Abstract
Apathy is a reduction in goal-directed activity in the cognitive, behavioral, emotional, or social domains of a patient’s life and occurs in one out of three patients after stroke. Despite this, apathy is clinically under-recognized and poorly understood. This overview provides a contemporary introduction to apathy in stroke for researchers and practitioners, covering topics including diagnosis, neurobiological mechanisms, associated consequences, and potential treatments for apathy. Apathy is often misdiagnosed as other post-stroke conditions such as depression. Accurate differential diagnosis of apathy, which manifests as reductions in initiative, and depression, which manifests as negative emotionality, is important as it informs prognosis. Research on the neurobiology of apathy suggests that there are few consistent associations between stroke lesion location and the development of apathy. These may be resolved by adopting a network neuroscience approach, which models apathy as a pathology arising from structural or functional damage to brain networks underlying motivated behavior. Importantly, networks can be affected by physiological changes related to stroke, including the acute infarct but also diaschisis and neurodegeneration. Aside from neurobiological changes, apathy is also associated with other negative outcome measures such as functional disability, cognitive impairment, and emotional distress, suggesting that apathy is indicative of a worse prognosis following stroke. Unfortunately, high-quality trials aimed at treating apathy are scarce. Antidepressants may have limited effects on apathy. Acetylcholine and dopamine pharmacotherapy, behavioral interventions, and transcranial magnetic stimulation may be more promising avenues for treatment.
Introduction
Apathy is a behavioral syndrome characterized by a loss of motivation that occurs in one-third of patients after stroke.1,2 Post-stroke patients with apathy suffer from greater functional impairment and demonstrate slower recovery times to normal functioning.3,4 Furthermore, apathy is a risk factor for incident vascular disease, dementia, and mortality.5,6 Despite high prevalence and an impact on outcomes after stroke, apathy remains poorly understood. It is also under-recognized, although the extent of this is unknown. This leads to a dearth of treatment approaches. This overview provides a contemporary introduction to apathy in stroke for researchers and practitioners, covering topics including diagnosis, neurobiological mechanisms, associated consequences, and potential treatments for apathy. The search strategy and selection criteria for papers referenced in this overview can be found after the “Discussion” section.
Diagnostic criteria for apathy
Diagnostic criteria for apathy.
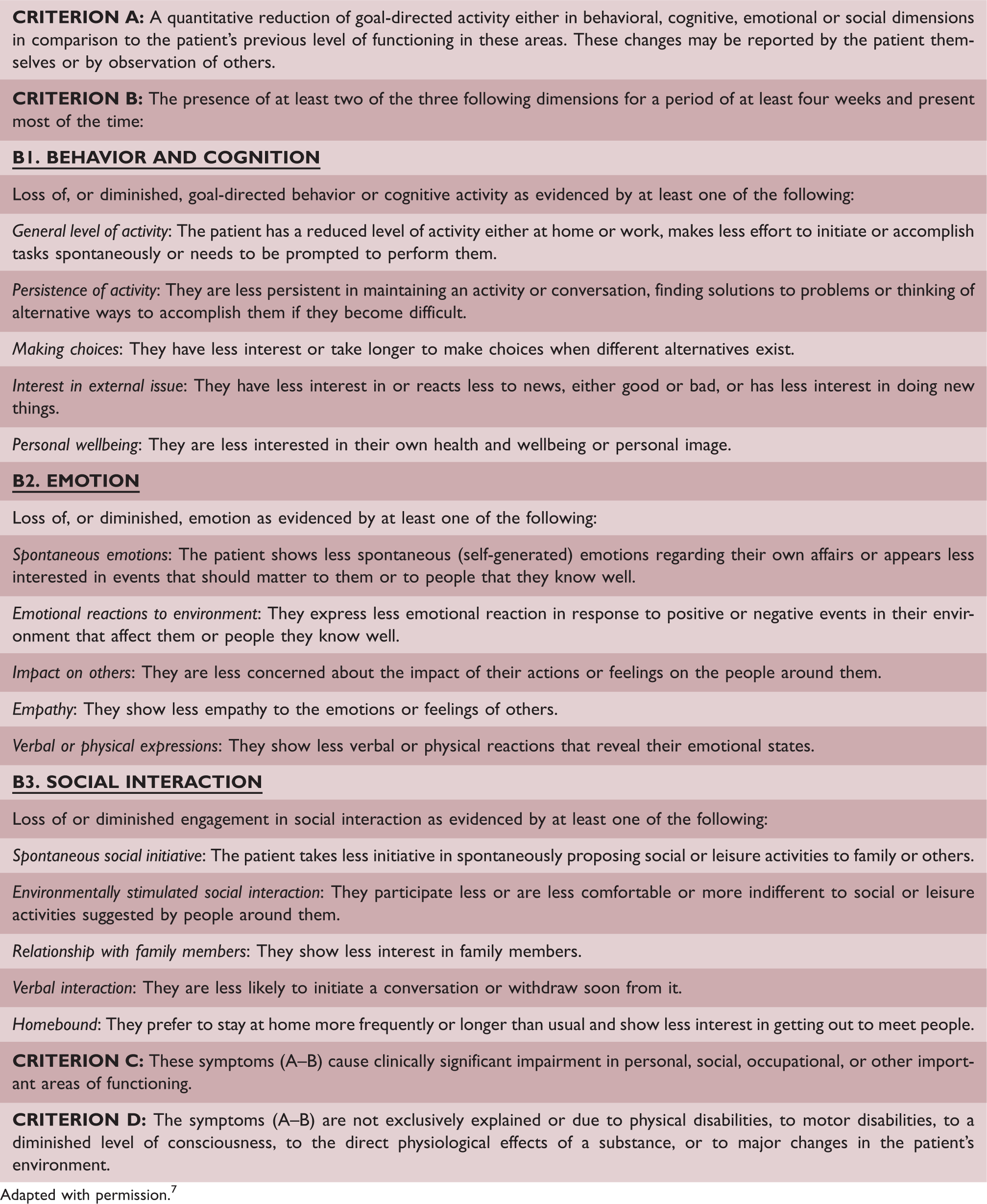
