Abstract
Background
Variants of the Circle of Willis (vCoW) may impede correct identification of ischemic lesion patterns and stroke etiology. We assessed reclassifications of ischemic lesion patterns due to vCoW.
Methods
We analyzed vCoW in patients with acute ischemic stroke from the 1000+ study using time-of-flight magnetic resonance angiography (TOF MRA) of intracranial arteries. We assessed A1 segment agenesis or hypoplasia in the anterior circulation and fetal posterior cerebral artery in the posterior circulation. Stroke patterns were classified as one or more-than-one territory stroke pattern. We examined associations between vCoW and stroke patterns and the frequency of reclassifications of stroke patterns due to vCoW.
Results
Of 1000 patients, 991 had evaluable magnetic resonance angiography. At least one vCoW was present in 37.1%. VCoW were more common in the posterior than in the anterior circulation (33.3% vs. 6.7%). Of 238 patients initially thought to have a more-than-one territory stroke pattern, 20 (8.4%) had to be reclassified to a one territory stroke pattern after considering vCoW. All these patients had fetal posterior cerebral artery and six (30%) additionally had carotid artery disease. Of 753 patients initially presumed to have a one-territory stroke pattern, four (0.5%) were reclassified as having more-than-one territory pattern.
Conclusions
VCoW are present in about one in three stroke patients and more common in the posterior circulation. Reclassifications of stroke lesion patterns due to vCoW occurred predominantly in the posterior circulation with fetal posterior cerebral artery mimicking multiple territory stroke pattern. Considering vCoW in these cases may uncover symptomatic carotid disease.
Introduction
Identifying stroke lesion patterns and affected territories supplied by neck arteries is an essential component in determining ischemic stroke etiology. More-than-one territory stroke patterns suggest a proximal embolic source, e.g. of cardiac or aortal origin, whereas single territory stroke patterns point to large or small vessel disease. 1 Assigning stroke lesions to the corresponding arterial territories is usually carried out using pre-set brain maps due to practicality. 2 However, since the Circle of Willis (CoW) connects all three major cerebral territories (left anterior, right anterior, and posterior), classifying stroke lesion patterns based solely on brain maps without considering variants of the Circle of Willis (vCoW) may lead to false assignment of affected arterial territories. 3 Taking vCoW into account may lead to clinically relevant reclassifications of stroke patterns.
Aims
Establishing the frequency of different vCoW could help to distinguish stroke locations that are more prone to misclassifications. Here, we analyzed the frequency of two vCoW in patients with ischemic stroke using time-of-flight magnetic resonance angiography (TOF MRA): (a) agenesis or hypoplastic A1 segment of the ACA in the anterior circulation (anterior vCoW) and (b) fetal posterior cerebral artery (fPCA) in the posterior circulation. Furthermore, we examined stroke lesion patterns (classified as one and more-than-one territory stroke pattern) in patients with complete Circle of Willis (cCoW) and vCoW. Finally, we examined the rate of reclassifications from more-than-one territory to one territory and vice versa, following reattributions of stroke lesions to the corresponding cerebral artery after identifying vCoW.
Methods
Study design and study population
MRI data were obtained from the prospective observational cohort study 1000+ (Clinical trials.gov identifier: NCT00715533), which was approved by the ethics committee of the Charité – Universitätsmedizin Berlin (EA4/026/08). The study enrolled consecutively admitted patients with transient ischemic attack or ischemic stroke to our stroke unit (Department of Neurology, Charité – Universitätsmedizin Berlin, Campus Benjamin Franklin, Berlin, Germany) from February 2011 to April 2013. 4 The inclusion criteria were onset of acute ischemic stroke symptoms within the last 24 h of admission to the emergency department, meeting general MRI eligibility criteria and age ≥18 years. 4 Additionally, only patients with a complete MRA were included into the current subanalysis.
