Abstract
This study made the first attempt to combine resting respiratory sinus arrhythmia (RSA) and cortisol to provide an explanatory mechanism for the effect of acute stress on emotion-induced blindness (EIB) from the perspective of vagus nerve activity and stress hormone responses. For this purpose, resting electrocardiogram (ECG) signals were recorded first. Participants underwent both the socially evaluated cold-pressor test and control treatments 7 days apart and then completed the EIB task. Heart rate and saliva samples were collected over time. The results demonstrated that acute stress promoted the overall detection of targets. Resting RSA and cortisol levels predicted the stress-induced changes in EIB performance under the negative distractor condition at lag2 negatively and positively, respectively. These findings indicate that the effect of stress on EIB was partially contributed by cortisol, which is more relevant to negative distractor conditions. Resting RSA, as an indicator of inter-individual differences, further provided evidence from the perspective of the trait emotional regulation ability based on the vagus nerve control. In general, resting RSA and cortisol changes over time exhibit different patterns of influence on stress-induced changes in EIB performance. Thus, this study provides a more comprehensive understanding of the effect of acute stress on attentional blindness.
Introduction
Stress permeates modern life. Acute stress activates the sympathetic–adrenal–medullary (SAM) axis and the hypothalamus–pituitary–adrenal (HPA) axis to secrete the increased catecholamines and glucocorticoids (mainly cortisol) (Allen et al., 2014), and is generally known to induce a switch from goal-directed behaviour control by prefrontal cortex (PFC) regulation towards stimulus-directed rapid emotional responses control by the amygdala (Arnsten, 2009; Hermans et al., 2014; Shields et al., 2016; Vogel et al., 2016). This allows individuals to temporarily prioritise rapid responses (especially to emotionally salient stimuli) over thoughtful higher-order cognitive functions. Indeed, when individuals dynamically process consecutive information in a short period, they are often disturbed by emotional information, which results in blindness to the target. Therefore, this study examines the effect of acute stress on emotional distractor-induced attentional blindness. Resting respiratory sinus arrhythmia (RSA) and cortisol were measured to investigate their relationship with stress-induced changes in emotion-induced blindness (EIB) performance from the aspect of vagus nerve activity and stress hormone responses.
EIB
Emotionally arousing stimuli prioritise attentional selection; when it appears as a to-be-ignored critical distractor item in rapid serial visual presentation (RSVP), it usually draws attention from the bottom-up and interferes with the processing of the target and then induces a transient deficit in the detection of single targets. This is known as EIB (Most et al., 2005; Wang et al., 2012). Studies have shown that the initial attention capture for the distractors is primarily determined by the physical salience and that emotional salience always exerts a potent interference after the initial stage; that is, the negative distractors could further enter the highly limited stage for working memory storage compared with the neutral distractors, which is a crucial stage for inducing attentional blindness (Baker et al., 2021; Hoffman et al., 2020).
The effect of acute stress on EIB
Existing studies have focused on the effects of acute stress on the attentional blink (AB; Raymond et al., 1992), which is a form of functional attentional blindness, resulting from a trade-off between two targets with limited available resources at the relatively late central processing stage (Chun & Potter, 1995; Nieuwenstein et al., 2009). Kan et al. (2019) set the first target as emotional and the second target as neutral and found that stress reduced the AB effect at lag2, independent of the target emotionality. Their subsequent study using the same AB paradigm combined with event-related potential (ERP) measures indicated that stress-reduced emotional AB was mainly related to the facilitation of the early selective attention process of the second target (Kan, Wang, et al., 2021). In other words, individuals under stress could better select the target among task-irrelevant interference (filler items) for refined processing in working memory.
Notably, emotional stimuli in AB are the targets that need to be reported, whereas emotional stimuli in EIB are the critical distractors that need to be ignored. Most et al. (2010) found that a sense of unease, as a social emotion, correlated with EIB performance decrements induced by negative distractors. While anxious anticipation does not affect EIB task accuracy, it takes longer for individuals to recover from the interference of negative distractors (Haddara et al., 2019). EIB reflects target processing under the interference of an emotional distractor in RSVP. Previous studies have also investigated the influence of stress on the interference effect in Stroop, explained through cue utilisation account with the belief that stress withdraws attentional resources from task-irrelevant stimuli, thereby allowing limited attentional resources to be more focused on the current target (Booth, 2019; Callaway & Dembo, 1958; Chajut & Algom, 2003; Easterbrook, 1959; Hoskin et al., 2014; Momin et al., 2020). Some studies also arrived at the contrasting conclusion and found the detrimental effect of stress on tasks requiring interference inhibition (Kan, Xue, et al., 2021; Sanger et al., 2014). In general, the influence of stress on EIB still needs to be further investigated.
Acute stress affects EIB in relation to RSA
The inconsistent target processing may have to do with the parasympathetic nervous system (PNS) activities. Specifically, the neurovisceral integration model indicates that the PFC connects with the PNS efferent to the heart via the vagus nerve and regulates PNS activities (Thayer et al., 2009, 2012). In other words, higher resting PNS activity indicates greater activity in the PFC (Thayer et al., 2012). Previous studies have suggested that individuals who upregulated their executive control network (PFC regulation) and downregulated their salience network (amygdala and related subcortical structures) under stress showed improved target detection (Kohn et al., 2017). Therefore, we speculated that the inter-individual difference in PNS activities might provide a potential explanatory mechanism for understanding target processing in EIB under stress.
RSA is considered to be a valid indicator of PNS activities via the vagus nerve control of the heart (Berntson et al., 1993; Porges, 2001, 2007). This is a cardiorespiratory phenomenon characterised by heart rate or R-R interval fluctuations, which are phase-related to inhalation and exhalation, respectively (Grossman & Taylor, 2007). It is generally believed that high resting RSA represents the greater coordination of physiological responding (Weissman & Mendes, 2021) and is thus associated with better emotional regulation and adaptive response ability to stress (Beauchaine & Thayer, 2015; Geisler et al., 2013; Porges, 2007; Thayer & Lane, 2000). Therefore, according to the polyvagal theory, vagal withdrawal during stress is particularly pronounced in individuals with high resting-state RSA, as RSA is inhibited during stress (Porges, 2001).
However, to date, no studies have focused on the relationship between resting RSA and target processing under the interference of emotional distractors. Previous studies have shown that high resting RSA exhibited a less negative facial response to negative stimuli (Demaree et al., 2004, 2006; Pu et al., 2010) and was related to the decreased negative emotions in response to daily stressors (Fabes & Eisenberg, 1997). The suppressed emotional response reduces the self-control capacity and further depletes the limited resources required for subsequent tasks. Thus, the high resting RSA individuals demonstrated an impaired working memory performance under the negative condition (Pu et al., 2010; Schmeichel, 2007). Therefore, we speculated that high resting RSA tends to be associated with worse target detection induced by stress.
Acute stress affects EIB in relation to cortisol
Cortisol is a core hormone in stress response, and its binding to receptors in the PFC and amygdala may play an important role in the effect of stress on EIB. However, it is still unclear whether stress exerts its effects on EIB primarily or entirely through cortisol. Prior studies on the relationship between cortisol levels, the detection of targets, and interference of emotional distractor illustrate the inconsistent conclusions. Acute stress could impair the top-down behaviour based on PFC functioning by upregulating cortisol levels (Porcelli et al., 2008; Vogel et al., 2016). However, it could also enhance the processing of emotional distractors during working memory, wherein individuals with high cortisol levels in response to stressors are less affected by emotional distractors (Oei et al., 2012). Hydrocortisone (cortisol) administration has also reduced selective attention towards emotional distractors (Oei et al., 2009; Putman et al., 2007, 2010).
