Abstract
Accessible and inclusive informed consent processes are crucial for ethical clinical research, yet little is known about how research staff implement these practices in Australia. This study aimed to explore current practice and perspectives of staff involved in informed consent processes for clinical research in Australia, including perceived barriers and enablers to facilitating accessible informed consent. To address this aim, we conducted a convergent parallel, mixed-methods study involving an online survey and semi-structured interviews (August to December 2024). Quantitative data were reported descriptively, while qualitative data were analysed using content analysis, with barriers and facilitators mapped to the Theoretical Domains Framework. Findings show that staff are motivated to employ a range of strategies to support their informed consent practice. The most frequently perceived facilitator to practice was providing information in varied formats, including the availability of video, audio, and language translations. However, there are challenges in offering information in multiple accessible formats due to limited skills, capability and resources (e.g. time, funding) to produce them. Industry leaders, regulators, funders and research organisations must lead and resource the delivery of accessible informed consent with focussed research staff training, funding accessibility, simplifying materials, promoting alternative formats, updating ethics guidance and convening shared-learning collaboratives with research participants to build sector confidence and ensure equitable, empowered consent.
Keywords
Introduction
Informed consent is a foundational ethical requirement for clinical research originating from the Nuremberg Code and the Declaration of Helsinki (Bierer, 2025). Recent updates to global and local standards, including the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use – Good Clinical Practice (ICH-GCP, 2025) and Australia’s National Statement (2025) have emphasised the need for greater accessibility and inclusion in informed consent processes for a wider and more diverse population (example Section 4, page 70). Many countries, including the UK and Australia, are adopting more inclusive consent documents, indicating a shift towards a recipient centred approach to engaging participants in research (O’Sullivan et al., 2022; Ong et al., 2024; Taylor et al., 2021). Equally, support for collaborative research designs with the priority community (both co-design and participatory), involving consent strategy design and implementation of processes, is growing (Slattery et al., 2020; Wiles et al., 2022). Resources such as the ‘Guidelines for Tailoring the Informed Consent Process in Clinical Studies’ (I-Consent guidelines) have emerged that aim to support researchers in adopting accessible and inclusive informed consent processes (Fons-Martinez et al., 2022). This shift is occurring to ensure more equitable research participation and to improve the quality and applicability of research outcomes. As a result, it necessitates a re-evaluation of current practices and identification of strategies to implement these evolving standards effectively.
Defining accessibility, inclusion and equity in informed consent
The principles of accessibility, inclusion and equity are interconnected and underpin ethical, effective informed consent practices (Beauchamp and Childress, 2019; Tan, 2019). ‘Accessibility’ involves presenting information and communications so that all potential participants, irrespective of ability or background, can access, understand and meaningfully engage with the informed-consent process (Rios et al., 2016). This requires adapting both format and delivery to meet diverse needs, including language, literacy levels, sensory abilities, cognitive processing, cultural factors and compatibility with assistive technology (Pandya-Wood et al., 2017). ‘Accessible’ is used to refer to materials themselves (e.g. videos, plain language summaries, translations) and ‘accessibility’ when referring to the broader concept, evaluation, policy or system to enable access. ‘Inclusion’ focuses on creating a process where participant’s feel respected, supported and valued. It recognises and accommodates diverse backgrounds and perspectives and helps individuals feel empowered to participate (Fortune et al., 2021). Key to both accessibility and inclusion is ‘equity’ which promotes fairness by tailoring the information exchange to each participant’s needs and where required, providing additional support to ensure everyone has a fair opportunity to understand and engage (Tan, 2019). Understanding how research staff navigate these principles in practice is crucial for improving informed consent processes in clinical research (Fortune et al., 2021; Griffith, 2022; Jawa et al., 2023).
Factors impacting comprehension and information accessibility
Accessible information and communication promotes agency and empowerment, yet evidence suggests that many participants struggle to understand consent materials. Studies in the UK, USA and Australia have consistently shown that Participant Information and Consent Forms (PICFs, also known as PICs) often require tertiary level education for comprehension (Flory and Emanuel, 2004; Nathe and Krakow, 2019; Nishimura et al., 2013; Symons et al., 2022; Symons and Davis, 2022; Tam et al., 2015). This high literacy demand poses a significant barrier to informed consent, particularly for diverse populations (Foe and Larson, 2016). Moreover, the length and complexity of these documents often deter participants from reading them thoroughly, with over 50% of participants reporting they do not read the information sheet in its entirety (Douglas et al., 2021; Geier et al., 2021; McNutt et al., 2008; Ozhan et al., 2014). Lengthy and complex PICFs correlate with reduced comprehension (Flory and Emanuel, 2004; Nathe and Krakow, 2019; Sharp, 2004), with participants preferring concise documents of no longer than four pages (Antoniou et al., 2011; Kirkby et al., 2012).
When faced with complex consent materials, participants may place greater reliance on the verbal explanation and other supplementary information provided by researchers. This tendency is particularly pronounced among participants with lower literacy levels (Manta et al., 2021). In such cases, a trusting relationship becomes crucial, as participants are less likely to ask fewer clarifying questions and more inclined to follow the perceived guidance of the researchers conducting the informed consent process (Manta et al., 2021). Clear, empathetic communication and the establishment of trust is required, as is the address of power imbalances during the informed consent process. Accessibility throughout the process can be strengthened by considering factors that affect communication and access to information such as literacy, language, culture, age and socioeconomic status (Shah et al., 2024).
Proposed interventions to improve information accessibility
Several interventions have been proposed to improve accessibility and comprehension of PICFs including simplifying and shortening content (Ong et al., 2024; Symons et al., 2022), using bullet points, larger fonts, diagrams and offering audio-visual formats (Gesualdo et al., 2021; Synnot et al., 2014). Participants value flexibility in how much information they consume and the ability to choose formats to suit their needs (Symons et al., 2022). Despite these advancements, questions remain about whether research staff possess the knowledge, skills, resources and capacity to implement accessible and inclusive practices. Institutional rigidity, time pressures and limited training will undermine these efforts unless research organisations address them. Implementation science offers tools to investigate these challenges, notably the Theoretical Domains Framework (TDF), which identifies determinants influencing practice change (Atkins et al., 2017; Michie et al., 2005). Although the TDF has been widely applied in healthcare (Shah et al., 2024), its use in clinical research, particularly around informed consent, is limited (Newlands et al., 2021). Applying TDF can highlight the key barriers and facilitators at individual and organisational levels, informing strategies to enhance accessibility and equity in consent processes (French et al., 2012).
