Abstract
This paper seeks to address the challenges with applying a right to withdraw to pluripotent stem cell (PSC) research. PSC lines are unique in that they can be expanded indefinitely, disseminated globally, transformed into multiple derivatives, and employed as therapeutic products, rendering withdrawal not only logistically unfeasible, but also a substantive risk to research stability. Following an analysis of whether the classical right to withdraw can be suitably modified to address the tensions between donor freedoms and transformative biomedical research, we argue for a recalibration of donor control. Specifically, we present a hybrid model that combines up-front broad consent (including an expression of initial preferences for currently known ethically contentious areas of stem cell research), ongoing dynamic consent, and robust governance. While the paper reviews existing consent and withdrawal models, its primary contribution is to show why these models fail to adequately address the PSC-specific tension between respect for donor autonomy and provision of research certainty and to propose a hybrid, autonomy-preserving alternative that is designed to deliver greater research stability than a modified right to withdraw.
Introduction
Stem cell research sits at the forefront of biomedicine, offering avenues for basic research in biochemistry and biology, drug screening, toxicological studies, disease modeling, and cell therapy (Cerneckis et al., 2024; Kim et al., 2022; Kobold et al., 2023; Mahla, 2016; Moradi et al., 2019; Romito and Cobellis, 2016; Sayed et al., 2016; Shi et al., 2017; Shoji et al., 2024). While stem cell lines can be expanded indefinitely in vitro and have become crucial for in-depth cellular, developmental, pathological, and toxicological analyses, they also highlight a fundamental tension in research ethics in relation to the moral right of donors to withdraw after providing biological materials (Lewis and Holm, 2024; Ravn et al., 2023, 2024; Rosema et al., 2025).
Historically, withdrawal was formulated in contexts where participants faced direct physical and/or psychological risks, ensuring they could exit a study immediately, unconditionally, and without detriment or penalty. Yet, the situation radically changes once donors’ tissues are reprogramed into pluripotent stem cell (PSC) lines.
Unlike conventional immortalized lines, PSCs—including embryonic stem cells and induced pluripotent stem cells—are inherently capable of self-renewal and can differentiate into any cell type. This potency opens up an even broader range of applications, from organoid development to patient-specific regenerative therapies (Cerneckis et al., 2024; Kim et al., 2022; Shoji et al., 2024; Wobus and Boheler, 2005). Once a PSC line is established, it may be distributed to various labs or commercial entities, each refining culture conditions or genetically modifying the cells to address different research questions. Over time, the chain of derivation expands, leading to advanced organoids (including neural and gonadal organoids), stem cell-based embryo models, or partial transplants in animal models (Shoji et al., 2024). Organoids are often considered prime examples of advanced downstream derivation from PSC lines (Bartfeld and Clevers, 2017). A single donation can spawn numerous organoids across multiple labs. These organoids can be repurposed for drug screening, toxicity evaluations, partial integration with animal or synthetic systems, or even for clinical trials (Moradi et al., 2019; Shoji et al., 2024; Vogt et al., 2025).
If a classical withdrawal right were exercised strictly within such an extended derivation chain context, it would imply tracing, recalling, and destroying cell lines, associated data, and any derivatives that had already been widely shared and embedded in research projects and clinical applications across multiple jurisdictions. In short, unraveling entire collaborative networks and downstream derivation chains. The right to withdraw has, thus, been argued to be problematic in this context because it: (a) is not practically possible to fulfill; and (b) creates significant uncertainty for stem cell researchers (Lewis and Holm, 2024; see also Rosema et al., 2025). 1
Although conventional biobanks often have guidelines and standards stipulating how and when donors may withdraw, PSC research frequently occupies a regulatory gray area (Moradi et al., 2019). Although European Commission and European Union regulations regarding standards of informed consent for the donation and use of human tissue do not explicitly cover research-intended uses outside of a clinical trial setting, many jurisdictions in the EU/EEA focus on the initial consent required to derive or use a PSC line, but not on possible expansions, sub-lines, or specialized derivatives like organoids or stem cell-based embryo models (Lewis and Holm, 2024). By contrast, some guidelines specify that once tissue has been transformed or integrated into a cell line, donors no longer retain a straightforward right to withdraw. For instance, the UK Human Tissue Authority’s Code of Practice (HTA) (2020)—under which the UK Stem Cell Bank’s Code of Practice for the Use of Human Stem Cell Lines (2010) falls—suggests that the withdrawal of consent prohibits researchers from deriving any more stem cell lines from the remaining donated tissue (if any), but those stem cell lines that have already been derived, including any data and information associated with them, do not have to be recalled or destroyed. 2 In addition, across the EU, references to data protection regulations highlight that data already gathered remains valid for scientific usage, even if the research subject withdraws from future studies (see European Union Regulation (EU) 536/2014).
By shifting attention toward the pluralistic and open-ended potential of PSC-based research, and by examining the ways in which this research recalibrates the normative considerations that have tended to underpin debates about withdrawal, we extend the analysis of the moral right to withdraw in several ways. First, we situate withdrawal within a broader framework of extended derivation chains, thereby articulating the central normative tension on which the question of a right to withdrawal in stem cell research turns; namely, the tension between respecting donor autonomy and providing a level of certainty for stem cell researchers operating in large-scale, multi-jurisdictional collaborations (see also Rosema et al., 2025). Second, we articulate how functionally and structurally more advanced derivatives prompt deeper questions about the moral significance of donated biomaterial—particularly when these derivatives approximate distinct organ functions or approach morally contentious thresholds—issues previous withdrawal debates typically did not anticipate (Lewis and Holm, 2022, 2024). Third, we evaluate proposals for adapting or modifying the right to withdraw—from reason-giving requirements (Edwards, 2005; Eriksson and Helgesson, 2005), cooling-off periods, and prospective-only restrictions on future use (Holm, 2011) to complete waivers (Chwang, 2008)—arguing that no adaptation or modification is, in principle, capable of satisfactorily addressing the tension between respect for donor autonomy and support for research utility. 3 Indeed, we stress the near-impossibility of withdrawing cells once they have spawned multiple sub-lines, organoids, chimeric constructs, and other derived products (see also Lewis and Holm, 2024; Ravn et al., 2023, 2024).
