Abstract
Aims
This study was performed to determine the short-term risk-benefit profiles of patients treated with oral anticoagulation for acute ischemic stroke or transient ischemic attack using a multicenter, prospective registry.
Methods
A total of 1137 patients (645 men, 77 ± 10 years old) with acute ischemic stroke/transient ischemic attack taking warfarin (662 patients) or non-vitamin K antagonist oral anticoagulants (dabigatran in 205, rivaroxaban in 245, apixaban in 25 patients) for nonvalvular atrial fibrillation who completed a three-month follow-up survey were studied. Choice of anticoagulants was not randomized. Primary outcome measures were stroke/systemic embolism and major bleeding.
Results
Both warfarin and non-vitamin K antagonist oral anticoagulants were initiated within four days after stroke/transient ischemic attack onset in the majority of cases. Non-vitamin K antagonist oral anticoagulant users had lower ischemia- and bleeding-risk indices (CHADS2, CHA2DS2-VASc, HAS-BLED) and milder strokes than warfarin users. The three-month cumulative rate of stroke/systemic embolism was 3.06% (95% CI 1.96%–4.74%) in warfarin users and 2.84% (1.65%–4.83%) in non-vitamin K antagonist oral anticoagulant users (adjusted HR 0.96, 95% CI 0.44–2.04). The rate of major bleeding was 2.61% (1.60%–4.22%) and 1.11% (0.14%–1.08%), respectively (HR 0.63, 0.19–1.78); that for intracranial hemorrhage was marginally significantly lower in non-vitamin K antagonist oral anticoagulant users (HR 0.17, 0.01–1.15). Major bleeding did not occur in non-vitamin K antagonist oral anticoagulant users with a CHADS2 score <4 or those with a discharge modified Rankin Scale score ≤2.
Conclusions
Stroke or systemic embolism during the initial three-month anticoagulation period after stroke/transient ischemic attack was not frequent as compared to previous findings regardless of warfarin or non-vitamin K antagonist oral anticoagulants were used. Intracranial hemorrhage was relatively uncommon in non-vitamin K antagonist oral anticoagulant users, although treatment assignment was not randomized. Early initiation of non-vitamin K antagonist oral anticoagulants during the acute stage of stroke/transient ischemic attack in real-world clinical settings seems safe in bleeding-susceptible Japanese population.
Keywords
Introduction
Cardioembolic stroke is the most severe ischemic stroke subtype. Nonvalvular atrial fibrillation (NVAF) has become more common as a cause of cardioembolism, mainly in the elderly population throughout the world.1,2 It has been well demonstrated that warfarin reduces recurrent ischemic stroke or transient ischemic attack (TIA) in patients with NVAF, but such patients, especially Asian patients, have a high risk for intracranial bleeding.3,4
Non-vitamin K antagonist oral anticoagulants (NOACs), also known as direct oral anticoagulants, have been shown to be effective for secondary prevention with infrequent bleeding complications in patients with NVAF and previous stroke/TIA in global randomized, controlled trials.5–9 However, a problem with these trials was lack of data on the risk-benefit profile of early anticoagulation after stroke, because acute stroke/TIA patients were excluded from enrollment.5–8 In addition, differences in conditions between trials and real-world practice, as well as Asian ethnic features including susceptibility to bleeding, 4 could cause unexpected postmarketing results for NOACs in Asian stroke care.
Aims
To resolve the above clinical questions, the prospective, multicenter, observational, Stroke Acute Management with Urgent Risk-factor Assessment and Improvement (SAMURAI)-NVAF Study was designed. In the initial report from the study, early initiation of NOACs after ischemic stroke/TIA seemed safe during acute hospitalization. 10 The aim of the present study was to determine the risk-benefit profile within three months after receiving warfarin or NOACs in stroke/TIA patients enrolled in the registry.
Methods
In the SAMURAI-NVAF Study, 1192 NVAF patients who were hospitalized within seven days after onset of ischemic stroke/TIA between September 2011 and March 2014 were enrolled in 18 stroke centers balanced regionally across Japan (Online Supporting Information Appendix, Participating Sites and Investigators). NVAF was diagnosed on 12-lead electrocardiogram or 24-h or longer monitoring for AF detection during acute hospitalization or from previous medical documents. The rationale and design of the study have been described elsewhere. 10 Study procedures were reviewed and approved by local Ethics Committees.