Rationale and study aims
The increasing emphasis on accessibility and inclusion in research, coupled with limited evidence on how research staff navigate these expectations, necessitates a systematic exploration of contemporary informed consent practices. This study seeks to answer the question: How do clinical research staff in Australia perceive and implement accessible and inclusive informed consent processes, and what barriers and facilitators influence their practices?
This study aims to:
Identify perceived barriers and facilitators to participant information access,
Classify these barriers and facilitators using the TDF,
Explore staff approaches to accessibility, including adaptations and challenges,
Highlight practice gaps and recommend strategies to address these gaps.
This study, part of a broader participatory research project, aims to inform future training, resource development, and procedural reforms to strengthen ethical and equitable informed consent practices.
Methods
Study design
The study utilised a convergent parallel design, in which data were collected from both a survey and semi-structured interviews concurrently, analysed separately, and then merged for interpretation (Zhang and Creswell, 2013). In Part 1, both quantitative and qualitative data were collected via an online survey. The survey consisted of 22 multiple-choice questions (close-ended) and 6 open-ended questions (free text). The survey was open to research staff who conduct or facilitate informed consent processes, including investigators, study coordinators, nurses and research assistants. Staff working in the clinical research sector across Australia took part between August and December 2024 (see additional details in the ‘Survey Design’ section below). Part 2 was designed to deepen our understanding of the findings from Part 1 by exploring the experiences and attitudes of a subset of survey participants undergoing a semi-structured interview. By capturing both quantitative and qualitative data, the study aimed to integrate findings and enrich the outcomes obtained (Andrew and Halcomb, 2007; Zhang and Creswell, 2013).
The study received ethics and research governance approvals in May and July 2024 respectively (SVHM HREC 084/24; ID: 2024/PID00103). Throughout the study, consultations were held with experts in the clinical trials sector, leading disability advocates and community organisations, and individuals with varied lived experiences and areas of expertise. The reporting of this mixed-methods study has been guided by the Checklist for Reporting Results of Internet E-Surveys (CHERRIES; Eysenbach, 2004), Standards for Reporting Qualitative Research (SROR; O’Brien et al., 2014) and the Good Reporting of A Mixed Methods (GRAMM) Study Checklist (O’Cathain et al., 2008).
Participant recruitment
Purposive sampling was utilised to directly engage with Australian research and clinical trial institutes within our national networks (>80 sites across all states and territories). Sites were asked to alert their research staff using ethics-approved invitation materials, which provided details about the study and the electronic survey link containing the information sheet and consent form. Networks could distribute the information in varied ways, including email, newsletter, printable flyer and online posts. Snowball sampling was also used with permission for sites to send the study invitation to other networks and affiliated research services. Those willing to participate in Part 1 read an electronic information statement on the first page of the survey, which included a link to download the full study information sheet and consent form. Participants were advised that implied consent would be obtained if they agreed to proceed to the survey. At the end of the survey, details of the lead researcher were provided to register interest in Part 2. Participants who agreed and contacted the lead researcher for Part 2 were sent an invitation to participate. Nineteen of the 20 volunteers were interviewed. Interviews were conducted in sequential stages until data saturation was achieved. Data saturation was determined by the researcher completing interviews and using the principles of information power (Malterud et al., 2016).
Part 1: Survey data collection and analysis
Each participant completed an online survey containing questions related to demographics, training, the types of information sources provided to participants (i.e. verbal, written, video, website), steps and approaches taken, perceptions of factors potentially affecting their practice (including participant-related, self, environmental and process-related aspects), methods for assessing understanding, perceived barriers to adopting new information delivery formats, monitoring consent or willingness to participate, and suggestions for improvement of their practice (Supplemental File 1). The survey was informed by several sources: a previous UK-based informed consent research study and research staff survey (O’Sullivan et al., 2021), international best practice guidelines (Summarised in Supplemental File 2), and TDF domains (Atkins et al., 2017). It was developed by 3 researchers and 2 consumer representatives with lived experience of low vision and expertise in access and inclusion, reviewed by 10 research partners from community support or disability services, and piloted with a further 5 consumers (male and female, varying ages, varied backgrounds). The survey items were checked for accessibility and reading ease (Flesch reading ease: 72.92, Flesch-Kincaid Grade level: 6, indicating relatively low reading difficulty). The final survey was administered using Qualtrics software (Qualtrics, Provo, Utah). All surveys were completed anonymously. Upon completion of the survey, respondents were asked to contact the lead researcher directly (email and phone number provided) if they wished to participate in a semi-structured interview. No contact details of the survey respondents were collected or recorded.
Survey data were exported from Qualtrics to Microsoft Excel for cleaning. This data was then imported into Minitab version 21.4 data analysis software. Descriptive data summarised demographic-related questions using percentages representing the total sample. Where possible, Fisher’s exact test was used to examine associations between categories (
Part 2: Semi-structured interviews and analysis
Semi-structured interviews enabled the exploration of participants’ experience with informed consent processes, including perceived value to participants, skills needed, challenges and facilitators. Interviews were conducted between July and October 2024 via one-on-one Zoom teleconferencing. The duration of interviews ranged from 30 minutes to 1 hour and 15 minutes (median of 50 minutes). Each was then transcribed verbatim and de-identified before further analysis. Transcripts were not sent to interviewees for verification. The interviewer (FOH) is a clinician-researcher with 25 years of experience encompassing qualitative research methods and clinical trials. The interview guide was informed by our previous qualitative research (Johansen et al., 2024; Paynter et al., 2023), and the Theoretical Domains Framework (TDF; Atkins et al., 2017; French et al., 2012; Supplemental File 1). Two consumer representatives reviewed the interview questions for comprehension and phrasing and checked accessibility and reading ease scores. Eight research collaborators from four organisations reviewed the interview guide. There was flexibility in the order of the questions asked, depending on the flow of the conversation with each interviewee.
