Abstract
Background
Case-fatality rates after aneurysmal subarachnoid hemorrhage have decreased over the past decades. However, many patients who survive an aneurysmal subarachnoid hemorrhage have long-term functional and cognitive impairments.
Aims
We sought to review all data on conventional brain MRI obtained in the chronic phase after aneurysmal subarachnoid hemorrhage to (1) analyze the proportion of patients with cerebral infarction or brain volume changes; (2) investigate baseline determinants predictive of MRI-detected damage; and (3) assess if brain damage is predictive of patient outcome.
Summary of review
All original data published between 1 January 2000 and 4 October 2017 was searched using the PUBMED, EMBASE, and Web of Science databases. Based on preset inclusion criteria, 15 from 5200 articles were included with a total of 996 aneurysmal subarachnoid hemorrhage patients. Quality assessment, risk of bias assessment, and level of evidence assessment were performed. The results according to aim, with levels of evidence, were: (1) 25 to 81% of aneurysmal subarachnoid hemorrhage patients show infarcts (strong); there is a higher ratio of cerebrospinal fluid-to-intracranial volume in patients compared to controls (strong); (2) there is a negative relation between age (moderate), DCI (low) and brain volume measurement outcomes; (3) lower brain parenchymal volume (strong) and the presence of infarcts or infarct volumes (moderate) are associated with a worse outcome.
Conclusion
Patients after aneurysmal subarachnoid hemorrhage may demonstrate brain infarcts and decreased brain parenchyma, which is related to worse outcome. Thereby, both brain infarcts and brain volume measurements could be used as outcome markers in pharmaceutical trials.
Systematic Review Registration
PROSPERO CRD42016040095
Introduction
Case-fatality rates after aneurysmal subarachnoid hemorrhage (aSAH) have decreased over the past decades. Today, approximately one-third of patients die within three months after the event. 1 Of the patients who survive, many have long-term functional and cognitive impairments.2,3
Common determinants of poor functional outcome are the impact of the acute hemorrhage, also known as early brain injury (EBI),4,5 bleeding related acute ischemia,6,7 and complications such as rebleeding of the aneurysm, procedure-related complications,8,9 delayed cerebral ischemia (DCI),10–13 and hydrocephalus. 14 It remains unclear, however, how these events lead to MRI changes in the chronic phase after aSAH, and how the MRI changes relate to adverse functional outcome.
Previous MRI research has focused on the assessment of focal brain injury or brain volume changes occurring after aSAH. As well, the relation between brain injury and clinical- or neuropsychological outcome has been investigated. In the present review, we grouped the results of studies performed in this area with the purpose to provide insight for future research. The following aims were formulated: (1) to analyze the proportion of aSAH patients with cerebral infarction or brain volume changes in the chronic phase; (2) to investigate baseline determinants predictive of MRI-detected damage; and (3) to assess if MRI-detected damage is predictive of patient outcome.
Methods
Search strategy
A literature search was conducted using the PUBMED, EMBASE, and Web of Science databases and included articles published between 1 January 2000 and 4 October 2017. The search variables were: (magnetic resonance OR MRI) AND (subarachnoid hemorrhage OR SAH) OR (DCI OR DCI OR DIND). Eligible articles were identified according to PICO criteria. Patient population (P): (1) ≥10 patients included with aSAH, defined as the presence of subarachnoid blood shown by CT or lumbar puncture and the presence of an aneurysm on CT angiography, magnetic resonance angiography, or digital subtraction angiography; (2) MRI scans performed >21 days after aSAH. Intervention (I): imaging sequences were either T1-weighted imaging, T2-weighted imaging, proton density-weighted imaging (PD), and/or fluid-attenuated inversion recovery (FLAIR). Comparator (C): N/A. Outcomes (O): MRI brain damage, either infarcts or brain volume change, >21 days after aSAH. Exclusion criteria were (1) article not reporting original data; (2) article written in another language than English, French, German, or Dutch; (3) conference abstract; (4) cause of the hemorrhage unknown or other than aneurysmal in >5% of patients; (5) animal study. Two authors (J.D.V. and L.v.d.K) screened on title and abstract. In case of disagreement, a consensus reading was performed. Subsequent full text screening was conducted by B.S., and exclusion was performed in consensus with J.D.V. If more than one article used the same or overlapping patient population and the same outcome measures, only the report with the largest patient population was included. If an article reported on MRI findings but the timing of imaging was unclear, the corresponding author was contacted for additional information. In case of no response, the article was excluded. The reference lists of included articles were hand-searched for additional eligible articles. A flowchart of the screening process can be found in Figure 1. The review methodology was published in the PROSPERO database (CRD42016040095). The PRISMA 2009 checklist for transparent reporting of systematic reviews and meta-analyses was applied.