Stroke severity was assessed using the National Institutes of Health Stroke Scale (NIHSS) with higher scores indicating greater severity (range: 0–42). Functional outcome at discharge was evaluated by modified Rankin Scale (mRS), a seven-point scale ranging from “0” (no neurological deficit) to “6” (death). We also recorded history of arterial hypertension, diabetes mellitus, hypercholesterinemia, coronary heart disease, atrial fibrillation, and smoking. Only patients with a diffusion weighted imaging (DWI) lesion that were previously included in the substudy by Erdur et al. (n = 1000) were analyzed in this substudy. 3
Neuroimaging and assessment of the variants of the Circle of Willis
All exams were acquired using 3-Tesla magnetic resonance imaging (MRI); Tim Trio 3T whole-body system (Siemens Healthcare, Erlangen, Germany) using a 12-channel receive radiofrequency (RF) coil (Siemens Healthcare). Extracranial neck arteries were either assessed by MRA, computed tomography angiography (CTA), or duplex ultrasound. The CoW was assessed using TOF MRA; voxel size 0.52 × 0.52 × 0.65 mm, matrix size 312 × 384 × 127 voxels, TR/TE = 22/3.86 ms, respectively, acquisition time 3:50 and flip angle 18°.
LSM mapped and characterized all lesions on DWI, FLAIR, and anatomical variations of cerebral vessels while blind to all clinical data except name and sex under continuous supervision by HE. All inconclusive imaging findings were discussed by HE, LSM, JBF and CHN for definitive allocation.
Stroke lesion patterns and vCoW were previously evaluated in a substudy by Erdur et al. using the data from the 1000+ study. 3 The current work represents a subanalysis of this data set. We examined the frequency of two vCoW that occur most commonly.5,6 In the anterior circulation, we assessed the agenesis or hypoplasia of the A1 segment of the anterior cerebral artery (ACA) – anterior vCoW. This variant was identified when one A1 segment was significantly narrower than the contralateral A1 segment or absent. Consecutively, blood supply to the A2 segment ipsilateral to the underdeveloped A1 segment was manly provided from the contralateral A1 segment via anterior communicating artery (AComA). In the posterior circulation, we defined fPCA where posterior communicating artery (PComA) was significantly larger than the P1-segment of the ipsilateral posterior cerebral artery (PCA). Vice versa, the adult PCA was defined as P1-segment of the PCA being larger than the ipsilateral PComA. In cases with nearly the same diameters of the PComA und P1-segment, the posterior circulation was labeled as transitional, as previously described in the literature. 7
We conducted separate comparisons of patients with vCoW and cCoW in the posterior and anterior circulation in terms of demographics, medical history, stroke severity (NIHSS on admission), and early functional outcome (mRS at discharge). Patients with only transitional posterior circulation were excluded from the comparison of vCoW due to the small sample size and because it was not possible to assign transitional PCA to either adult PCA or fetal PCA variant, owing to distinct hemodynamics of each variant. Patients with fPCA on one side and either adult or transitional PCA on the other side were considered as fPCA patients for this particular analysis.