Qualitative content analysis
Qualitative content analysis was selected to analyse and interpret the textual content of our qualitative data from both open-ended survey responses and interview data. Each data set was analysed separately to identify patterns, concepts, themes, and with subsequent triangulation to interpret our findings with existing literature and theory (Bazeley, 2021). The lead researcher (FOH) led the qualitative data analysis, with supervision and collaboration with two other researchers (CP, TS), each with extensive experience in conducting interviews and evaluating qualitative data. This group undertook a modified Framework Analysis approach (Ramanadhan et al., 2021), and this was guided by the cohort’s homogeneity, as well as the lead researcher having implicit knowledge of the research environment and reflexivity. However, to mitigate risk of bias, the group participated in ongoing discussion throughout the analysis.
The Framework Analysis process involved five key steps. Step 1 was data familiarisation with the group reviewing the cleaned interview transcripts. Step 2 focussed on the group developing a coding scheme. The coding scheme involved pre-defined high-level categories that were driven by deductive analysis based on the underlying research aims and implementation theory (described in more detail below). Step 3 was indexing where all cleaned transcripts were imported into Nvivo software (QSR International Pty Ltd, Melbourne University licence) and the coding structure was applied to the entire data set. Data was reviewed for content and coded for correspondence with or exemplification of the identified categories. Under these categories, data excerpts were organised into subcategories following inductive coding principles (Bazeley, 2021; Elo and Kyngäs, 2008). Initial open coding was performed by the lead researcher followed by a second round of coding involving the other two analysts working collaboratively. Step 4 involved charting and organising data based on patterns across the data set. Step 5 involved mapping and interpretation where the group looked at the range and nature of relationships across and between codes (Gale et al., 2013). Data were systematically synthesised and displayed based on predetermined categories and concept subcategories.
During deductive coding, barriers and facilitators were categorised and mapped to the TDF domains (Atkins et al., 2017). Under these top-tier categories were second tier category descriptors or generic codes (linked to the TDF domain) and under these, third-tier subcategories. Data excerpts were organised under the subcategories which were open codes derived using an inductive content analysis approach and were framed around current practices, adaptations and modifications to informed consent practice that were described by research staff (Fereday and Muir-Cochrane, 2006). Data were triangulated across study parts to help identify practice gaps and interpret the findings (Bingham and Witkowsky, 2022). Findings were also interpreted with reference to implementation theory and existing literature.
Results
Participant demographics
