Abstract
The purpose of this study is to examine the importance that real patients attach to their right to withdraw from an on-going feasibility randomised trial (RCT) evaluating types and timings of breast reconstruction (two parallel trials) following mastectomy for breast cancer. Our results show that, while some respondents appreciated that exercising the right to withdraw would defeat the scientific objective of the trial, some patients with a surgical preference consented only given the knowledge they could withdraw if they were not allocated to their preferred treatment.
Introduction
While there has been some philosophical work published on the conditions associated with exercising the right to withdraw from research with a view to protecting the internal validity of controlled trial results (Edwards 2005; Edwards 2011), there is little in-depth empirical work on the topic. Surveys of clinical trial participants simply report how well they recall their right to withdraw (Edwards et al. 1998). However, there is no published research into how those invited to take part view their right to withdraw and whether reserving such a right influences their decision to consent.
The purpose of this study is to examine the importance that real patients attach to their right to withdraw from an on-going feasibility randomised trial (RCT) evaluating types and timings of breast reconstruction (two parallel trials) following mastectomy for breast cancer. The described QUEST Perspectives Study (QPS) was embedded in a surgical RCT, called QUEST (Quality of life after mastectomy and breast reconstruction) (Winters, 2013). The QUEST trials were two parallel studies that were investigating the impact on Patient Reported Outcomes (Health Related Quality of Life (HRQL) and satisfaction) of different types (QUEST Trial A) or timings (QUEST Trial B) of breast reconstructions. QUEST aimed to establish the feasibility of women accepting recruitment into larger randomised controlled trials and comprised 15 UK centres. QPS evaluated the perceptions of women who were invited to take part in the QUEST trials, including their understanding of recruitment and the randomization processes, in order to inform on-going recruitment.
Methods
Recruitment and procedure
Ethical approval for the QUEST trials and the QUEST perspectives study (QPS) was granted by South West 2 Research Ethics Committee on 9th November 2010. All eligible patients approached to participate in QUEST were simultaneously also invited to be involved in the QPS. Those women who consented to the QPS, both decliners and acceptors of the QUEST trial, were invited to participate in a qualitative interview at one month following their surgery. Interviews took place over the telephone with either NB or LM. Interviews were audio-recorded with participants’ permission and transcribed verbatim. The interviews explored the women’s understanding of the QUEST trials, the process of randomisation, clinical equipoise each on the part of the patient and the surgeon, and their decision-making processes in accepting or declining to enter the QUEST trial.
Participants
In total, 96 patients were eligible to enter QPS, and 57 (55%) consented to take part, of whom the 39 who responded to the invitation took part in qualitative interviews. The present analysis focuses on those interviews in which the issue of the right to withdraw from the QUEST trials arose spontaneously (n = 12). The socio-demographic characteristics of this subset of the interviewees are summarised in table 1.