Flowchart.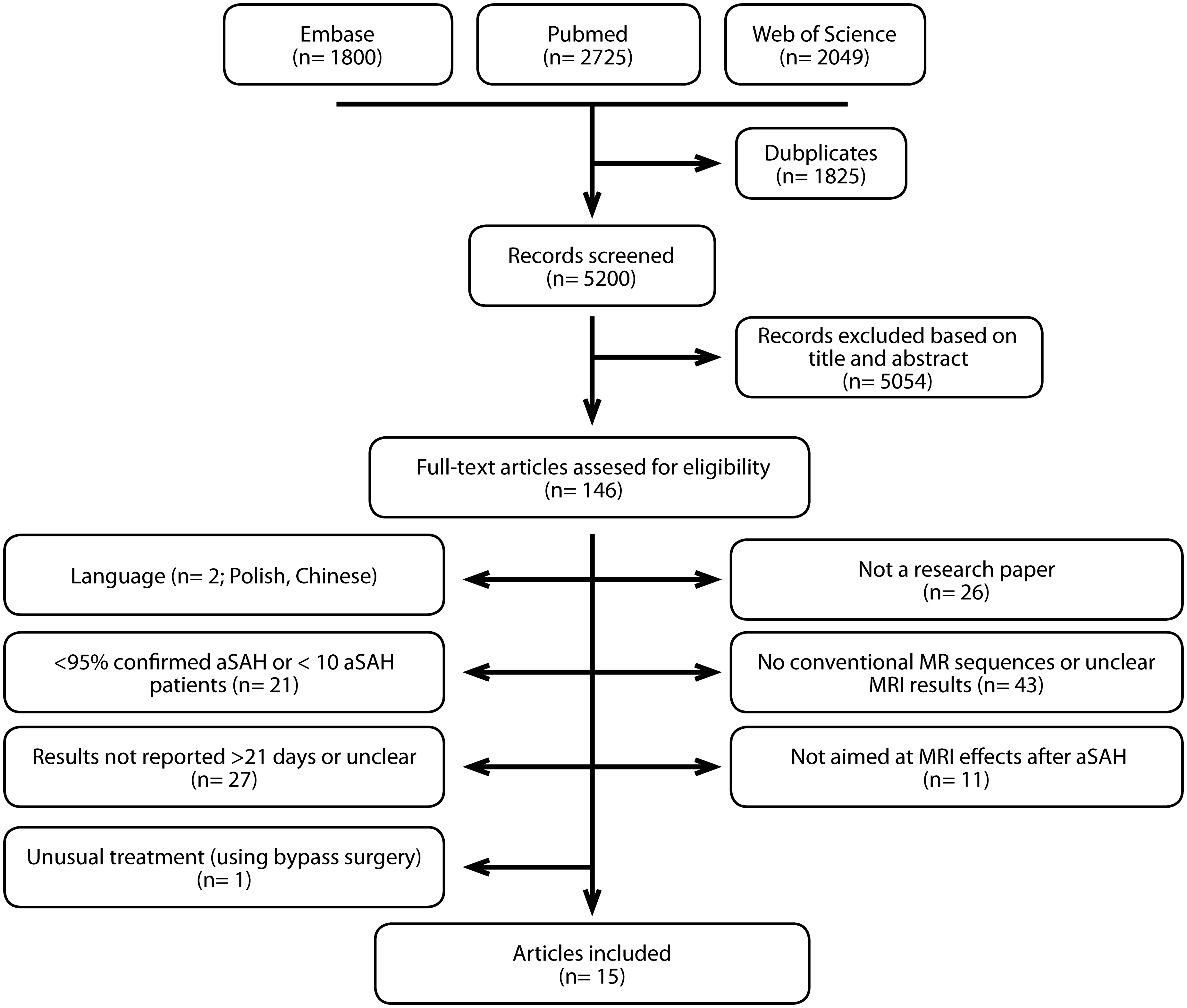
Quality assessment, risk of bias assessment, and level of evidence assessment
Quality assessment was conducted based on a previously published scoring system15,16 adapted to fit the studies within the scope of this review (Supplemental Table 1). Two authors (B.S. and J.D.V.) conducted the assessment independently, and disagreement was resolved by consensus. A high-quality article was predefined as a score between 12 and 18. A score below 12 was considered to reflect a low-quality article.
Risk of bias assessment was performed by two authors (B.S and J.D.V.) to evaluate external validity, internal validity, and overall risk of study bias (Supplemental Table 2). 17
The level of evidence (strong, moderate, weak, low, or inconsistent) for a certain finding was determined based on study quality and number of studies supporting the finding (Supplemental Figure 1).
Data extraction and analyses
The following baseline characteristics were extracted: number of patients who received MR imaging, age, sex, clinical condition at admission, i.e. Hunt and Hess scale (H&H) 18 or World Federation of Neurosurgical Societies SAH grading system (WFNS), 19 amount of blood on admission CT, i.e. Hijdra scale 20 or Fisher grade, 21 type of aneurysm treatment, the timing of the MRI, MRI sequences, and field strength.