Hyperintense lesions in the DWI and low apparent diffusion coefficient (ADC) were considered as acute ischemic lesions. Chronic ischemic lesions were assessed using fluid attenuated inversion recovery (FLAIR) sequence. Only acute ischemic lesions were considered for determining stroke patterns and affected arterial territories. Arterial territories were divided into left and right anterior (middle cerebral artery (MCA) + ACA territory of each side, originating from the internal carotid artery (ICA)) and posterior territory (PCA territories and vertebrobasilar system). Allocating stroke lesions to the corresponding arterial territories was initially performed using brain maps according to Tatu et al. with no regard to possible vCoW.2,3
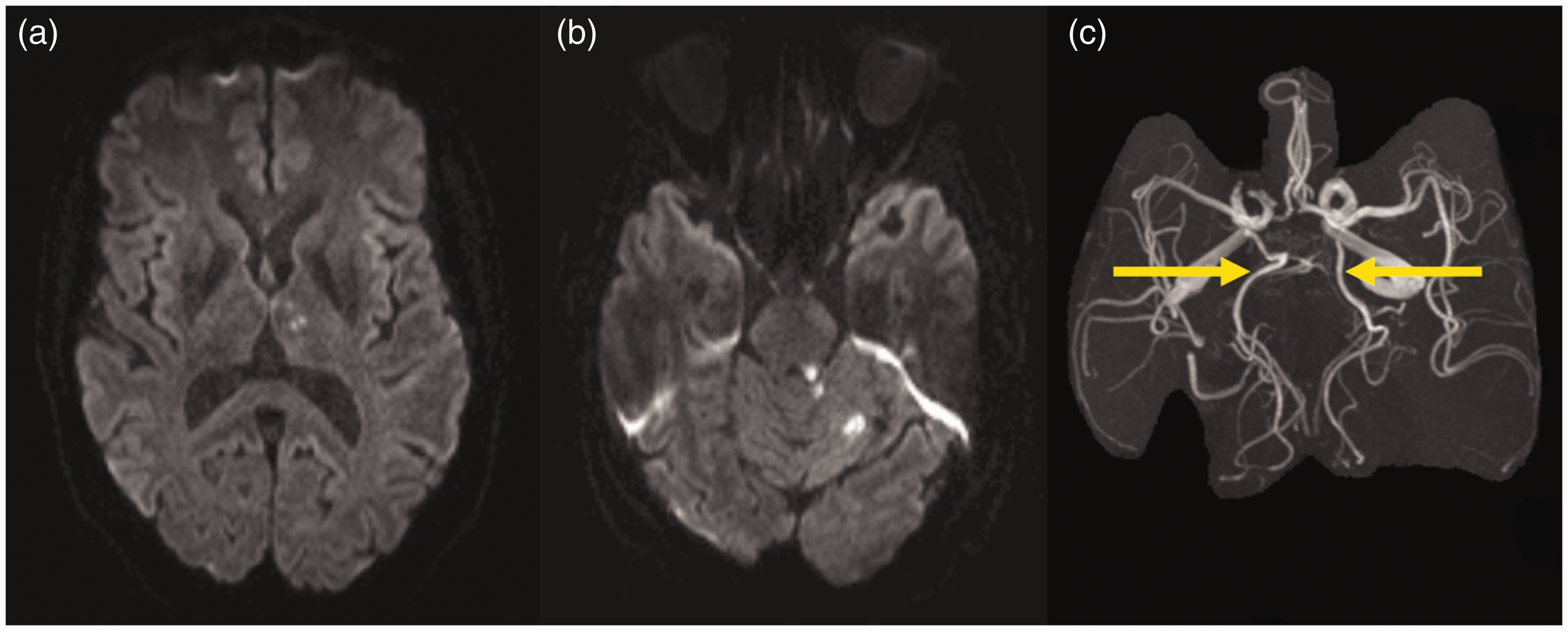
Stroke lesion patterns were classified as one and more-than-one territory stroke pattern. We examined the frequency of different stroke patterns with no regard to vCoW. After identifying vCoW, we examined the rate of reclassifications from more-than-one territory to one territory stroke pattern. For example, in a patient with ipsilateral temporal and occipital lesions, the lesions were labeled as more-than-one territory pattern. This had to be corrected to a one territory pattern after ipsilateral fPCA was identified (Figure 1). Reversely, reclassification from one to more-than-one territory pattern was conducted in cases where simultaneous PCA strokes and cerebellar strokes were seen and had been considered to belong to the same (posterior) territory. However, in patients with fPCA, the PCA stroke belonged to the anterior circulation, and consequently, a reclassification into more-than-one territory group was conducted (Figure 2). In addition, we examined how often ipsilateral fPCA is present in patients with isolated lesions in the PCA territory. In the presence of fPCA, isolated lesions in the PCA territory were re-allocated from the posterior to the (ipsilateral) anterior vascular territory.