Baseline characteristics, features of index events, acute care process, and outcomes
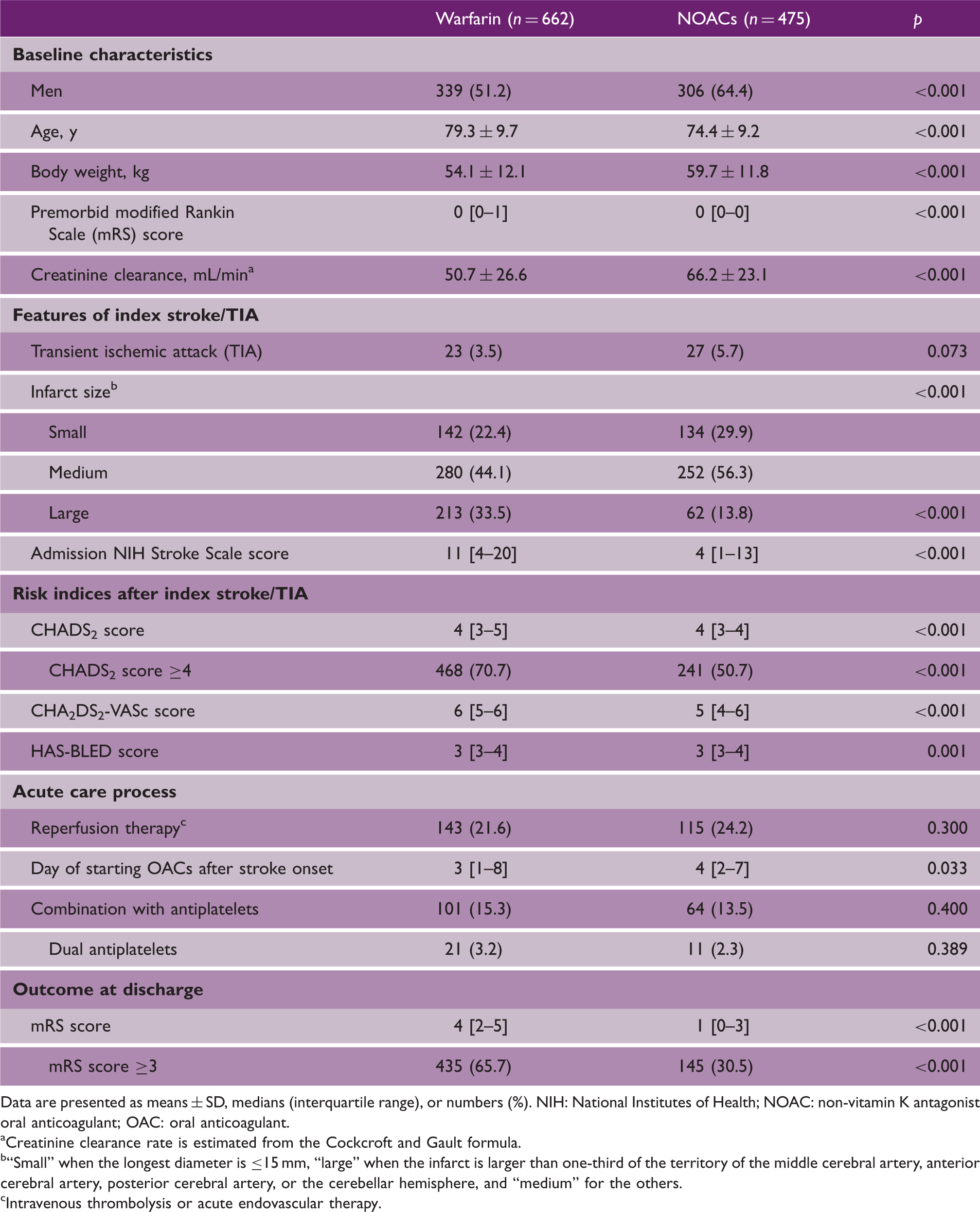
Data are presented as means ± SD, medians (interquartile range), or numbers (%). NIH: National Institutes of Health; NOAC: non-vitamin K antagonist oral anticoagulant; OAC: oral anticoagulant.
Creatinine clearance rate is estimated from the Cockcroft and Gault formula.
“Small” when the longest diameter is ≤15 mm, “large” when the infarct is larger than one-third of the territory of the middle cerebral artery, anterior cerebral artery, posterior cerebral artery, or the cerebellar hemisphere, and “medium” for the others.
Intravenous thrombolysis or acute endovascular therapy.
The target intensity of the prothrombin time (PT)-international normalized ratio (INR) was principally 2.0 to 3.0 for patients younger than 70 years and 1.6 to 2.6 for those 70 years or older according to Japanese guidelines. 14 Dosages of NOACs were determined based on renal function, age, and others according to each medical package insert. The official daily doses in Japan were: dabigatran 300 mg or 220 mg; rivaroxaban 15 mg or 10 mg (based on results of a domestic trial 15 ); and apixaban 10 mg or 5 mg.
The primary efficacy outcome was stroke or systemic embolism within three months. The primary safety outcome was major bleeding within three months defined according to the International Society on Thrombosis and Hemostasis statement, 16 including fatal bleeding, symptomatic bleeding in a critical area or organ, or bleeding causing a fall in hemoglobin level of 2.0 g/dl or more or leading to transfusion of two or more units of whole blood or red blood cells. Secondary outcomes included: (1) any ischemic events, including recurrence of ischemic stroke or TIA, systemic embolism, acute coronary syndrome, aortic dissection, aortic aneurysm rupture, peripheral artery disease requiring hospitalization, venous thromboembolism, and revascularization such as carotid endarterectomy, carotid artery stenting, and percutaneous coronary intervention; (2) ischemic stroke or TIA; (3) intracranial hemorrhage, including intracerebral, subarachnoid, and subdural hemorrhages; and (4) mortality from any cause. Observation was discontinued when the patients developed any primary or secondary endpoint, as their anticoagulants were principally changed after events.