To address the first aim, data on the number of patients with a cerebral infarct were extracted, and the proportion (with 95% CI) of patients with an infarct was calculated. Additionally, the number of infarcts per patient and infarct size was extracted. As well, brain volume measurement results were extracted. Due to the heterogeneity of the used measurements, it was chosen to focus on the cerebrospinal fluid/intracranial volume ratio (CSF/ICV) or measures related to it, and on the ventricular-to-intracranial-width ratio (VIWr). The incorporated measures related to the CSF/ICV were brain parenchymal volume (BPV), lateral ventricle volume, and peripheral CSF volume.
To address the second aim, data on determinants and their relation to cerebral infarct or brain volume changes were extracted. The parameters investigated were: age, the clinical condition on admission, and the amount of extravasated blood on admission CT. For brain volume changes, two additional determinants could be investigated: preoperative- or symptomatic hydrocephalus, and DCI.
To address the third aim, data describing the relation between brain damage and neuropsychological or clinical outcome were extracted.
Statistical analyses were performed using SPSS Statistics (SPSS, Inc., Chicago, IL, version 22).
Results
Level of evidence

Proportion of aSAH patients with cerebral infarction or brain volume changes
Cerebral infarction
Twelve studies, with a median number of 50 patients per study (range 20–138), reported on the presence of cerebral infarcts.24–28,30–36 Of 791 patients, 372 showed a cerebral infarct. The percentage of patients with an infarct ranged between 25 and 81 percent between studies (see Supplemental Table 3). The included studies varied in patient disease severity between WFNS I or II at admission or H&H grade 1 up to 4.31,36
Size of the deficits in patients with a cerebral infarct
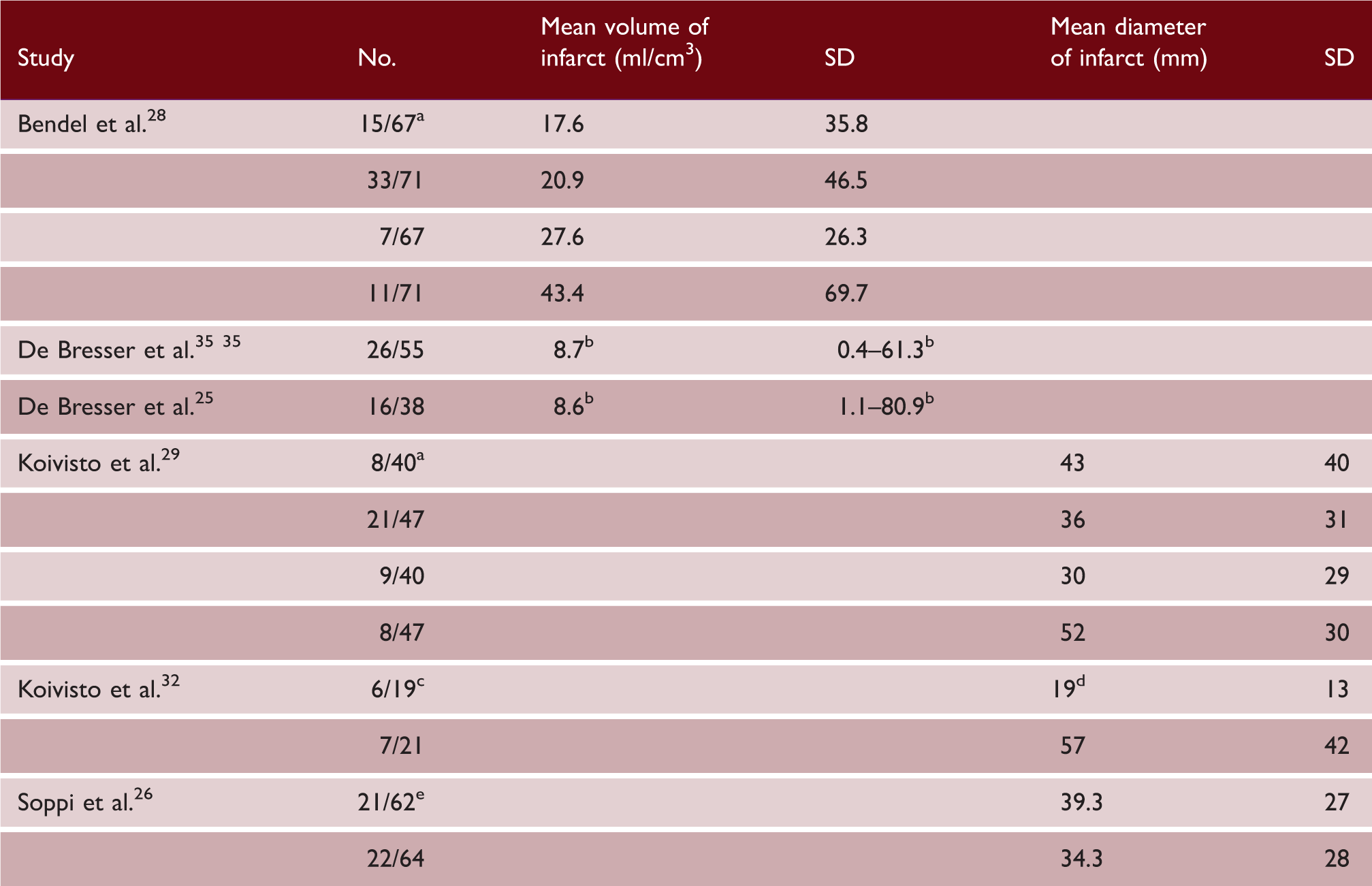
Note: In the articles it was not specified if, in case of multiple infarcts, a summation of the volumes or sizes was given or results of the largest infarct were given, unless stated otherwise.