One hundred forty-three people provided implied consent and proceeded to the online survey, with 95% (136/143) completing all multiple choice items. The mean age of the survey cohort (Part 1) was 43 years (SD = 10.3 years), and 76% were women. Most spoke English as their first language (86%) and worked in various settings (e.g. hospital and research settings). The cohort represented a variety of primary roles with Study Coordinators representing 27%, Principal Investigators 18%, Research Nurses 10% and the other roles including Sub-Investigators, Research Managers or Research Assistants. Nine respondents held multiple research roles. The number of years spent working in clinical research varied, with a good distribution across all categories for years of experience (range 2 to 21+ years; central tendency across 3–5 and 6–10 years categories). Most (66%) were involved in both interventional clinical trials and non-interventional studies. Diverse research interest areas were represented, including ophthalmology (30%), oncology (11%), infectious diseases (17%), cardiology (6%) and others. A subset of 19 participants from 20 volunteers (100% women) were interviewed for Part 2. After conducting 19 interviews, data saturation and information power were achieved.
Survey quantitative data (N = 136)
From the total sample (
Current accessible practices
Respondents were asked what information sources they offer participants as part of their usual informed consent practice. A third (38%,
Desired accessible practices: Perceived facilitators
We captured a clear gap between current practice and what respondents would like to offer if requisite resources were available. When asked what could improve accessibility in the process for research participants, 65% (
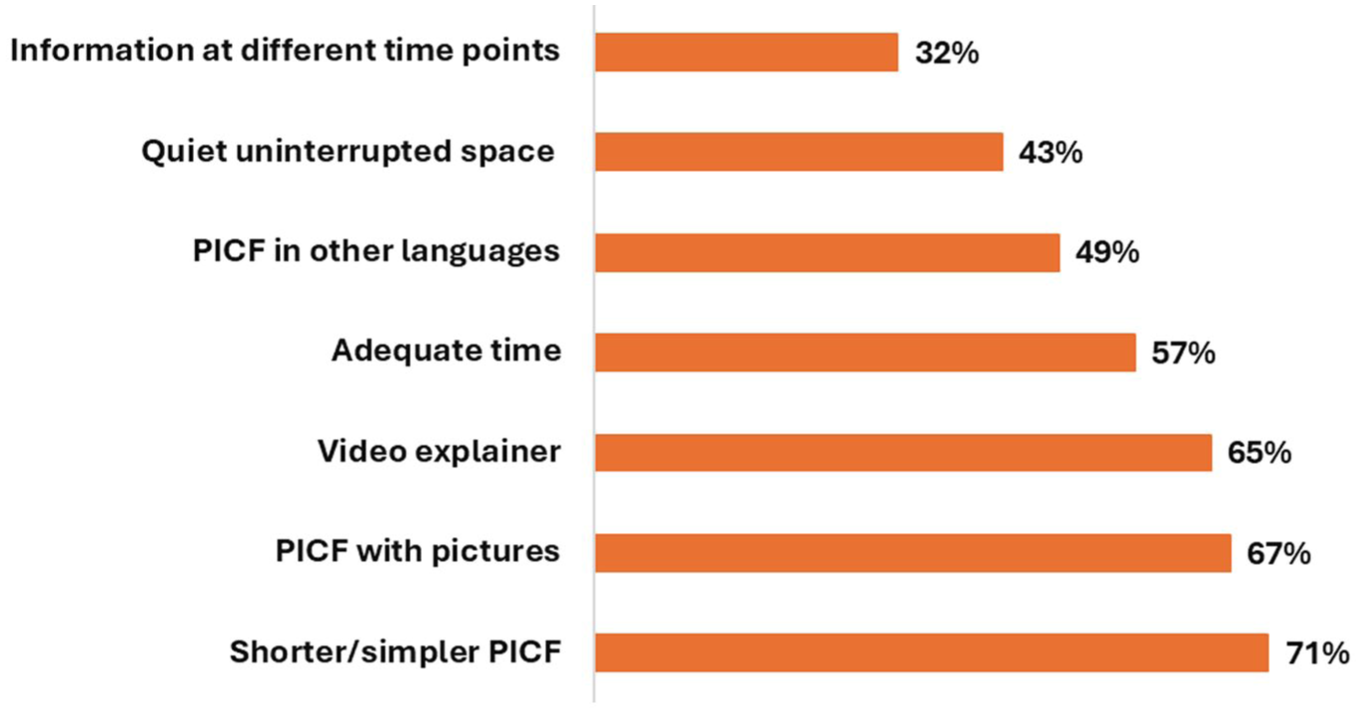
Leading facilitators to conducting an informed consent procedure from close-ended responses (
Current barriers
Respondents reported that diverse participant needs are difficult to meet when resources are limited. Challenges arose with unmet participant needs including language needs (e.g. not having access to language interpreters or translated materials; 43%,

Leading barriers to conducting an informed consent procedure from closed-ended responses (
Perceived barriers to adopting change to practices
While there is overwhelming support for improving accessibility in informed consent practice, the main hurdles perceived to implementing new information formats are rigid institutional policy and procedure guides (71%,
Qualitative data from the survey (n = 109) and semi-structured interviews (n = 19)
Interviews supplemented the open-ended survey data and enabled exploration of the factors that distinguish high-quality from low-quality informed consent processes. Insights helped to clarify and explain the reasons for the gap between current and desired accessible practices. Although the interview guide covered all TDF domains, responses clustered in five key domains: environmental context and resources, beliefs about capabilities, beliefs about consequences, skills and knowledge (see coding scheme presented in Table 1).
Coding scheme for qualitative data from open-ended survey responses (
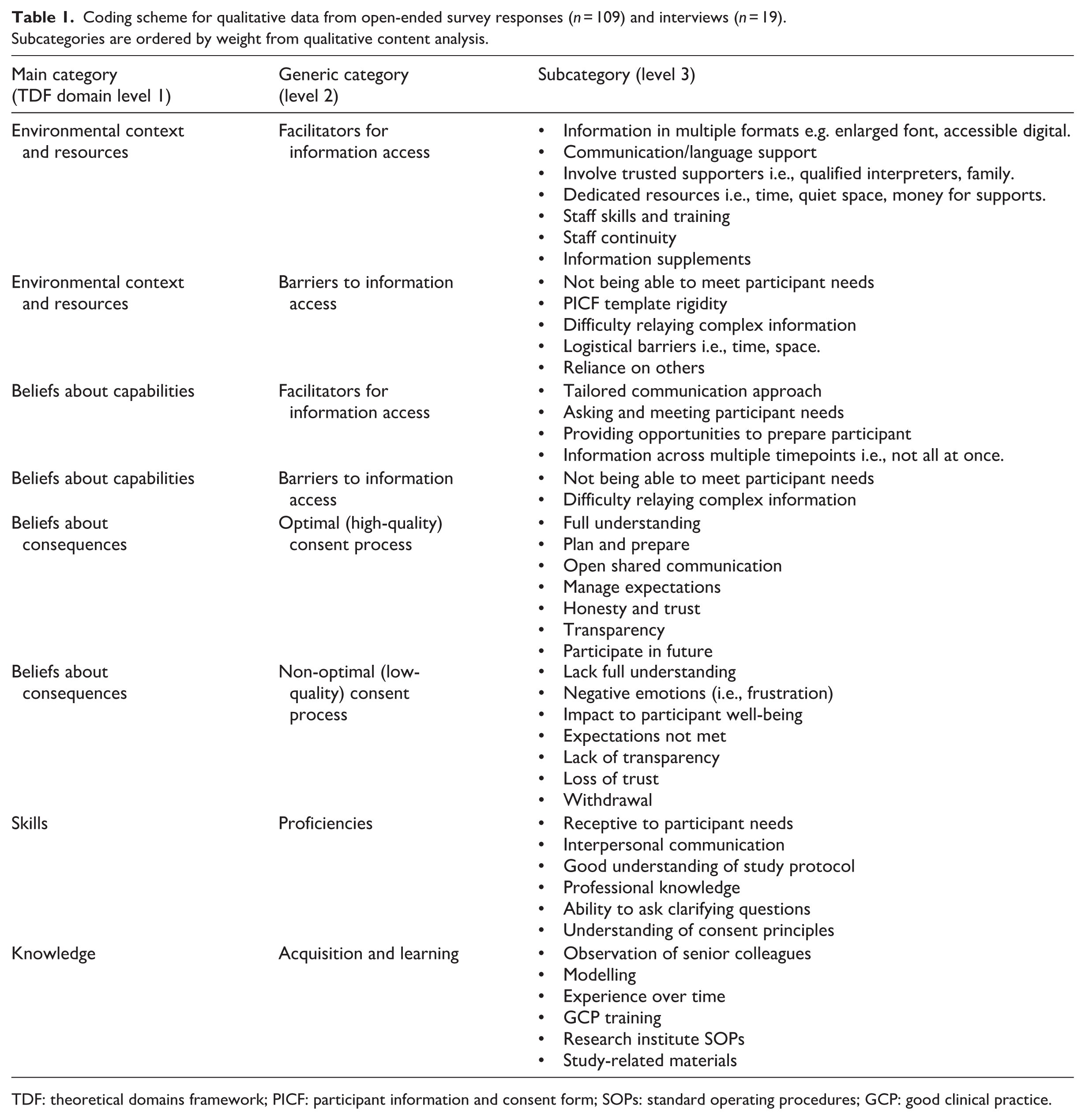
TDF: theoretical domains framework; PICF: participant information and consent form; SOPs: standard operating procedures; GCP: good clinical practice.