Adapted with permission. 7
Commonly used apathy scales.
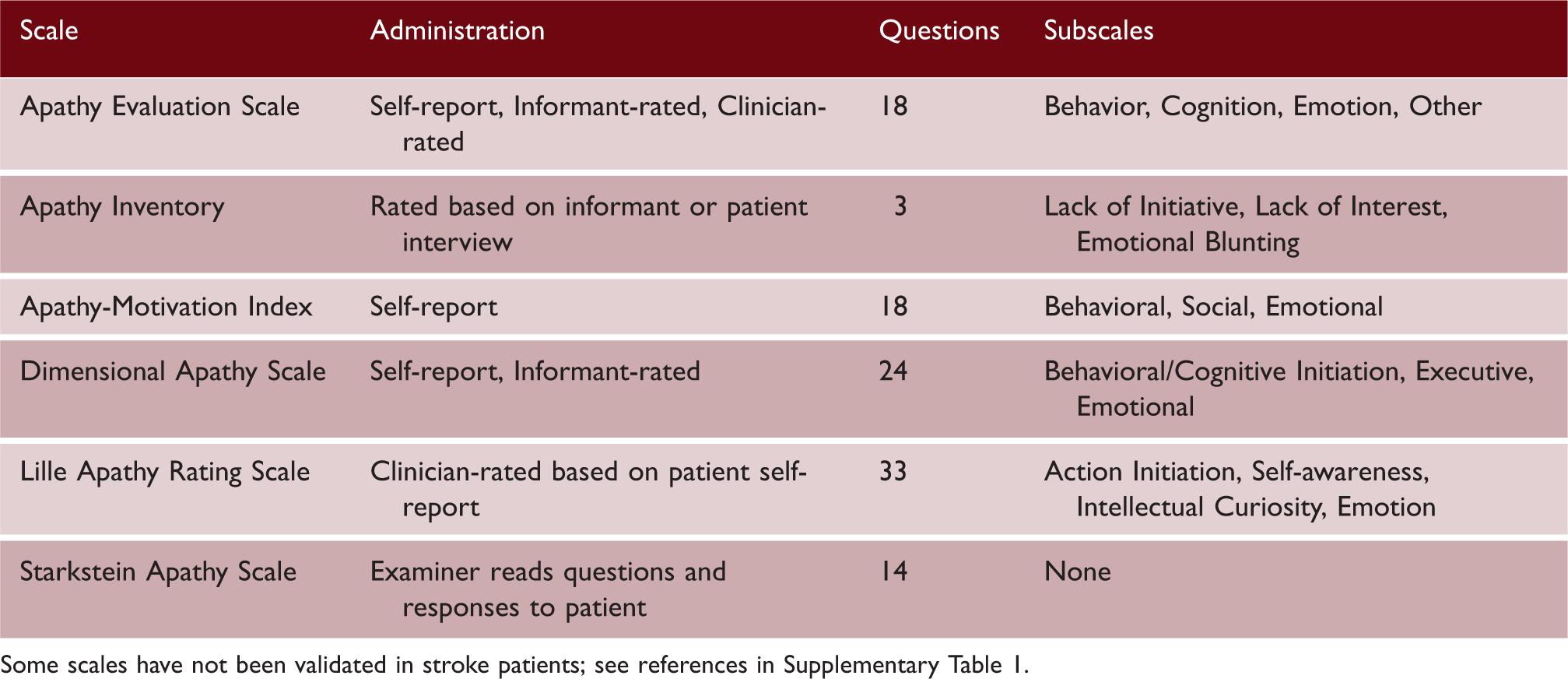
Some scales have not been validated in stroke patients; see references in Supplementary Table 1.
Apathy is described as a symptom in the International Classification of Diseases, Eleventh Revision under code MB24.4. Similarly, the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition acknowledges apathy as a symptom of other disorders, such as mood and neurocognitive disorders. 9 Unfortunately, neither classification system describes apathy as a syndrome, potentially limiting its recognition.
Differential diagnosis of post-stroke apathy
Diagnosing apathy may be complicated by other post-stroke neuropsychiatric symptoms, particularly depression. Core symptoms of a depressive episode, which can be triggered by stroke, include low mood and diminished pleasure (anhedonia).
9
Depressive symptoms such as anhedonia can appear behaviorally similar to apathy and may have a similar neurobiological basis, making them difficult to distinguish in practice (Figure 1(a)).
Apathy and depression are dissociable syndromes: (a) distinct and overlapping symptoms of apathy and depression. Apathy manifests as a reduction in goal-directed behaviors while depression is marked by negative emotionality. Overlapping symptoms include loss of pleasure and energy; (b) prevalence rates of post-stroke apathy and depression in studies with n ≥ 100. Patients present with either apathy or depression, with a minority showing comorbid symptoms. Distinguishing between the two informs prognosis and treatment.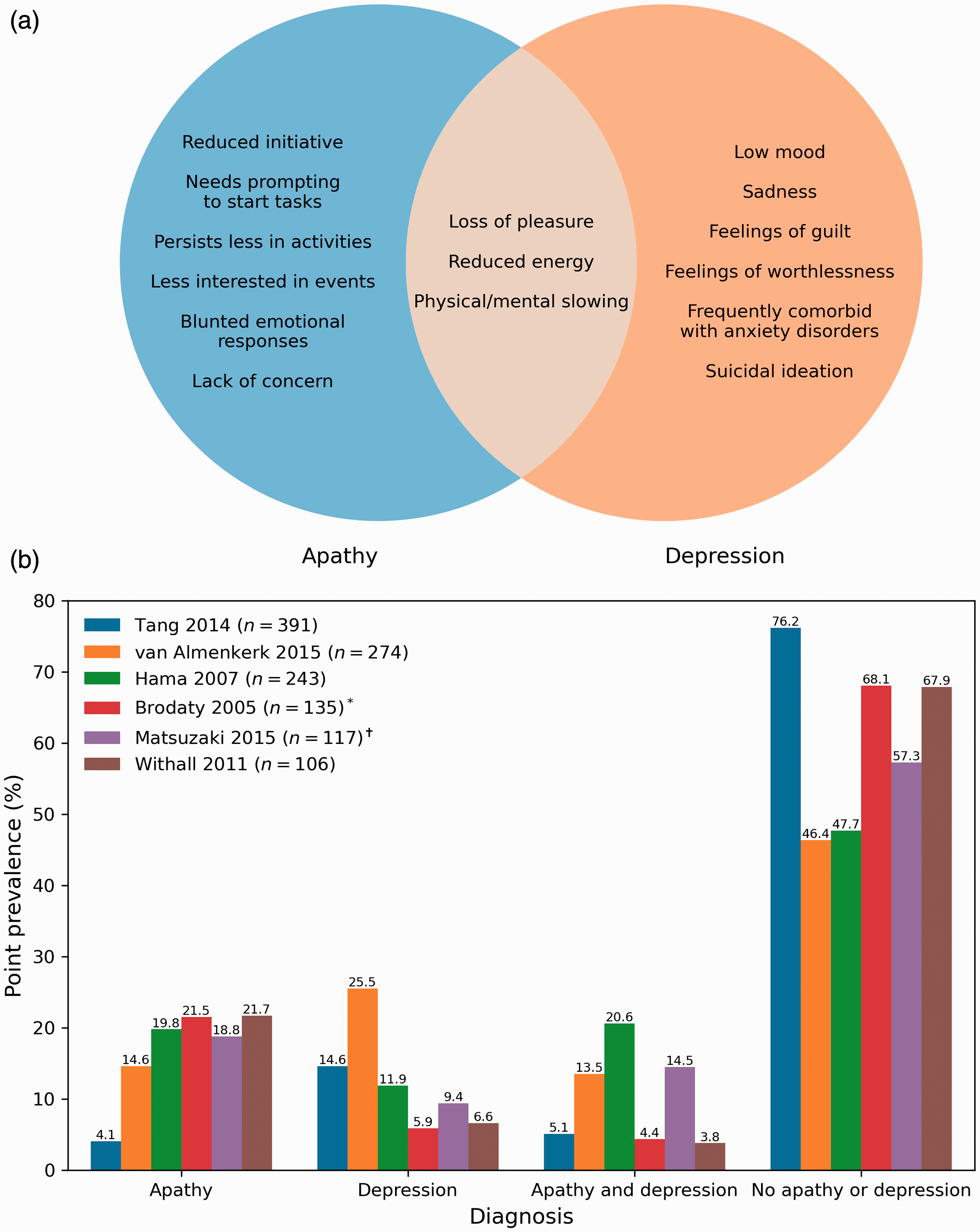
Despite shared symptoms, post-stroke apathy and depression are dissociable syndromes with different prevalence rates (Figure 1(b)). Stroke patients tend to develop either apathy or depression, with a minority showing symptoms of both (Figure 1(b)). Furthermore, apathy and depression have different trajectories10,11 and effects on outcomes such as functional disability12,13 and cognition.6,14,15
Negative emotionality is a key characteristic of depression that distinguishes it from apathy. Depressed patients may present with pessimism and hopelessness, while those with apathy show a lack of emotional distress. 16 Depressed patients can also actively engage in avoidant behavior, resisting socializing and treatment attempts, while apathetic patients are passive and indifferent to these activities. 16
Post-stroke fatigue may also be a potential comorbidity with post-stroke apathy. Fatigue can be defined as a subjective feeling of extreme and persistent physical/mental tiredness, weakness, or exhaustion. 17 Although fatigue has a similar behavioral manifestation to apathy (i.e. less energy), preliminary findings indicate that they are not correlated and do not interact, 18 suggesting that the two are independent.