No.: number of patients with an infarct / total MR population.
Results were presented based on vascular territory and treatment method (i.e. coiling or clipping), in descending order; infarcts in parental artery territory in coiled aSAH patients, infarcts in parental artery territory in clipped aSAH patients, infarct in vascular territories other than the parental territory in coiled aSAH patients, and infarcts in vascular territories other than the parental territory in clipped aSAH patients.
Median and 10th–90th percentile.
Patients presented by treatment method; surgical and endovascular.
Size of infarction is size of largest deficit in case of multiple deficits.
Patients presented by treatment strategy; enteral and intravenous administration of nimodipine.
Brain volume changes
Six articles, with a median number of 65.5 patients per study (range 38-88), reported on brain volume changes.22–25,29,35 In four of the six articles, patients were compared to age- and sex-matched controls.22–24,35
Brain volume measurement outcomes
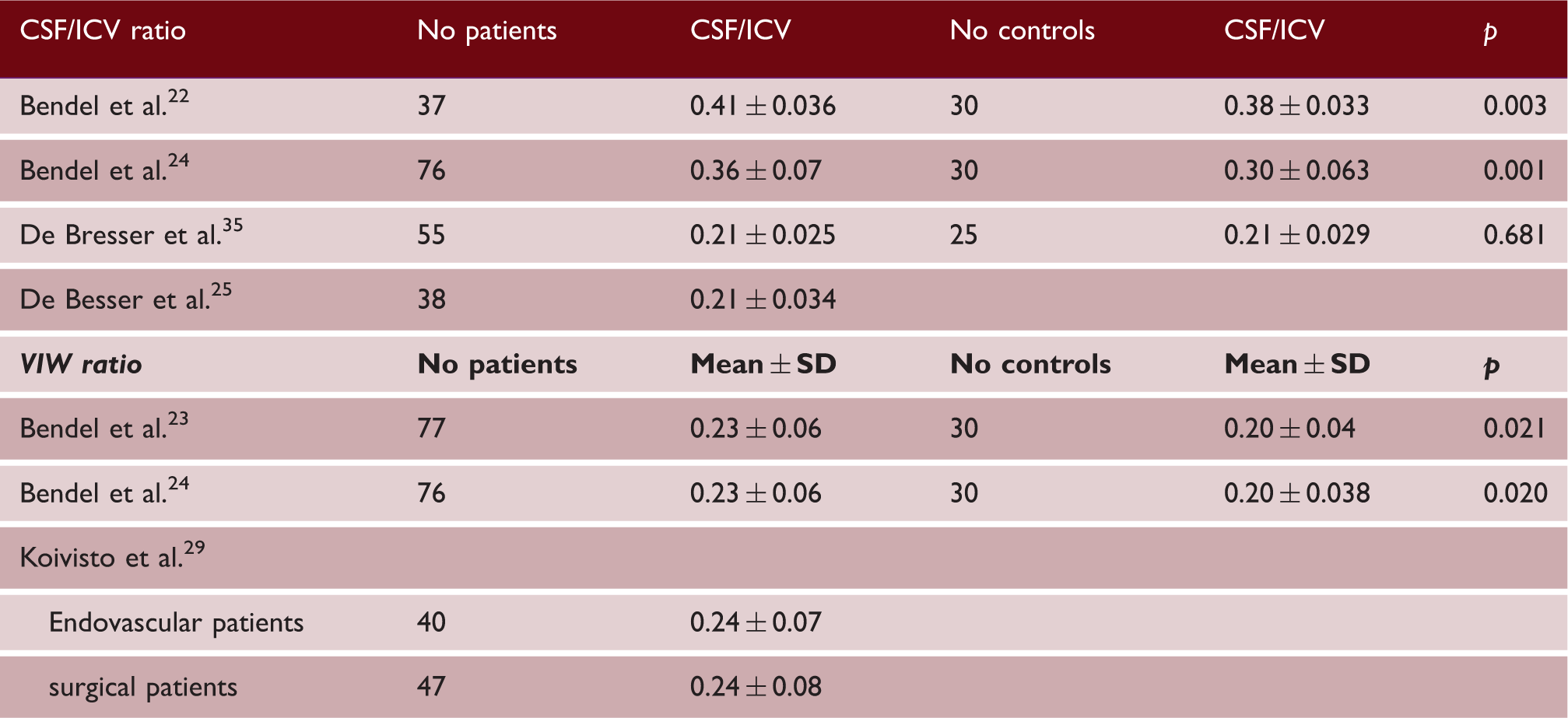
CSF/ICV; cerebrospinal fluid / intracranial volume; VIW: ventricular-to-intracranial width.