Participants’ socio-demographics.
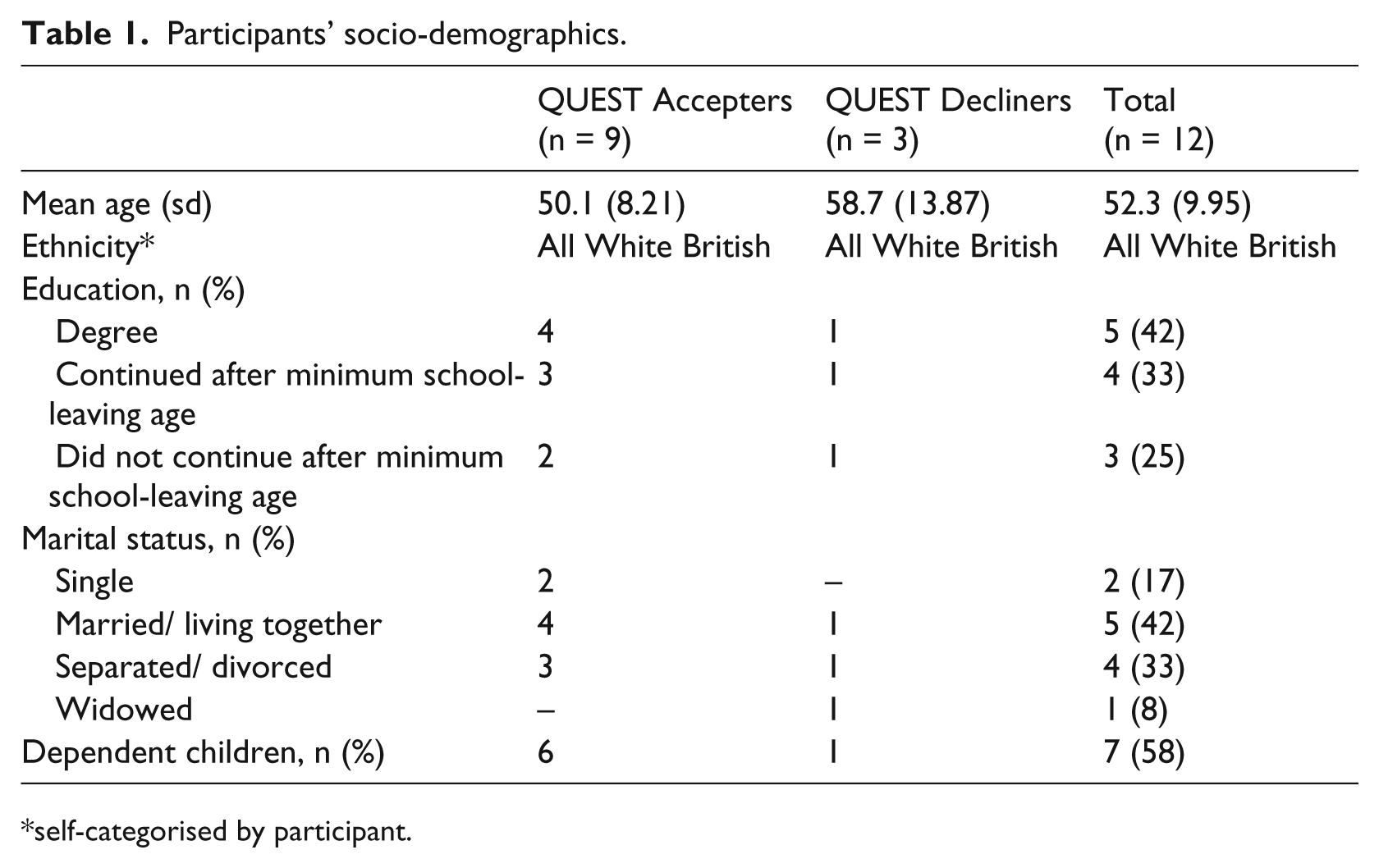
self-categorised by participant.
Data analysis
Two authors (NB and LM) initially independently coded the transcripts. Thematic analysis enabled codes to be conceptualised into common themes. Emerging themes were subsequently discussed and guided by SE. Further development of the themes occurred by NB and LM returning to the original interview transcripts. NB and LM coded the data independently at first, with consensus agreement through discussion where initial coding differed. Participants’ right to withdraw was a key finding of the analysis with the details thereof elaborated in this paper. Other findings are presented in depth elsewhere.
Results
The data relating to the overall theme of the right to withdraw from the QUEST trial are presented as themes with qualitative quotes for illustrative purposes.
1. Right to withdraw could defeat the objective of trial
Participants discussed the objectives of the QUEST trial and why it was conducted using randomisation. One decliner struggled to rationalise her right to withdraw from the QUEST study with the need to accept randomisation. Note that this respondent understood the process of randomisation but not its scientific purpose in removing selection bias)
… [the right to withdraw] wasn’t explained, let’s go back to the implant thing, maybe some people wouldn’t want that anyway and if they were then selected to have one, have they got the opportunity to say, no I don’t want that.
To change their mind, yes.
And the same thing, that destroys the objective though I suppose. If I had taken part and I came up with the decision that I didn’t want, do I then say, no I don’t want that.
Yes, and then so you wouldn’t be…
So me deciding what I’m having done anyway.
A QUEST acceptor also articulated this issue, although her uncertainty appeared to arise from the advice given by the healthcare professionals (HCPs) involved with the trial rather than from inferring it for herself from the information provided. This respondent understood the process of randomisation but not necessarily its rationale.
I said if they choose for me and I don’t want that can I back out of the trial at any time so they said well there’s no point in you really doing the trial if you’re going to back out.
2. Right to withdraw from trial relevant when not allocated to treatment preference
The right to withdraw was seen by respondents as most relevant after randomisation and before surgery and so related to the direct result of randomisation for treatment allocation. For example, one QUEST acceptor stated that she would have withdrawn from the study if she had not been allocated her preferred option, although she lacked a clear understanding of randomisation and her (false) perception was that the outcome of randomisation was either being allocated to mastectomy with or without a reconstruction (see Figure 1).
So I think it’s very difficult for any woman to come to that situation and not have some preconceived idea of what outcome they would most likely have and to think, ‘Okay I’m going to have this. I’m going to have a reconstruction,’ and then be told, ‘Well actually you’re not going to,’ I think most people would just opt out at that stage, to be honest.