Notably, we cannot ignore the particularity of attention processing in the RSVP paradigm, in which there is fierce competition for attentional resources between rapidly presented stimuli in millisecond time frames. Previous studies have found a beneficial effect of cortisol on changes in stress-induced AB, but they did not focus on the cortisol activity throughout the entire experiment. Specifically, Schwabe and Wolf (2010) simply divided participants into high and low cortisol response groups, and Kan et al. (2019) supplemented the role of cortisol in the existing facilitation effect of stress on AB. Overall, we preliminarily speculated that cortisol would decrease attention engagement towards negative distractors and would thus relate to better performance. Moreover, studies have demonstrated that stress regulation could increase PFC activity and reduce stress response (Baeken et al., 2014; Pulopulos et al., 2020). In this study, we also sought to examine whether better stress regulation abilities among individuals with high resting RSA interact with lower cortisol responses to be associated with poor stress-induced EIB performance changes.
The present study
The present study investigated the influence of acute stress on EIB, and resting RSA and cortisol provide a fully explanatory pathway for understanding this effect. Based on existing studies, we hypothesised the following: (a) acute stress would improve the target accuracy; (b) high resting RSA would predict the decreased stress-induced changes in EIB performance, especially for the negative distractor conditions; (c) cortisol levels would positively predict the increase in stress-induced changes in EIB performance; and (d) individuals with high resting RSA who have a lower cortisol stress response would be associated with the worse performances when stressed.
Method
Participants and design
A priori power analysis was conducted for a 2 × 2 × 3 within-subjects, repeated-measures analysis of variance (ANOVA) with a medium effect size of at least 0.25 (f) and 95% statistical power using G*Power 3.1 software (Faul et al., 2007). This analysis suggested a minimum sample size of 18 participants. Considering that regression analysis was also involved in the subsequent data analysis, another priori power analysis was also run for multiple regression analysis with a medium effect size (f2 = 0.15) and 80% statistical power, which suggested a minimum sample size of 55 participants. Therefore, we collected as much data as possible to test the study effects. In total, 67 healthy undergraduates recruited from Shaanxi Normal University and Xi’an Polytechnic University participated in exchange for monetary reward. They had normal or corrected-to-normal vision (right-handedness) and provided written informed consent. However, 2 weeks before the first experiment, the subjects completed the State-Trait Anxiety Inventory (STAI) and Beck Depression Inventory (BDI) through an online scale collection platform to avoid the possible impact of known trait factors (Booij et al., 2015; Vreeburg et al., 2010). Participants with a score of 35–50 on the Trait Anxiety Inventory were deemed to have non-trait anxiety, and scores below 14 on the BDI indicated the absence of depressive symptoms (Beck et al., 1988; Shek, 1993; Spielberger, 1983). Furthermore, only individuals with a body mass index (BMI) between 18 and 27 kg/m2 were included in this experiment. Given the sensitivity of stress hormones (Kudielka et al., 2009), all participants were asked not to take any medicine during the last week prior to the experiment and no drinking, eating, or strenuous exercise was allowed within 3 hr before the experiment. No participant had a history of hypertension and heart disease. Female subjects were also not allowed to participate in the experiment during the menstrual period. The study conformed to the principles of the Declaration of Helsinki (World Medical Association, 2013) and was permitted by the Academic Committee of the school of psychology, Shaanxi Normal University in China.
To minimise the impact of individual differences on the experimental results, a within-participant design was performed. All subjects completed two experimental sessions; that is, they need to, respectively, perform an EIB task after acute stress treatment and control treatment 7 days apart. The order of the two sessions was also counterbalanced across participants. Two parallel versions of the EIB task composed of different image materials were available. The order of two parallel versions of the task was also counterbalanced across the two experiments. Specifically, the participants randomly completed one version of the EIB task in the first experiment and the other version in the second experiment.
All experiments were conducted in the afternoon between 14:00 and 18:00 to control for the effect of diurnal rhythm of cortisol (Izawa et al., 2010). The six subjects were excluded because they were unable to complete the two experiments on time. Data from a total of 61 subjects were included. A sensitivity power analysis was further conducted for multiple regression analysis (α = .05, power = .8). The results indicated that the minimum effect size detectable was 0.133, which was a medium effect size. The sample characteristics are depicted in Table 1.
Descriptive sample characteristics.
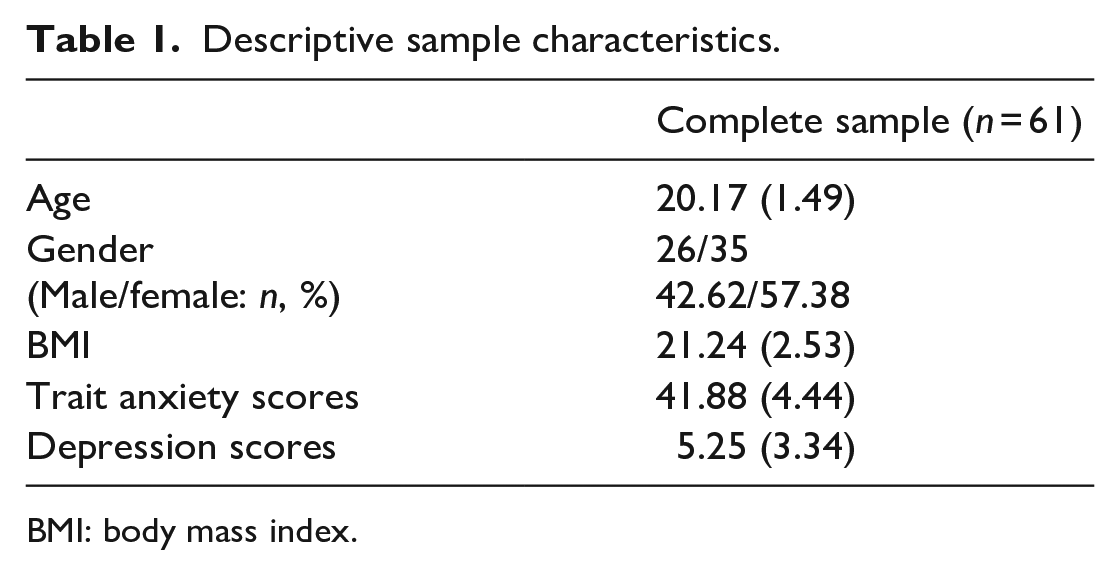
BMI: body mass index.
Acute stress induction
The socially evaluated cold-pressor test (SECPT) was used to induce participants’ psychophysiological stress experience, which has been shown to be effective in activating the acute stress response-related autonomic nervous system and the HPA axis (Schwabe et al., 2008; Schwabe & Schächinger, 2018). Under the stress condition, participants thoroughly immersed their hands, including their wrists, in ice water (0°C–3°C) and were recorded by a camera. The water temperature was continuously monitored by a thermometer. During the immersion, there were also two unfamiliar experimenters (of whom one was of the opposite sex for the participants) in white laboratory coats who blankly stared at the participants during the process without saying a word. The participants were required to face the camera. The camera and the two experimenters were positioned in equilateral triangles to ensure that the participants felt the pressure from both the camera recording and the experimenters’ observation. The participants did not know how long they had to keep their hands in the water but were informed that they could remove them at any time if they could not tolerate it after 3 min.
Under the control condition, the participants were asked to immerse their hands in warm water (35°C–38°C) for 3 min. Only one familiar experimenter, dressed in casual clothes and with a neutral expression, accompanied the participants to complete the immersion task, and there was no camera.
Acute stress response measures
Subjective response
The Positive Affect and Negative Affect Scale (PANAS) (Watson et al., 1988) was used to measure the subjects’ emotional changes during the experiment, in the same way as Kan, Wang et al. (2021). It contains 20 items scored on a five-point scale, split into two dimensions, with 10 positive and 10 negative items.