Environmental context and resources
Assimilating with quantitative survey data, barriers and facilitators within informed consent practice regarding environmental context and requisite resources were perceived at both an organisation level and an individual level respectively (Table 1). Research staff described a dynamic series of informed consent steps (Supplemental File 3). Steps are influenced by resources at hand and importantly, access to trusted supporters of the participant for language and communications support. Often informed consent processes involved multiple stages of communication and contact points with prospective research participants. Actions that align with best practice standards yet are not often accurately reflected in study protocols, particularly schedule of assessments.
Systemic and logistical challenges were highlighted, particularly when thinking about barriers to introducing practice changes. Research staff noted ‘systemic stuckness’ (survey P13) caused by academic and research constraints, resulting in a perceived lack of power to advocate for change. This lack of power was also influenced by research sponsors, ethics and governance bodies that stipulated PICF requirements. Access to varied accessible information templates, devices, technologies to share these formats, and communication supports enhance practice. However, not having access to requisite resources to be able to meet participant requirements hinders practice.
Beliefs about capabilities
Communication was a strong theme across both survey and interview data, with resonance around research staff’s beliefs about their capabilities and confidence. The ability to adapt communication methods strengthens over time, with some research staff admitting that they need more support to develop their capabilities. Reliance on trusted supporters to facilitate the informed consent practice is common, as is tailoring communications approaches, particularly when engaging with people who identify with a disability. Research staff noted that having an early conversation with participants about their needs, including how much information they require and in what format was highlighted as helpful. Beliefs about capabilities were also linked to the TDF domain’s skills and knowledge explained below.
Skills
Interviewees were asked to consider the skills required to deliver an informed consent procedure. The leading skill identified was the ability to be receptive to participant needs. Related, interpersonal communication skills were also highly cited as being critical to delivering an accessible informed consent process. Interviewees described scenarios and experiences that highlight the importance of emotional intelligence and receptive communication in facilitating an engaging informed consent process. They strongly conveyed the importance of establishing that the participant has understood the information shared with them. To facilitate this understanding, interviewees acknowledged that they employ several strategies, such as tailoring their language and communication style (reported by 13 of the 19 interviewees), encouraging questions, or asking participants to summarise the key information (talk-back method; interviewees P14, P16, P19). These findings were corroborated by open ended survey responses.
However, other interviewees (P2, P3, P4) acknowledged that this process could put participants on the spot and make them feel uncomfortable. The ability to discreetly check understanding was viewed as a specialised skill. Several interviewees suggested indirect methods including ‘tuning in to the participant’ and using their expertise in ‘reading the participant’ to confirm understanding (P1, P2, P3, P5, P13, P14, P16). This allowed reframing the information differently. Table 2 presents varied quotes about these skills, exemplified by this quote. P2:
Main categories with illustrative quotes from content analysis of semi-structured interviews (

Knowledge
Both survey and interview data highlighted six main ways research staff acquire knowledge to support their informed consent practice (Tables 1 and 2). The key learning area is ‘on-the-job’ experiential training with observing senior experienced colleagues and modelling favourable behaviours and approaches as core components. As interviewee P1 stated: ‘
Six interviewees noted that reflective practice was not merely a skill but a valuable means to identify improvements in individual practice. This involves being receptive to ongoing self-assessments, learning directly from participants/patients, and reflecting on could have been accomplished differently (noted by interviewee P2, P3, P12, P13, P15, P19). While many did not recall receiving formal informed consent training, interviewees frequently mentioned completing Good Clinical Practice training and exposure to Standard Operating Procedures at the commencement of their research role. Two out of the 19 people interviewed, and a small proportion from the wider survey cohort, mentioned additional sources of information to support their practice and address knowledge gaps in engaging with people with disabilities. For example, seeking information and guidance from established community support organisations.
Beliefs about consequences
The positive benefits of an optimal and accessible informed consent process are in enhancing the experience for participants in seven key areas (subcategories; Table 1). Interviewees consistently cited that a high-quality process enabled participants to gather a ‘full understanding’ of the requirements, evaluate their commitment, and determine whether they felt comfortable participating. The process also required the delivery of information in an engaging, open and transparent manner to ensure there were ‘no surprises’ for the participant. Building trust and communicating honestly were frequently noted (Table 2). In contrast, when asked about the consequences of a poorly delivered informed consent process interviewees expressed impacts to participants in directly opposing areas. These included a lack of understanding, experiencing negative emotions or having a negative impact to emotional well-being, participant expectations not met, loss of transparency around the study requirements, loss of relational trust, withdrawing from the study and also being reluctant to participate in future research (Table 2).
Synthesis of results to identify practice gaps and strategies
The integration of mixed methods data across Parts 1 and 2 of this study has highlighted four main gaps in informed consent practices (Table 3). First, there is a need for more focussed formal informed consent training, and second, this training should include effective communication. Third, there is a need to build capacity and capability in providing information in varied formats. This specifically pertains to investing the funds, time and skills required to produce engaging audio-visual materials and to build knowledge in providing information in other languages. Finally, there were procedural changes were identified to improve participant experience, particularly stakeholder support for simplifying the PICF, adopting flexible methods for obtaining and documenting consent (e.g. offering verbal, audio-recorded, implied and written), and standardising monitoring (e.g. scheduled repeat consent discussions and routine checks of understanding to reduce information overload).
Knowledge gap areas and strategies proposed to address these gaps, to improve informed consent practice, by research staff and implementation science models.