Reclassification from more-than-one to one territory stroke pattern. Reclassification from one territory to more-than-one territory stroke pattern.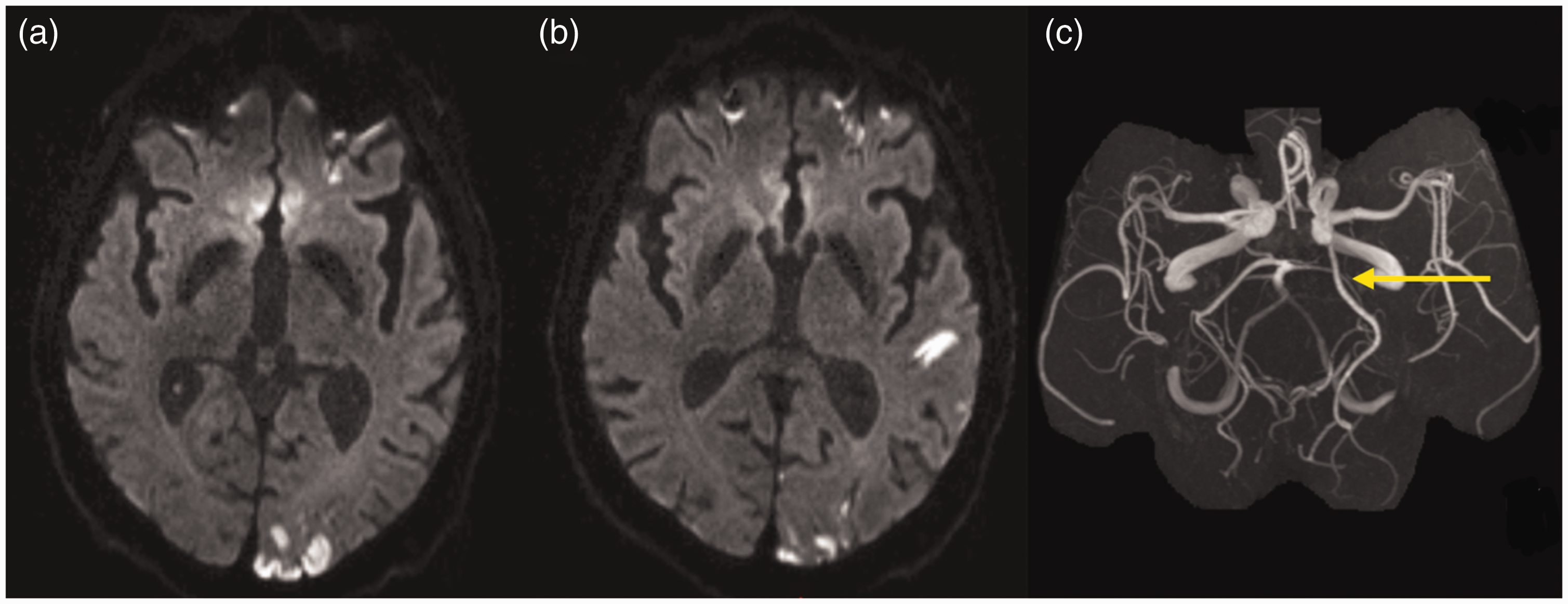
Statistics
SPSS Version 25.0. (IBM, Armonk, NY, USA) was used for statistical analyses. Metric data are presented as medians with interquartile range (IQR) due to non-normal distribution and were compared using Mann-Whitney U-Test. Categorical data were compared using chi-squared test or Fisher’s exact test as appropriate. All tests were performed at α = 0.05 level of confidence and were two sided.
Results
Characteristics of the study population
Of 1000 included patients, 991 had complete data sets of MRAs fully eligible for further analyses. MRA was not available in nine patients, either due to motion artifacts or because examination had to be stopped prematurely. Cases without MRA were excluded from further analyses. Of these 991 patients, 448 (45.2%) were female and median age was 74 years (IQR: 66–81). History of arterial hypertension was present in 86.2%, 29.7% had atrial fibrillation, 26.3% had diabetes mellitus type 2, and 18.8% had a history of coronary heart disease. Furthermore, 20.1% of patients reported being a smoker. Median NIHSS on admission was 4 (IQR: 2–8).