This paper has three aims. First, it synthesizes a fragmented literature on withdrawal by organizing existing proposals (i.e. reason-giving, cooling-off periods, prospective-only withdrawal, and waiver models) around a single core tension that PSC research brings into sharp relief: how to respect donor autonomy while providing the stability and certainty required by multi-jurisdictional, resource-intensive research and downstream translation. 4 Second, it extends this debate by arguing that no modification of the classical withdrawal right can, even in principle, resolve that tension once PSC lines enter extended derivation chains. Third, to meet the practical realities of indefinite cell proliferation, constantly changing and increasing derivation pathways, and the potential deeper moral significance of stem-cell-based derivatives, we argue for a recalibration of the donor control that the classical right to withdraw functions to enable. Specifically, we propose a positive alternative to the right to withdraw: a hybrid model combining up-front broad consent and an elicitation of preferences for currently known ethically contentious areas of stem cell research with ongoing dynamic consent and structured governance (Figure 1). This shifts debates from the question of whether withdrawal can be saved to the question of what donor control should look like when withdrawal is not practically realizable.
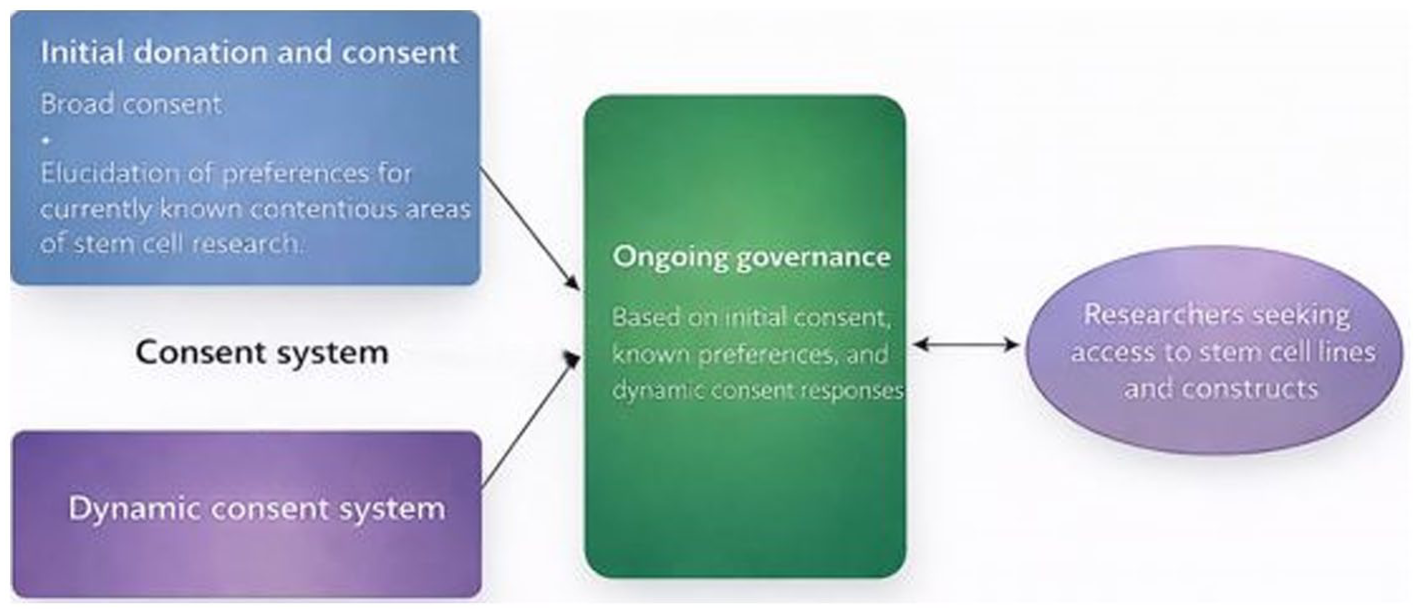
Proposed model for balancing donor autonomy and scientific utility in the absence of a right to withdraw.
The paper proceeds by: (i) clarifying the classical withdrawal right and why PSC research undermines its core assumptions (§1); (ii) evaluating proposed modifications to the classical right to withdraw and explaining why each fails to resolve the central tension between respect for donor autonomy and provision of research certainty (§2); and (iii) developing a hybrid model of donor control built around initial broad consent, ongoing dynamic consent, and governance as an alternative to withdrawal (§§3–4) with suggestions for future research (§5).
The classical right to withdraw: Core principles and challenges
The right to withdraw from research gained traction through ethics codes and legal instruments designed to protect participants in clinical trials from potential harm or exploitation. It was already enunciated in the Nuremberg Code’s Principle 9 (1947) and reached something close to its current form in the World Medical Association’s Declaration of Helsinki, originally published in 1964 and most recently amended in October 2024. The latter states that “the potential participant must be informed of the right to refuse to participate in the research or to withdraw consent to participate at any time without reprisal” (World Medical Association, 2024: Art. 26). Similarly, UNESCO, in its Universal Declaration on Bioethics and Human Rights (2005: Art. 6, s.2), states that “consent may be withdrawn by the person concerned at any time and for any reason without any disadvantage or prejudice.” Furthermore, the Council of Europe, in its Additional Protocol (2005: Art 13, s.3) to the Oviedo Convention (1997), asserts that “persons being asked to participate in a research project shall be informed of the rights and safeguards prescribed by law for their protection, and specifically of their right to refuse consent or to withdraw consent at any time without being subject to any form of discrimination, in particular regarding the right to medical care.” Thus, we see that the right to withdraw has typically been formulated around a few key principles (see also Eriksson and Helgesson, 2005; Helgesson and Johnsson, 2005; Holm, 2011):
In clinical contexts—such as drug trials or invasive procedures—these features make intuitive sense, ensuring that participants retain full control over bodily risk and can exit a study if side effects, discomfort, fear, or other motivating factors arise (Eriksson and Helgesson, 2005; Holm, 2011; Hug et al., 2012). In addition, non-waivability is key feature of the classical right because, as one of us previously argued, “if I cannot withdraw myself and my body from clinical research then I have bound myself in some kind of quasi-slavery” (Holm, 2006: 57–58).
With the storing of human biological samples for future research use, the notion of withdrawal becomes more complex (Eriksson and Helgesson, 2005; Helgesson and Johnsson, 2005; Holm, 2011; Hug et al., 2012). When donors contribute blood, tissue, or cells to biobanks, they often face minimal ongoing physical or immediate psychological risk. However, the storing of biomaterials engenders other kinds of risk. For example, how genetic or other personal data might be used over time. Thus, rather than the right to bodily integrity, the principle of respect for autonomy becomes, from a donor’s perspective, the primary normative consideration underpinning a right to withdrawal. However, it is unclear exactly how strong such a right will be. Unless we believe that autonomy always overrides other moral considerations, there will be instances where other normatively compelling factors require consideration (Holm, 2011: 272). For instance, physically recalling stored samples or halting ongoing analyses can be significantly more difficult, and give rise to more substantive normative considerations, than simply ceasing an active intervention on one’s body (Eriksson and Helgesson, 2005).
