Abstract
Background
Health-related quality of life measured with the EuroQol Group 5-Dimension Self-Report Questionnaire was one of the secondary outcomes in the Multicenter Randomized Clinical trial of Endovascular treatment for Acute ischemic stroke in the Netherlands (MR CLEAN). We reported no statistically significant difference in EuroQol Group 5-Dimension Self-Report Questionnaire score between the intervention and control groups, but deaths were not included.
Aims
Reanalyze the effect of intra-arterial treatment for large vessel occlusion in acute ischemic stroke patients on health-related quality of life in more detail. We now include patients who died during follow-up.
Methods
The EuroQol Group 5-Dimension Self-Report Questionnaire questionnaires were obtained 90 days after treatment. We used the Dutch tariff to derive a utility index from the EuroQol Group 5-Dimension Self-Report Questionnaire score. Treatment effect was estimated with the Mann–Whitney U test and linear regression. The effect of treatment on the distribution of EuroQol Group 5-Dimension Self-Report Questionnaire dimension scores was assessed with ordinal logistic regression.
Results
We obtained EuroQol Group 5-Dimension Self-Report Questionnaire scores from 457 (91.7%) of the 500 patients, including 108 who died before follow-up. Median EuroQol Group 5-Dimension Self-Report Questionnaire score in the intervention group was 0.57, and 0.39 in the control group (p = 0.03). Treatment effect estimated with linear regression was 0.07 (95%CI: −0.001 to 0.143). Treatment specifically affected EuroQol Group 5-Dimension Self-Report Questionnaire dimensions “mobility” (OR: 0.43, 95%CI: 0.29–0.66), “self-care” (OR: 0.60, 95%CI: 0.41–0.89), and “usual activities” (OR: 0.53, 95%CI: 0.36–0.79).
Conclusion
Treatment had a limited effect on quality of life, as measured with the EuroQol Group 5-Dimension Self-Report Questionnaire. Nevertheless, patients with acute ischemic stroke caused by an intracranial occlusion in the anterior circulation, who had intra-arterial treatment, experience better health-related quality of life than patients without intra-arterial treatment.
Trial Registration
URL: http://www.isrctn.com/ISRCTN10888758
Unique identifier: ISRCTN10888758
Keywords
Introduction
In MR CLEAN, a a multicenter randomized clinical trial of endovascular treatment for acute ischemic stroke, the health-related quality of life measured with the EuroQol Group 5-Dimension Self-Report Questionnaire (EQ-5D-3L) at 90 days was one of the secondary outcomes. The reported difference in the EQ-5D-3L score between the intervention group (intra-arterial treatment) and the control group was not significant: adjusted difference 0.06 (95% CI −0.01 to 0.18). 1 Patient who died before assessment of the EQ-5D-3L were not included in the analyses. This was the default statistical approach. However, in the EQ-5D valuations of health states, which make use of the TTO method, the state of death is given the anchor value of 0. We felt that these valuations should be included in order to reflect the total loss of quality of life due to stroke.
The rather small and nonsignificant effect of treatment on the EQ-5D-3L was remarkable since all clinical (modified Rankin Score, NIH Stroke Scale, and Barthel Index) and radiological outcome scores favored the intervention.
Aims
The purpose of this study was to reanalyze the effect of intra-arterial treatment for intracranial large vessel occlusion in acute ischemic stroke patients on health-related quality of life. In the present study we included patients who died during follow-up. We estimated the effect of IAT on the total score, the EQ-5D-3L, and the score on its dimensions. Furthermore, we studied the relation with other clinical outcome measures like the modified Rankin Score, the NIH Stroke Scale, and Barthel Index.
Methods
Study design
Patient eligibility and methods of the MR CLEAN trial have been reported previously.1,2 In short, MR CLEAN was a randomized clinical trial of IAT versus no IAT along with best medical care in patients with a proximal intracranial arterial occlusion in the anterior circulation demonstrated on vessel imaging, treatable within 6 h after symptom onset.