Physiological measures
The electrocardiogram (ECG) 100C amplifier module based on BIOPAC MP160 (BIOPAC Systems, Inc., CA, USA) was used to continuously record the ECG data. Three Ag–AgCl disposable electrodes were placed in a standard lead II configuration. ECG signals were acquired and processed using AcqKnowledge 6.0 software. ECG data were acquired online at a 1,000 Hz sampling rate with 0.5–35 Hz bandpass offline filtering. A combination of both visual checks and automated identification was used to process the artefact (e.g., temporary loss of signal or an unusual R-R interval) of the ECG signal. The missed beating within the spurious R-R interval was adjusted using the interpolation method (Berntson et al., 1997). Specifically, the R-wave time series was converted to interbeat intervals and sampled at 4 Hz to produce equal time intervals. A fast Fourier transformation was submitted for a spectral power analysis to calculate RSA. RSA was defined as the power density in the 0.15–0.40 Hz frequency band (Berntson et al., 1997). A natural log conversion of high-frequency heart rate variability (HF-HRV) was performed to approximate a normal distribution and was expressed in ln units (ms 2 ).
A resting ECG was recorded for a 10-min baseline period, of which the last 5 min were used for the RSA analysis. Participants were required to maintain silence and spontaneous breathing while viewing a fixation point presented on the monitor.
After collecting the resting data, ECG signals were continuously recorded from the SECPT/control task until the end of the entire experiment. The average heart rate was calculated during the baseline period, SECPT/control task period, the first three blocks of EIB period (first 3b), and the last three blocks of the EIB period (last 3b). The RSA was also analysed during the SECPT period to calculate RSA reactivity, which represents the withdrawal of the vagus in response to the stressor.
Saliva sample collection
The participants were required to hold the swab from Salivette collection devices (Salivette, Sarstedtstr.1 D-51588, Germany) under tongue for 45 s. The collected saliva samples were stored in a freezer (−20°C). Thawed saliva samples were centrifuged at 3,000 r/min for 20 min to extract the supernatant. The concentration of cortisol in saliva sample was detected by sandwich enzyme-linked immunosorbent assay (ELISA) in vitro. The inter-assay coefficients of variability were 10%. The intra-assay coefficients of variability were 7%.
EIB task
Two parallel versions of the experimental task, with only different image materials, were used in this experiment. The experimental task consists of emotional critical distractors, targets, and filler items. The negative and neutral distractor images were drawn from International Affective Picture System (IAPS) (Lang et al., 2008). 1 We also supplemented the materials with the same type of images from the internet. Specifically, the negative distractor images were those involving war, bleeding, and injured people or animals, and also fierce, scary animals. Neutral distractors were normal images of people or animals that do not involve anything scary or bloody, but nothing pleasurable either. As the filler items and target images were architectural and landscape images, we chose distractors by excluding images that contained a lot of architectural or landscape information to avoid confusion. The stimuli supplemented from internet were made publicly available at https://osf.io/eqhx8/.
Targets and filler items were landscape and architecture images selected from copyright-free website (https://pixabay.com/). All images were 420 × 315 pixels colour graphs (visual angle 8.6° × 7°). A total of 100 negative and 100 neutral distractor images, and 150 rotated target images were included. 2 An additional 20 participants (15 females; Mage = 20.68 years, SD = 1.55, range: 18–22) re-evaluated the distractor images to ensure their validity in eliciting the corresponding emotional experience. Specifically, all the emotional distractor images were re-evaluated for pleasure and arousal during RSVP on a nine-point scale. The independent sample t-test results have shown that the pleasure of negative distractors (M = 3.311, SD = 0.446) was significantly lower than that of neutral distractors (M = 6.356, SD = 0.711), t(198) = −36.100, p < .001. The arousal of negative distractors (M = 8.003, SD = 0.411) was significantly higher than that of neutral distractors (M = 2.734, SD = 0.752), t(198) = 61.157, p < .001. The target accuracy was also evaluated in the absence of critical distractors during RSVP to ensure that subjects themselves had no difficulty in judging the orientation of target. The accuracy between the architectural images (M = 0.988, SD = 0.029) and landscape images (M = 0.985, SD = 0.035) was not significant, t(298) = −0.706, p = .480. The filler items were 750 upright landscapes and architecture images, which randomly called in two parallel versions.
Participants sat in front of a 24-in monitor, 70 cm away (refresh rate: 100 Hz). The task was presented via E-prime 3.0 software. Each trial started with a white fixation cross lasting 1,000 ms against a black background, and 17 images were presented sequentially for 100 ms each. Each trial contained one distractor (negative, neutral, or distractor-absent condition, the distractor was a substitute to filler items), one target (landscape or architecture images of left or right arrows), and 15 filler items (upright landscape or architecture images). The distractor was randomly presented at the third, fourth, and fifth positions, and the target always appeared two or eight items (lag2, lag8) after the distractor. Participants were required to identify the orientation of the targets by pressing the left or right arrow key at the end of image presentation (see Figure 1). The positions of the distractor and target were counterbalanced between all conditions. The formal experiment consisted of 300 trials and was segmented into six blocks. Participants took a fixed rest lasting 90 s between each block to ensure consistency in the time points for saliva sample collection time points. They were also required to perform 16 practice trials (except for the negative distractors condition) before the formal experiment.
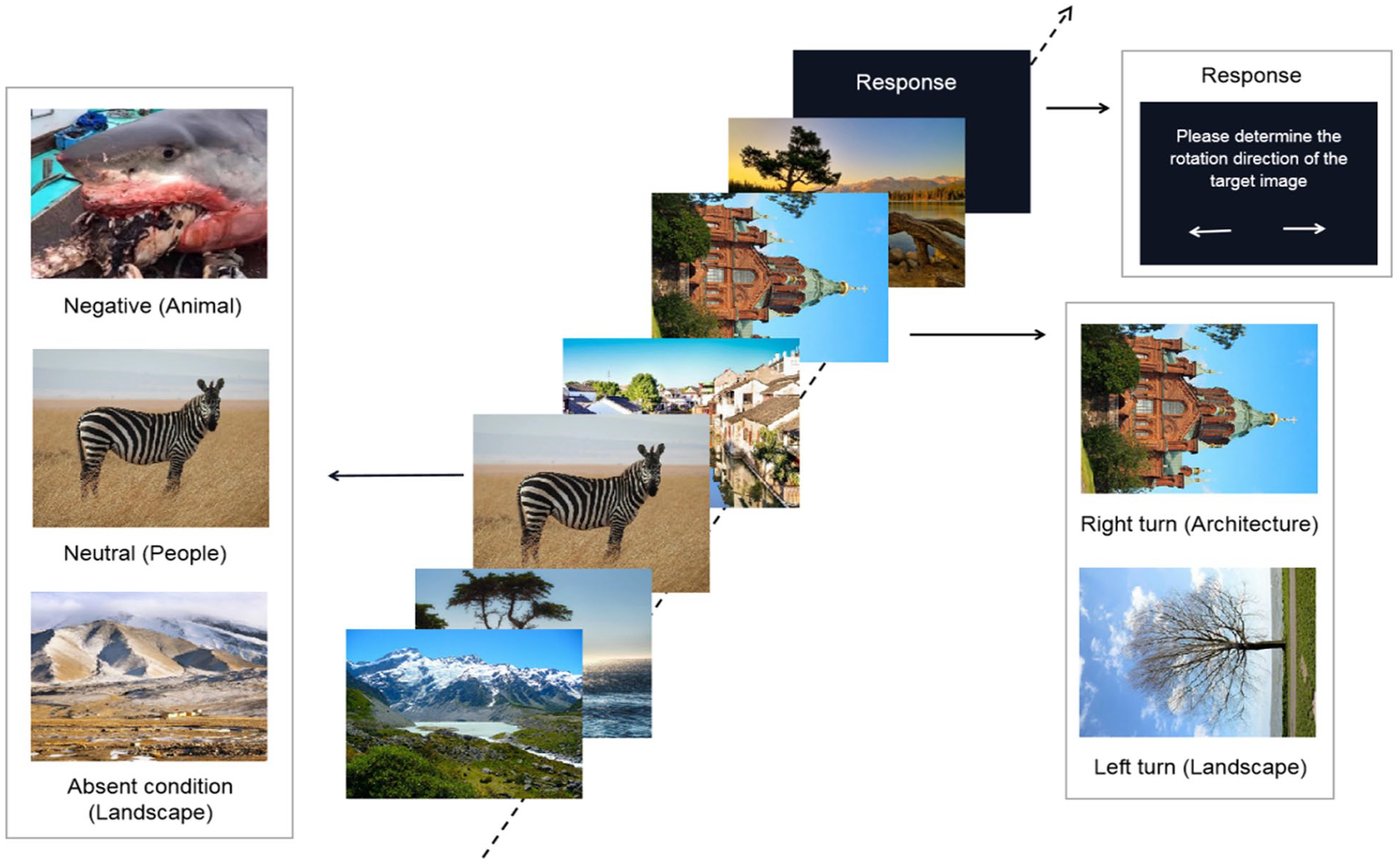
An illustration of a sample trial in experiment.