The classical right—encompassing immediate effect, unconditionality, and non-waivability—often proves practically unworkable once a cell line has been shipped to multiple research institutions or used to establish large data sets (see also Eriksson and Helgesson, 2005; Helgesson and Johnsson, 2005; Holm, 2011; Hug et al., 2012). Attempting to retrieve or destroy all instances of a widely shared cell line may be literally impossible (Lewis and Holm, 2024). Furthermore, it is not obvious that either respect for donor autonomy or a donor’s bodily integrity justifies a right to complete withdrawal if completeness is taken to include withdrawal of all data, all samples, and all derivatives collected and developed before the moment of withdrawal (see Holm, 2011). Thus, on the one hand, and unless donated biomaterial has been used outside the terms of the original consent, there seems to be an important distinction between having the right to withdraw the “basic” tissue and cells from future use (Holm, 2006, 2011), and withdrawing a stem cell line and associated derivatives that have been produced from the donated cells (Holm and Lewis, 2022).
On the other hand, as the chain of derivation gets increasingly longer, and the functional and structural properties of stem cell-based derivatives become increasingly complex and start to impinge on the thresholds of what some might consider to be ethically permissible (i.e. neural organoids, embryo models, reproductive organoids, or more complex and/or holistic somatic organoids), it is more likely that donors will imbue these derivatives with moral value for functional, genetic, and meaning-based reasons (Lewis and Holm, 2022). In other words, as empirical studies indicate, rather than relating to stem cell-based derivatives merely as tissue samples or immortalized cell lines, donors are likely to treat the boundary between derivative and body as increasingly blurred when those derivatives become progressively more mature and complex (Boers et al., 2018, 2019; Hyun, 2017), and when certain models, for example, recent stem cell-based embryo models, offer the possibility of more advanced development and thereby begin to challenge legal definitions and currently accepted regulatory standards (see, e.g. Lewis and Holm, 2025; Rivron et al., 2023).
This points toward a tension between categorizing and treating stem cell lines and their associated derivatives as “objective” biomaterial over which—we would expect—those who have willingly and in full knowledge donated tissue to have few downstream moral concerns, and responding to them as “hybrids” (Boers et al., 2019) whose symbolic, functional, and, ultimately, moral value is such that donors should be accorded a greater level of control over their use than we can expect a classical right of withdrawal to deliver (Lewis and Holm, 2022). And such a classical right cannot be expected to afford such control precisely because stem cell lines and their associated derivatives—despite potentially being imbued with moral significance—are, in part, biotechnological artifacts; that is, the products of complex technical interventions and manipulations and a substantive amount of resource investment.
From a normative perspective, the characterization of stem cell lines and derivatives as biotechnological artifacts undermines a donor’s claim to exclusive control through a putative right to withdraw because tracing, retrieving, and destroying widely shared PSC lines and PSC-based derivatives is practically unfeasible and because uncertainty surrounding the donor’s continuing power to recall can hamper the stable environment needed for large-scale research and the commercial development of stem cell-based products (Holm and Lewis, 2022; Lewis and Holm, 2024; Ravn et al., 2023, 2024; Rosema et al., 2025). If a donor can abruptly insist on the destruction of PSC lines and derivatives that have become a de facto standard for a particular field, then commercial or academic partners may forfeit significant investments of time and money. Stem cell researchers have expressed their concern that indefinite withdrawal options might sabotage the viability of translational pipelines designed for cell-based therapies, because such options would make any estimation of a future “return on investment” much more uncertain (Lewis and Holm, 2024; Ravn et al., 2023, 2024). Thus, bridging the divide between support for research utility and support for donor control (i.e. ensuring a level of respect for autonomy and the promotion of trust in biomedical research, see Eriksson and Helgesson, 2005; Helgesson and Johnsson, 2005) demands carefully calibrated frameworks that address the open-ended nature of stem cell applications while offering donors meaningful input and recourse if they oppose certain uses (for moral reasons or otherwise; Pamies et al., 2022; Ravn et al., 2024; Rosema et al., 2025).
Modifying the right to withdraw: Insufficient in and of itself
Several proposals have been offered for modifying the classical right to withdraw, that is, adapting one or more of its key conditions—immediacy, unconditionality, and non-waivability—typically to better suit the realities of biomaterial donation and biobanking (see Chwang, 2008; Edwards, 2005; Eriksson and Helgesson, 2005; Helgesson and Johnsson, 2005; Holm, 2011; Hug et al., 2012).
Edwards (2005), for instance, has argued against the unconditionality of the right to withdraw, claiming that a participant requesting withdrawal should provide an explanation given that it would be irrational for someone who had already exercised their autonomy on the question of donating their biomaterial to then withdraw. However, in making this claim, Edwards assumes that the participant is informed of the types of studies they will be involved in, that their decision to participate in those types of studies is voluntary, and that the background conditions against which the initial decision to participate was made have not changed.
Typically, stem cell research adopts a broad consent approach, which operates on a scale ranging from consent to certain types or classes of research and clinical applications within initial broadly defined boundaries to blanket (i.e. unrestricted) consent to any kind of use, be it research, clinical or commercial (see Hansson et al., 2006; Helgesson, 2012; Hofmann, 2009; Petrini, 2010; Steinsbekk and Solberg, 2015). This means that, depending on the terms of the initial consent, biobanks, researchers, and clinicians can use a donor’s biospecimens and data without obtaining new consent. One of the issues with applying not only the model of broad consent, but also Edwards’ argument to a stem cell research context that entails extended and often unpredictable derivation chains is that all future types or classes of research use, and their specific risks and ethical salience, cannot be fully known and thereby communicated to a donor at the time when broad consent is obtained (Lewis and Holm, 2022). In other words—as the history of stem cell research shows us—even if the types and classes of research use are broadly categorized and communicated to the participant at the time of initial consent, there is no guarantee that these broadly defined types and classes are exhaustive or that a donor won’t rationally revise their values and preferences over time in light of developments in stem cell research (for details, see Lewis and Holm, 2022). The issue here is not just that donors may be ignorant of the causal links or entailing relationships between their donation and all possible stem cell-based research activities; it could also be that the technical and functional complexity of stem cell-based derivatives increases beyond the scope of what could be anticipated at the time of initial broad consent, for instance, with derivatives recapitulating specific parts of a donor’s body or biologically-cum-conceptually approaching morally contentious thresholds (e.g. “human sentience” in the case of neural organoids or chimeric models or “human embryo” in the case of stem cell-based embryo models). If we accept that certain stem cell-based derivatives—particularly neural and gonadal organoids, stem cell-based embryo models and chimeric models—can have moral value because of their “hybridity,” and if we accept that the moral value of those derivatives is likely to increase as they become structurally and functionally more complex, then we cannot draw reliable inferences about the (ir)rationality of a donor’s request for withdrawal downstream of the initial donation.
