Abstract
This case study considers LGBTQ+ participation in cancer clinical trials as an issue of justice and data equity in research ethics. Although representation of LGBTQ+ people in clinical trials varies depending on the type of cancer, routine data collection of gender and sexual orientation in clinical trials is inconsistent, leading to underrepresentation in routine reporting. Improving LGBTQ+ participation in cancer clinical trials ensures more ethical and inclusive research, supporting the principle of justice, and enabling more accurate data collection practices to support LGBTQ+ health. This case study draws on findings from a pilot study involving a scoping review and key informants’ interviews in Australia to understand barriers and facilitators to LGBTQ+ participation in cancer clinical trials. A key issue identified is that gender and sexual orientation are not routinely collected in cancer clinical trials, and that while most professionals working in cancer clinical trials do not object to collecting this data, further education and resourcing is needed to support them to do so effectively. The case study provides key recommendations for improving LGBTQ+ participation in cancer clinical trials, including supporting minimum data requirements for gender and sexual orientation in cancer clinical trials, providing further training and support for workforce capacity, and upskilling ethics committees regarding LGBTQ+ data equity.
Keywords
Introduction
The principle of justice in research ethics requires fair access to the benefits of research, including equitable participation in research that may yield health benefits (National Health and Medical Research Council, 2025). Ensuring that lesbian, gay, bisexual, transgender, queer, and other gender and sexuality diverse (LGBTQ+) individuals are included in clinical trials is critical to ensuring scientific progress and upholding the principle of justice, as an essential part of the right to health (United Nations, 1966). Clinical trials should neither exclude sexuality and gender diverse people explicitly, through direct exclusion criteria; nor implicitly, through the use of exclusionary cisnormative and heteronormative language (Drysdale et al., 2024). It is also essential that LGBTQ+ participation is accurately documented so that rates of inclusion can be assessed, thereby contributing to LGBTQ+ health equity (Saxby and Hammoud, 2025). Without accurate data on sexuality and gender, cancer clinical trials risk reinforcing cisnormative and heteronormative biases, limiting the applicability, and effectiveness of trial outcomes in these communities.
A growing literature over the past decade has recommended standardised data collection of sex, gender, and sexual orientation, including the use of a two-step gender identity question to be able to accurately and sensitively determine cisgender and transgender identity (Cahill et al., 2014; Callander et al., 2021; Lombardi and Banik, 2016; Reisner et al., 2014). In Australia, best practice in data collection on sex and gender, sexual orientation, and trans and gender diverse classifications are set out in the “ABS Standard,” or the Standard for Sex, Gender, Variations of Sex Characteristics and Sexual Orientation Variables (Australian Bureau of Statistics, 2020), and endorsed by the National Health and Medical Research Council (National Health & Medical Research Council, 2024). However, it is not clear how the ABS Standard has been implemented, including in cancer clinical trials.
Research indicates LGBTQ+ individuals have unique experiences with cancer compared to cisgender and heterosexual people (Drysdale et al., 2021). Trans and gender diverse individuals in particular may encounter barriers due to gender affirming hormone use or exclusionary language in clinical trials, especially for cancers affecting reproductive or genital systems (Lee and Streed, 2024). Despite assumptions of underrepresentation, a 2012 US survey found higher participation rates among lesbian, gay, and bi+ individuals in cancer clinical trials, possibly due to later cancer diagnoses, meaning LGBQ+ people had fewer evidence-based treatments or had exhausted available treatment options (Jabson and Blosnich, 2012). However, there remains a lack of documented participation in clinical trials among trans and gender diverse people (Ludmir et al., 2020). These inequities for LGBTQ+ people build on existing gender disparities in the collection of sex and gender in medical research (Carcel et al., 2024). Ensuring equal trial participation for LGBTQ+ people and making their participation visible is crucial for equity, non-discrimination, and research integrity, allowing for a thorough analysis of sex, gender and sexuality-based impacts on trial outcomes.