Statistics
Data are presented as means ± standard deviation (SD), median values (interquartile range), or numbers (%). Variables were compared between warfarin and NOAC users using χ2 tests, unpaired t tests, and Wilcoxon’s tests, as appropriate. The cumulative rates of primary and secondary events were calculated within three months after initiation of oral anticoagulants using a Cox proportional-hazards model adjusted by potential confounding factors (sex, age, CHADS2 score, admission National Institutes of Health Stroke Scale (NIHSS) score, creatinine clearance). In consideration of the small number of outcomes, complementary analyses using propensity scores as an adjustment covariate were added. To develop the propensity scores, control variables listed in Table 1 were included. All statistical analyses were performed with JMP 11·0·2 statistical software (SAS Institute, Inc., Cary, NC). A p < 0·05 was considered significant.
Results
Of 1192 patients, 55 patients not taking oral anticoagulants after the index stroke/TIA mainly due to severe neurological deficits were excluded; thus, the remaining 1137 patients (645 men, 77 ± 10 years old) were studied. Of these, three-month data for 38 patients taking warfarin, nine taking dabigatran, seven taking rivaroxaban, and two taking apixaban were not available mainly due to dropouts from the follow-up survey; for these patients, subacute data until around a month after registration were used
Types of events.
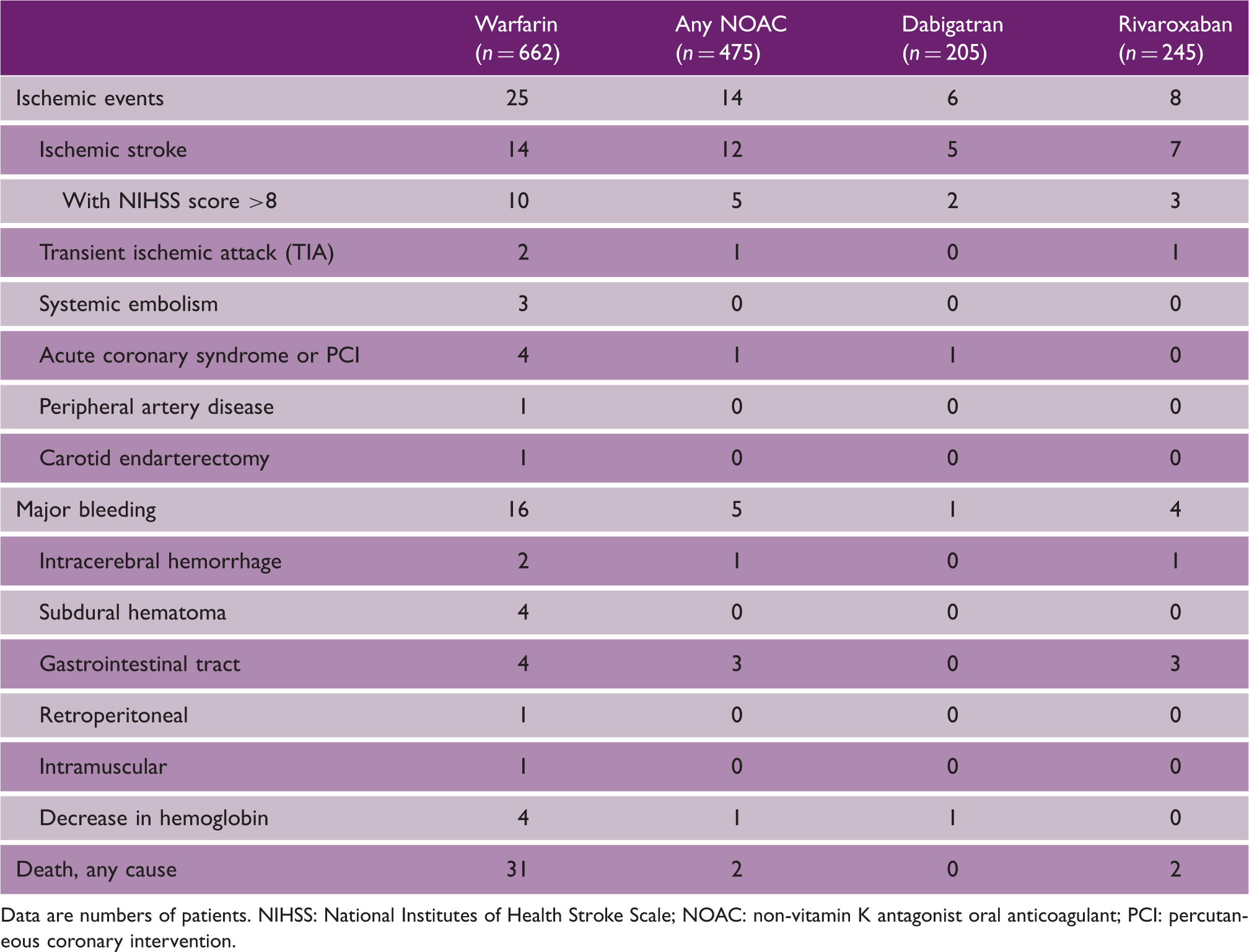
Data are numbers of patients. NIHSS: National Institutes of Health Stroke Scale; NOAC: non-vitamin K antagonist oral anticoagulant; PCI: percutaneous coronary intervention.