Nevertheless, even if a donor is acting rationally and thereby making a genuine autonomous decision in choosing to withdraw, we might still think it reasonable to seek an explanation for their choice as well as inform the donor of the risks posed by their withdrawal to stem cell research (see, e.g. Holm, 2011). In this vein, Eriksson and Helgesson (2005) argue that those wishing to withdraw their samples from biobank research should present “sufficient reason” for doing so and that the sufficiency of such a reason should be judged by those with “genuine, deeply felt concerns that are not based on misconceptions.” On the one hand, such an approach sustains the fundamental option to withdraw whilst providing some certainty that the donor’s decision to withdraw is suitably rational and not ill-informed. On the other hand, it can only deliver the certainty required by stem cell researchers if the researchers themselves—or those designated to protect researchers’ interests—are the ones who judge whether the reasons given are sufficient. As one of us has previously argued (Holm, 2011), this seems naïve and/or dangerous. It is naïve to think that stem cell researchers—with a vested interest in promoting a climate of research certainty—will assess the rationality of a donor’s decision to withdraw with a sufficient degree of normative disinterest (as typically required by most reasonable accounts of ethical justification). In addition, such an approach is dangerous as it risks the use of power to potentially override genuinely autonomous decisions that are rationally grounded in substantive moral, religious, and/or cultural objections to certain stem cell-based research projects or derivatives. Furthermore, as argued elsewhere, it is unlikely that such paternalistic approaches to donor autonomy will do much to promote public trust in stem cell research—another factor on which the stability and continuity of stem cell research depend (Lewis and Holm, 2022).
An alternative challenge to the classical right to withdraw turns on the immediacy component. For example, one of us has previously suggested that if the goal of withdrawal is to stop researchers from using a donor’s samples, then a system could be devised where the donor must either reconfirm their decision to withdraw after a specified timeframe or indicate that they have changed their mind to withdraw (see Holm, 2011). This cooling-off framework respects donor autonomy and other potential donor rights in the sense that it allows donors to prohibit the use of their samples and associated data during the cooling-off period whilst also providing them with the option to fully withdraw (i.e. to have their biosamples and associated data recalled and/or destroyed) should they not change their mind during that period. Furthermore, such a framework may offer a degree of stability for stem cell research to the extent that the presence of a cooling-off period may promote genuinely autonomous decisions to withdraw and, by implication, mitigate the number of decisions that merely reflect transient donor mental states, external pressures, or misinformation. However, if a PSC line or stem cell-based derivative has already been widely distributed, even a structured waiting period may fail to prevent confusion over identifying and tracing the donor’s cells through the entire chain of derivation (Lewis and Holm, 2024). Indeed, it may be practically impossible for all donor-related PSC lines and derivatives thereof to be traced during the cooling-off period, especially if the timeframe is relatively short. In addition, in the case of PSC derivatives that have already been established, but the development of which the donor objects to, the cooling-off period may present a barrier to the donor’s control precisely because it is the existence of such entities—and not solely their continued use—that matters to the donor. More importantly, mere modifications to the immediacy condition cannot get round the problem that withdrawal of PSCs from a global chain of derivatives is not practically possible to fulfill. Furthermore, so long as full withdrawal is an option available to donors (i.e. at the end of the cooling-off period), stem cell science is still likely to face significant uncertainty.
To address this, we might consider modifying the right to withdraw to only allow for the prohibition of further expansions and derivations of their cell lines and the use of existing lines and derivatives in new research projects (Holm, 2011). This prospective withdrawal approach draws on the logic in data protection norms, where an individual may—by withdrawing consent—have their data deleted (assuming that the only lawful basis for continued processing is consent) but cannot revoke the lawfulness of data processing that took place before withdrawal (see, e.g. Regulation (EU) 2016/679; for a nuanced account of post-withdrawal data processing under Regulation (EU) 2016/679, see European Data Protection Board (EDPB), 2020: §§117–120). A similar principle might be applied to biological materials: donors, upon objecting to aspects of ongoing or anticipated stem cell research, might exercise the right to prohibit any new expansions, derivations, distributions, or research using their cell lines without destroying cell lines and PSC derivatives that have already been developed or ceasing research that is already under way or completed. However, forward-looking withdrawal does little for those donors who strongly oppose how their cell lines are currently being used, rather than how they might be used in the future. For instance, a donor who learns that their PSC-derived neural organoids have been combined with the brain of a monkey in a way they find deeply objectionable might consider that partial “forward-only” restrictions are inadequate. If labs are already conducting a study that goes against the donor’s moral beliefs, halting only future expansions may not address the donor’s sense of immediate wrongdoing or complicity in—what they see as—ethically impermissible research. This gap leaves donors who harbor urgent or severe objections dissatisfied and risks eroding public trust (Hug et al., 2012).
By establishing that only future expansions or derivative uses can be curtailed—rather than retroactively destroying or invalidating PSC lines and derivatives already in circulation—researchers gain a degree of stability for ongoing studies, investments, and collaborations. Once a donor initiates forward-looking withdrawal, labs know that any existing applications of the cell line are permitted to continue to completion, thus reducing the risk of abrupt terminations and severe disruptions mid-stream (Holm, 2011). This clarity may be especially attractive in large-scale, resource-intensive stem cell projects, where new funding or commercial partnerships frequently hinge on guaranteed access to the cell lines in question. Nonetheless, while this approach preserves existing work, it does not eliminate uncertainty. Researchers will be keenly aware that a donor may, at any time, impose partial restrictions that halt subsequent expansions or commercialization efforts, possibly undermining the viability of planned projects. This can be particularly consequential in translational contexts where stem cell lines or PSC derivatives are propagated with an eye toward clinical application or market authorization; even if ongoing studies can proceed, prospective bans on future modifications or sub-lines might deter investment or complicate regulatory pathways.
What we can infer from these different proposals for the modification of the classical right to withdraw is that so long as the modified right is still non-waivable, stem cell research is likely to face significant uncertainty.