Frequency of vCoW
Comparison of the demographics and cardiovascular risk factors in patients with posterior vCoW (fPCA) and posterior cCoW (adult PCA). Patients with transitional posterior circulation (n=21) were excluded from this analysis.
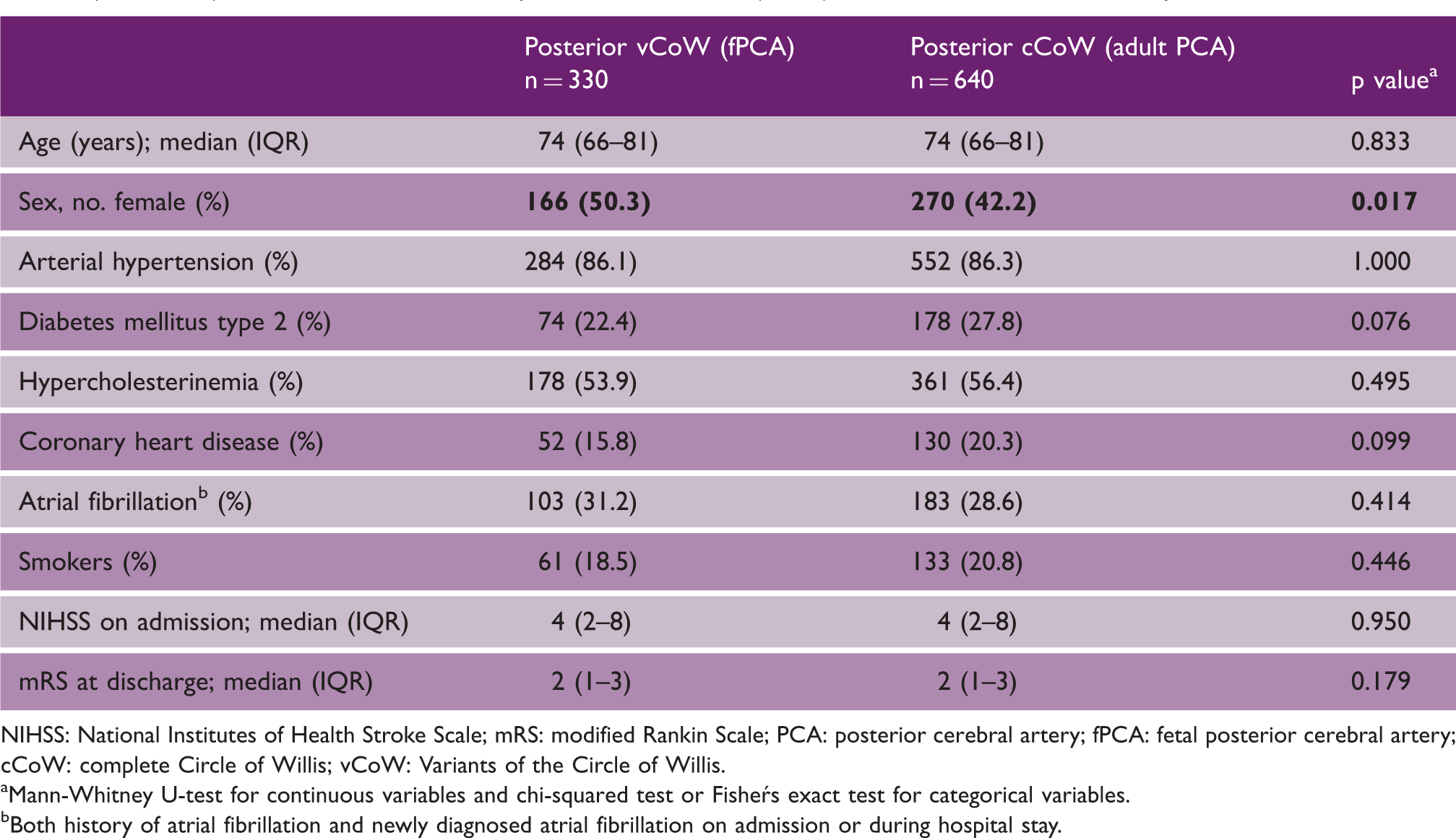
NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin Scale; PCA: posterior cerebral artery; fPCA: fetal posterior cerebral artery; cCoW: complete Circle of Willis; vCoW: Variants of the Circle of Willis.