Case study
This case study reports on a pilot study with two aims. The first aim was to develop a clear understanding of the state of LGBTQ+ data collection in cancer clinical trials in New South Wales (NSW), Australia’s most populous state. The second aim was to develop recommendations for practicable actions to support healthcare workers and researchers to improve inclusive LGBTQ+ data collection in cancer clinical trials in NSW. Understanding the participation rates of LGBTQ+ communities in cancer clinical trials in NSW is difficult due to lack of standardised reporting. Statewide clinical trial reporting systems and local health districts have electronic medical systems that currently do not capture data on gender and sexuality consistently (NSW Health, 2022).
To study these aims, we undertook a scoping review to map what is known about the collection of gender and sexuality data in cancer clinical trials, and key informant interviews with professionals (n = 10) working in cancer clinical trials to understand current practices and attitudes, and barriers and facilitators. The full details of these components were published in a publicly accessible report (Smith et al., 2025).
In summary, the scoping review addressed the question “What is known about the collection of gender and sexuality data in cancer clinical trials, current practices, and perceptions among healthcare professionals regarding barriers and facilitators to collecting this data?.” We examined peer-reviewed academic literature published from 1 January 2019 to 3 June 2024, yielding six publications that fit the search eligibility criteria (see Figure 1 and Table 1). Qualitative structured interviews were conducted with professionals working in cancer clinical trials (“key informants”) to document their perspectives about the state of data collection regarding gender and sexuality, and perceptions about barriers and enablers for improving the inclusion of LGBTQ+ people in cancer clinical trials. Data were analysed utilising interpretive description, with a focus on informing an understanding of barriers and enablers of LGBTQ+ inclusion in cancer clinical trials. Ethical approval was received from the UNSW Human Research Ethics Committee (iRECS6660) and the ACON Research Ethics Review Committee (202405).

Study selection for the scoping review.
Scoping review search terms and databases searched.

Findings
Scoping review