Three articles reported the VIWr (Table 3).23,24,29 Two studies found a significant higher mean ratio in aSAH patients compared to controls.23,24 The third study reported results found in endovascular- and surgical-treated patients. 29
Baseline determinants predictive of MRI-detected damage
Determinants of cerebral infarction
Determinants of cerebral infarcts and brain volume measurements
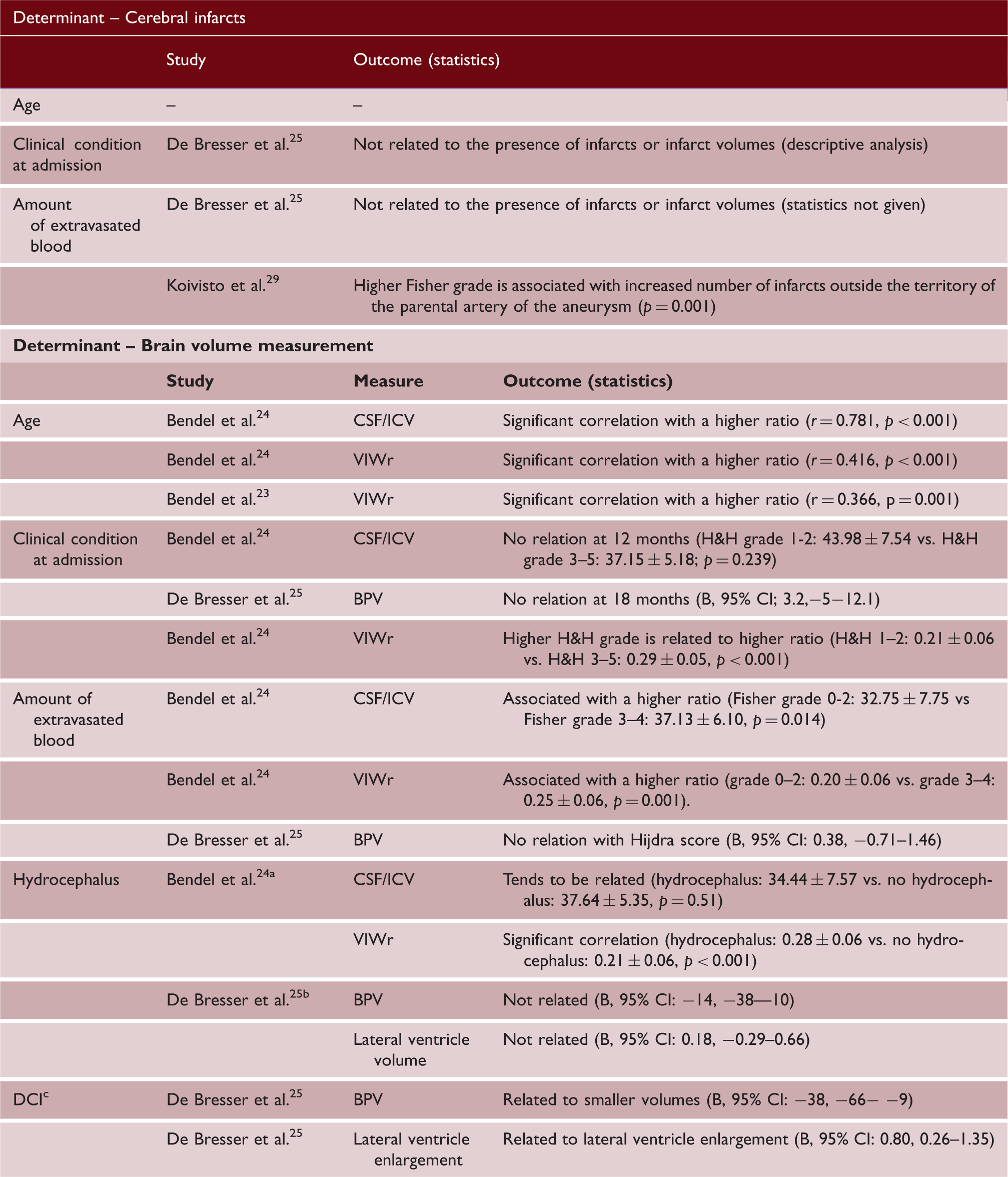
Preoperative hydrocephalus on admission CT.
