Abstract
The transfer of biological materials is pivotal in health research, especially in clinical trials conducted in sub-Saharan Africa (SSA). Standard material transfer agreements (MTAs) have been developed to exercise due diligence during the transfer of biological materials. Recent controversial conduct in research in SSA highlights the need for robust regulation, especially concerning biological sample transfer. This study evaluated the sufficiency of Zambia’s existing regulations concerning MTAs using the South African regulatory framework as a comparator. A qualitative study approach was adopted utilizing a desk review. Thematic analysis was conducted using relevant, publicly accessible national regulatory documents. All necessary approvals were obtained. Both countries have established provisions for MTAs in health research, although the legal status of the respective provisions differ. Zambia’s MTA is based on the National Health Research Act No. 2 of 2013 and mandates that while samples and data remain the property of the Zambian government, research participants must share in future benefits that arise. In contrast, South Africa’s MTA (2018) is a framework agreement, outlining the contractual aspects of biological sample sharing in research. It highlights ownership of samples as resting with the “donor,” benefit sharing, an ongoing consent process, secondary use of materials and dispute settlement. Both nations emphasize intellectual property rights, privacy with respect to samples and associated data. In Zambia, it is a legal requirement for contracting parties to sign a MTA. Ownership of biological samples vests with the Zambian government. On the other hand, the South African MTA (2018) is regarded as a regulatory framework and is contentious especially with respect to ownership and inclusion of the REC as a contractual party. In both countries, enforcement and implementation remain critical concerns in collaborative research involving sharing of biological samples. Consequently, the MTAs need to be reviewed. Reaching consensus is a research ethics imperative.
Keywords
Background
International collaborative health research is growing exponentially including on the African continent. The collection, storage and transfer of biological materials represent important components for such research and are crucial for medical innovation. Material Transfer Agreements (MTAs) facilitate biological material transfer and are pivotal in the governance of collaborative research, especially in sub-Saharan Africa (Nielsen et al., 2018). However, when biological samples are transferred abroad, several concerns arise including controversial concepts like ownership, custodianship, consent and benefit sharing. Furthermore, concerns about national sovereignty and national interest have led to the creation of regulatory instruments (Rusakaniko et al., 2019). The potential for commercialization of such research escalates these concerns (Blakeley et al., 2019; Moodley and Kleinsmidt, 2020).
Material Transfer Agreements are legal instruments that govern the transfer of biological materials in collaborative research (Brett-Major, 2020; Bubela et al., 2015). In Zambia, historically, these transfers were regulated by health research ethics committees (HRECs). However, international governance frameworks have led to stricter legal frameworks at country level, as outlined in the National Health Research Act (National Health Research Act, 2013). Material Transfer Agreements govern the transfer of tangible research materials, including reagents, cell lines, plasmids, vectors, and associated data (Autm.net, 2024), and range from simple documents to complex agreements. These agreements may include provisions for intellectual property rights (Autm.net, 2024), and may also include clauses for regulating ownership and commercialization.
There are several typical examples of MTAs that have been developed in collaborative research, such as the Uniform Biological Material Transfer Agreement (UBMTA) developed by the National Institutes of Health (Autm.net, 2024) and the Standard Material Transfer Agreement 2 (SMTA2) developed by the World Health Organization (WHO, 2016). While these agreements govern and facilitate the sharing of biological samples and data (Brett-Major, 2020; Margoni, 2013), there are ethical concerns surrounding their application (Kalkman et al., 2019). This study explored the governance of biological sample sharing in health research in Zambia and South Africa and determined if current laws and guidelines were sufficient to safeguard the rights and well-being of health research participants and research institutions.
The governance of research in sub-Saharan Africa is often attributed to suboptimal regulatory systems, guidelines and laws (Bain et al., 2022; Thurtle et al., 2021). To address this, many countries have enacted laws in recent years to regulate health research, including the transfer of biological materials. South Africa and Zambia were selected as case studies due to the high scores they attained for key parameters in a survey conducted by the World Health Organization African Region (WHO AFRO) and the European and Developing Countries Clinical Trials Partnership (EDCTP) to demonstrate strengthening of national health research systems in sub-Saharan Africa (Rusakaniko et al., 2019). Laws, MTAs and research ethics guidelines have also been developed. However, it is uncertain to what extent current governance mechanisms are enabling ethical conduct and safeguarding research participants in collaborative research involving biological samples. Research regulatory authorities often struggle with implementation of MTAs, regulations and guidelines. This paper is based on an evaluation of the adequacy and sufficiency of the Zambian MTA, focusing on the South African context as comparator. The work has the potential to raise awareness about important ethics issues implicit in MTAs, surface existing contentions and stimulate regulatory reform where necessary. This is especially important as the African Union encourages adoption of access and benefit sharing agreements (African Union Commission, 2015).