The cumulative rate of stroke/systemic embolism at three months was 3.06% (95% CI 1.96%–4.74%) in warfarin users and 2.84% (1.65%–4.83%) in NOAC users (Table 3). The event curve showed a steep slope in the initial weeks (Figure 1(a)). After multivariable adjustment, the risk did not differ significantly between warfarin users and NOAC users (adjusted hazard ratio (HR) 0.96, 95% CI 0.44–2.04). In the subgroups, there was a significant interaction between the treatment effects and body weight (p = 0.036, Figure 2(a)).
Cumulative rates for the primary outcomes. (a) Stroke or systemic embolism, (b) Major bleeding. Choice of anticoagulants (warfarin or non-vitamin K antagonist oral anticoagulants) was depended on the discretion of the physicians in charge (not randomized). OAC, oral anticoagulant; NOAC: non-vitamin K antagonist oral anticoagulants. Relative risk of the primary outcomes by subgroup. (a) Stroke or systemic embolism, (b) Major bleeding. Choice of anticoagulants (warfarin or non-vitamin K antagonist oral anticoagulants) was depended on the discretion of the physicians in charge (not randomized). CI, confidence interval; mRS, modified Rankin Scale; and NIH, National Institutes of Health; NOAC: non-vitamin K antagonist oral anticoagulants. Primary and secondary outcomes Choice of anticoagulants (warfarin or non-vitamin K antagonist oral anticoagulants) was depended on the discretion of the physicians in charge (not randomized). Adjusted by sex, age, CHADS2, admission National Institutes of Health Stroke Scale score, and creatinine clearance. NIHSS: National Institutes of Health Stroke Scale; NOAC: non-vitamin K antagonist oral anticoagulant; CI: confidence interval; TIA: transient ischemic attack. p < 0.05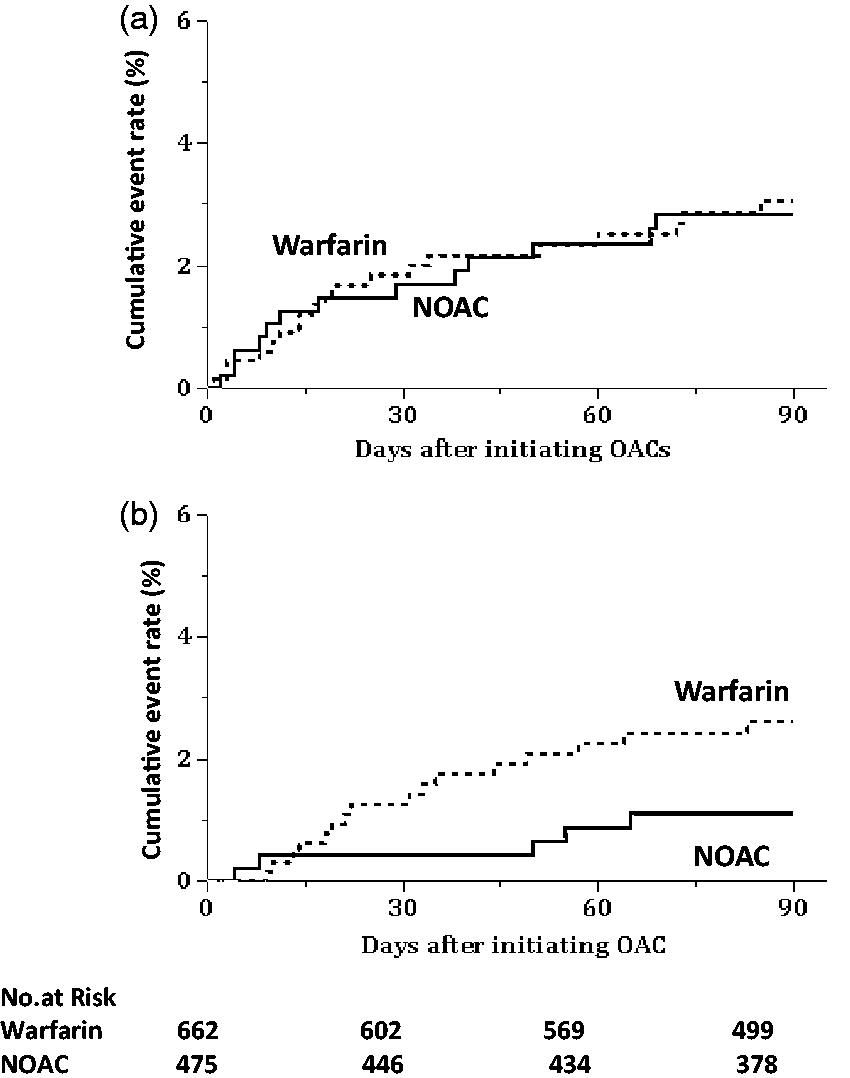
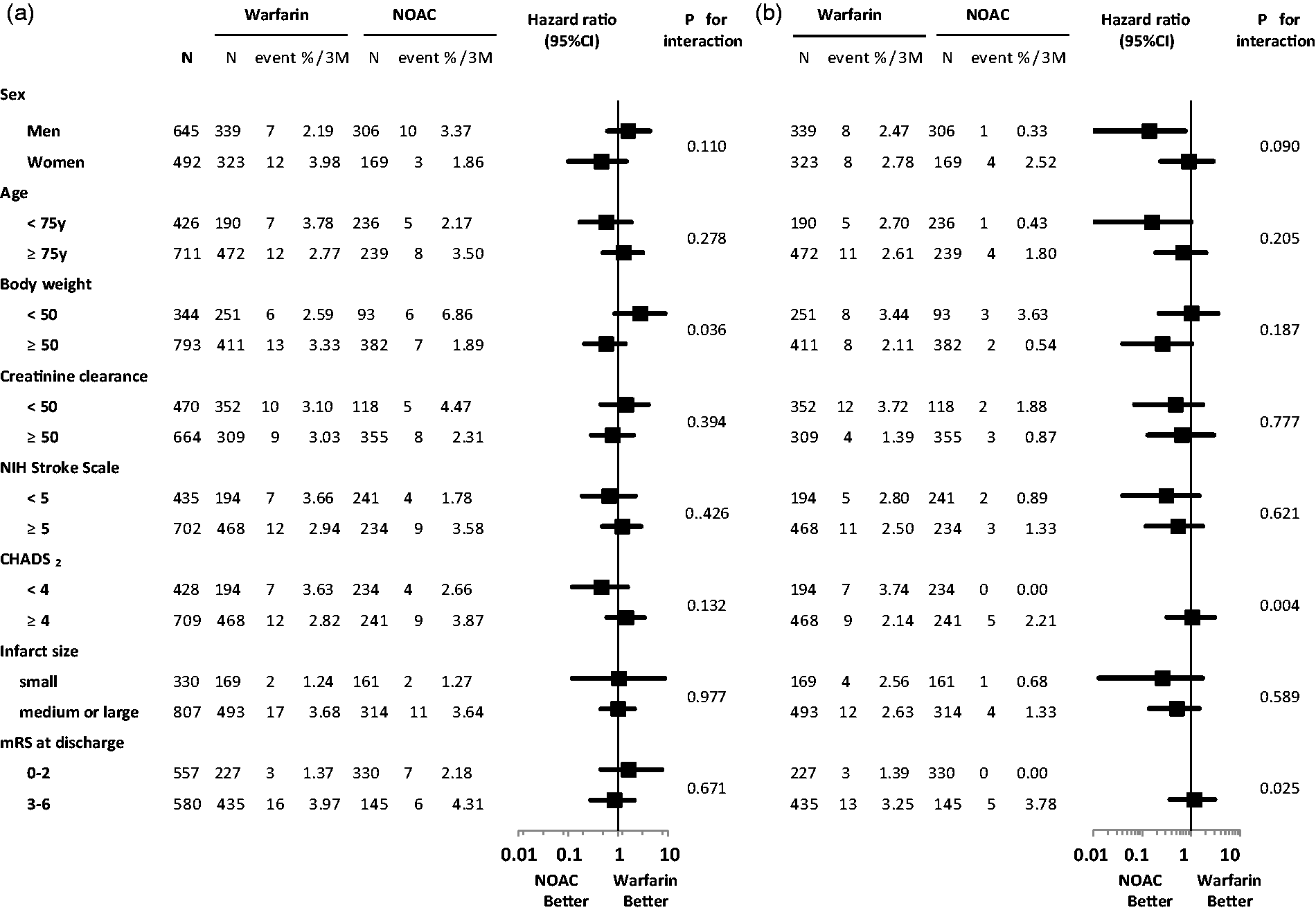
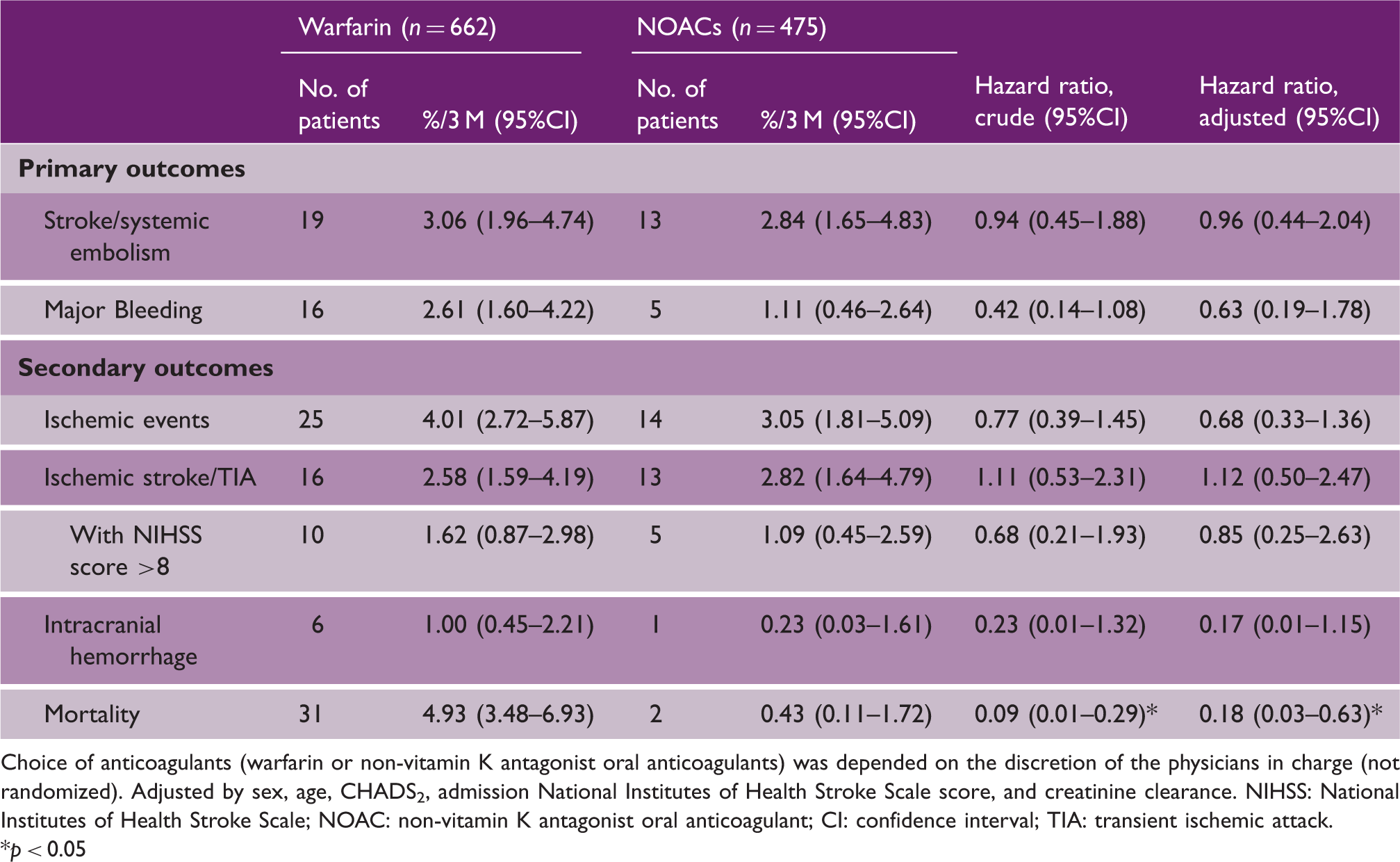
The cumulative rate of major bleeding at three months was 2.61% (95% CI 1.60%–4.22%) in warfarin users and 1.11% (0.14%–1.08%) in NOAC users (Table 3). Events did not occur within the first several days and then occurred in a relatively linear fashion (Figure 1(b)). After multivariable adjustment, the risk of major bleeding did not differ significantly between warfarin users and NOAC users (HR 0.63, 95% CI 0.19–1.78). In the subgroups, major bleeding did not occur in NOAC users with CHADS2 scores <4 or those with discharge mRS scores ≤2, and the interactions between the treatment effects and these two scales were significant (p = 0.004 and p = 0.025, respectively, Figure 2(b)).
Of the secondary outcomes, the cumulative rate of any ischemic events and that of ischemic stroke/TIA, as well as that of ischemic stroke with the initial NIHSS score >8, were similar between warfarin users and NOAC users (Table 3, Figure 3). Intracranial hemorrhage tended to be less common in NOAC users (0.23% (95% CI 0.03%–1.61%) than in warfarin users (1.00% (0.45%–2.21%). Mortality was less common in NOAC users (0.43% (0.11%–1.72%) than in warfarin users (4.93% 3.48%–6.93%). Apixaban users did not develop any primary or secondary outcome events.