It has been suggested that donors might contractually waive any future right to withdraw, thereby relinquishing control once, for instance, their cell line is derived (Chwang, 2008; Helgesson and Johnsson, 2005). This approach mirrors the logic behind other everyday agreements—such as non-refundable purchases or binding service contracts—where individuals voluntarily surrender certain rights in exchange for clarity, stability, or other perceived benefits. In the stem cell context, permanently ceding the right to withdraw could eliminate the persistent uncertainty that troubles both researchers and commercial partners. By guaranteeing that no abrupt recall can occur, research institutions and funders gain confidence to invest resources in developing new lines, derivatives, and therapeutic products. Nonetheless, several ethical and practical concerns arise. First, an unconditional waiver may leave donors exposed to unforeseen research directions that contravene their deeply held moral, religious, or cultural values, desires, and motivations. Given the unpredictability of cutting-edge stem cell research, it is impossible to anticipate all possible future uses. Cell lines might be integrated into morally contentious experiments, such as creating advanced neural organoids, stem cell-based embryo models, or interspecies chimeras. Donors who discover these applications only later might wish to withdraw, but under a full waiver, they lack any mechanism to intervene (Holm, 2011). Second, a permanent surrender of rights potentially magnifies power imbalances in research, particularly if donors come from marginalized communities with limited bargaining power (Ravn et al., 2023). When participants are pressured—either directly or by broader socioeconomic factors—into signing waivers for the sake of medical care, financial incentives, or perceived altruistic obligations, the voluntariness of their consent is questionable. There is also a debate about whether waivers of this nature genuinely align with the principle of autonomy. While some note that consenting adults may legitimately alienate certain rights if it serves their interests or moral commitments (Helgesson and Johnsson, 2005; Manson and O’Neill, 2007), we have previously argued that donors cannot fully comprehend how their cell lines might be used—meaning that it is practically and conceptually impossible to obtain genuinely informed, up-front consent to blanket or broad range of research uses (Lewis and Holm, 2022). As a result, a waiver-based model would come at the considerable cost of respecting donor autonomy, which, by its dynamic, iterative and relational nature, entails the ability to revisit and potentially modify decisions in light of new information and/or changing values (ibid).
Protecting donors’ interests in the absence of a right to withdrawal – Multilayered consent
As already discussed, the normative challenge with stem cell research is finding a way to honor donors’ genuine freedom of choice—allowing them to make binding decisions—about how their cell lines should be used whilst providing researchers with a high level of certainty and administrative processes related to donor control that are feasible to implement.
We’ve already seen that the best way to promote research utility, stability, and certainty in stem cell science is to either deny a right to withdraw or have donors waive such a right once cell lines have been derived from their donated biomaterials. The question, then, is whether we can abandon the notion of withdrawal and still preserve the sort of donor control that the classical right to withdraw functioned to enable?
When previously analyzing the limits and limitations of consent frameworks in stem cell and organoid research contexts, we emphasized the iterative, relational, context-sensitive, and value-laden dimensions of donor decision-making throughout stem cell and organoid research (Lewis and Holm, 2022). Specifically, we argued for the notion of “authentic choice”—an account of autonomy in which genuine self-governance in an open-ended, dynamic stem cell research context depends not merely on initial competence, voluntariness, and information disclosure, but also on a donor’s evolving capacity to reflect on and potentially revise their values over time in light of developments in stem cell and organoid research. In short, we proposed that if we are to genuinely respect donor autonomy in a stem cell or organoid research context, then we need to adopt a diachronic, longitudinal, and relational approach to the exercise of autonomy (ibid.). Given the “hybridity” of PSC derivatives and thereby the moral, symbolic, and practical significance of such entities for those that have donated, frameworks are required that enable donors to remain engaged and retain meaningful influence if and/or when previously unforeseen morally, religiously, culturally, or other sensitive uses arise. Ultimately, without ongoing participant involvement—and without acknowledging how donors’ moral attitudes may shift—stem cell and organoid activities risk undermining the very autonomy they are required to protect.
Even though we argued that models of consent preclude adequate engagement with donor autonomy in stem cell and organoid contexts because they are primarily concerned with facilitating informed consent—the conditions for which cannot be equated with the conditions for the exercise of autonomy—we acknowledged a notable exception in the model of dynamic consent (Lewis and Holm, 2022).
Dynamic consent refers to a personalized, online or IT-based communication and interactive decision-making interface that facilitates not only donor decision-making, but also communication and information transfer between researchers and donors (Budin-Ljøsne et al., 2017; Kaye et al., 2015; Steinsbekk and Solberg, 2015; Teare et al., 2015). The interface and the principles on which it is based were developed, in part, because focus groups found that some donors wanted more information about, and stronger oversight concerning the uses of, their samples and data (Prictor et al., 2018). At the same time, developers of dynamic consent acknowledged that donor decision-making in these respects could be better supported by bolstering communication and engagement between researchers and their donors (Prictor et al., 2018).
Dynamic consent offers a way to bridge the gap between a stem cell donor’s ongoing moral interests and the open-ended nature of pluripotent (or otherwise immortal) cell lines. Rather than treating consent—and any potential withdrawal—as a single event, dynamic consent allows participants to engage iteratively over the course of their donated biomaterial’s lifespan, receiving updated information about newly proposed uses of their cell lines and providing or withholding consent on a case-by-case or ongoing broad basis. Thus, dynamic consent seems uniquely suited to PSC-based research because it acknowledges that new, morally-sensitive applications—like creating advanced neural organoids or organoid-based xenotransplantation—might not have been foreseeable at the time of initial donation. As a result, donors can choose to opt in or out of specific lines of research without withdrawing entirely.
As argued elsewhere, dynamic consent, in principle, supports a longitudinal and diachronic approach to donor autonomy (Lewis and Holm, 2022). However, whilst dynamic consent preserves the sort of donor control that the classical right to withdraw functioned to enable, it shifts the constellation of procedures and principles typically underpinned by the right to withdraw. First, it moves the emphasis from recalling samples to demarcating future uses that were often unforeseen at the time of initial consent. Under a dynamic framework, the donor retains partial control without needing to destroy or halt all existing work. In this sense, it functions like prospective withdrawal but addresses one of its key problems; namely, prior to new procedures or applications getting underway, donors will have already been informed about them and will have thereby already had the opportunity to decline.
Second, by regularly updating donors on advanced or potentially sensitive PSC derivatives, dynamic consent offers granular engagement, providing donors with the opportunity to continuously calibrate their participation as science evolves. This ongoing awareness replaces the traditional assumption that either the donor withdraws once and for all or they remain perpetually bound to the broad or blanket permission provided at the time their donation was made (see Lewis and Holm, 2022).