No articles identified in our scoping review focused on Australia. Articles included in our review reported current practices (data collection, annotation of records, trial documentation, and trial processes) related to gender and sexuality diversity (See Table 2 for included articles; Cathcart-Rake et al., 2019; Geffen et al., 2023; Jones et al., 2020; Kamen et al., 2022; Patel et al., 2023; Witham et al., 2020). Overwhelmingly, routine or reported gender and sexuality data collection was low. Each article indicated facilitators and barriers to gender and sexuality data collection, either directly related to participation in cancer clinical trials or in findings on broader oncology practice (recruitment, biobanking) relevant to data collection aspirations and practices. The articles indicated a range of factors impacting gender and sexuality data collection: aspirational principles, systemic and/or structural issues, healthcare professionals’ traits and experiences, and population and/or partner outreach efforts. Taken together, they indicated a whole-of-system response was required to increase LGBTQ+ participation in cancer clinical trials.
Articles included in the scoping review.
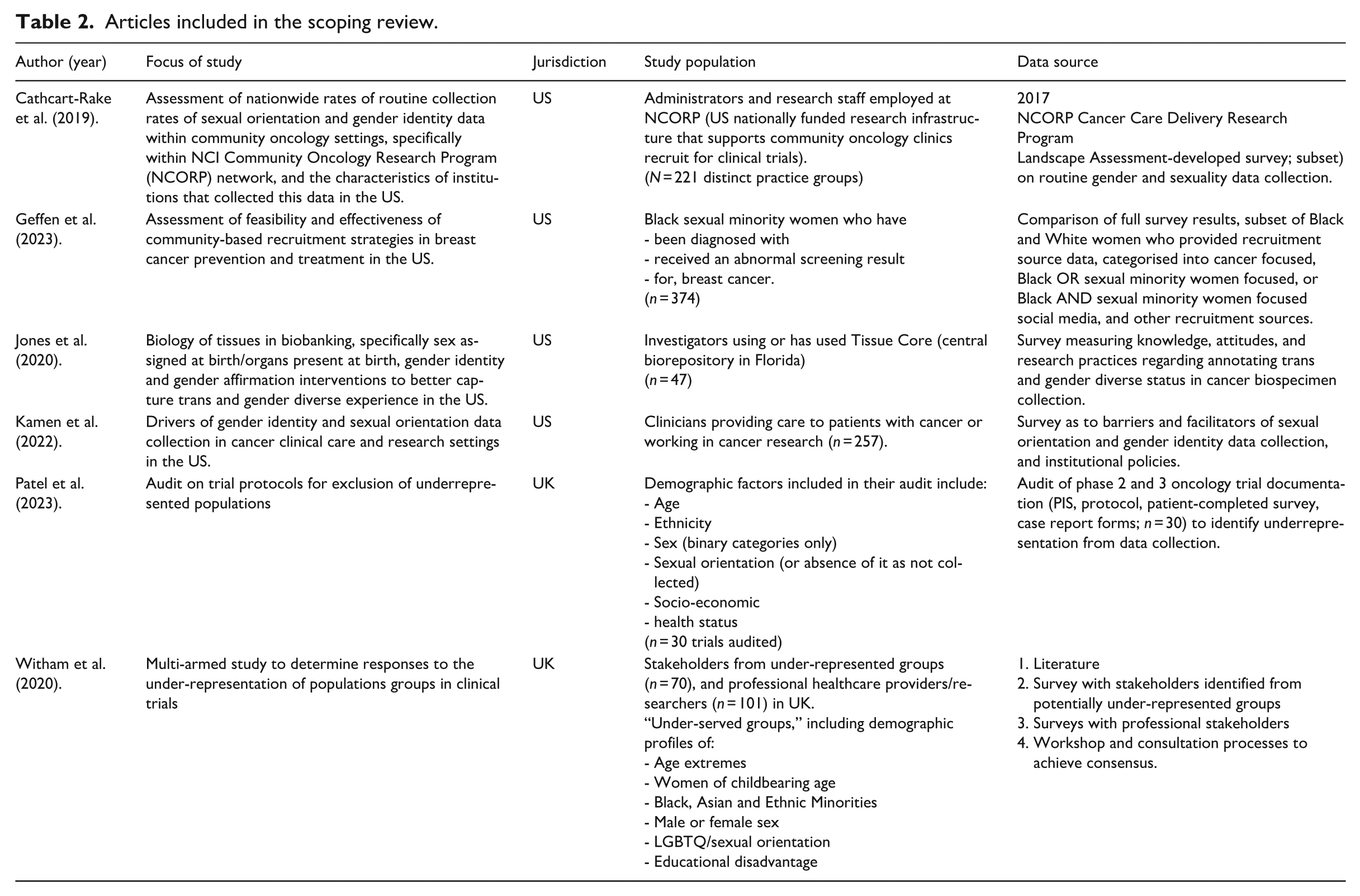
Institutional data collection in cancer care and research was significantly associated with healthcare professional’s belief that knowing patient gender identity and sexual orientation was important for high-quality care provision (see e.g. Kamen et al., 2022
Interview results
We interviewed 10 key informants who had a range of roles working in cancer clinical trials, including patient-facing roles (nursing or doctor), clinical trial coordination, and head of departments. Key informants were diverse in gender (five cis men, four cis women, and one non-binary), most worked in major city regions (three in inner regional areas), and all at publicly funded clinics. We identified a range of barriers and facilitators in current practices with respect to collecting gender and sexual orientation data in cancer clinical trials.
Barriers: Lack of training and cis/heteronormative standards
Gaps in training and knowledge were evident with informants reporting not having received training regarding gender identity and sexual orientation, and potential impacts on healthcare and engagement. This lack of training was reflected in some key informants never having considered whether gender identity and sexual orientation data collection were important.
