingredient in the decoction. 11 Third, the overall methodological quality of all 10 studies was low. All 10 studies had some concerns in reporting bias, and none of the studies was at low risk of bias overall. Fourth, only six trials reported adverse events, but these were not systematically documented. Only one of the modafinil trials assessed its cost-effectiveness, modeled using data from Australia; it is unclear whether the same economic model would be applicable in other settings and countries. In sum, these factors result in downgrading the quality of the evidence and prevent us from being able to make firm recommendations about the pharmacological management of PSF.
Strengths and weakness of this review
Previous reviews have examined the evidence on PSF interventions, focusing on studies published in English-language databases,6,9,10 or a single Chinese herbal medicine.11,12 This review examined completed trials not exclusively published in the English-language databases, thus addressing the language bias of previous reviews and extended geographical location to search and include trials from Chinese databases. This allows a more comprehensive investigation of PSF treatment and provides insights into different Chinese herbal medicines that are used in Asia and Asian populations. Furthermore, two independent review authors screened citations to reduce the risk of inadvertently excluding relevant trials. Two reviewers also extracted data and assessed the risk of bias, thus minimizing errors in the review process. 13 We were able to screen all the identified trials, including those written in other languages as all had English versions available. However, it is possible that studies that were not indexed in databases we searched might have been overlooked, and we were not able to include ongoing trials that did not have registered protocols; thus, we invite readers to contact us if they are aware of any studies that have been missed.
Review results compared to previous reviews
This up-to-date review has extended the findings from previous smaller reviews to all pharmacological interventions, including Chinese herbal medicines, providing more evidence of the possible beneficial effect on reducing fatigue severity among stroke fatigue across different types of treatments.
Conclusion
Implication for clinical care
There is insufficient evidence to support a particular pharmacological treatment for PSF or to support a change in current clinical practice or guidelines. This information is helpful for clinicians and patients, and will avoid the unnecessary prescription of drugs that are unlikely to be of benefit. Anecdotally, people with PSF are often offered antidepressants empirically on the basis that their fatigue might be part of a depressive disorder—our review does not support this approach, unless they have a definite mood disorder.
Implication for research
The six ongoing placebo-controlled RCTs address several methodological concerns we identified in the published trials, including small sample size and single-center design. For instance, four ongoing Chinese herbal medicine trials proposed larger sample sizes ranging from 112 to 432 Chinese patients.36–38,40 However, two of these trials did not mention the treatment duration,36,37 and one trial did not specify the study design to be double-blinded. 37 None of these Chinese trials outlined plans for assessing adverse events or follow-up. MIDAS is conducting a phase 3, placebo-controlled, double-blinded, RCT trial with a larger sample size of 300 participants, investigating modafinil treatment for an extended duration of 56 days compared to the previous phase 2 trial. 35 One trial tests a new drug that has not been studied for PSF, BP1.3656, among 54 patients in France and Switzerland. 39 Further trials are needed to explore this drug’s efficacy in larger sample sizes, other stroke types, and different populations. While most of these ongoing trials (five trials) are multicenter, it remains important to test the efficacy of the drugs in diverse populations, including Chinese herbal medicines in other ethnic groups. It is worth noting that, except for the MIDAS trial, none of the other five trials included data analysis plans in their protocols.
Based on the methodological limitations in these ongoing trials, more trials are still needed to examine these drugs to comprehensively understand their efficacy, immediate and long-term effects, safety, and cost-effectiveness. First, larger, multicentre trials and broader inclusion criteria are needed to test drug treatment for PSF. Data in this review can be used to inform power calculations for bigger trials. Second, it is crucial that future trials are robustly designed to minimize bias, improve their study design’s validity, and provide comprehensive details on their protocols, including the details of the randomization process and blinding, choosing appropriate analysis methods, and ensuring adherence to the pre-specified plan. Third, baseline characteristics need better characterization (including fatigue duration, mood, and aphasia). Fourth, for outcome measure, trialists should carefully consider which aspects of fatigue, such as severity, impact, and type (i.e. mental or physical), explain the rationale for selecting their outcome measures appropriate for their research question, 42 taking into account the mechanism of actions of the treatment and which aspect of fatigue is most likely to be improved. In addition, trials should report outcomes several weeks or months after treatment ends, not just at the end, to determine whether any treatment effects are long-lasting. Fifth, important secondary outcomes (i.e. mood, anxiety, quality of life, and return to work) should be collected by all trials. Adverse events should be systematically recorded at every follow-up. More evaluations on cost-effectiveness are needed to provide more information on the affordability, applicability, and policy decision in pharmacological interventions for treating PSF in clinical settings.