Mann-Whitney U-test for continuous variables and chi-squared test or Fishers exact test for categorical variables.
Both history of atrial fibrillation and newly diagnosed atrial fibrillation on admission or during hospital stay.
Comparison of the demographics and cardiovascular risk factors in patients with anterior vCoW and anterior cCoW.
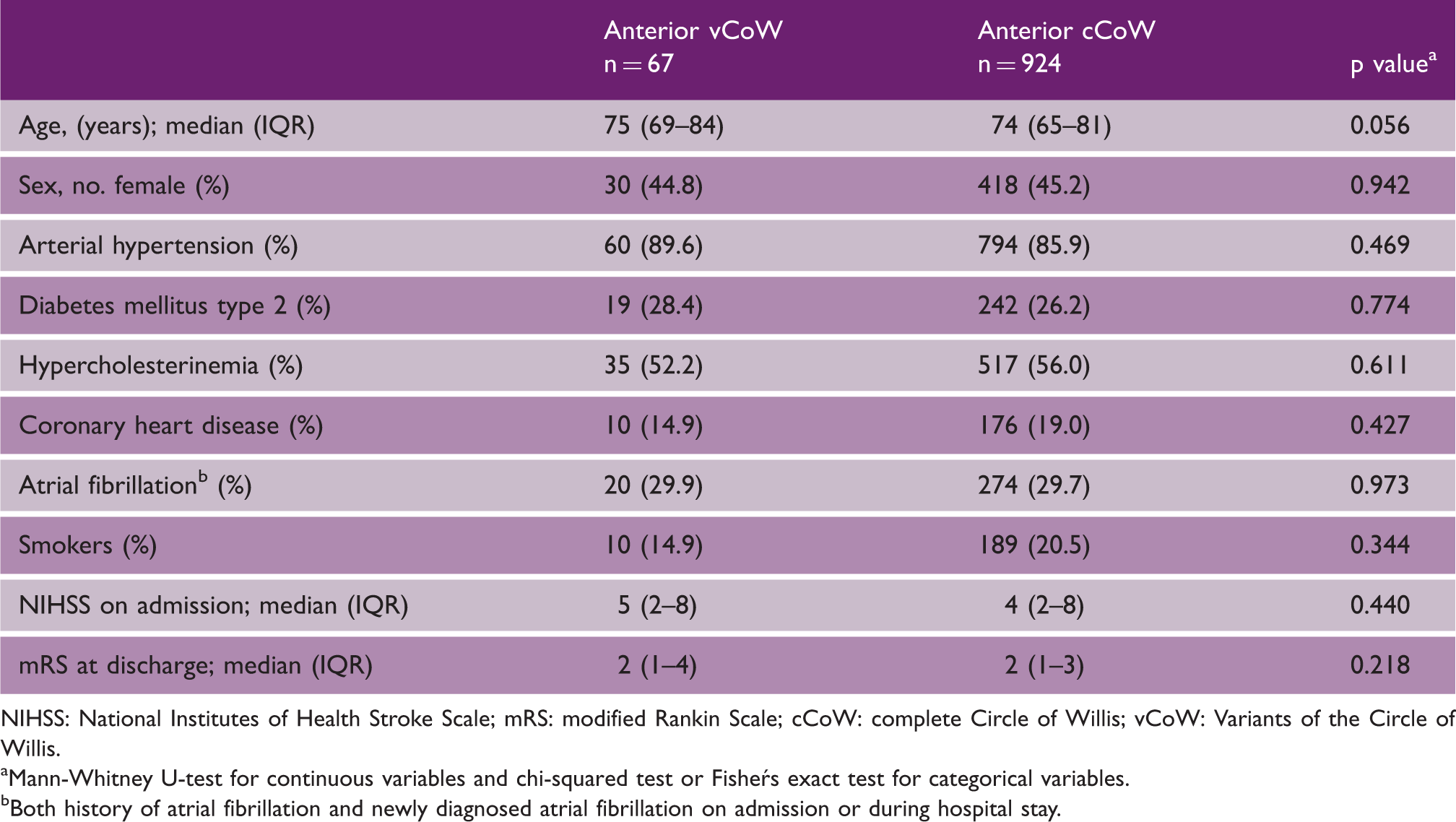
NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin Scale; cCoW: complete Circle of Willis; vCoW: Variants of the Circle of Willis.