General procedure
Participants read and signed an informed consent form and rested for 15 min before starting the formal experiment. Resting ECG signals were acquired for 10 min. Then, the first saliva sample (sC1) was collected from the participants, after which they completed the PANAS (P1). After the practice experiment and understanding the RSVP task procedure, they were randomly assigned to the SECPT or control treatment condition. The second saliva sample (sC2) was collected immediately, and the PANAS (P2) was completed. Following this, participants performed the EIB task and underwent the third saliva sample collection (sC3) after the first three blocks. Finally, the fourth saliva sample (sC4) was collected from the participants, after which they again completed the PANAS (P3). ECG signals were also continuously recorded from the SECPT/control task until the end of the entire experiment (see Figure 2).
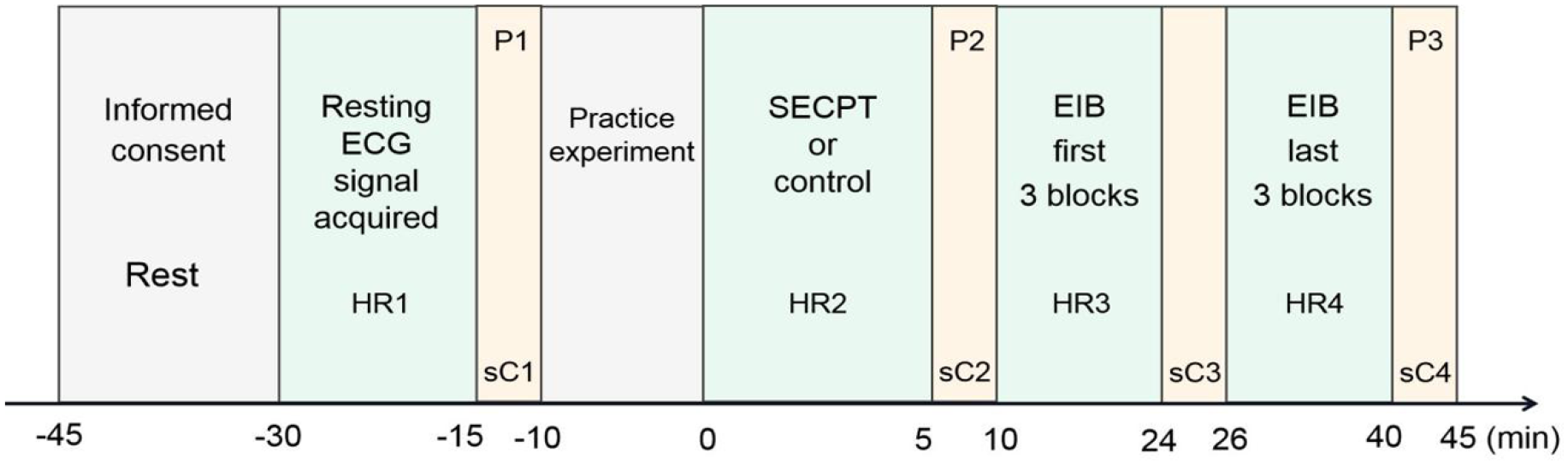
Schematic diagram of the overall experimental timeline. “HR” stands for heart rate.
Statistical analyses
Subjective assessments were analysed using 2 × 3 repeated-measures ANOVA with treatment (SECPT, control condition) and time (pre-SECPT, post-SECPT, and post-EIB). A repeated-measures ANOVA with treatment (SECPT, control condition) and period (resting, SECPT/control task, the first three blocks of EIB, the last three blocks of EIB) was conducted on the collected heart rate. We examined changes in cortisol concentration over time using 2 × 4 repeated-measures ANOVA with treatment (SECPT, control condition) and time (pre-S, post-S, post-EIB 3b, post-EIB 6b). Performance in the EIB task under stress was analysed with a 2(treatment: SECPT, control) × 3(critical distractors: negative, neutral vs absent) × 2(lag: lag2, lag8) rmANOVA. For all, the data were analysed with SPSS statistics 21. All reported p-values are two-tailed, and partial-eta
2
(
To simultaneously investigate the roles of resting RSA and cortisol in the effect of stress on EIB, Pearson correlations were first performed to examine the associations between the primary study variables. The cortisol changes over time were calculated by the area under the curve with respect to ground (AUCg) under stress and control condition, respectively (Pruessner et al., 2003). The AUCg was defined below:
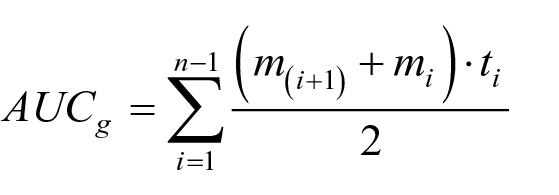
In the formula above, mi represents the salivary cortisol concentration in one measurement, ti is the time distance between measurements, and n is the total amount of measures. The stress-induced cortisol changes were calculated by subtracting AUCg-control from AUCg-stress. RSA reactivity was obtained by subtracting the RSA during SECPT from the resting-state RSA. Changes in heart rate were calculated in the same way as cortisol, and stress-induced heart rate changes were calculated by subtracting AUCg-control from AUCg-stress. The stress-induced EIB performance change was also represented by subtracting ACCcontrol from ACCstress under different conditions. False discovery rate (FDR) correction for p values was performed by running a script in MATLAB2020b to control the false-positive rate. Hierarchical multiple regression analyses were further executed. All indicators were standardised prior to entry into the regression equation.