Third, a common argument in favor of the right to withdraw is that it helps sustain public trust (Helgesson and Johnsson, 2005). Dynamic consent seems to offer a way to address this more effectively by allowing donors to have real-time input if new, potentially controversial uses of their cells arise (Kaye et al., 2015; Kraft et al., 2018). The approach thus fosters transparency and removes the necessity for a retroactive right of withdrawal.
Compared to a single, all-encompassing right to withdraw, dynamic consent seems to confer greater stability and predictability on stem cell projects. Rather than having to fear an outright demand for the recall or destruction of cell lines, derivatives, and associated data, researchers know that donors will be notified of materially different or anticipated ethically sensitive uses of their cells, at which point the donor may revise or refine consent. This process allows labs to clarify and compartmentalize risk: even if a donor later objects to a specific future application, approved or ongoing studies can continue. In addition, because dynamic consent allows for the parceling out of donor objections, it also gives researchers advance notice of where potential moral, religious, and/or cultural friction might arise. For instance, if a lab plans to transition from simple tissue culture to complex organoid derivation or neural grafts in animals, it can access the dynamic consent platform to identify how many participants have permitted or declined such uses at the time of donation before the lab invests heavily in the new direction, reducing wasted resources and last-minute disruptions (Kaye et al., 2015; O’Doherty et al., 2011). Furthermore, the trust-building effect of dynamic consent can contribute to overall research certainty. Proponents have argued that the facilitation and maintenance of an ongoing connection with donors might assist researchers with fostering trust, especially as dynamic consent aims to promote improved understanding of research uses and a stronger sense of collaboration between donors and researchers (Prictor et al., 2018). By making donors feel more included and respected on a continuous basis, the model, in principle, is capable of mitigating the risk of abrupt or wholesale withdrawal that may have arisen from dissatisfaction or mistrust.
Although dynamic consent plays a central role in our hybrid model, it does not eliminate the need for an up-front consent framework. Stem cell research involving PSC lines and their derivatives is, as already discussed, characteristically open-ended and cumulative, with multiple downstream projects, transfers between laboratories, and re-uses of derived materials that are often unforeseeable at the time of donation. If case-by-case consent was required, many stem cell repositories and projects would likely suffer undue administrative burdens and operational delays, and donor attrition and unresponsiveness could introduce systematic biases, particularly in longitudinal or cohort-based studies (Hansson et al., 2006; Helgesson, 2012; Petrini, 2010). Broad consent is, therefore, practically defensible as an initial permission structure for PSC research, not because it fully captures the principle of respect for donor autonomy (for details, see Lewis and Holm, 2022), but because it can support the sustainability of PSC research whilst allowing donors to permit and restrict the use of their biomaterial in those types and classes of research that can be reasonably anticipated at the time of donation.
However, broad consent is—from an autonomy perspective—fragile if it is treated as a blanket waiver of all downstream moral engagement on the part of the donor (for details, see Lewis and Holm, 2022). The central worry is not merely that donors cannot be adequately informed about all future studies and potential types of research use, but that donors may—at the time of donation—have stable, deeply-held values and preferences about some types of use that are already known to be ethically contentious in PSC and organoid research—for example, neural organoids approaching greater functional complexity, human–animal chimera research, embryo-like models, certain commercialization pathways, and military applications. In such cases, a broad-consent-only approach risks coercing donors into moral complicity with future uses that they would have refused had they been given the option, which undermines consent’s autonomy-respecting function. This is precisely why our hybrid model treats broad consent not as a sufficient solution to the central tension between donor autonomy and research stability but as the groundwork that needs to be built upon with: (i) up-front preference elicitation for ethically sensitive categories of research that can be reasonably anticipated at the point of donation; and (ii) dynamic consent for ethically salient downstream developments that cannot be anticipated at the time of up-front broad consent.
This justification also bears on the withdrawal problem. If donors are provided with an up-front opportunity to register their preferences on currently anticipated ethically sensitive uses, then they may be less likely to face the all-or-nothing dilemma of either remaining broadly committed to the continued use of their cell lines and any associated derivatives or seeking to withdraw entirely once they hear about a use they find morally objectionable. Therefore, initial preference elicitation supports donor autonomy and trust while also providing researchers with greater predictability than a system in which ethically contentious uses trigger donor conflict only after significant investment and scientific dependence have accumulated.
In terms of what the combination of up-front broad consent and initial preference elicitation might procedurally look like, donors would, at the time of donation, provide broad consent to types and classes of research use typical of current consent frameworks in stem cell research, for example, derivation of cell lines, storage, quality control, distribution of PSC lines and associated data, basic research, disease modeling, drug screening and toxicological studies, organoid development, research in precision, personalized, or regenerative medicine, including use of PSC lines and associated derivatives in clinical trials. They would also be invited to express their preferences regarding currently anticipated ethically sensitive categories. A workable preference structure is not a protocol-by-protocol menu, but a set of reasonably general categories that correspond to reasonably stable clusters of known or anticipated ethical concern in the PSC field. Examples might include: (i) derivation and use of neural organoids either in general or beyond specified maturation and/or complexity thresholds; (ii) creation and manipulation of stem cell-based embryo models; (iii) transplantation or integration of PSC-derived material into animals, including chimeric research; (iv) genome editing beyond disease-modeling corrections; (v) commercial licensing and partnerships, especially where international transfers of lines and/or associated data are involved. For each category, donors could choose a default stance such as “permit,” “restrict,” or “ask me again if this arises,” with the final option functioning as one of the triggers for recontact under the dynamic consent dimension of our proposed hybrid model (see §3.1 for further details).
To support donor autonomy and promote the implementation of the preference elicitation component, there appear to be two main requirements. First, the system should be designed so that donors are not overwhelmed: preference elicitation should be limited to categories that are ethically salient, currently known or reasonably foreseeable, and likely to be value-sensitive across diverse donor populations. Second, preferences must be actionable to the extent that downstream projects can be screened against the donor’s stated permissions and restrictions.
It is also important to acknowledge that, in principle, initial broad consent plus preference elicitation strengthens research stability. It provides a reasonably clear network of permissions and restrictions for day-to-day research activity, while identifying in advance those donors whose preferences will constrain sensitive uses. This allows researchers to plan prospectively rather than discovering conflicts only after investing heavily in new projects or protocols.
Having clarified why the hybrid model begins with broad consent plus preference elicitation, we now turn to the key implementation question: how dynamic consent can be operationalized in PSC research in a way that preserves donor autonomy while supporting research stability.