Symptomatic hydrocephalus defined as a decreased level of consciousness with increased bicaudate index that was larger than the upper 95th percentile for age.
Defined as new focal deficits during admission or decreasing level of consciousness with new infarcts on CT.
CSF/ICV; cerebrospinal fluid/intracranial volume; VIWr: ventricular-to-intracranial width ratio; BPV: brain parenchymal volume; DCI: delayed cerebral ischemia.
One study found no relation between the amount of extravasated blood on admission CT and the presence of cerebral infarcts or infarct volumes (descriptive analysis), 25 while the second study did find a significant association with a higher Fisher grade. More specifically, a higher Fisher grade was associated with an increased number of infarcts outside the territory of the parental artery of the aneurysm (p = 0.001). 29
Determinants of brain volume changes
Table 4 summarizes the results found on this topic. In short, three studies, with a median of 76 patients (range 38–77), reported on this subject.23–25 A significant correlation of age with brain volume changes was found in two studies (CSF/ICV: r = 0.718, p < 0.001; CSF/ICV: r = 0.416, p < 0.001; VIWr: r = 0.366, p = 0.001).23,24
No relation between clinical grade at admission and brain volume changes was found in two studies (CSF/ICV: p = 0.239; BPV: B (95%CI); 3.2 (−5 – 12.1)).24,25 One study on the other hand, did find more brain atrophy in patients with higher H&H grade (VIWr: p < 0.001). 24
The amount of extravasated blood on admission CT was found to be associated with brain atrophy in one study (CSF/ICV: p = 0.014; VIWr: p = 0.001), 24 whereas another did not find a relation (BPV: B (95% CI); 0.38 (−0.71−1.46)). 25
One research group reported that patients with hydrocephalus on admission CT tended to have lower brain volumes (CSF/ICV: p = 0.51; VIWr: p < 0.001). 24 Another study reported that symptomatic hydrocephalus was not related to volume measurements (BPV: B (95% CI);−14 (−38 – 10); lateral ventricle volume: B (95% CI) 0.18 (−0.29 – 0.66)). 25
Lastly, it was reported that DCI is related to lower BPVs and lateral ventricle enlargement, which would result in a higher CSF/ICV (B (95% CI): −38 (−66− −9); B (95% CI): 0.80 (0.26–1.35)). 25
Damage detected on MRI predictive of patient outcome
Clinical or neuropsychological outcome – Cerebral infarct
MRI damage predictive of patient outcome
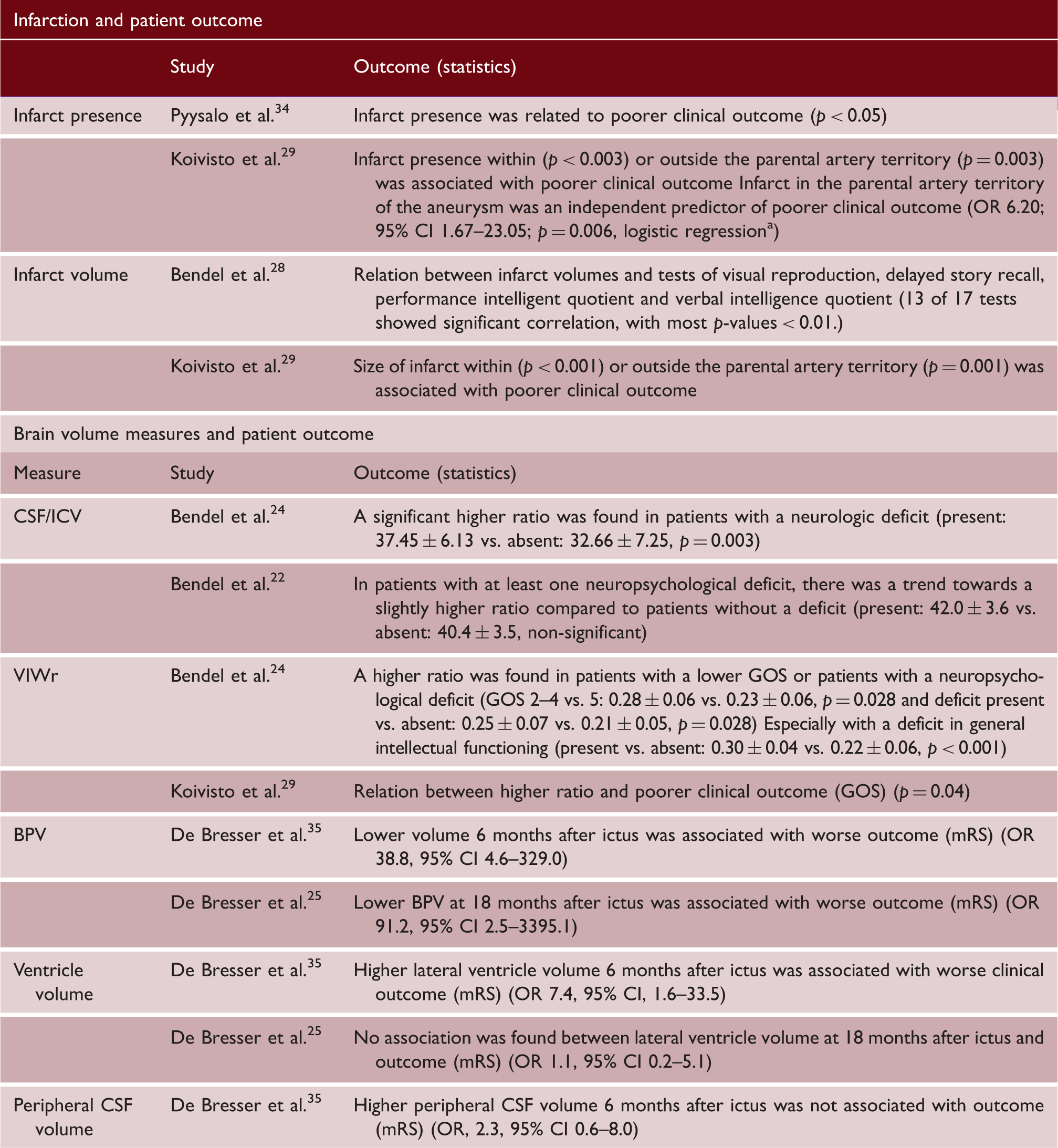
Variables used in the model: size of the ischemic lesion in the parental artery territory or in any other location; presence of an ischemic lesion in the parental artery territory or in any other location; deficit due to preoperative intracerebral hematoma and higher VIWr.