Mann-Whitney U-test for continuous variables and chi-squared test or Fishers exact test for categorical variables.
Both history of atrial fibrillation and newly diagnosed atrial fibrillation on admission or during hospital stay.
Affected arterial territories
Altogether, 386 patients had lesions in the right anterior territory, 424 had lesions in the left anterior territory, and 382 patients had lesions in the posterior territory. Of patients with lesions in the posterior territory, 84 had isolated lesions in the PCA territory.
Stroke lesion patterns
More-than-one territory stroke pattern was seen in 238 (24.0%) patients. In patients with fPCA, more-than-one territory stroke patterns according to Tatu maps were less common than in patients with adult PCA (61/330 (18.5%) vs. 172/640 (26.9%), p = 0.014). In patients with anterior variants, frequency of more-than-one territory stroke pattern was similar to patients without anterior cCoW (19/67 (28.4%) vs. 219/924 (23.7%), p = 0.459).
Reclassifying stroke patterns
Of 238 patients initially categorized as having more-than-one territory stroke pattern, 20 (8.4%) were reclassified into having one territory lesion pattern after correcting for vCoW. All of these patients had fPCA. Stroke lesions originally presumed to belong to the vertebrobasilar territory had to be re-assigned to the ICA territory. More-than-one arterial territory stroke lesion had only been mimicked since all lesions were distal to a common (ICA) artery (Figure 1). Of the 20 patients affected by the reclassification from more-than-one to one territory pattern, six (30%) had a severe symptomatic carotid stenosis. Out of 84 patients with isolated lesions in the PCA territory, 11 had fPCA (13.1%). These cases were reclassified from posterior to anterior arterial territory. No clinically relevant ipsilateral ICA stenosis was found in any of these patients.
Reclassification from originally presumed one territory to more-than-one territory stroke pattern was indicated in 4/753 (0.5%) patients. All these patients showed vCoW in the posterior circulation (Figure 2).
Discussion
In this cohort of patients with acute ischemic stroke, MRA revealed at least one vCoW in 37.1% patients with most variants observed in the posterior circulation (33.3% compared to 6.7% in the anterior circulation). The adjustment of large cerebral vessel territories after considering vCoW led to several reclassifications of lesions patterns with relevance for the assumed ischemic stroke etiology.