Implementing dynamic consent in PSC research: Feasibility and design requirements
Although dynamic consent, in principle, provides for a level of donor control that could warrant the abolition of the right to withdraw in biomaterial donation contexts (for further details, see Lewis and Holm, 2022), the question remains whether it can address two of the main problems engendered by the right to withdraw in stem cell research contexts; namely, that withdrawal is not practically possible to fulfill and creates significant uncertainty for stem cell researchers.
We start by addressing the certainty question. Although it is obvious that dynamic consent facilitates a greater level of certainty for stem cell research than unconditional, immediate, and non-waivable right to withdraw, there is a question of whether, in principle, it can provide more stability and certainty than prospective-only (i.e. forward-looking) withdrawal. As already mentioned, if a donor has donated biomaterial according to a broad or blanket consent framework, there is the risk with prospective-only withdrawal that they may abruptly impose restrictions on the use of their cells or derivatives at any time. By contrast, under a dynamic consent model researchers have the opportunity to receive advance notice of donor prohibitions, which means that, so long as researchers plan far enough in advance and are primed to inform donors of particularly contentious or sensitive potential avenues of research, they can assert some control over the risk of non-participation and thereby engage and invest in specific studies or derivations once there is sufficient PSC lines to make the research viable. Thus, although a dynamic consent approach does not provide stem cell researchers and investors with absolute certainty, it is, at least in principle, capable of facilitating a greater level of stability and certainty than prospective-only (or, indeed, retrospective) withdrawal.
We have already explored the factors that render classic withdrawal in PSC research practically impossible to fulfill. By comparison, when it comes to practicality, even advocates of dynamic consent recognize that this approach is not without its drawbacks. First, given that study-specific decision-making would be made available to donors, there is the risk that this could lead to a diversion of resources away from research activities, thereby affecting the ability of a lab to operate at full capacity (Mikkelsen et al., 2019). Second, it would necessitate detailed tracking and compartmentalization of cell lines and derivatives in line with an evolving matrix of donor permissions; again, potentially adding to the administrative workload of individual labs and multi-institutional consortia (Lewis and Holm, 2024). Third, repeated requests for consent to new uses could potentially overwhelm or fatigue participants, undermining the very autonomy dynamic consent seeks to protect (see Lewis et al., 2023; Ploug and Holm, 2015).
Therefore, a frequent worry about dynamic consent is not its ethical rationale but its feasibility in practice: that is, how it can be implemented across biobanks, laboratories, and downstream applications without becoming administratively brittle or generating undue pressure for researchers. In practice, implementation challenges tend to cluster around four areas. First, infrastructure and interoperability. Dynamic consent has to connect to existing biobank information systems, laboratory information management systems, and downstream access control systems, so that permissions are checked at the point of sample or data release (Kaye et al., 2015; O’Doherty et al., 2011). Second, traceability. Where PSCs generate extended derivation chains, restrictions on use ought to track derivatives and datasets, including across institutions and jurisdictions (Lewis and Holm, 2024). Third, issues concerning operational burden and cost; that is, staffing and general coordination demands will likely increase with scale (Mikkelsen et al., 2019). Fourth, participant experience and equity. Frequent prompts can give rise to consent fatigue (Lewis et al., 2023; Ploug and Holm, 2015), and dynamic consent systems can exclude donors who aren’t able to access digital-only platforms (Prictor et al., 2018).
These concerns do not defeat dynamic consent in the sense that they render the approach practically impossible in a similar way to classic withdrawal but they do imply certain design requirements. To support autonomous decision-making, mitigate against consent fatigue and the routinization of consent in general, and reduce operational and resource burdens for researchers, one requirement is a tiered preference taxonomy at the initial broad consent stage that is granular enough to capture ethically salient differences without necessarily requiring donors to respond to every protocol-level variation (see, e.g. Ploug and Holm, 2015). For example, as we proposed in the previous section, at the time of initial broad consent, donors should also set default preferences for certain ethically contentious categories of research. On that basis, with ongoing dynamic consent, donors are approached or recontacted only when a proposed use pertains to a type or class of research use that they had not previously consented to, goes against a previously defined preference, or exceeds an ethically relevant threshold that was not foreseen at the time of initial (broad) consent (Kaye et al., 2015).
A second requirement is for prospective effect rules that, in practice, could support research continuity without, again, engendering operational and resource pressures. In short, changes to a donor’s preferences or broad consent should normally apply to future uses, not to studies or other uses already underway. Institutions can seek to operationalize this through, for example, time-limited consent-cum-preference stability windows (e.g. the duration of a defined protocol cycle or project). Finally, to address concerns regarding interoperability and traceability, the implementation of dynamic consent should be auditable. For instance, researchers, governance bodies, and downstream users of PSC derivatives across jurisdictions should be able to readily access something like a digital consent ledger that combines data relating to participant preferences, consents (both initial and downstream), and restrictions across biobank, laboratory, and institutional information management systems, and which records the state of the donor’s consent each and every time the ledger is accessed (see, e.g. Barnes et al., 2025; Charles et al., 2024; Mamo et al., 2020).
Evaluations of prototype, proof-of-concept, or minimally viable dynamic consent technologies are currently underway, though the current evidence base is extremely limited (see, e.g. Charles et al., 2024). Furthermore, with the currently popular approach to developing dynamic consent platforms using blockchain-based technologies (see, e.g. Mamo et al., 2020), the issue, as Charles et al. (2024) have noted, is that different products are being developed to address specific issues, and, therefore, it is, in practice, difficult to evaluate dynamic consent technologies as a whole, and especially against the specific requirements for dynamic consent engendered by PSC research. As a result, perhaps a more pragmatic way to develop evidence on the feasibility of dynamic consent in PSC research would be via a staged pilot study or set of studies. For example, a single biobank or other stem cell repository could begin with a limited set of ethically salient categories (e.g. animal integration, advanced neural organoids, stem-cell-based embryo models, and commercial licensing), implement trigger-based recontact and prospective effect rules, and evaluate administrative and operational burdens, donor comprehension and engagement (including attrition and preference-change rates), and research stability (e.g. delays and redesigns attributable to consent changes). Such a pilot would provide at least some empirical assessment of whether dynamic consent could potentially deliver meaningful donor control while providing the predictability that long-term PSC research requires.
Although dynamic consent can, in principle, facilitate a greater level of certainty for stem cell research than an unconditional, immediate right to withdraw, this stability depends on implementation choices, particularly clear thresholds for what counts as a materially different or ethically salient use, trigger-based recontact, and prospective application of preference changes. Without such features, dynamic consent risks reproducing the unpredictability it is intended to reduce.