Cumulative rates for the secondary outcomes. (a) Ischemic events, (b) Ischemic stroke or TIA, (c) Intracranial hemorrhage, (d) Mortality. Choice of anticoagulants [warfarin or non-vitamin K antagonist oral anticoagulants) was depended on the discretion of the physicians in charge (not randomized). OAC, oral anticoagulant; TIA, transient ischemic attack; NOAC: non-vitamin K antagonist oral anticoagulants.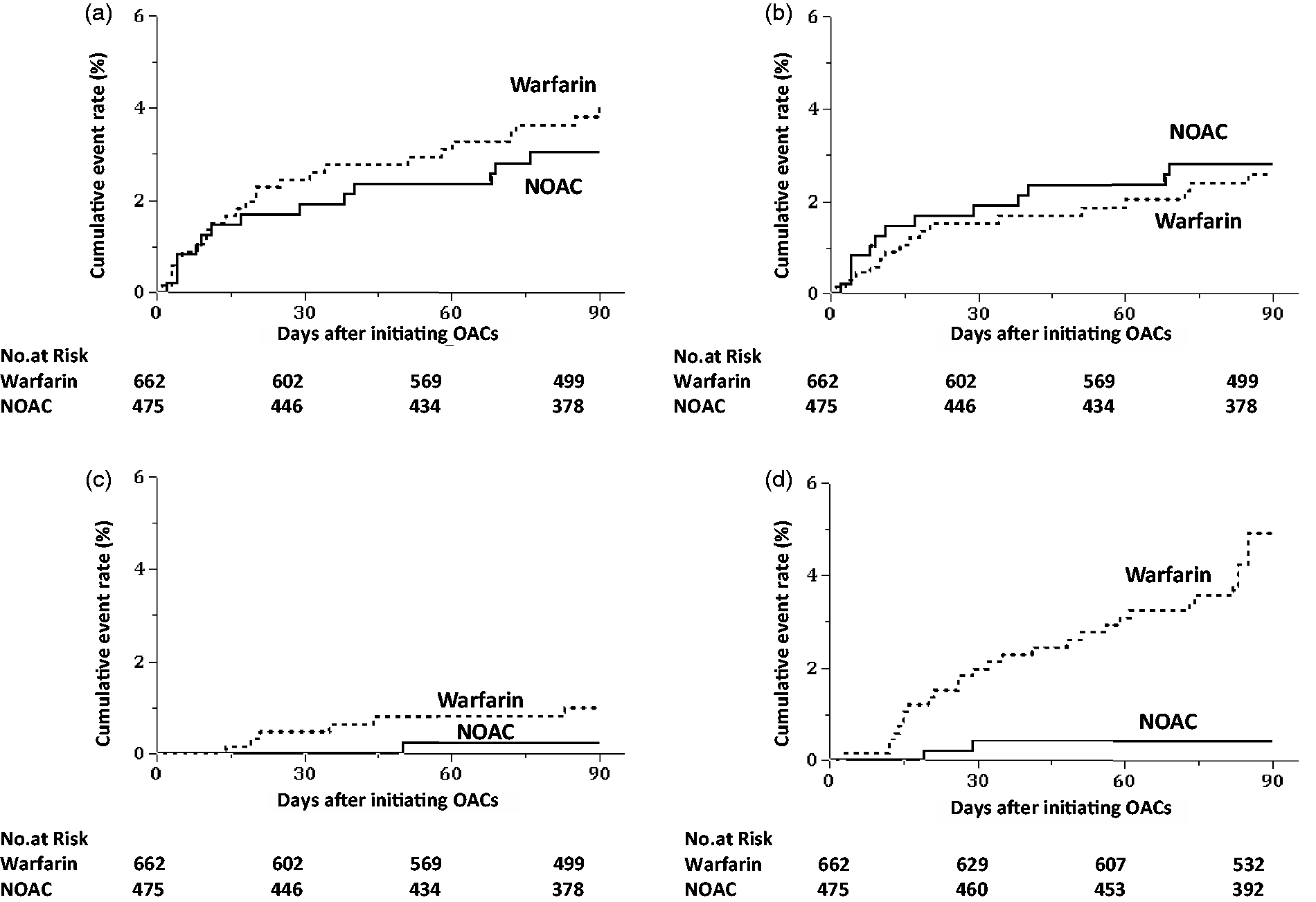
Primary and secondary outcomes were also compared using the propensity score analysis. Intracranial hemorrhage (HR 0.15, 95% CI 0.01–1.01) and mortality (HR 0.32, 95% CI 0.05–1.14) tended to be less frequent in NOAC users than in warfarin users (Figure S1).
Discussion
Conditions for secondary stroke prevention in daily clinical settings differ greatly from the ideal trial setting with respect to patients’ background characteristics, medication compliance, and intensity of anticoagulation. The SAMURAI-NVAF Study was performed to elucidate real-world risks and benefits of anticoagulation including NOAC use, mostly initiated quite early after stroke/TIA onset. In the present study, the risk-benefit profiles of warfarin vs. NOACs within the initial three months were determined. The first major finding was that both the cumulative rate of stroke/systemic embolism in warfarin users and that in NOAC users (≈3% for both) were not high compared to those in the previous trials and observations. The second major finding was that major bleeding, in particular intracranial hemorrhage, was relatively less common in NOAC users than in warfarin users, although the differences were not significant. These two findings suggest that the risk-benefit profile of NOACs in major clinical trials was reproducible in clinical settings for acute stroke/TIA patients. The third finding was that major bleeding did not occur in NOAC users with CHADS2 scores <4 or those with discharge mRS scores ≤2.
The present cohort had several features that may have caused different risk-benefit profiles of anticoagulation from those in major clinical trials. Early initiation of oral anticoagulation, 17 as well as a purely Japanese cohort known to have a high prevalence of intracranial hemorrhage due to susceptibility to intracranial vasculopathy, high sodium intake, and other factors,4,18 might increase the bleeding risk. On the other hand, the low target intensity of PT-INR for warfarin in aged patients and the low official dosage of rivaroxaban, which are both unique to Japan, mainly due to the fear of bleeding,14,15 might weaken the benefit of anticoagulants. In fact, plasma concentrations of rivaroxaban in acute stroke patients in our hospital were relatively low compared with those in the major trial.19,20 We thus hypothesized that our patients might show divergent risk-benefit profiles from those reported previously.