Results
Subjective stress responses: PANAS
The main effect of treatment was significant, F(1, 60) = 9.080, p = .004,
For the negative affect, treatment interact with time points was significant, F(2, 120) = 13.752, p < .001,
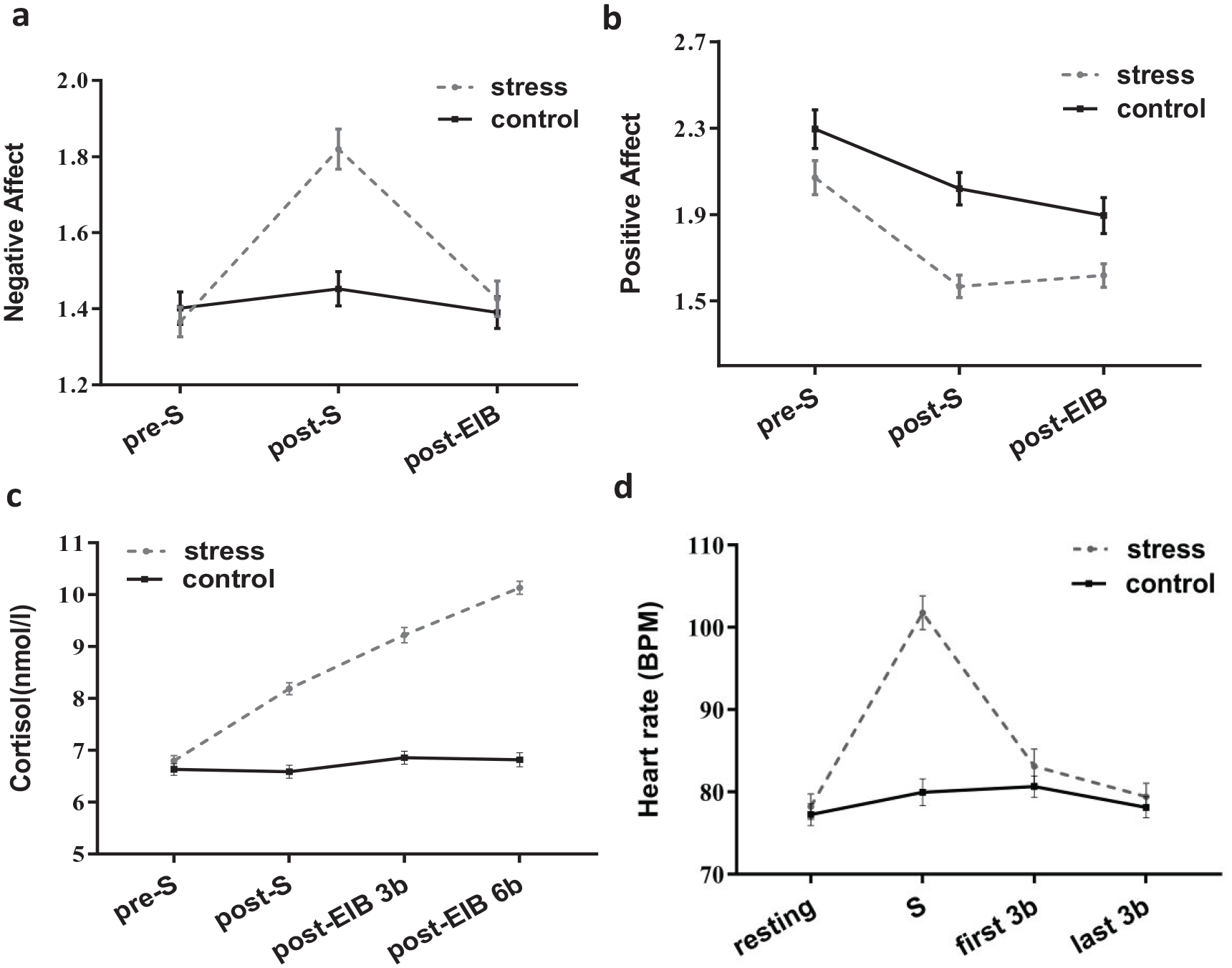
(a) Negative affect scores for the stress and control treatments varied with time. (b) Positive affect scores for the stress and control treatments varied with time. (c) Cortisol levels for the stress and control treatments varied with time. “S” stands for SECPT or control task. “3b” stands for three blocks in EIB and “6b” stands for six blocks in EIB. (d) Heart rate for the stress and control treatments varied with time. “First 3b” stands for the first three blocks of EIB period and “last 3b” stands for the last three blocks of the EIB period. Bars represent standard errors of mean.
Heart rate
The main effect of the treatment was significant, F(1, 60) = 25.078, p < .001,
Treatment significantly interacted with the period, F(3, 180) = 19.762, p < .001,
Salivary cortisol
The main effect of treatment was significant, F(1, 60) = 629.997, p < .001,
There was a significant interaction between treatment and time points, F(3, 180) = 76.634, p < .001,
EIB performance: accuracy of targets
The main effect of critical distractor emotionality was significant, F(2, 120) = 35.527, p < .001,
Critical distractor emotionality was significantly interacted with lag, F(2, 120) = 25.970, p < .001,
Target accuracy at each condition (M ± SD).
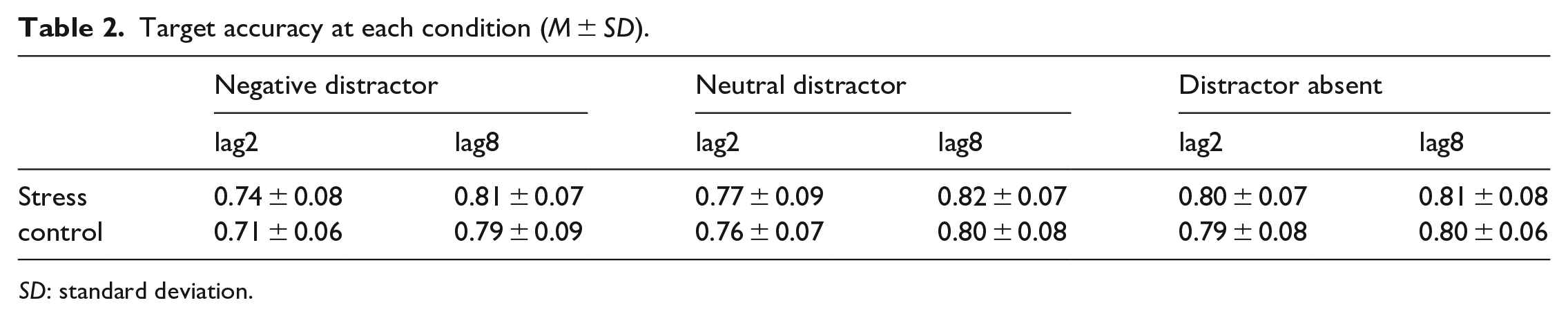
SD: standard deviation.
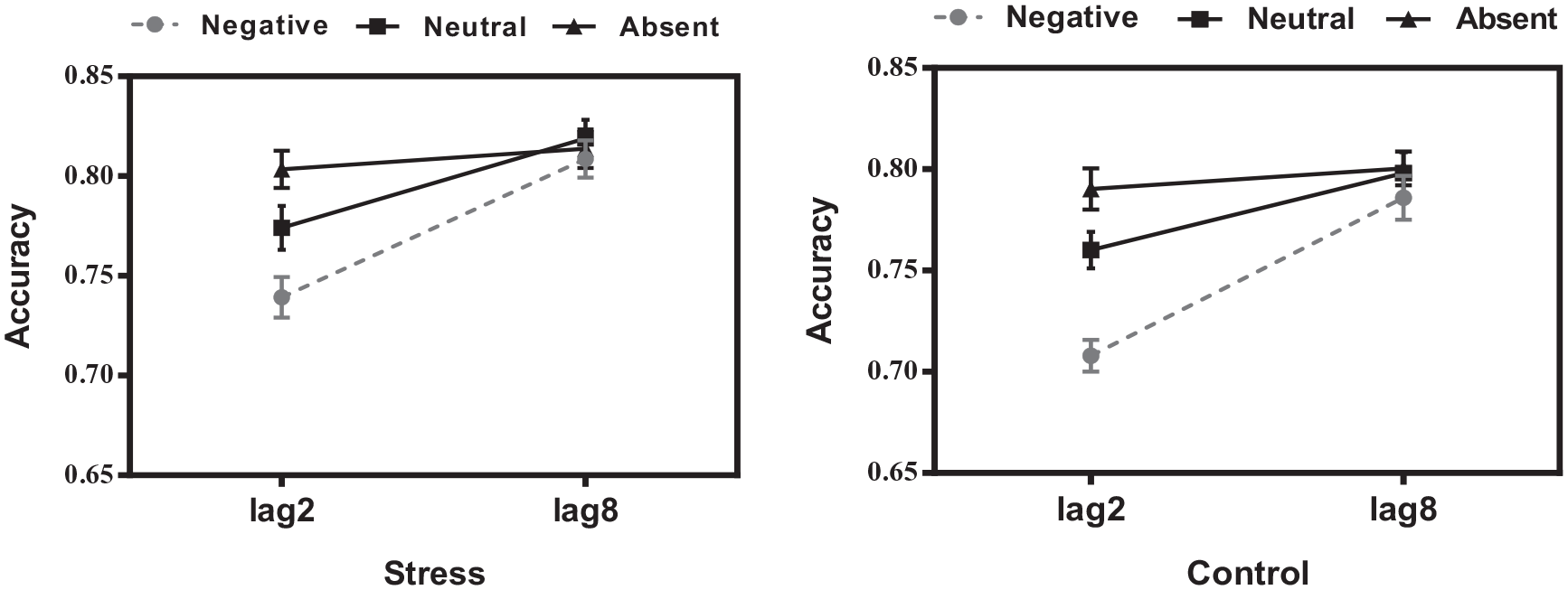
(Left) The target accuracy under stress treatment. (Right) The target accuracy under control treatment.