CSF/ICV; cerebrospinal fluid/intracranial volume; VIWr: ventricular-to-intracranial width ratio; mRS: modified Rankin Scale; GOS: Glasgow outcome scale.
Two studies, with 88 and 138 patients, reported on the relation of infarct size and outcome.28,29 Size of infarcts in the parental artery territory (p < 0.001) or in another vascular territory (p = 0.001), was significantly associated with poorer clinical outcome. 29 A significant relation between infarct volume and deficits in several tests of visual reproduction, delayed story recall, performance intelligent quotient and verbal intelligence quotient was reported (most p-values < 0.01). 28
Clinical or neuropsychological outcome – Brain volume measurements
Table 5 shows all results on this topic. In short, four studies with a median of 65.5 patients (range 38–88) reported on correlations between brain volume measurements and clinical outcome.24,25,29,35 The reported relations were as follows; a positive relation for BPV (at 6 ± 2 months OR 38.8, 95% CI 4.6–329.0; and at 18 months OR 91.2, 95% CI 2.5–3395.1),25,35 a positive relation for VIWr (GOS 2–4 vs. 5; 0.28 ± 0.06 vs. 0.23 ± 0.06, p = 0.028 and univariate analysis, p = 0.04),24,29 a positive relation for lateral ventricle volume (OR 7.4, 95% CI 1.6–33.5), 35 no relation for lateral ventricle volume (OR 1.1; 95% CI 0.2–5.1), 35 and no relation for peripheral CSF volume (OR 2.3, 95% CI, 0.6–8.0). 35 Sample size differences between the studies investigating the relation between lateral ventricle volume and outcome may have contributed to conflicting differences; the sample size was 55 in the study where a positive relation was found compared to 38 in the negative study.
Two studies, with 37 and 76 patients, reported on brain volume measures and neuropsychological outcome.22,24 In short, there was a positive relationship and a tendency towards a positive relationship between CSF/ICV and neurologic deficits (present vs. absent; 37.45 ± 6.13 vs. 32.66 ± 7.25, p = 0.003 and 42.0 ± 3.6 vs. 40.4 ± 3.5, non-significant).22,24 A higher VIWr was found in patients with any neuropsychological deficit (present vs absent; 0.25 ± 0.07 vs. 0.21 ± 0.05, p = 0.028), especially with a deficit in general intellectual functioning (present vs. absent; 0.30 ± 0.04 vs. 0.22 ± 0.06, p < 0.001). 24
Discussion
The following findings, summarized according to the aims of the review, were found: (1) 25–81% of aSAH patients show infarcts; there is a higher ratio of cerebrospinal fluid-to-intracranial volume in patients compared to controls; (2) there is a negative relation between age and DCI versus brain volume measurement outcomes; (3) lower BPV and the presence of infarcts or infarct volumes are associated with a worse outcome.