Ischemic stroke recurred within 7 to 14 days in 3.0% of cardioembolic stroke patients with heparin or heparinoid use in a meta-analysis of seven trials involving 4624 patients. 17 The rate of recurrent ischemic stroke within 30 days after cardioembolic stroke was 5.3% from the Rochester Epidemiology Project medical records 21 and 6.2% from the Japan Standard Stroke Registry. 22 The rate within 90 days in stroke patients with atrial fibrillation was 10% in the data from the Virtual International Stroke Trials Archive (VISTA) 23 and 7.6% in the Early Recurrence and Cerebral Bleeding in Patients With Acute Ischemic Stroke and Atrial Fibrillation (RAF) study. 24 The present three-month rate of stroke/systemic embolism and that of ischemic stroke/TIA were ≈3% or less with both warfarin and NOACs. The present rates were higher than those in major trials including chronic stages of stroke/TIA,5–9 but somewhat lower than most of the previous rates from acute patients. A reason for the relatively low rates in the present study was that severe patients who developed recurrent stroke before initiating oral anticoagulants were excluded. Some fatal ischemic events might not have been accurately diagnosed, especially in the present warfarin users, since they often had high mRS scores and might often have missed close inspection for fatal events.
The rate of intracranial bleeding within the initial 7 to 14 days was 2.5% in the above meta-analysis from seven trials. 17 The rate within 90 days was 3% in the VISTA 23 and 3.6% in the RAF. 24 Intracranial hemorrhage occurred in only 0.23% of the present NOAC users, and its relative risk was only 17% of that in warfarin users. Thus, NOAC appears to be relatively safe for acute stroke patients.
The subgroup analyses of major bleeding might be biased due to both the small event number and nonrandomized method. The advantage of NOACs over warfarin in preventing major bleeding was clear when the CHADS2 score was 0–1, and it was somewhat unclear when the score was 3–6, in both the single trial and a meta-analysis of major trials;25,26 these findings were similar to the present study. The interaction between anticoagulant effects and independence after stroke has never been determined. The present finding that major bleeding did not occur in NOAC users with mRS scores ≤2 suggested the safety of early NOAC use after mild stroke.
Although not statistically significant, sex showed an interesting interaction with treatment effects; stroke/systemic embolism tended to be less common in women (HR 0.46, 0.10–1.45) and major bleeding was less common in men (HR 0.14, 0.01–0.76) during NOACs than during warfarin (Figure 2). Women with atrial fibrillation were reported to be at greater risk of ischemic stroke without warfarin, 27 as well as during warfarin anticoagulation.28,29 As a possible cause, women spent more time below the therapeutic range of PT-INR than men in a large database involving 4060 patients. 28 In contrast, a meta-analysis of major trials did not show sex differences in the risk of ischemia and showed a smaller risk of major bleeding in female NOAC users than male users. 29 These data, including the present insignificant tendency, suggest the superiority of NOACs to warfarin in preventing ischemia for women.
The present study has the strength that the real-world effects of oral anticoagulation during the initial years of the NOAC era were clarified by the government-funded registration study. All the participating 18 institutes were known to have systematic stroke care units or stroke units and departments of cardiology as well. Thus, results of the study seem to be representative of the actual conditions of Japanese NVAF patients with recent ischemic stroke/TIA. However, this strong point caused several limitations. First, the choice of anticoagulants depended on the discretion of the physicians in charge, and therefore, stroke severity and other background characteristics differed greatly much between warfarin users and NOAC users, as described elsewhere. 10 To minimize the influence of different background characteristics on the interpretation of results, both an analysis using Cox proportional-hazards models and a propensity score analysis were done and showed similar results. However, because of extremely different backgrounds and the small number of outcome events, these analyses might be biased. Statistical difference in mortality between groups did not remain significant in the propensity score analysis. Since most of causes of death were not ischemia or bleeding, choice of anticoagulants did not seem to contribute to death much in the present cohort. Second, intravenous heparin was used during the acute stage for many warfarin users and some NOAC users as a bridging therapy, and heparin affected the risk-benefit profile. Acute heparin use in the present cohort was safe for patients without major artery occlusion. 30 Third, data on apixaban and edoxaban were incomplete because of the timing of their commercial approval, and comparisons between dabigatran and rivaroxaban were difficult due to the small patient numbers and small event numbers.
In conclusion, early initiation of NOACs during the acute phase of stroke/TIA seems safe at up to three-month follow-up in bleeding-susceptible Japanese population. We will continue the follow-up of the present subjects for two years and ascertain the long-term risk-benefit profile.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by a Grant-in-Aid (H23-Junkanki-Ippan-010) from the Ministry of Health, Labour and Welfare, Japan, Grants from the Japan Agency for Medical Research and Development (AMED: 15ek0210051h0001, 15ek0210042h0001), and an Intramural Research Fund (H23-4-3) for Cardiovascular Diseases of the National Cerebral and Cardiovascular Center.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