Apathy itself may present differently based on the underlying neurological disease. More patients with mixed dementia, which includes individuals with cerebrovascular disease, may show deficits in behavioral initiation compared to patients with other neurocognitive diseases. 8 Conversely, mixed dementia patients also show the lowest proportion of emotional impairment compared to other disorders except major depression. 8 Although these patients did not have stroke per se, stroke pathology is highly prevalent in mixed dementia. 19
Prevalence and natural history of post-stroke apathy
Apathy presents in approximately one-third of stroke patients, with symptoms beginning as early as four days post-stroke. 20 Longitudinal research suggests that most post-stroke patients have a constant level of high (7%), moderate (33%), or low/no (50%) apathy for up to a year after stroke, with a minority improving (7%) or worsening (7%). 4 The prevalence of apathy increases by ∼10% five years following stroke, 21 although this may be an underestimate as patients with apathy may be more likely to drop out of longitudinal studies. 4
Neurobiological mechanisms underlying apathy
Post-stroke apathy is increasingly recognized as a consequence of neurobiological changes triggered by a stroke. Apathy is traditionally described as the result of damage to specific brain structures related to GDB such as the basal ganglia and prefrontal cortex. 22 If this lesion-deficit view of apathy was true, one would expect a clear relationship between lesion location and apathy. However, no common localizations across stroke studies have been found,2,23 suggesting that relationships between structural damage and functional deficits are more complex than initially thought.
A recent theoretical approach has recontextualized apathy in cerebrovascular disease as the product of damage to brain networks underlying GDB. 24 This network-based framework suggests that post-stroke apathy follows focal lesions in key network regions or diffuse cerebrovascular pathology disrupting connections within networks. Acute infarcts to core brain regions underlying GDB can result in apathy, recapitulating the lesion-deficit view. Alternatively, diffuse white matter damage due to cerebral small vessel disease can lead to network disruption, 25 explaining associations between MRI markers of small vessel disease and apathy. 26
This framework also suggests that post-stroke phenomena such as functional diaschisis and secondary neurodegeneration drive increases in apathy over time.
27
These phenomena can occur distal to an acute infarct, leading to clinical symptoms that appear inconsistent or unexpected if only considering a focal lesion. Importantly, secondary neurobiological changes can propagate through structural and functional connections within the brain, potentially leading to apathy if affecting GDB-related networks (Figure 2). As a corollary, apathy may improve in response to restorative mechanisms such as adaptive plasticity and functional remapping.
27
A network-based model of how stroke leads to apathy. Apathy can result from focal lesions disrupting key network regions or from diffuse damage such as white matter ischemia. Acute strokes may not immediately lead to apathy symptoms if not occurring in brain regions supporting goal-directed behaviors but can result in the delayed onset of apathy. Infarcts may lead to disrupted functioning in connected areas, known as diaschisis. Over time, secondary neurodegenerative processes lead to atrophy in regions structurally connected to the infarct, which propagates diaschisis-related deficits further throughout the brain. This network-based spreading of stroke-related pathology may explain why apathy can occur in individuals who do not have acute infarcts in motivation-related regions, and why apathy symptoms worsen in some patients over time.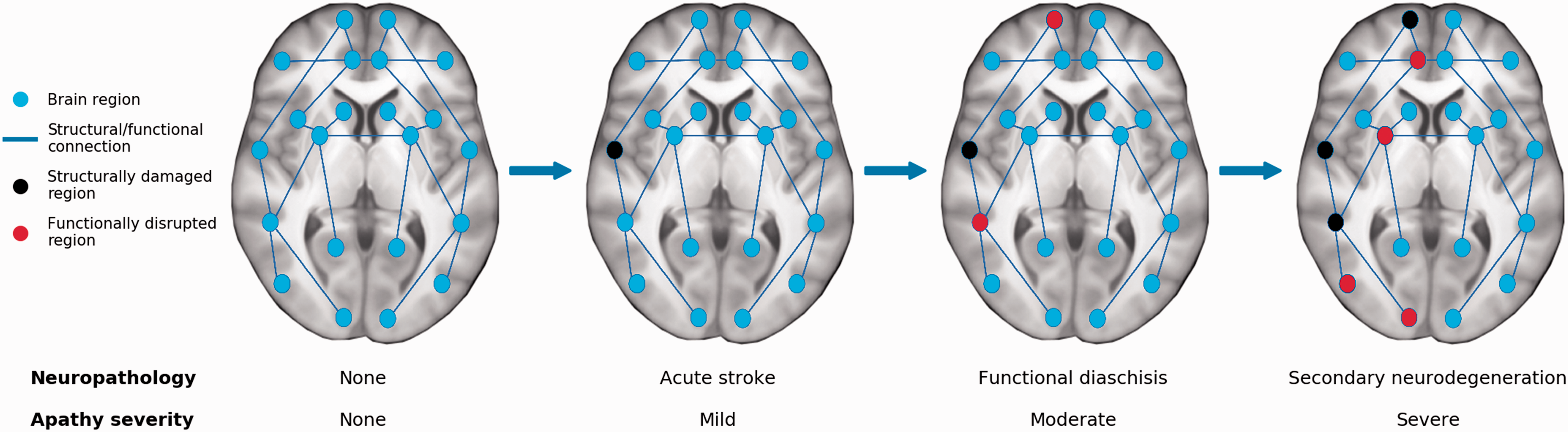