… I actually think, for me, because it’s a time where you’re so desperate to stay in control anyway that I was quite clear for myself I think that had it come back with a different option to the one that I actually ended up with, I might have withdrawn from the study at that stage.
If you’d been randomised and the different option had come up, did you tell me a little bit earlier that you would have then…
I think in all likelihood I would have withdrawn from the trial and that’s the thing, it’s like I do understand why it has to be done in that way but I wonder how many people actually withdraw at that stage because it is one of those things.
3. Right to withdraw not relevant
For some respondents, the right to withdraw was irrelevant to their decision whether or not take to part in the QUEST trial.
A: because participant was in equipoise
Two women with full understanding of randomisation were clearly in equipoise between treatments (completely uncertain which option would be best for her), and hence did not have a preference for either surgical option offered in the QUES T trial.
… In fact in my case because they did say that if I changed my mind whether it was the QUEST, or if I had to suddenly say “oh I must go down that way” then that would have been okay I could have just dropped out of it all, but when it got nearer the time which was only like weeks anyway, I studied it more and read everything I actually, you won’t believe this, I couldn’t even make me mind up even if I wasn’t on QUEST and even if the surgeon said “which one would you want?” - I couldn’t even tell you now which I would have preferred.
… I think it was one of those really busy days when I did ask a few questions but I think I understood it quite clearly so I didn’t really ask a lot to be honest and I can’t remember what I did ask. I can remember being in the office and she did say, ‘You can withdraw at any point.’
Yes so you understood that.
So I didn’t feel under pressure. I thought, ‘Well I can go with it and if then I feel as though I don’t want to proceed with it then I can stop.’ So I didn’t feel as though I was tied.
So if one of the options had come out you could have changed your mind?
Yes [the surgeon] has said all along, ‘Whatever you decide, it’s your decision. Please don’t worry, it won’t affect your treatment or anything like that. If you want to withdraw then just withdraw.’
B: because participant was committed to the trial despite having a preference
Although aware of their right to withdraw, two acceptors claimed that they would not have exercised it even if they had not been allocated their preferred surgical option. They expressed different reasons for showing this apparent commitment to the QUEST trial, and only the second respondent, PA15, seemed to fully understand the process of randomisation, although she expressed a certain belief in the ‘equivalence’ of the treatment options (as if there were evidence that the options were equally good) which could have indicated a lack of understanding of uncertainty through the absence of evidence and hence a therapeutic misconception associated with the trial.
…it was my decision in the end and I could go along with what I wanted. It wouldn’t make no difference about the trials, if I was randomised for a different type and I didn’t like it, she said that’s no problem, she said you don’t have to go on the trials but I agreed in the end. I thought about it and I agreed. I thought yes it’s a better outcome and for people to understand more about breast cancer.
…she did say to me any time if you don’t want to be random picked it doesn’t matter. You don’t have to be on the trials and we’ll do the procedure whatever you want anyway but then I decided. They said to me I could go away and think about it and come back later, which we did, and I said we’ve discussed it and we’ve talked about it and we’ve looked at the pros and cons and we decided yes we’re going to go along with it.
Had I had a strong view that way then the chance that it would have come back without an implant would obviously have compromised my wishes as a patient but I was told from very early on if you have a strong view and it comes back with what you don’t want you can opt out of the trial at any time… Anyway it came back and it wasn’t an implant so that was fine.
What do you think you would have done if it had come back with the other option? Would you have…
I probably would have gone with it anyway because I absolutely trust the team that are looking after me. I think that was the most important thing that you trust the team that are caring for you and my surgeon was absolutely adamant that both outcomes would be as good.
4. Ability to withdraw affected decision to participate or not
For one QUEST decliner, it was evident that her decision not to take part in the trial was affected by her lack of knowledge about her right to withdraw from the trial. However, she had no understanding of randomisation either.
It was also taken out of my hands, I felt out of control completely. Although I felt out of control anyway I needed to keep some kind of control somewhere and then I thought well, I wouldn’t have any at all and I don’t know whether…it wasn’t explained to me if I could disagree with the computer.
Okay, that wasn’t explained, alright okay.
I didn’t know that I could say actually no, I don’t agree with that and I suppose I should have been and in retrospect, later on, you think a little bit better about it, do you know what I mean? But at time I was thinking well I don’t want a computer telling me what kind of surgery or treatment they’re going to do for me and then nobody said to me yes but you can disagree.
Yes, so that wasn’t made clear.
But that wasn’t made clear to me, I just thought that would be it, it would come back down, the computer would say do this and that would be where I’d have to do it no matter what.
Similarly, for two QUEST acceptors, their knowledge about their ability to withdraw influenced their decision to consent to the trial. However, only the second respondent, PB08, fully understood the process of and rationale for randomisation.