Outcome measures
EQ-5D-3L was scored at 90 days after inclusion. The EQ-5D-3L consists of a descriptive system with five dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. Each dimension has three levels: 1, no problems; 2, some problems; 3, extreme problems. When three levels are included a total of 243 (35) possible health states are defined. The EQ-5D-3L score was converted into a utility index through a country-specific value set (the Dutch tariff).3,4 The Dutch tariff was established on the basis of the time trade-off method. The values represent the preferences of the Dutch population. Patients who died before the follow-up interviews at 90 days received a zero score on the EQ-5D-3L, an anchor score in the valuation. Scores range from −0.33 to 1.00, with higher values indicating a better quality of life. A single experienced investigator, who was unaware of the treatment assignments, interviewed the patient, proxy, or health care provider by telephone to assess the EQ-5D-3L.
Statistics
Difference in EQ-5D-3L utility scores between treatment arms was compared with the Mann–Whitney U test. Treatment effect on the EQ-5D-3L utility score was estimated with the use of linear regression.1,2 The effect of treatment on the EQ-5D-3L was adjusted for age, NIH Stroke Scale at baseline, time from stroke onset to randomization, previous stroke, atrial fibrillation, diabetes mellitus, and occlusion of the internal carotid artery terminus.
We assessed the effect of treatment on the distribution of each of the dimension scores with ordinal logistic regression. To validate the EQ-5D-3L assessment we correlated (Spearman) it with the score on the modified Rankin Score at 90 days, the NIH Stroke Scale at one week, and the Barthel Index at 90 days. All analyses were performed with Stata/SE statistical package, version 14.1.
Results
Baseline characteristics
Clinical characteristics at baseline and outcomes by availability of EQ-5D-3L assessment
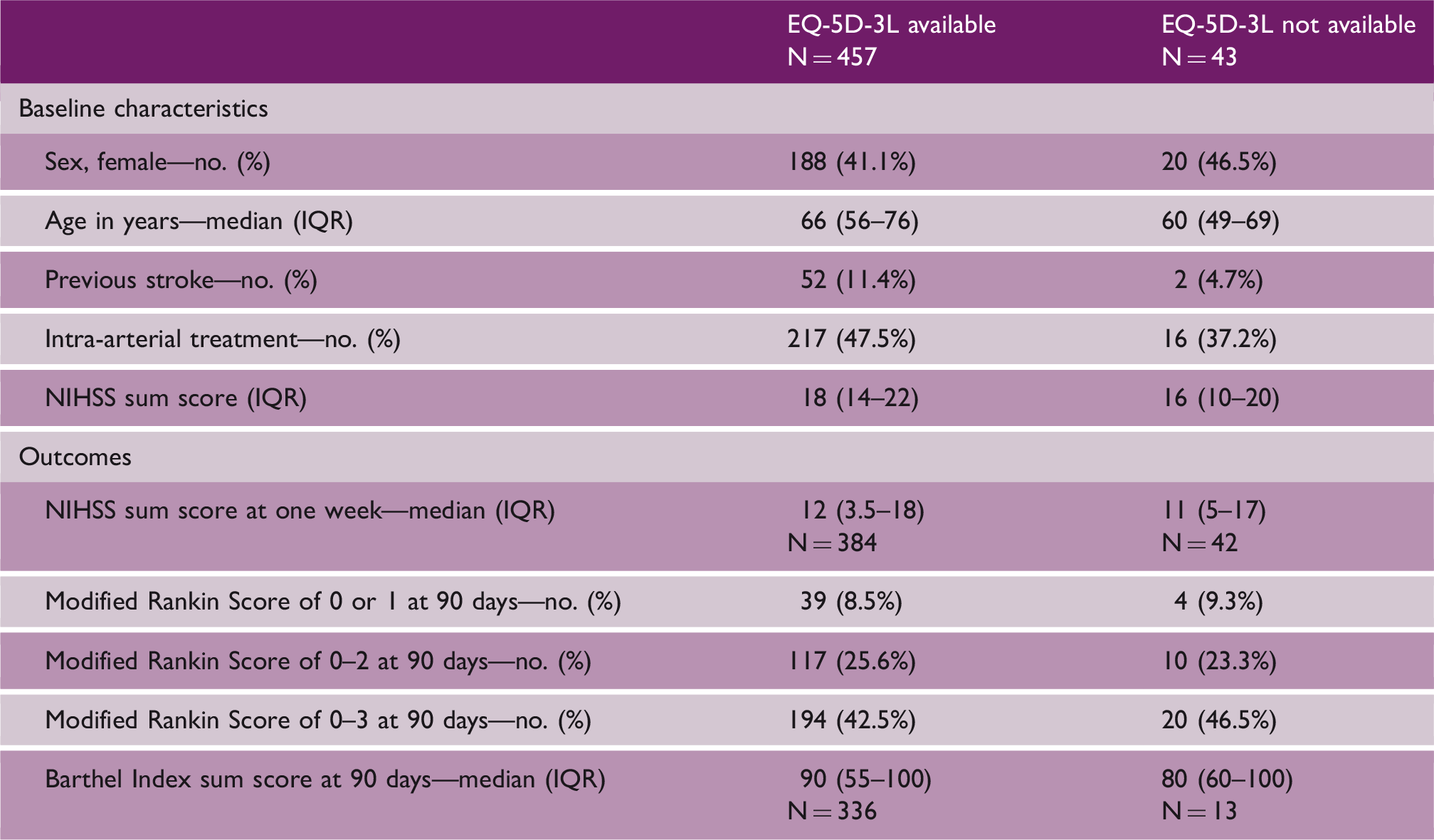
EQ-5D-3L: EuroQol Group 5-Dimension Self-Report Questionnaire; IQR: interquartile range.