Control analysis
We conduct control analysis to eliminate important potential effect of repeated-measures designs.
Order effects
We included order (stress-control vs control-stress) as a between-subjects variable in the rmANOVA. Among them, 30 participants performed the stress task first and the remaining 31 subjects performed the control task first. The results show that the order main effect was also not significant, F(1, 59) = 0.252, p = .617,
Practice effects
We ignored the treatment variable and included a session (the first RSVP task vs the second RSVP task) as a within-subjects variable to perform the rmANOVA. The session main effect was not significant, F(1, 60) = 0.983, p = .326,
Role of cortisol and resting RSA on stress-induced RSVP performance
Pearson correlation analysis
We mainly focused on the correlation between stress reactivity-related physiological and biochemical responses, and stress-induced changes in EIB performance. Pearson correlation analysis results with 95% bootstrapped CIs suggested that the changes in cortisol concentration (ΔAUCg-cortisol) were positively correlated with stress-induced changes in target accuracy under negative condition at lag2, r = .362, 95% confidence interval (CI) = [0.126, 0.549], p = .016. Resting RSA was negatively correlated with stress-induced changes in target accuracy under negative condition at lag2, r = −.330, 95% CI = [−0.564, −0.095], p = .024. In addition, we also found a significant positive correlation between resting RSA and RSA reactivity, r = .585, 95% CI = [0.412, 0.720], p < .001, between resting RSA and the changes in heart rate (ΔAUCg-heart rate), r = .307, 95% CI = [0.059, 0.534], p = .026, and between RSA reactivity and the changes in heart rate (ΔAUCg-heart rate), r = .309, 95% CI = [0.067, 0.513], p = .026. These results demonstrated that high resting RSA was associated with the greater withdraw of vagus nerve and cardiovascular response during stress. Heart rate was negatively correlated with cortisol level over time, r = −.270, 95% CI = [−0.497, −0.027], p = .046. The results suggested the contrary effect between SAM and HPA axes in this study. The results were illustrated in Table 3 and Figure 5.
Pearson correlations of study variables.
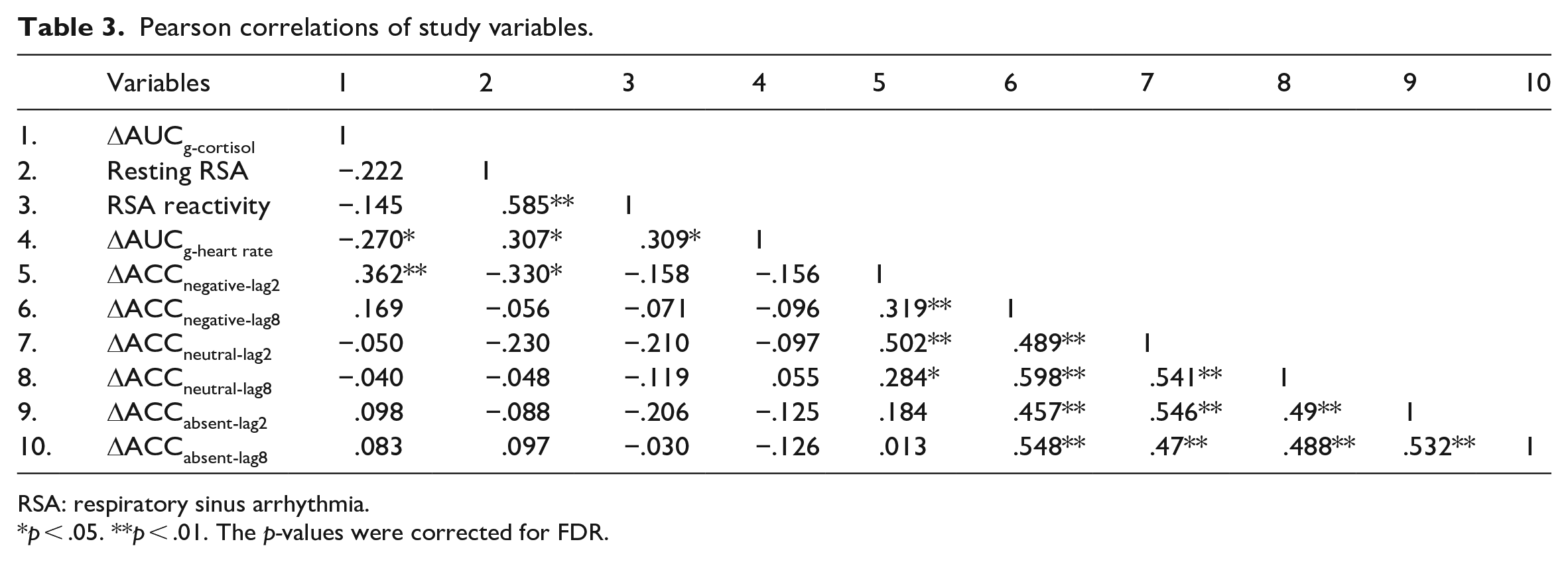
RSA: respiratory sinus arrhythmia.
p < .05. **p < .01. The p-values were corrected for FDR.