The anterior cingulate cortex (ACC) and nucleus accumbens may be core network regions supporting GDB, as damage to these structures is associated with apathy across neurological disorders. 28 These core regions are embedded in large-scale functionally connected networks that underlie GDB-related cognitive functions such as reward-based decision-making, attentional control, and reinforcement learning.24,28 Disruption to these networks may lead to cognitive deficits that manifest behaviorally as apathy (Supplementary Table 2). 24
This network-based conceptualization of apathy explains a wide range of findings in patients with post-stroke apathy, although further testing of it is necessary. Future work could investigate time since stroke in conjunction with more precise localizations of the acute infarct and any white matter ischemia, given the specificity of the neuroanatomical networks and neurophysiological processes that may be related to post-stroke apathy. 24
Effects of apathy on functional outcomes
Post-stroke apathy is associated with functional disability, including reductions in basic activities of daily living such as eating or dressing and slower functional recovery over time.2,3,12 Factors underlying these relationships remain unexplored, although a potential explanation is that motivational deficits impair recovery by reducing engagement in rehabilitation programs. Alternatively, apathy could precede sedentary behavior, explaining associations between apathy and incident vascular disease. 5
Apathy is associated with general cognitive deficits, with post-stroke apathy patients scoring ∼2–3 points lower on the Mini-Mental State Examination. 2 Apathy has also been associated with impairments in specific cognitive domains such as verbal learning, short- and long-term verbal recall, semantic fluency, abstract reasoning, and attention and concentration.11,14,15,29 This suggests that some post-stroke patients with apathy suffer from cognitive impairment, particularly in executive and memory-related domains, supporting the notion that similar neurobiological networks underlie cognitive function and motivated behavior.24,26 These cognitive deficits may have functional consequences. Apathetic patients show diminished instrumental activities of daily living, which are tasks that require planning such as shopping and housekeeping.29,30 Furthermore, apathy is associated with worse scores on dementia scales and a higher risk of incident dementia, suggesting that apathy may be symptomatic of prodromal vascular dementia.6,31
Finally, although apathy can co-occur with depression, apathy may be a risk factor for developing subsequent depression. 10 Apathy is also associated with suicidal ideation independently of depression three months after stroke. 32 This suggests that apathy can not only occur in tandem with depression but also exacerbate it.
Treatments for post-stroke apathy
There is a lack of high-quality evidence to guide management of post-stroke apathy. Treatment can be considered under pharmacological, behavioral, and other approaches. Given the importance of treatment, we have included all trials that were found (Search strategy and selection criteria).
Pharmacological approaches for treating apathy
Antidepressants such as selective serotonin reuptake inhibitors (SSRIs) may be prescribed for apathy in clinical practice due to shared symptomatology with depression. Unfortunately, little evidence suggests that antidepressants are effective for treating apathy in the absence of additional depressive symptoms, with some research suggesting that certain antidepressants exacerbate effort-based decision-making deficits. 33
One double-blind placebo-controlled trial examined the effects of the SSRI escitalopram (10 mg/day in patients ≤65 or 5 mg/day in patients >65) in apathy-free stroke patients. 34 After 12 months, the 51 participants receiving escitalopram were 3.47 times less likely to develop apathy compared to the 47 on placebo. 34 This suggests that escitalopram reduces apathy risk, though future trials are needed to determine whether it ameliorates existing post-stroke apathy.