Reports on the frequency of vCoW are available from autoptic examinations and neuroimaging studies using CTA or MRA.5,8–11 Depending on the method, the criteria used for defining variants and the examined population previous studies reported frequencies of any vCoW ranging from 45% to 88%.8–15 Our findings on the frequency of fPCA are in accordance with most previous studies, which found fPCA in around 20–30% of cases.9,15–19 The higher frequency of fPCA on the right side found in our study was also reported by two earlier studies in populations with cerebrovascular diseases.9,16 In line with earlier studies, we found a lower frequency of vCoW in the anterior circulation compared to the posterior circulation.9,13,15 Similar to the prevalence of 6.7% of agenesis or hypoplasia of the A1 segment of the ACA in our study, other imaging and autopsy studies reported the prevalence of this variant ranging from 9.5 to 11.7%.6,9,15,20
To our knowledge, this is the first study that examined rates of reclassification of stroke patterns in patients with vCoW. The detection of vCoW led to stroke pattern reclassification in 8.9% of patients. The majority were reclassified from more-than-one territory pattern to one territory stroke pattern. A minority of patients was reclassified from one to more-than-one-territory lesion pattern after taking vCoW into consideration. We showed that more-than-one territory stroke pattern is overdiagnosed in a relevant number of patients when vCoW is not taken into account. Consequently, proximal embolic sources may be preferably searched for as an underlying cause of the ischemic stroke. Although the combination of having stroke in the anterior (MCA + ACA) and PCA territories in the setting of a fPCA and symptomatic carotid stenosis was rare in our study (six out of nine hundred ninety-one cases), it is important to identify such patients, as they may benefit from carotid endarterectomy in cases of carotid artery disease. Current guidelines on the early management of patients with acute stroke recommend performing CTA and MRA in selected patients with suspected large vessel occlusion (i.e. patients with severe neurological symptoms, expressed by a higher NIHSS), who might benefit from mechanical thrombectomy. 21 Hence, intracranial vessel imaging may not be performed in the acute phase in all patients, especially those with minor stroke symptoms. Without intracranial CTA or MRA in the acute phase, vCoW may be missed and stroke patterns may not be correctly classified. Etiological assessment of stroke lesions requires a comprehensive approach and should not be based solely on stroke patterns. We consider CTA or MRA of intra- and extracranial arteries as an indispensable part of diagnostic workup in all patients presenting with acute ischemic stroke. Failure to accurately determine the underlying cause of stroke may not only affect acute care but could also compromise choosing the optimal secondary prevention.
Some limitations must be taken into account, when interpreting our findings. Firstly, our study was retrospective, and the data were collected at a single site, potentially limiting generalizability of our findings. Secondly, only patients who were eligible for MRI were included in our study, possibly introducing a selection bias. Thirdly, the by and large single-person assessment may have introduced bias. Finally, our study did not control for intracranial vessel occlusions and stenoses, which could potentially influence the diameter of intracranial vessels and consequently change the anatomy of the CoW.
In conclusion, vCoW occur in more than a third of patients with ischemic stroke and are more common in the posterior than in the anterior circulation. Patients with vCoW in the posterior circulation are particularly prone to misclassifications of stroke patterns. We emphasize the importance of conducting intracranial vessel imaging in all stroke patients, independent of stroke severity, to facilitate correct classification of stroke etiology, and therefore provide optimal treatment.
Footnotes
Data availability statement
Anonymized data from the present study will be shared on reasonable request from any qualified researcher for well-defined research questions. Please contact the corresponding author.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Audebert reports receiving personal fees from Bayer Vital, Boehringer Ingelheim, Bristol Myers Squibb, Novo Nordisk, Pfizer, and Sanofi, outside the submitted work. Dr. Fiebach reports receiving personal fees from Abbvie, AC Immune, Artemida, Bioclinica, Biogen, BMS, Brainomix, Cerevast, Daiichi-Sankyo, Eisai, F.Hoffmann-La Roche AG, Eli Lilly, Guerbet, Ionis Pharmaceuticals, IQVIA, Janssen, Julius clinical, jung diagnostics, Lysogene, Merck, Nicolab, Premier Research, and Tau Rx, outside the submitted work. Nolte does not report conflicts of interest with respect to the submitted work. Dr. Nolte received research grants from German Ministry of Research and Education, German Center for Neurodegenerative Diseases, German Center for cardiovascular Research, and speaker and/or consultation fees from Boehringer Ingelheim, Bristol-Myers Squibb, Bayer, Daiichi Sankyo, Pfizer Pharma, Abbott and Alexion. Dr. Erdur, Dr. Galinovic, Dr. Milles, Dr. Rangus and Dr. Villringer declare that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