Some key informants stated, “I treat everyone the same,” indicating they did not believe knowledge of a patient’s gender identity or sexual orientation was important to delivering person-centred care or to trial participation: I don’t think there’s [anyone who has not participated in a trial because of] not being treated appropriately because of your sexual affiliation or gender or whatever. So, I just don’t think we can do better. I think we do well. (P10)
However, others explained barriers for patients to LGBTQ+ participation in cancer clinical trials, and that they or their colleagues may require training to best support LGBTQ+ people or collect LGBTQ+ demographic data. Beyond general LGBTQ+ inclusivity training, informants indicated training topics that would be helpful. For example, some were uncertain about management of hormone therapy trans patients may be taking and its impact on trial eligibility. Key informants were unsure about the potential impacts on treatment and had not considered potential psychosocial implications if patients were asked to cease hormone therapy as part of trial participation.
Key informants reported awkwardness discussing sex and contraception. Some were familiar with the issues involved in cancer clinical trials related to chemotherapy, where discussion of safer sex practices is standard to protect patients’ sexual partners from cytotoxic agents, but this information had not been tailored for LGBTQ+ people, and may require training to address.
Key informants noted organisational practices and record keeping systems were not set up to consider patients’ gender and sexuality. Consequently, minimal approaches are used: for example a single question about sex or gender, or assuming gender based on presentation.
It’s usually just their gender, but we don’t really ask sexuality, identification or orientation. So, if patients don’t tell us we wouldn’t know they are gay or they are lesbians. So that’s the thing if they don’t tell us, we wouldn’t know. (P05)
Key informants who attempted to use LGBTQ+ inclusive language in their consultations to avoid making assumptions explained it could be challenging to maintain as a standard approach with all patients, especially as they were not supported to manage difficult conversations.
Trying to reflect back, when I was more junior, I asked questions, particularly to older people, one about pronouns and about relationships. . . but the older males feel quite confronted with a question about sexuality. So, you know, I backed off a little bit with that. (P11)
Key informants who identified the importance of inclusive language explained it was common to see exclusionary language used in cancer clinical trial information or they found it difficult to implement inclusive language in consultations. Exclusionary language included trial eligibility or information referring to a binary of men or women but failing to clarify if it was inclusive of trans people, implying sexual organs exclusively belong to specific genders (e.g. implying only men have testes), and using language that assumes the gender of a partner (e.g. assuming a woman who is married has a husband).
Cancer clinical trial protocols and participant information statements were also described as excluding LGBTQ+ people through the explicitly exclusionary language used. This highlighted the lack of consideration of trans people, particularly trials in sex organ cancers: the language we use in clinical trial protocols does exclude patients . . . for example, that in a prostate cancer trial, patients must be male. I mean, I personally think [gender is] irrelevant as long as they have a prostate. (P11)
Facilitators: Support for inclusive practices and enhanced data collection standards
All key informants were supportive of including standardised questions about LGBTQ+ status on patient intake forms or during history taking, and suggested ways to improve LGBTQ+ inclusivity.
They supported the use of inclusive signs or symbols (e.g. badges, flags and lanyards) to support patients feeling comfortable to disclose. Some key informants proactively used signs and symbols to indicate LGBTQ+ allyship and inclusivity, a strategy that is evidence based (Haire et al., 2021).
Increasing the capability of the cancer clinical trial workforce to cater specifically for LGBTQ+ patients was viewed by many as part of providing person-centred care, and identified it as improving overall quality of care: I want patients feel comfortable being treated and respected. I don’t want to make any [mistakes] . . . If they don’t feel comfortable to tell us some very private information, we may miss that data collection as well. (P05)
Improved care requires respect and trust, which ultimately improves care and cancer outcomes. Key informants reported that ensuring people feel supported is critical and should be present when referring people to cancer clinical trials: You have to make people feel supported; they need to feel comfortable to share information with you . . . the rapport really kicked off [with a patient] once we broke through the ice with her saying something like that’s my partner, not my friend. (P06)
Multiple points in the system were identified by key informants where data could be collected to improve cancer care and trial participation. If sexual orientation and gender identity questions were mandated as a requirement of running cancer clinical trials, it would be easily implemented in intake forms: In the waiting room, while they’re filling out forms. I mean, [we ask] what’s your age, what’s your date of birth, where do you live, male or female? Are you Aboriginal background or not? That’s probably where we should be having a question. It shouldn’t be well into the interview, well into this process to say, “oh, hold on, how do you want to be managed?” (P08)
Participants talked about history taking as the opportunity for learning about patients’ sexuality through questions about social support. Others talked about the formal trial enrolment when forms are filled out as being the “natural” place, while a third group relied on information collected through referrals. It was also suggested that standardising or even mandating the approach for all clinics would make it easier to implement, as it would simply become a routine part of cancer clinical trials.