Two double-blind placebo-controlled trials have evaluated the efficacy of nefiracetam, which enhances monoaminergic, cholinergic, and GABAergic signaling to treat post-stroke apathy. The first was conducted in patients with post-stroke depression and apathy and assigned patients to placebo (n = 22), 600 mg/day nefiracetam (n = 26), or 900 mg/day nefiracetam (n = 22). 35 After 12 weeks, the patients receiving 900 mg nefiracetam had a greater reduction in apathy compared to those receiving 600 mg nefiracetam or placebo. The second study examined post-stroke apathy patients and assigned participants 900 mg/day nefiracetam (n = 6) or placebo (n = 7), but did not find a statistically significant decrease in apathy after 12 weeks. 36 The conflicting results of these studies should be interpreted with caution due to small samples.
One open-label trial examined the acetylcholinesterase inhibitors galantamine and donepezil in treating apathy in 26 cognitively impaired stroke patients. 37 Thirteen patients were administered galantamine on a dosing regimen starting at 4 mg twice per day (b.i.d.) before increasing to 8 mg b.i.d. and then 12 mg b.i.d. in four-week increments. Remaining patients were administered donepezil starting at 5 mg/day before increasing to 10 mg/day in six-week increments. After 12 weeks, the entire sample showed a non-statistically significant decrease in apathy compared to baseline. 37 Acetylcholinesterase inhibitors warrant further consideration, however, given the small sample in this study and positive results in dementia studies. 38
These inconclusive results preclude the recommendation of any pharmacological treatment for apathy in stroke, although research in other neurological diseases may provide avenues for future investigation. For instance, acetylcholinesterase inhibitors show promise in treating apathy in dementia, 38 while dopamine has been suggested as a treatment for apathy in Parkinson’s disease given the role of this neurotransmitter in motivated decision-making. 39 Theoretical work predicts that dopamine-based improvements in apathy are paralleled by increased reward sensitivity during behavioral tasks and improved functional connectivity within fronto-striatal networks. 24
Behavioral approaches for treating apathy
Neuropsychological advice can be provided to patients with apathy in the context of more general rehabilitation procedures and can be delivered individually or in formal group settings. Patients can be engaged in goal setting with an emphasis on planning future goals and evaluating success to help re-establish GDB. 40 Complimentary approaches include problem-solving, wherein a patient selects an activity and makes a plan to achieve it while self-monitoring the process and outcome. 34 Behavioral activation can be combined with cognitive-behavioral therapy which explores psychological issues that may prevent GDB engagement. Approaches should foster a sense of the self, belonging, and respect and be tailored toward settings, where patients can derive enjoyment from exercises and activities such as planned outings consistent with personal backgrounds. 41
Few trials have evaluated behavioral approaches for treating apathy after stroke. One study on post-stroke patients without apathy found that 56 patients undergoing problem-solving therapy were 1.84 times less likely to developing apathy compared to 47 patients on placebo, though risk was even lower for patients receiving escitalopram. 34 A randomized trial examined strategy training, wherein patients are coached to focus on self-selected activity goals and encouraged to derive strategies to address performance in pursuit of those goals, to treat apathy in cognitively impaired stroke patients. 40 After three months, 15 patients undergoing strategy training had significantly lower apathy compared to 15 in a control condition. 40 Group differences were also found after six months, although these were not statistically significant. Another randomized trial examined the efficacy of a group-based approach to promote activity for treating post-stroke symptoms including apathy in 186 patients. 41 Groups met in a community-based setting twice a week for 3 h each, during which they engaged in exercise, project-based activities, and planned outings. Apathy decreased over the course of the 12-month intervention compared to baseline, although changes were not statistically significant. Apathy was significantly lower after a 15-month follow-up, however, suggesting that motivation continued to improve after the intervention.