The wide range (25–81%) in aSAH patients demonstrating brain infarcts (strong level of evidence) underscores the heterogeneity of patient populations in the included articles. Regarding brain volume changes, it was found that aSAH patients have a lower brain volume compared to controls (strong level of evidence).22–24 Unfortunately, due to the continuous nature of a volume measurement and the effect of age on such a measurement, a cut-off value for presence or absence of brain volume loss could not be established. Therefore, the proportion of aSAH patients with brain volume loss could not be calculated. However, in general, the decreased brain volume in aSAH patients as compared to controls does bring support for the EBI theory. Further support for the EBI theory is found in two of the studies included in this review. One study found brain atrophy independent of infarct presence, 22 and a second study quantified global ventricular and sulcal enlargement. 24
Regarding the question whether there are determinants predictive of MRI-detected damage, our search taught us that a patient’s age is negatively related to brain volume (moderate level of evidence), and that DCI relates to smaller BPV and lateral ventricle enlargement (low level of evidence). The first, although yielding limited evidence, is not surprising. It is well known that some quantity of brain volume loss is encountered with increasing age. 37 The second finding could be attributed to two things. One, in the study investigating this topic, the presence of new infarcts was included in their DCI definition. 25 Logically, this would lead to a negative relation of DCI with brain volume due to volume loss within the infarcted area. Two, pronounced EBI, resulting in an increase of global parenchymal loss, may increase the likelihood of developing DCI. Besides the relation of age and DCI with MRI-detected damage, we were not able to identify any other positive or negative determinant. In fact, most investigated determinants produced conflicting results. This may be due to heterogeneity of the used research methods. Across studies, different tools or scoring systems were used for both determinants and outcome measures which made the results difficult to summarize and may have introduced inadequacies. For example, two scoring methods were used to assess the amount of extravasated blood, i.e. Fisher grade and Hijdra scale. Of these two, the Hijdra score is thought to be more precise since it takes into account specific anatomic locations and it is shown to have a better interobserver agreement than the Fisher scale. 38 The same accounts for the discrepancies in findings between CFS/ICV and VIWr. In general the laborious volume measurements are thought to be more precise than the simple planimetric measure of the VIWr.
Our assessment of the relationship between MRI-detected damage and patient outcome, resulted in the findings that BPV and VIWr are related to clinical- and neuropsychological outcome (strong level of evidence), and that the presence of infarcts or infarct volumes are significantly correlated with clinical or neuropsychological outcome (moderate level of evidence). This latter finding is not surprising, since neurologic deficits are expected when infarcts occur in critical brain areas. It is known that tests may be difficult to perform in aSAH patients due to test difficulty, poor clinical condition, or severe headache during mental exercise. 3 Therefore, studies investigating these properties may be skewed towards patients with better clinical outcome due to selection bias. Furthermore, a simple clinical tool such as the GOS can underappreciate the neuropsychological impact due to the gross subdivision into categories. For instance, patients with GOS “5” (i.e. mild or no disability) might still have subtle cognitive deficits and a need to apply adaptive strategies in daily life.3,27,39
A limitation of this review is that only cerebral infarcts and brain volume changes were investigated. Not only infarcts but also many different focal injury entities can occur, including white matter lesions, superficial siderosis, and leukoaraiosis. Taking these lesions into account might lead to stronger levels of evidence. However, the manner of data presentation in the included studies did not allow for a thorough evaluation of this.
Another limitation is that most studies showed skewedness towards a good recovery for the included patients. This is probably caused by the fact that patients had to endure scanning sessions and other follow-up investigations to be able to participate. Moreover, a few studies used data of MRI-sequences which were only obtained when the patient voluntarily stayed longer in the scanner, resulting in selection bias.22–24 As well, a few studies even stated a good clinical condition or functional recovery as a inclusion criterium.25,35 Thereby, it is likely that the general patient outcome in clinical practice, and thereby most likely the MRI results, is worse than the presented results.
Future directions
The findings of this review are relevant as they underscore that not only focal brain injury plays an important role in the course of the disease, but presumably EBI also attributes significantly to brain damage as was proven by brain volume loss irrespective of brain infarcts. The results regarding possible determinants of infarct development or brain volume loss, and the relation between brain damage and neuropsychological or clinical outcome, however, were scarce and heterogeneous of character. On the one hand, this shows the complexity of the effects of the disease with research focusing on different parameters, while on the other hand, it emphasizes the need to conduct research on a uniform basis to be able to draw solid conclusions.
Therefore, in future work the entity of EBI, its effect on volume changes and, in continuation, its effect on patient outcome should be investigated. Of equal importance is the question how to intervene in this process to constrain global neuronal loss. In addition, standardized research needs to be conducted to draw rigid conclusions regarding determinants of brain injury and/or brain volume measurement outcomes, and the relation with clinical- or neuropsychological outcome. Only when understanding the source of the damage and the subsequent effects on patient level, therapies can be targeted to prevent any additional damage and with that improve patient outcome.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results has received funding from the European Research Council under the European Union's Horizon 2020 Programme (H2020)/ERC grant agreement n°637024.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