EQ-5D-3L
The median EQ-5D-3L score in the intervention group was 0.57, and in the control group 0.39 (p = 0.03, Mann–Whitney U test). In the intervention group 49 of 217 patients died (22.6%) and were given a zero score on the EQ-5D-3L, compared to 59 of 240 patients (24.6%) in the control group. Treatment effect estimated with linear regression was 0.07 (beta coefficient, 95% CI −0.001 to 0.143). The adjusted value was 0.06 (beta coefficient, 95% CI −0.005 to 0.128). Treatment effect was seen in the EQ-5D-3L dimensions “mobility,” “self-care,” and “usual activities.” We observed no significant treatment effect for the dimensions “pain/discomfort” and “anxiety/depression” (Figure 1).
Effect of treatment on distribution of scores in the EQ-5D-3L dimensions. EQ-5D-3L: EuroQol Group 5-Dimension Self-Report Questionnaire.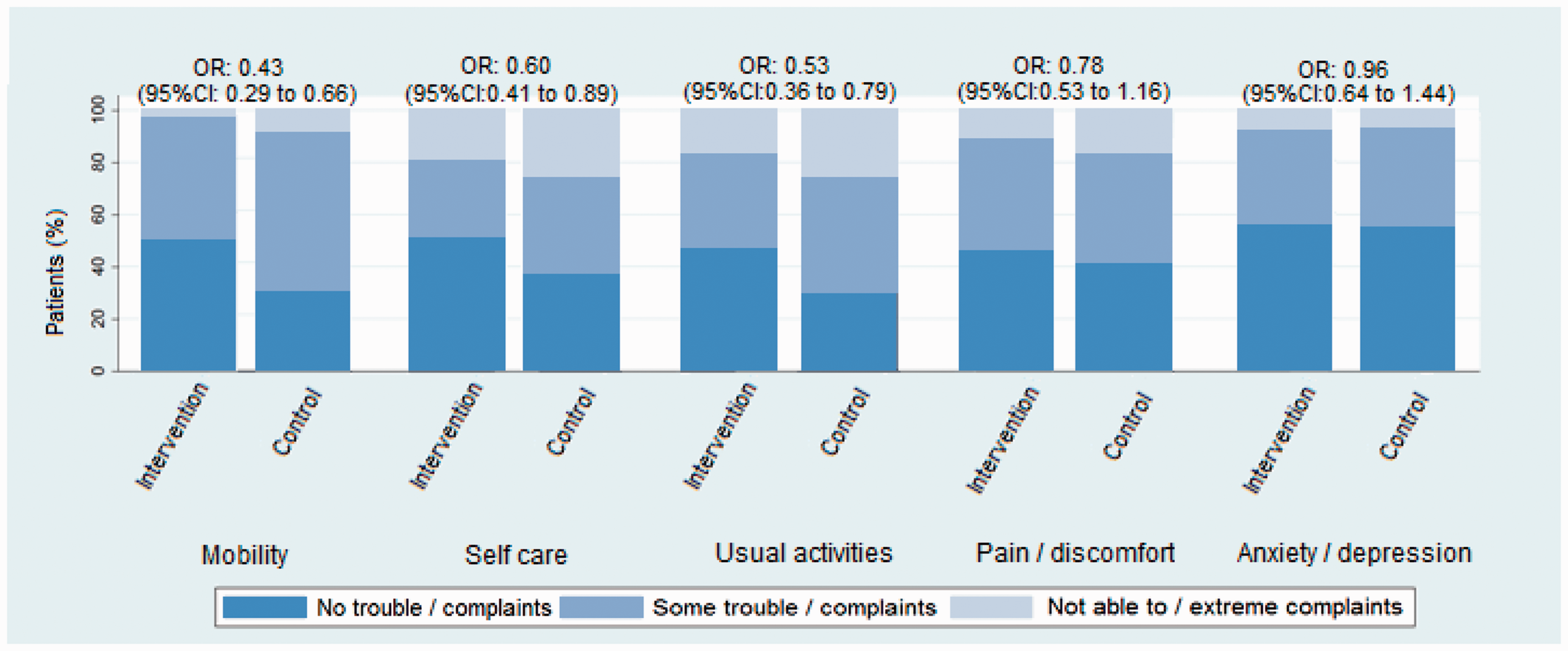
Validation
The correlation between the EQ-5D-3L score and the modified Rankin Score at 90 days was strong (rho = 0.67, p < 0.01), and the same was true for the correlation with the Barthel Index at 90 days (rho = 0.45, p < 0.01). The correlation with the NIH Stroke Scale at one week was moderate (rho = 0.30, p < 0.01).
Discussion
In this updated analysis of MR CLEAN, the effect of intra-arterial treatment on the EQ-5D-3L score, which now incorporated mortality, was significant with the Mann–Whitney U test. Treatment effect estimated with linear regression was smaller than the difference between the median scores, because regression models essentially compare means and the distribution of scores was skewed and bimodal. Compared to the previously published results, the beta coefficient is unchanged, but now with a smaller 95% confidence interval. Our reanalyzed EQ-5D-3L scores are similar to the results found in other RCTs of intra-arterial treatment for acute ischemic stroke. REVASCAT reported a treatment effect of 0.13 (95% CI 0.03–0.23) 5 and the ESCAPE trial of 9.4 (95% CI 3.5–15.2). 6 However, the ESCAPE trialists used the EQ-5D visual-analog scale, which has a range of 0–100 and not the EQ-5D utility index. The SWIFT PRIME and EXTEND-IA trials did not report the EQ-5D as a secondary outcome.7,8