Other approaches for treating post-stroke apathy
Repetitive transcranial magnetic stimulation (rTMS) has been used to treat post-stroke apathy. One randomized sample of chronic stroke patients showed that high-frequency rTMS over the ACC and medial prefrontal cortex improved apathy after five days in those receiving treatment (n = 7) compared to sham stimulation (n = 6). 42 A case study suggested that rTMS-based decreases in apathy are paralleled by increasing interhemispheric connectivity. 43 Both studies utilized small samples, however, necessitating replications in larger trials.
Discussion
Apathy is a quantitative reduction in GDB and is a common but under-studied syndrome following stroke. We provided an overview on apathy in stroke, highlighting contemporary issues on definitions, diagnosis, neurobiology, consequences, and treatments. A diagnosis of apathy may be complicated by symptoms of fatigue and especially depression, which may appear behaviorally similar to apathy. Apathy may be the result of damage to neural networks underlying cognitive functions that support motivated GDB. Post-stroke neurophysiological processes may lead to structural and functional network changes, leading to longitudinal changes in apathy. Post-stroke changes to these networks may explain why some patients show improving or worsening apathy over time, although further testing is required.
Finally, apathy is associated with numerous behavioral, cognitive, and emotional concomitants, impacting quality of life and functional outcomes. These emphasize the importance of treating apathy, but unfortunately, pharmacological and behavioral interventions have yielded inconclusive results. Although some treatments, such as dopamine pharmacotherapy and rTMS, show promise for treating apathy, no treatment can be fully recommended.
Clinical recognition of post-stroke apathy is important, as it informs outcomes and treatment approaches. Patients with apathy may recover functional abilities more slowly and could be at-risk for future vascular events, dementia, and mortality, stressing the importance of early detection and continued monitoring. Little evidence suggests that SSRIs effectively treat apathy and should only be used in patients with concomitant depression. It should be noted, however, that our non-systematic review may have led to bias, such as in article inclusion, and further systematic analyses may be required to assess treatment efficacy.
Research on neurobiological mechanisms of apathy should consider adopting the network-based framework for investigating the presentation and development of apathy. Focal lesions or peripheral degeneration in brain networks should be examined in conjunction with different behavioral manifestations of apathy. Epidemiological and outcome research on apathy could also benefit from better stroke and apathy subtyping. Finally, validating apathy measures in stroke patients would improve study reliability and help characterize specific motivational deficits in post-stroke patients. These could then inform approaches for treating apathy, such as pharmacological interventions to target neurotransmitter systems, behavioral interventions to target cognitive-behavioral symptoms, and brain stimulation approaches that target distinct brain networks.
Search strategy and selection criteria
PubMed was searched for articles published in English between 1 January 1970 and 31 December 31 using the following terms: (apath* OR indifferen* OR abuli* OR motivat*) AND (stroke OR infarct OR cerebrovascular* OR lacun* OR infarct* OR small vessel disease* OR white matter hyperintens* OR white matter lesion* OR white matter disease* OR microbleed* OR ischemia OR ischaemia OR haemorrhag* OR hemorrhag* OR perivascular space* OR leukoaraiosis OR leukoencephalopath* OR age-related white matter damage OR vascular dementia). This returned 3210 results. Titles and abstracts were screened by one of the authors (JT) for relevance to the topics covered in this review. Full-text articles of relevant articles were retrieved and reviewed. Reference lists from these articles were screened for other eligible studies. Further relevant studies were also taken from the authors’ own published works.
Supplemental Material
sj-pdf-1-wso-10.1177_1747493021990906 - Supplemental material for Apathy after stroke: Diagnosis, mechanisms, consequences, and treatment
Supplemental material, sj-pdf-1-wso-10.1177_1747493021990906 for Apathy after stroke: Diagnosis, mechanisms, consequences, and treatment by Jonathan Tay, Robin G Morris and Hugh S Markus in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This work is funded by a Priority Programme Grant from the Stroke Association (2015-02) and National Institute of Health Research (NIHR) Biomedical Research Centre Dementia and Neurodegeneration Theme (146281). JT is supported by a Cambridge International Scholarship from the Cambridge Trust. HSM is supported by the NIHR Cambridge Biomedical Research Centre and an NIHR Senior Investigator Award.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