…did you have any concerns about any potential disadvantages in taking part in the study at all?
Not really because they said [nurse’s name] who was the lady who has been doing who met me and talked through this again with me, she just said you know at any point if you are not happy you can withdraw. So that was an option, if I decided a few weeks down the line that I wasn’t happy, I knew I had the option.
The respondent who did seem to gain full understanding of randomisation stated:
If I had a particular choice, if at the beginning and I did to be honest, when I was being selected I was keeping my fingers crossed that I would be following a certain path, the nurse could have come back and told me actually you’re following the other path in which case maybe I would have been a bit disappointed..
… it’s all a bit of time ago. From what I remember, I could’ve pulled out at any time from the trial anyway. I wouldn’t have wanted to and I don’t think I would have done, but I was fully aware that I could pull out at any time which was fair and I did appreciate that. That really swayed it for me, signing up in the first place.
That you knew that you had options all the way along.
Yeah.
Discussion
Our results show that, while some respondents felt that exercising the right to withdraw would defeat the scientific objective of the trial, some patients with a surgical preference consented only given the knowledge they could withdraw if they were not allocated to their preferred treatment.
This is consistent with other studies on open preference trials and the problem of ‘resentful withdrawal’ following randomisation where consent rates may be directly proportional to attrition rates (King et al. 2005). This phenomenon may be in part resolved through partial preference randomization trial designs, which showed increased recruitment to randomisation with a mean of 63% (range 26 – 88%) in five surgical trials.
This highlights persistent issues of retained patient preferences despite the communication of trial rationale and clinical equipoise. These preferences should be assessed prior to consent and randomization and evaluated in terms of their potential contributory effects to treatment outcomes, which at present remain uncertain (BMJ, 2008 Preference Collaborative Review Group, Torgerson DJ). The Preference Collaborative Review Group performing a meta-analysis of patient preference trials showed that the effect size of treatments in patients who were randomized to their preferred treatment was significantly greater, than in patients indifferent to their treatment allocation. This increases our understanding and shows that preferences can affect outcomes, especially where preferences concentrate on one treatment potentially confounding un-blinded randomized studies.
As this data set is part of a larger study of patients participating in QUEST, the responses are somewhat limited in important ways. For example, no respondent discussed the right to withdraw without needing to give any reason, only the right to withdraw after randomisation and before surgery. This limitation in responses could have been partly due to the questions asked of respondents and the type of trial under study. However, these responses are unusually based on the views of real patients presented with real options and so they are authentic in a way surveys of the public may not be. The challenges in the field of breast reconstruction are embedded in informed consent processes synonymous with patient choice. Current clinical evidence is poor where studies have either not evaluated the effects of breast reconstruction surgery on patients’ HRQL or have used poor study or HRQL methodology. Despite clinical equipoise based on two systematic reviews and a recently published Phase 2 surgical study, retained patient preferences remain a potential bias.
It is very interesting to note that not one of the respondents mentioned the any conditions under which their right to withdraw might be exercised. The orthodox wording in information sheets states that participants retain a right to withdraw from a trial “at any time without needing to give researchers a reason” (GAfREC 2011). This patient perception may be related to the ‘irreversible’ nature of the surgical intervention, which is synonymous with the ethical dilemmas and challenges faced by surgical trials. Interviewees were not asked specifically about their understanding of the right to withdraw during these qualitative interviews and all instances in this analysis represent times where they mentioned it spontaneously. All instances related to withdrawal after allocation and prior to surgery. Although some participants mentioned other disadvantages of the trial, none spoke of these in relation to wanting to withdraw from the trial.
We might expect people’s views on the right to withdraw to be heavily influenced by the context of the trial where either treatment arm can be selected outside the trial. Both randomisation arms comprised well-established types of breast reconstructions potentially associated with clinician and patient bias despite clinical equipoise. The QUEST trials were the first multicentre feasibility trials showing levels of patient preference of 60%. More research is thus needed to investigate the influence of different approaches to the right to withdraw on recruitment rates to other sorts of trials. Indeed, a recent survey of ethics committees in the UK NHS reported sympathy with the need to reduce the problem of missing data to better protect the internal validity of trials where we might expect a systematic difference associated with the trial treatment between those who withdraw and those who do not. For example, obesity trials are particularly vulnerable to this form of bias (Li et al. 2013).
Footnotes
Declaration of conflicting interest
The author declares that there is no conflict of interest.
Funding
This research was funded by the Bupa Foundation as part of the QUEST studies (A and B). SJLE is funded by the National Institute for Health Research (NIHR) with support from the Centre for the Advanacement of Sustainable Medical Innovation.