Recommendations
LGBTQ+ populations face a range of implicit barriers to accessing clinical trial participation that may be addressed by training of staff and improving data collection processes so that LGBTQ+ populations are visible. Thus, explicitly including gender and sexual identity data in cancer clinical trials is essential to increasing accurate representation of LGBTQ+ people in research and clinical practice. Greater support of researchers and clinicians will improve the ethical collection of reliable data. This includes the need to disaggregate gender from sexuality, and recognising the diversity of populations hidden by the term “under-served” populations. Best practice in data collection on sex and gender, sexual orientation, and trans and gender diverse classifications are set out in the ABS Standard (Australian Bureau of Statistics, 2020). This standard recommends use of a gender two-step question (sex reported at birth and gender identity) to collect data on gender diversity. There is a clear need for training the cancer clinical trial workforce to ensure awareness of gender diversity, impacts of gender affirming interventions, and the complex relationships which impact cancer clinical outcomes. Standardising collection of sex, gender, and sexual orientation data in clinical trials will normalise asking these questions with the aim of improving inclusiveness of care and cancer clinical trial participation.
Although there is a growing literature on the use of standardised LGBTQ+ data collection (Cahill et al., 2014; Callander et al., 2021; Lombardi and Banik, 2016; Reisner et al., 2014), it is necessary to monitor how data standards are implemented in specific research areas. Beyond adding in these standard questions, it is also vital to consider how gendered norms and language operate within data collection. As this case study has identified, the ways in which anatomy and specific types of cancer are gendered impact LGBTQ+ inclusivity, with implications for the way inclusion and exclusion criteria are framed, for interactions with hormonal treatment, and discussion of sexual practices and assumptions about partners and families. Ensuring justice and data equity related to LGBTQ+ populations requires attending to these specific nuances in different areas of research focus.
Based on the findings of this pilot study (Smith et al., 2025), our actionable recommendations are provided in Table 3. To support workforce training and organisational capacity to work with LGBTQ+ populations, providing cancer clinical trial teams in Australia with a consolidated list of existing LGBTQ+ inclusive resources and training packages will be necessary (Cathcart-Rake et al., 2019; Jones et al., 2020; Kamen et al., 2022). National ethical bodies and funders are increasingly expecting reporting on sex, gender, and sexuality variables in medical research (Carcel et al., 2024; National Health & Medical Research Council, 2024), but it will be necessary to upskill human research ethics committees to promote standards (Australian Bureau of Statistics, 2020) and to promote LGBTQ+ inclusive research practices. There is also scope to mandate minimum datasets, including the collection of sex, gender, and sexuality. Finally, more specific co-designed resources, including guidelines and training packages should be developed for cancer clinical trials. Achieving improved LGBTQ+ data equity in cancer clinical trials will ensure more ethical research practices and also contribute towards improving health outcomes for LGBTQ+ people in cancer clinical care (Saxby and Hammoud, 2025).
Practical strategies to improve inclusion of LGBTQ+ people in cancer clinical trials.

Footnotes
Acknowledgements
We thank all the stakeholders who took part in our interviews. We also thank Gianfranco Giuntoli and Emma Kirby for their contributions to this project.
Ethical considerations
Ethical approval was received from the UNSW Human Research Ethics Committee (iRECS6660) and the ACON Research Ethics Review Committee (202405).
Consent to participate
All key informants provided informed consent prior to participating in an interview.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Cancer Institute NSW and ACON.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Bridget Haire is an editor-in-chief and Anthony K. J. Smith is an associate editor of Research Ethics.
Data availability statement
Research data are not shared.