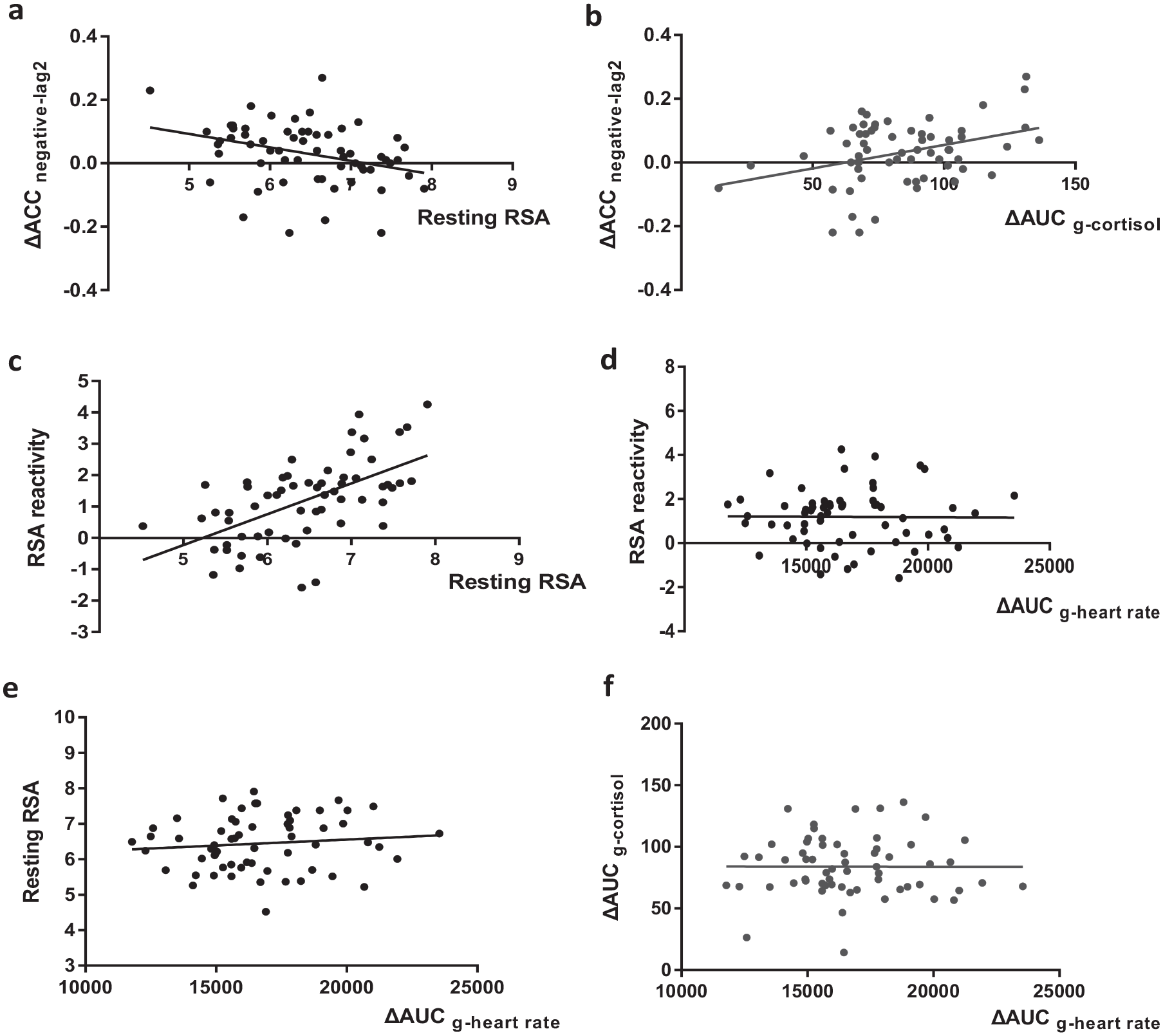
Scatter plots for correlation analysis. (a) Negative correlation between resting RSA and stress-induced changes in EIB accuracy under the negative distractors at lag2. (b) Positive correlation between ΔAUCg-cortisol and stress-induced changes in EIB accuracy under the negative distractors at lag2. (c) Positive correlation between resting RSA and RSA reactivity. (d) Positive correlation between ΔAUCg-heart rate and RSA reactivity. (e) Positive correlation between ΔAUCg-heart rate and resting RSA. (f) Negative correlation between ΔAUCg-heart rate and ΔAUCg-cortisol.
Hierarchical regression analysis
We performed a hierarchical regression analysis to investigate whether participants with high and low resting RSA levels exhibit differential performance in target processing during RSVP under different cortisol levels. The correlation analysis results were also referenced. Resting RSA and cortisol concentration were entered into the regression equation in the first step. Two-way interactions for cortisol × resting RSA were added in the second step. The results showed that the cortisol (p = .012) and resting RSA (p = .045) significantly predict the EIB performance. We did not find a significant interactive prediction between cortisol and resting RSA (p = .473). These results indicated that the resting RSA was not interacting with cortisol to predict the RSVP performances, but they could predict task performance separately. The results were shown in Table 4.
Hierarchical regression for stress-induced changes in target accuracy under negative condition at lag2: The predicting effects of resting RSA and cortisol.
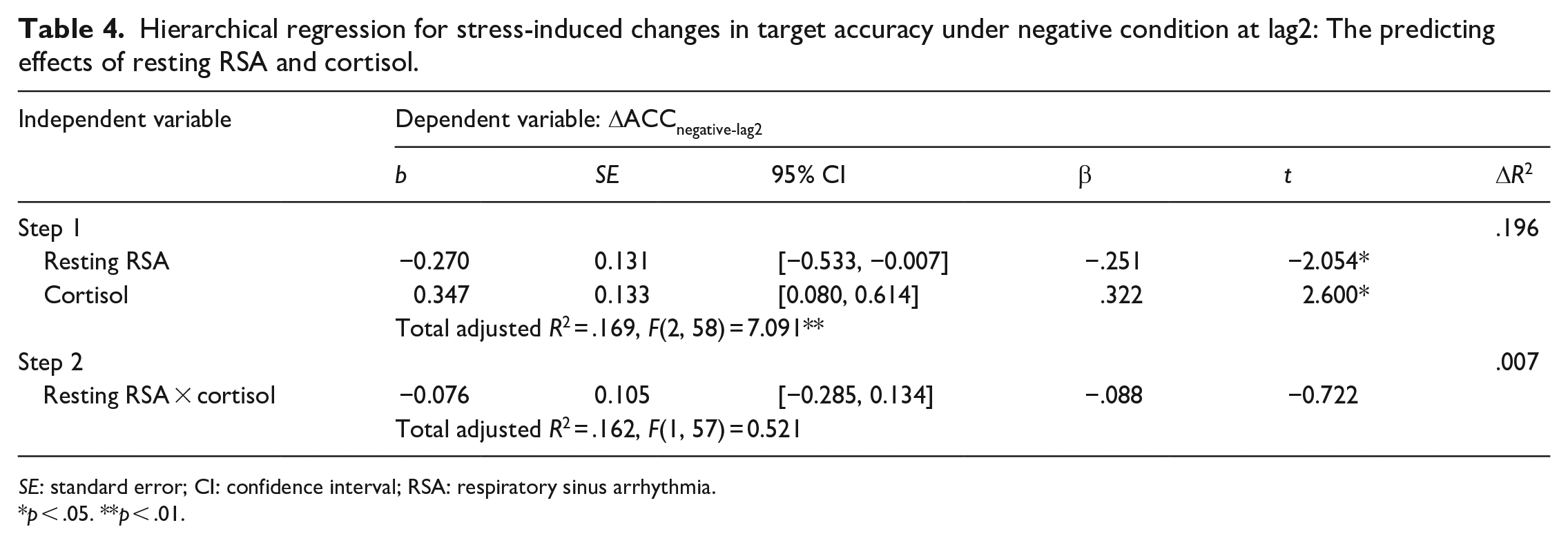
SE: standard error; CI: confidence interval; RSA: respiratory sinus arrhythmia.
p < .05. **p < .01.
Discussion
This study filled the gap in the field of acute stress affecting EIB and focused on resting RSA and cortisol levels in relation to stress-induced changes in EIB performance. The greater subjective negative emotional response and the higher heart rate and cortisol levels under the SECPT treatment indicated the effectiveness of stress induction. The results of this study revealed that stress reduced EIB overall, which may be associated with adaptive resource reallocation. Moreover, resting RSA and cortisol negatively and positively, respectively, predicted changes in the target accuracy induced by stress under the interference of negative distractors at lag2. Resting RSA did not interact with cortisol to predict task performance. This study provides a more comprehensive understanding of attentional blindness under acute stress.