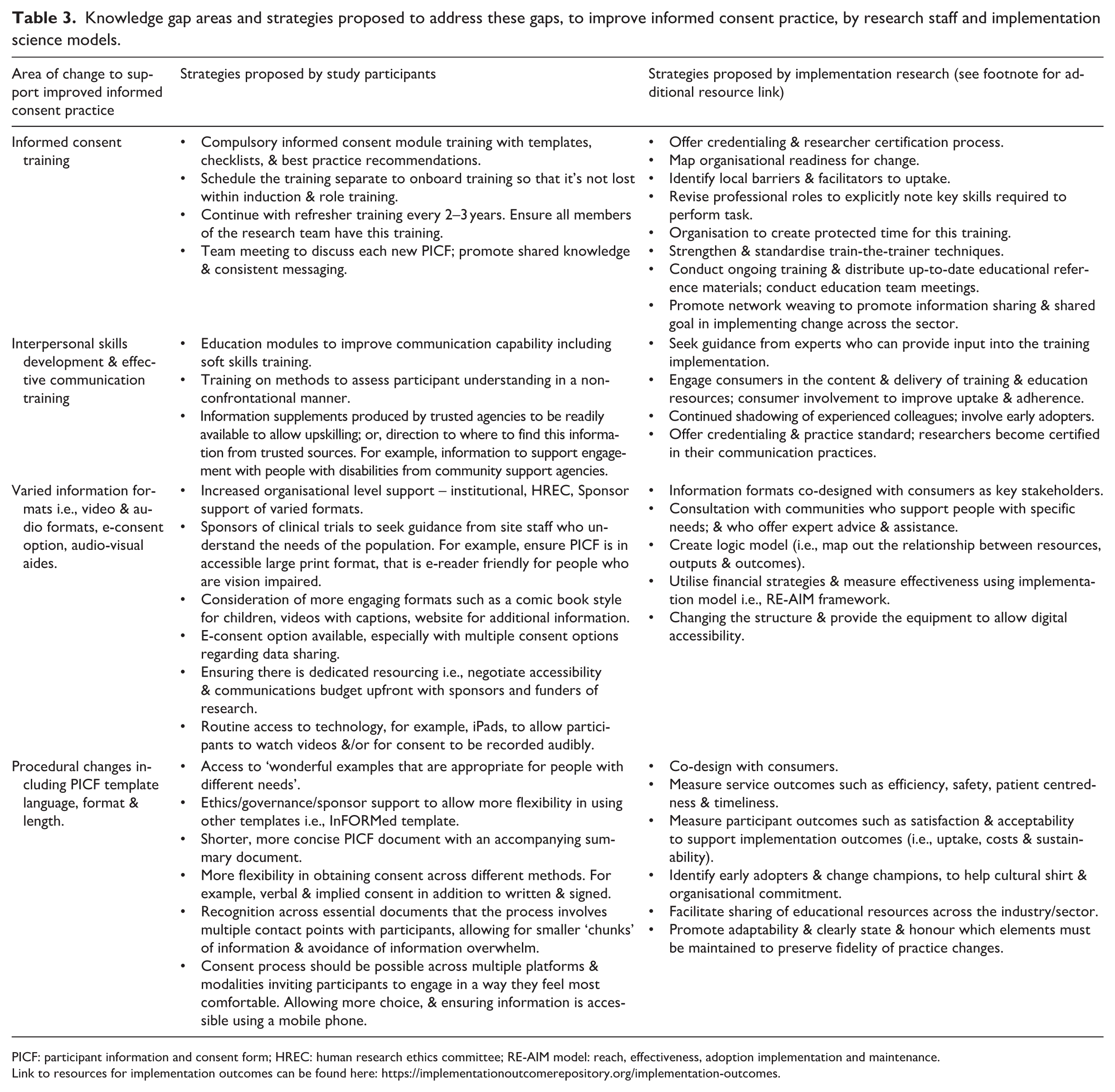
PICF: participant information and consent form; HREC: human research ethics committee; RE-AIM model: reach, effectiveness, adoption implementation and maintenance.
Link to resources for implementation outcomes can be found here: https://implementationoutcomerepository.org/implementation-outcomes.
Formal and focussed training on informed consent processes was widely acknowledged as necessary. It was recommended that training be staged separately from onboarding training (i.e. so that it is not lost in the induction process) and that it be repeated periodically. Indeed, Good Clinical Practice training is repeated every 3 years as an industry standard, yet recollection of specific informed consent components was rarely reported by interviewees. Regarding communication training, this was perceived as valuable for building the skills and knowledge to tailor and adapt communication styles to meet varied participant needs, including managing emotional situations, and addressing diverse literacy and learning needs. As P3 stated: ‘
The study cohort perceived information supplements as helpful for meeting differing information needs. This included offering an Easy-Read version, summary of the key points (‘quick reference’), video, audio formats, e-consent options and other audio-visual aides. The option of conveying information in video format, with captions as needed (i.e. for people with vision impairment or those with cognitive processing difficulties), was strongly supported. Yet whilst offering information in alternative formats was positively viewed, barriers to implementing them in routine practice were evident. For example, perceived lack of skills in producing videos, production costs, access to and cost of equipment to show videos, and perceived resistance from commercial sponsors of trials in providing resources to create and use them. Organisational, institutional and procedural changes are proposed by research staff in our study to address challenges with resource shortages and practice gaps. These and other strategies to address identified gaps in skills and knowledge by the study cohort are highlighted in Table 3. Additional strategies proposed by implementation science approaches are also shown in Table 3. The overall perceived benefits of these improvements aim to ensure that all participants experience a consistently high-quality and accessible process, with the opportunity to choose how they receive information and in what format, to suit their needs. Furthermore, it allows for flexibility in obtaining consent in varied ways.
Discussion
This mixed-methods study offers insights into the experiences and attitudes of research staff regarding accessible and inclusive informed consent processes. By applying the Theoretical Domains Framework (TDF), we identified five areas influencing practice, explored staff approaches to accessibility, and highlighted practice gaps. Four principal gaps in informed consent practice were identified. First, there is a clear need for more focussed, formal informed-consent training; second, that training must explicitly incorporate effective communication techniques. Third, capacity and capability to provide information in varied formats remain limited - notably the time, skills and resources required to produce engaging audio-visual materials and to deliver information in other languages. Finally, procedural aspects of the consent process, including PICF design and methods for obtaining consent, require reform to better support participant understanding and choice. Together, these findings point to the need for coordinated investment in training, accessible resources and procedural change to translate promising practices into routine, equitable consent processes.