The effect of treatment on the EQ-5D-3L is relatively small compared to the other clinical outcomes and we found no treatment effect for two out of the five EQ-5D-3L dimensions (“pain/discomfort” and “anxiety/depression”). Particularly the dimension “pain/discomfort” is originally defined as “pain or other complaints.” One could expect this dimension also covers neurological deficits that interfere less with mobility or self-care dimensions. However, our results indicate otherwise. This raises the question if the EQ-5D-3L reflects quality of life in the full range of limitations relevant to stroke patients. However, we showed that the EQ-5D-3L correlated well with other valid measures of functional outcome and slightly less with neurological deficits.
Our study has several limitations. First, the NIH Stroke Scale was done 1 week after treatment allocation and the EQ-5D-3L at 90 days. Second, there was no formal blinding for treatment allocation of interviewer and of course, patients could also be aware of the treatment they had received. Third, ideally we want to compare EQ-5D-3L scores for each patient before and after stroke, but EQ-5D-3L scores before stroke were not available. Last, in this study we only had three-month results. Long-term results will be presented in CLOT MR CLEAN (cost-effectiveness analyses and long-term follow-up in MR CLEAN). 9
In conclusion, we found that treatment had a limited effect on quality of life, as measured with the EQ-5D-3L. Nevertheless, we can conclude that patients with acute ischemic stroke caused by an intracranial occlusion in the anterior circulation, who had intra-arterial treatment within 6 h after stroke onset, experience a better health-related quality of life at three months after onset than patients without intra-arterial treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The MR CLEAN trial was partly funded by the Dutch Heart Foundation and by unrestricted grants from AngioCare BV, Medtronic/Covidien/EV3®, MEDAC Gmbh/LAMEPRO, Penumbra Inc., Stryker®, and Top Medical/Concentric. Erasmus MC received funds from Stryker® and Bracco Imaging® for consultations by DWJD. AMC received funds from Stryker® for consultations by CBLMM, YBWEMR, and OAB. MUMC received funds from Stryker® for consultations by WHZ.