In principle, dynamic consent stands out as both a practical and normative compromise. It avoids the extremes of either unconditional, immediate, full withdrawal—unworkable and normatively undesirable in these contexts—or forcing participants solely into an up-front broad consent framework that can lead to moral distress later if they object to new research directions. By enabling real-time communication, dynamic consent aims to support donors’ personal autonomy over the morally valuable derivatives of their donated biomaterial throughout the research lifespan whilst, in principle, providing a greater level of stability and certainty for the climate of stem cell research than can be provided by modifications to the right to withdraw. That said, if we are still unconvinced of the ability of dynamic consent to provide certainty for stem cell research, we can also seek to supplement the model of dynamic consent with a robust form of governance without necessarily undermining donor control.
Enhancing research certainty and stability through governance
In addition to modifying or reinterpreting the right to withdraw, some scholars emphasize the value of formal governance-based frameworks (Boers and Bredenoord, 2018; O’Doherty et al., 2011; Prainsack and Buyx, 2013). For instance, future (morally, religious, or culturally sensitive) developments in stem cell-based research and application are dealt with by a combination of ethical oversight and participant engagement, with the latter, for instance, consisting of groups of donors or the wider public engaging directly, deliberatively or representatively in the design and continuous adaptation of biobank governance (Boers et al., 2019). Not only do participant engagement and ethical oversight function as safeguards to ensure that the different interests of diverse stakeholders are taken into account when considering new avenues of research, they can contribute to decisions about which research activities should require new consent. In principle, governance models typically involve ethics committees, donor and public engagement, and consistent ethical oversight that can adapt to new developments in research. Such structures may reduce the burden on donors to monitor and keep abreast of potential developments that may directly relate to the use of their samples by ensuring that a trusted, forward-looking system of review is in place (Boers and Bredenoord, 2018). At the same time, oversight bodies are better placed to anticipate new research developments, assess whether previously unforeseen forms of usage align with consented terms or moral norms, and communicate these potentially morally, religious, and/or culturally sensitive uses to those who have donated. Although we have previously argued that governance-based frameworks can dilute personal autonomy by placing ultimate authority in committees or majority decisions that might not reflect individual preferences (Lewis and Holm, 2022), when combined with a dynamic consent framework, a governance model can, at least in principle, support research certainty without any significant effects to donor autonomy.
Under such a hybrid model, donors could initially choose broad categories of permitted research. Governance committees, in parallel, anticipate and address potential morally sensitive issues (e.g. shifting societal attitudes about neural organoids, chimeric xenotransplants, gene editing, and the moral status of embryo models) while evaluating the impact of donor (non-)participation on potential future studies that may or may not have yet been planned for. In short, the governance structure should act as a disinterested intermediary in the balance between donor autonomy and scientific utility.
One function of the right to withdraw that we have not discussed so far is that it functions as a circuit breaker if a research participant or cell donor comes to suspect that researchers are doing research beyond what they have permission to. It is a way for the research participants to close down the research of non-scrupulous researchers, or, at the very least, to disengage publicly from that research. If the right to withdraw is exchanged for multi-layered consent and ongoing governance in stem cell research, then it will fall to the ongoing governance structures to take robust action against researchers who use stem cell lines and constructs outside of the permissions and consents that have been given.
Limitations and future research
Our proposal specifies a hybrid ethical framework and identifies practical design requirements, but it is not a claim that dynamic consent and governance can be implemented at no cost or that a single approach will fit all jurisdictions and repositories of PSCs and their derivatives. Cross-institutional implementation will require careful harmonization of consent and preference categories and regulatory arrangements for material and data transfers, as well as empirical evaluation of donor experience and administrative and resource burden (for details, see Lewis and Holm, 2024). Future work should, therefore, prioritize the evaluation of extant (albeit currently nascent) dynamic consent technologies as well as pilot implementations and comparative assessments of different tiered preference taxonomies, participant recontact thresholds, time-limited consent-cum-preference windows, and auditing and traceability mechanisms (see §3.1), alongside continued normative work on governance standards, processes, and structures for ethically sensitive downstream PSC uses.
Conclusion
Stem cell research, particularly where pluripotent stem cell lines are concerned, necessitates a rethinking of the classical right to withdraw. Initially anchored in clinical contexts to safeguard against bodily integrity, this right is far less straightforward in situations where a single donor’s biological material can be continually expanded, shared among numerous laboratories, and incorporated into a constellation of ever-expanding derivatives and therapy-oriented pipelines. Maintaining an unconditional, immediate, and non-waivable right to withdraw in these circumstances is logistically impossible, undermines research utility and stability, and may not always serve the interests of donors themselves—especially once valuable downstream uses are underway.
Nonetheless, withdrawing the right altogether would risk undermining personal autonomy and trust in stem cell research. To navigate this tension, we have reviewed proposals for adapting or modifying the right to withdraw in ways that remain consistent with respect for donor autonomy. These include reason-giving frameworks, cooling-off periods, and the possibility of prospective restrictions instead of total recall. Each approach aims to preserve at least some degree of donor control while avoiding a complete reversal of valuable research initiatives. Additionally, the notion that donors might waive the right entirely, though attractive for researchers and commercial actors seeking certainty, raises significant concerns about inalienable human interests and broader inequalities of power.
Ultimately, we have argued for a hybrid of up-front broad consent, initial donor preferences for known ethically contentious areas of stem cell research, ongoing dynamic consent, and robust oversight as potentially the most normatively appropriate way to achieve a balance between donor claims and the long-term, open-ended nature of stem cell research (see Figure 1). Dynamic consent captures how donor preferences can evolve over time, allowing targeted objections to novel or morally challenging uses unforeseen at the time of initial broad consent and preference elicitation, rather than forcing donors and researchers into an all-or-nothing scenario. Similarly, governance-based models—whether centralized ethics committees or adaptive regulatory frameworks—can help anticipate and identify the boundaries of acceptable research, limiting the need for abrupt, retrospective recalls while fostering ongoing trust and dialog between donors and the scientific community.
By taking seriously the justifications for, and limits of, a right to withdraw, stem cell research can move toward a structure that neither shackles scientific progress with untenable recall demands nor leaves donors powerless once their biospecimens are in circulation. Such a recalibration requires open communication, transparent decision-making, and continued ethical scrutiny. Yet, by re-orienting donor control toward frameworks like dynamic consent and robust governance, both researchers and donors may be able to realize the full promise of stem cell lines without sacrificing genuinely autonomous decision making.
Footnotes
Ethical considerations
No ethical approval necessary.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