Current practices and perceptions
Australian research staff generally report high confidence in their informed consent practices. However, this confidence primarily stems from ‘on the job’ training through observation of senior colleagues, rather than formal, structured training opportunities. Aligning with UK studies, our findings highlight research staff generally equate more complex studies with more complex participant information sheets, requiring longer consent discussions (O’Sullivan et al., 2021).
While staff express confidence, our analysis reveals significant barriers and facilitators that influence the quality and accessibility of informed consent practices. To better understand these factors, we applied the TDF to our data.
Barriers and facilitators (TDF analysis)
Using the TDF, we identified key domains influencing informed consent practices; skills, knowledge, beliefs about capabilities, beliefs about consequences and environmental context and resources. Staff are motivated to enhance their capabilities yet there’s a significant lack of formal training in communication strategies and accessibility practices. Generally, research staff found it difficult to communicate for diversity. Those who engage regularly with people who have diverse needs develop the knowledge and skills to adapt their informed consent practice to suit the necessities of their participants. However, this often relies on self-education and advice from experienced colleagues or community experts.
Notably, no survey respondent or interviewee mentioned using any communication assessment tool, with options available including Self-Efficacy of Communication skills (SE-12; Axboe et al., 2016). Around one-third of research staff interviewed indicated that they shape or adapt their communication approach directly by asking participants their preferences. Participants may not know what accessible options are available in that context; therefore, offering several accessible formats signifies an intention to provide a more inclusive experience. It may also create an opportunity for parties (i.e. researchers, research participants, their families and trusted supports) to feel more comfortable in discussing and planning for the most effective supports.
Strong interpersonal communication is viewed as a necessary skill. However, lack of formal communication training was reported, with skills often honed from observation of experienced colleagues and modelling these behaviours. In another Australian-led study, it has been highlighted that there is a lack of standardised training across both written and verbal aspects of informed consent procedures, and that access to interpersonal interaction training is limited (Wells et al., 2024). These findings are supported by McWhirter and Eckstein (2018), who also showed some research sites adopt strategies to address research staff training needs, such as scheduling weekly team debrief and discussion meetings, or, less frequently, conducting role plays to highlight optimal versus non-optimal consent interactions (McWhirter and Eckstein, 2018). They also reported that some research staff tailor their communication approach to participants’ motivations, health and digital literacy, and access to technology, noting that a ‘one size fits all’ approach is rarely effective. Our findings echo those made by these two Australian-based studies, specifically, the need for research staff to be trained in effective communication strategies.
Accessible consent: Evidence and implementation barriers
Results revealed low current use of videos, audio tools, explanatory diagrams, flipcharts and websites for supplementary information. Production of PICF documents in multiple languages is limited. Research staff are more likely to offer a summary pamphlet or a plain language supplement than multimedia or translated materials. The principal perceived barriers to providing multiple accessible formats are existing institutional policies and procedures, lack of training, and resistance from HRECs, governance offices and sponsors.
The literature supports the potential value of accessible formats. Concise multimedia (e.g. short videos, narrated slides and simple animations) and interactive tools generally improve trial understanding and participant satisfaction more than text alone, particularly when paired with opportunities for questions or comprehension checks (Flory and Emanuel, 2004; Nishimura et al., 2013). Pictorial aids, diagrams and plain language summaries reliably increase comprehension and recall (Houts et al., 2006). Electronic and dynamic consent platforms can standardise content, integrate multimedia and provide audit trails, but they raise governance, privacy and equity concerns that require careful mitigation (Kaye et al., 2015).
Our study also highlighted several systemic and organisational constraints: lack of standardised training, resource limitations, time constraints and complex documentation and inflexible consent models. These findings align with previous work showing that organisational factors restrict flexible informed consent strategies (Alemayehu et al., 2018; Djurisic et al., 2017; Godskesen et al., 2023; Hallinan et al., 2016). Implementation science explains why effective interventions are not always adopted: inner setting inertia (i.e. policies and SOPs), limited implementation capacity (e.g. skills, time, funding), stakeholder caution (i.e. HRECs and sponsors), and technology specific challenges (i.e. workflow fit, interoperability, digital literacy) are recurrent themes (Damschroder et al., 2009; Greenhalgh et al., 2017). Remaining evidence gaps include the effects of multiple accessible formats on decisional capacity and agency, and how to scale inclusive digital solutions without worsening inequities.
Evidence informed solutions
Drawing on barriers and facilitators identified through our TDF analysis, recommendations made by research staff and insights from implementation research (Powell et al., 2015), we propose the following recommendations for various stakeholders:
Research organisations should deliver structured training on communication and accessibility, allocate resources for accessible materials, develop reusable templates and tools, and foster shared learning.
Ethics committees should encourage alternative formats and provide clear guidelines on PICF simplification and flexible consent options. Notably ethics committees in Australia are increasingly requesting alternative information formats (examples: Bellberry Limited and Royal Children’s Hospital Melbourne).
Research sponsors (funders) should prioritise consumer and community involvement in research designs and mandate accessibility budgets.
Industry regulatory bodies could support informed consent competency and credentialing opportunities.
Research staff should pursue communication and informed consent education, use readability and accessibility tools, and share resources.
All stakeholders should form learning collaboratives to share best practice and involve participants in design and evaluation.
Practical, low-cost steps to build capacity include short workshops on plain language drafting and basic multimedia production, creation of a central repository of templates and media assets, pooling translation resources (e.g. professional translation with community review), and prioritising short plain language summaries that can serve as scripts for short videos and audio narrations. Production can be kept simple (e.g. smartphone, tripod, good lighting, clear script) with materials stored centrally and adapted for study specific detail. These recommendations adopt implementation strategies, encompassing targeted training, facilitation, stakeholder engagement, audit and feedback and tailoring (Powell et al., 2015), which can be mapped to the priority TDF domains to address organisational rigidity, time pressures and limited skills. Addressing local barriers is essential to translate good intentions into sustained practice change.