Supplemental Material
sj-docx-1-wso-10.1177_17474930231196648 – Supplemental material for Systematic review: Pharmacological interventions for the treatment of post-stroke fatigue
Supplemental material, sj-docx-1-wso-10.1177_17474930231196648 for Systematic review: Pharmacological interventions for the treatment of post-stroke fatigue by Shuk Han Chu, Xu Zhao, Ahmad Komber, Joshua Cheyne, Simiao Wu, Eileen Cowey, Mansur Kutlubaev and Gillian Mead in International Journal of Stroke
Supplemental Material
sj-jpg-2-wso-10.1177_17474930231196648 – Supplemental material for Systematic review: Pharmacological interventions for the treatment of post-stroke fatigue
Supplemental material, sj-jpg-2-wso-10.1177_17474930231196648 for Systematic review: Pharmacological interventions for the treatment of post-stroke fatigue by Shuk Han Chu, Xu Zhao, Ahmad Komber, Joshua Cheyne, Simiao Wu, Eileen Cowey, Mansur Kutlubaev and Gillian Mead in International Journal of Stroke
Supplemental Material
sj-jpg-3-wso-10.1177_17474930231196648 – Supplemental material for Systematic review: Pharmacological interventions for the treatment of post-stroke fatigue
Supplemental material, sj-jpg-3-wso-10.1177_17474930231196648 for Systematic review: Pharmacological interventions for the treatment of post-stroke fatigue by Shuk Han Chu, Xu Zhao, Ahmad Komber, Joshua Cheyne, Simiao Wu, Eileen Cowey, Mansur Kutlubaev and Gillian Mead in International Journal of Stroke
Supplemental Material
sj-jpg-4-wso-10.1177_17474930231196648 – Supplemental material for Systematic review: Pharmacological interventions for the treatment of post-stroke fatigue
Supplemental material, sj-jpg-4-wso-10.1177_17474930231196648 for Systematic review: Pharmacological interventions for the treatment of post-stroke fatigue by Shuk Han Chu, Xu Zhao, Ahmad Komber, Joshua Cheyne, Simiao Wu, Eileen Cowey, Mansur Kutlubaev and Gillian Mead in International Journal of Stroke
Supplemental Material
sj-jpg-5-wso-10.1177_17474930231196648 – Supplemental material for Systematic review: Pharmacological interventions for the treatment of post-stroke fatigue
Supplemental material, sj-jpg-5-wso-10.1177_17474930231196648 for Systematic review: Pharmacological interventions for the treatment of post-stroke fatigue by Shuk Han Chu, Xu Zhao, Ahmad Komber, Joshua Cheyne, Simiao Wu, Eileen Cowey, Mansur Kutlubaev and Gillian Mead in International Journal of Stroke
Footnotes
Acknowledgements
The authors are very grateful to Dr Niall Anderson for his advice on the meta-analysis and Dr Marie Nilsson and Professor Angelika Sortberg for providing the trial’s data for the meta-analysis.
Correction (September 2023):
Article updated to correct Joshua Cheyne’s affiliation details.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.K. was supported by the Bashkir State Medical University Strategic Academic Leadership Program (PRIORITY-2030). S.W., member of DSMB of Large Artery Occlusion Treated in Extended Time With Mechanical Thrombectomy Trial (LATE-MT), received grants from National Natural Science Foundation of China (82171285) and Science and Technology Department of Sichuan Province (2021YJ0433I). S.H.C., X.Z., A.K., J.C., E.C., and G.M. have no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