The classical EIB effect
This study found the typical EIB effect consistent with numerous previous studies (e.g., Guilbert et al., 2020; Haddara et al., 2019; Kennedy et al., 2014; Macleod et al., 2017); that is, the negative critical distractors significantly impaired the target detection compared with the neutral and distractor-absent conditions. More severe blindness was also observed for the neutral and negative conditions compared with the distractor-absent condition. These results support the view that physical salience may determine the initial visual process and that emotionally arousing information plays a significant role in the late stage of fine processing, thereby inducing attentional blindness (Baker et al., 2021; Hoffman et al., 2020);). The occurrence of the EIB phenomenon illustrates that the task-relevant target detection (top-down) could not prevent the interference of emotional distractors. The rapid processing of emotional stimuli involves the activation of subcortical circuits in the amygdala (Méndez-Bértolo et al., 2016; Rafal et al., 2015). This circuit works in parallel with the striato-based visual pathway, which can encode information into the working memory, potentially breaking the temporal attention bottleneck for rapid processing, even when an individual is executing task-relevant target detection (Pourtois et al., 2013).
The overall effect of stress
This study found an overall improvement in the effect of acute stress on target detection in EIB, which conforms to the views of cue utilisation accounts (Callaway & Dembo, 1958; Lavie, 2005). Specifically, stress can further consume attentional resources and reduce the allocation of attentional resources to critical distractors. Attentional selectivity is promoted by stress when there are no available resources to process the task-irrelevant distractors, thus allowing attention to be more focused on the current target detection (Booth, 2019; Chajut & Algom, 2003; Hoskin et al., 2014). Moreover, this study revealed that acute stress could break through the temporal visual bottleneck and promote target detection under the interference of critical distractors, which is generally consistent with the blindness due to the trade-off between dual targets (Kan et al., 2019; Kan et al., 2021; Momin et al., 2020; Schwabe & Wolf, 2010). These results indicate that stress reduces the attentional blindness, regardless of whether top-down attentional processing is required for critical stimuli (people or animals). This may be because the prior critical stimuli, whether targets or not, take up almost the same amount of attentional resources, which may be sufficient to cause impaired detection of subsequent targets. Previous studies also suggested a similar cognitive processing mechanism of EIB and AB, namely, the attentional resources deficit leads to an insufficient encoding of information into the working memory (Macleod et al., 2017). In summary, this study extends previous studies on the effects of stress on attentional blindness, finding that stress reduces not only AB but also EIB.
Resting RSA negatively predicts stress-induced changes in EIB performance
This study demonstrated that resting RSA negatively predicted stress-induced EIB performance under negative distractor conditions at lag2. The high level of resting RSA is associated with less disturbed negative stimuli (Demaree et al., 2004; Thayer & Lane, 2000), indicating effective emotional regulation. The high resting RSA may be associated with better performance. However, it is important to note that not all emotional responses have negative effects on task performance. At the macro level, as the present findings showed that stress itself promotes target processing in EIB, the mitigative effect of individuals with high resting RSA is possibly associated with the reduced facilitation of stress on target detection in EIB.
Given that the RSVP paradigm involves stimuli competing for attentional resources over a very short period, we also interpret the negative prediction from the perspective of attentional resources. The available resources for targets are especially inadequate when the targets appear in the second position (lag2) after the critical distractors (McHugo et al., 2013). This was also reflected in our results that the target accuracy at lag2 was significantly lower than that at lag8, and that blindness recovered at lag8. People with high resting RSA are generally considered to play better the role of PNS for self-regulation (Butler et al., 2006; Geisler et al., 2013), and this process requires subjective efforts (Segerstrom & Nes, 2007). Particularly, under acute stress, people with high resting RSA may be more likely to regulate their emotions to avoid more severe imbalances in homeostasis. Therefore, they need to reserve additional resources for emotion regulation (Pu et al., 2010; Spangler et al., 2015). This self-emotion regulation impairs task performance by further consuming limited resources (Ortner et al., 2013). Although the limited resources were more concentrated under stress (Booth, 2019; Chajut & Algom, 2003; Hoskin et al., 2014; Sato et al., 2012; Tiferet-Dweck et al., 2016), we suggest that individuals with high resting RSA may be inclined to reserve partial task-relevant resources to support the extra self-emotion regulation process, which might be associated with poorer task performance. Furthermore, we believe that the negative distractor is also a type of “stressor” that requires active regulation on the part of the participants. Thus, this negative prediction was more significant under the negative distractor condition.
Cortisol positively predicts stress-induced changes in EIB performance
We believed that cortisol partially contributed to the influence of stress on EIB because of the positive prediction specific to the negative distractor condition. At first glance, the results suggest a contradiction with the conventional idea that stress boosts bottom-up emotional stimuli processing and attenuates up-down target detection. This surge in vigilance under stress is generally thought to be mediated by the release of norepinephrine by the activation of the SAM axis (Aston-Jones & Cohen, 2005; Cousijn et al., 2010; Valentino & Van Bockstaele, 2008). Previous studies have shown that the beta-adrenergic receptor blocker can inhibit SAM axis activity, thereby increasing cortisol levels and reducing the interference of emotional distractors (Oei et al., 2009). In this study, heart rate, as a cardiovascular activity indicator of the SAM axis, was negatively correlated with the cortisol released in response to stress, which suggests that there may be a similar inhibitory function of cortisol on emotional distractors. Several previous studies have also concluded that individuals with high cortisol levels pay less attention to task-irrelevant emotional distractors and are less disturbed (Oei et al., 2012; Putman et al., 2007, 2010; Putman & Roelofs, 2011). Thus, the positive prediction between cortisol and stress-induced improvement of EIB performance was related to the negative distractor condition.
Resting RSA and cortisol do not interact to predict stress-induced changes in EIB performance
Contrary to our original hypothesis, in this study, resting RSA did not interact with cortisol levels to modulate stress-induced changes in EIB performance. These results may suggest a dissociation of influence between state-dependent cortisol levels and trait ability reflected by resting RSA, most likely because the regulation of vagus on PNS activity is mainly reflected in the changes in cardiovascular responses to the stressor. Specifically, there was a positive correlation between resting RSA and RSA reactivity, indicating that individuals with high resting RSA were related to a greater withdrawal of vagus in response to the stressor, which was consistent with the polyvagus theory (Porges, 2001). This demonstrated that the regulation of stress in individuals with high resting-state RSA is mainly related to the changes in cardiovascular activity but not to the effect of cortisol levels. The positive correlation between resting RSA and heart rate also further supported the above findings. Furthermore, Kan et al. (2019) found that the cumulative effect of stress on promoting target processing in AB mainly occurred at the stage when cortisol took effect. Although we have not measured the saliva-alpha amylase to reflect the activity of the SAM axis, the results that heart rate did not correlate with stress-induced changes in EIB performance also partially supported the important role of cortisol. In general, resting RSA and cortisol levels over time have different patterns to exert an effect on stress-induced changes in EIB performance.
Limitations and future directions
There are certain limitations that need to be emphasised. First, the relationship between stress-induced changes in EIB performance with resting RSA and cortisol just reached a moderate degree of correlation (.3 < r < .6). Given that this exploration is preliminary, we consider the findings acceptable. Future studies need to more comprehensively enrich the interpretation of acute stress on attentional blindness. Besides, according to the polyvagal theory, RSA is generally inhibited during a stressed task but returns to its baseline state immediately after the task (Porges, 2001). Future studies can investigate the relation between state-dependent changes in cardiovascular activity (including baseline, response, and recovery periods) and attentional processing, according to the cardiovascular response model of stress. Moreover, the accuracy as an evaluation indicator to reflect the cognitive processing stage in EIB is limited. The N2, P3, and their regulatory component (such as early posterior negativity and late positive potential) representing the selective attention and the working memory encoding process could help comprehensively understand the stimuli processing during the capacity-limited stage in EIB (Kennedy et al., 2014; Macleod et al., 2017). Therefore, future studies can further combine ERP measures to verify the interpretation of the results involved in stage characteristics in this study.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Social Science Fund of China (BHA180160) and the National Natural Science Foundation of China (Grant No. 32071078).