Strengths and limitations
The strengths of this study include a convergent parallel research design that combined mixed-methods data from a national survey and semi-structured interviews. The study applied implementation theory in the assessment of barriers and facilitators, generating evidence around the main pressure points within informed consent practice. Equally, this theoretical approach allowed for tailoring of appropriate intervention strategies that might lead to improved practices. Another strength was using a previously validated survey, which we refined based on diverse consumer feedback and pilot testing. This process ensured face validity, comprehensive content coverage and clarity and conciseness of items to effectively capture intended responses. The chosen online platform was selected for its accessibility features. Additional strengths include the high number of respondents who completed all survey items, the high number of volunteers interviewed, and the outreach to sites across all states and territories. This success can be partly attributed to the various engagement strategies used to engage institutes and sites, such as presentations at staff meetings and personalised emails to key contacts at these sites. However, a limitation is that most of the respondents resided in Victoria (62%), which may limit the generalisability. Due to the nature of our recruitment approach, which relied on networks to facilitate recruitment, we are unable to determine the number of participants approached for our study. Finally, we adopted a range of strategies to ensure our consent process involved information across different mediums, and to ensure that survey participation was accessible.
Interestingly, we experienced challenges in obtaining ethical approval to use an alternative PICF template that featured simpler language and a more condensed legal section. Therefore, ironically, our own PICF was not as accessible in terms of language and format as we had hoped. Additionally, we cannot provide data on the acceptability of our implied consent process, PICF and survey platform.
Conclusions
This work provides insights into the conditions and challenges faced by research staff when striving to deliver accessible informed consent processes. A culture of inclusion is fostered by involving consumers early in the research planning phase and by implementing deliberate actions that meet diverse needs. Communication and information should be provided in multiple accessible formats that are both culturally and age appropriate and translated into a participants’ preferred languages. Industry leaders, regulatory bodies, funders and research organisations must lead and resource this change by mandating structured communication and informed consent training, funding accessibility budgets, providing simplified templates and alternative formats, strengthening ethics guidance and credentialing, and convening a shared-learning collaboratives that includes research participants. Coordinated leadership will build sector-wide confidence and competence, enabling researchers to adopt accessibility practices and strengthening equity in information access and empowered informed consent for research participants.
Supplemental Material
sj-pdf-1-rea-10.1177_17470161261427839 – Supplemental material for Accessible informed consent: A mixed methods study of Australian clinical research staff perspectives
Supplemental material, sj-pdf-1-rea-10.1177_17470161261427839 for Accessible informed consent: A mixed methods study of Australian clinical research staff perspectives by Fleur O’Hare, Camille Paynter, David Foran, Kelly Schulz, Myra B. McGuinness, Lauren Barina, Steven Y. C. Tong, Tanya Symons, Lauren N. Ayton and Tessa Saunders in Research Ethics
Supplemental Material
sj-pdf-2-rea-10.1177_17470161261427839 – Supplemental material for Accessible informed consent: A mixed methods study of Australian clinical research staff perspectives
Supplemental material, sj-pdf-2-rea-10.1177_17470161261427839 for Accessible informed consent: A mixed methods study of Australian clinical research staff perspectives by Fleur O’Hare, Camille Paynter, David Foran, Kelly Schulz, Myra B. McGuinness, Lauren Barina, Steven Y. C. Tong, Tanya Symons, Lauren N. Ayton and Tessa Saunders in Research Ethics
Supplemental Material
sj-pdf-3-rea-10.1177_17470161261427839 – Supplemental material for Accessible informed consent: A mixed methods study of Australian clinical research staff perspectives
Supplemental material, sj-pdf-3-rea-10.1177_17470161261427839 for Accessible informed consent: A mixed methods study of Australian clinical research staff perspectives by Fleur O’Hare, Camille Paynter, David Foran, Kelly Schulz, Myra B. McGuinness, Lauren Barina, Steven Y. C. Tong, Tanya Symons, Lauren N. Ayton and Tessa Saunders in Research Ethics
Footnotes
Acknowledgements
Our deepest gratitude to the participants in this study for their willingness to share their valuable insights, lived experiences and expert knowledge. We would like to acknowledge the valuable contributions of the following organisations who assisted with engagement and recruitment for this study: Cerulea Clinical Trials, Vision Australia, Blind Citizens Australia, Staphylococcus Aureus Network Adaptive Platform (SNAP) Trial Management Team and clinical trial sites and Consumer Reference Group, the Centre For Accessibility Australia, Lions Vision Trials, Australian Clinical Trials Alliance, ARCS Australia, Next Sense, AccessCR, Information Alternatives, VCCC Alliance, Bellberry Limited, Peter MacCallum Cancer Centre, Save Sight Institute, Queensland Eye Institute and Australasian College of Emergency Medicine.
Ethical approval
Ethical approval was granted by St Vincents Hospital Melbourne Ethics Committee on 28 May 2024 (REF: 084/24). Amendment to the survey was approved on 23 July 2024. Research Governance approval was granted by the Centre for Eye Research Australia office on 31 July 2024. Participants in this study provided implied consent before proceeding to complete the survey, and verbal consent before participating in a semi-structured interview. A plain language summary of the project with links to a copy of the Participant Information and Consent Form was available. A link to the study website was also available which provided additional information about the study.
Author contributions
All authors contributed to the study conception and design. Methodology by Fleur O’Hare, Lauren Ayton, David Foran, Tanya Symons and Kelly Schulz. Data collection by Fleur O’Hare, Steve Tong and Lauren Barina. Data analysis by Fleur O’Hare, Tessa Saunders, Camille Paynter and Myra McGuiness. The first draft of the manuscript was written by Fleur O’Hare and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: No funding was received to assist with the preparation of this manuscript. Fleur O’Hare is supported by a scholarship from the Australian National Industry PhD Programme (#34984). Lauren Ayton is supported by a National Health and Medical Research Council Investigator Grant (GNT#1195713). The Centre for Eye Research Australia wishes to acknowledge the support of the Victorian Government through its Operational Infrastructure Support Programme (VIC, Australia).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
This research will made available in a public repository provided by the University of Melbourne. The research can be found here: https://figshare.unimelb.edu.au/ and ![]()
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