Methods
This study constitutes a segment of a comprehensive investigation into the ethical and legal challenges associated with consent and the transfer of data and biological samples in Zambia. We conducted a documentary analysis of existing legislation and guidelines pertaining to MTAs in Zambia and South Africa. The study employed a qualitative research approach, utilizing an exploratory case study design (Priya, 2021; Yin, 2009), commencing with a desk review. There was an in-depth engagement with the regulatory frameworks to assess the extent to which they safeguard research participants and institutions. In South Africa, the primary documents reviewed included the National Health Act, 2003 (Act No. 61 of 2003) and the Material Transfer Agreement for Biological Materials (SA MTA Notice no. 719 in Government Gazette no. 41781 dated 20 July 2018). In Zambia, we examined four national documents: the Health Research Act (National Health Research Act, 2013), Statutory Instrument No. 92 on MTAs (2018), the official MTA form, and the guidelines for transfer of biological materials for research and quality assurance purposes. All documents were sourced from official government websites to ensure authenticity and traceability.
The analysis adhered to thematic analysis principles as delineated by Braun et al. (2022). The process involved the following steps:
Familiarization: Multiple readings of the documents to gain an overview of their content and structure.
Initial coding: Using Atlas.ti version 22, we uploaded the documents and generated initial codes. These codes reflected recurring ideas, requirements, and regulatory principles related to MTA content, ethical obligations, institutional roles, and compliance.
Theme development: Codes were grouped into broader themes using both deductive categories informed by the research question (e.g., consent, benefit sharing, oversight) and inductive themes that emerged during coding.
Theme refinement: Themes were reviewed against the coded data and full documents to ensure internal consistency and distinctiveness.
Theme naming: The essence of each theme was identified and the aspect of the data each theme captured determined. Clear and informative names for each theme were developed.
The final step consisted of a cross-case comparison: Themes were analyzed for similarities and differences between the two countries’ approaches to MTA governance.
This structured approach enabled us to systematically interrogate the content and intent of the regulations while being responsive to nuanced differences in context and ethics.
Validity and reliability
To ensure rigor and trustworthiness, the study employed multiple strategies: Document selection prioritized official, national-level regulatory instruments, thereby ensuring authenticity, representativeness, and credibility, in accordance with guidance on document analysis (Morgan, 2022). The use of thematic analysis was rendered transparent and reproducible through the application of qualitative data analysis software (Atlas.ti), facilitating an audit trail of codes and theme development. Investigator triangulation was utilized, involving both Zambian and South African legal and ethics scholars in data interpretation, which helped mitigate individual researcher bias. To support dependability, we applied a consistent analytical lens across both jurisdictions, employing the same thematic categories and coding procedures. Reflexivity was maintained by keeping analytical memos and reviewing assumptions during team discussions, particularly when resolving coding disagreements. While qualitative document analysis relies on interpretive judgment, these strategies enhanced the transparency, credibility, and confirmability of the findings.
Ethical considerations
The Zambian National Health Research Authority provided the authorization to conduct the research. Ethics approval was obtained from the University of Zambia Biomedical Research Ethics Committee (REC) reference number 1420-2020, and Stellenbosch University HREC Ethics Reference No: S20/10/278 (PhD). The study did not include interaction or intervention with human participants.
Results
This section focuses on the study findings based on themes that emerged from the analysis of the current regulatory documents in Zambia and South Africa.
While MTAs are legal documents establishing contractual relationships between partners, they exist in a specific context made up of several interrelated ethics issues stemming from the need to govern sovereign interests and protect vulnerable parties in a relationship (van Wichelen, 2023). Out of the eleven (11) major themes identified as the core areas that MTAs should cover, Zambia only had seven (7) in place, as indicated in Table 1.
Emerging themes: Similarities and differences between the material transfer agreements of Zambia and South Africa.
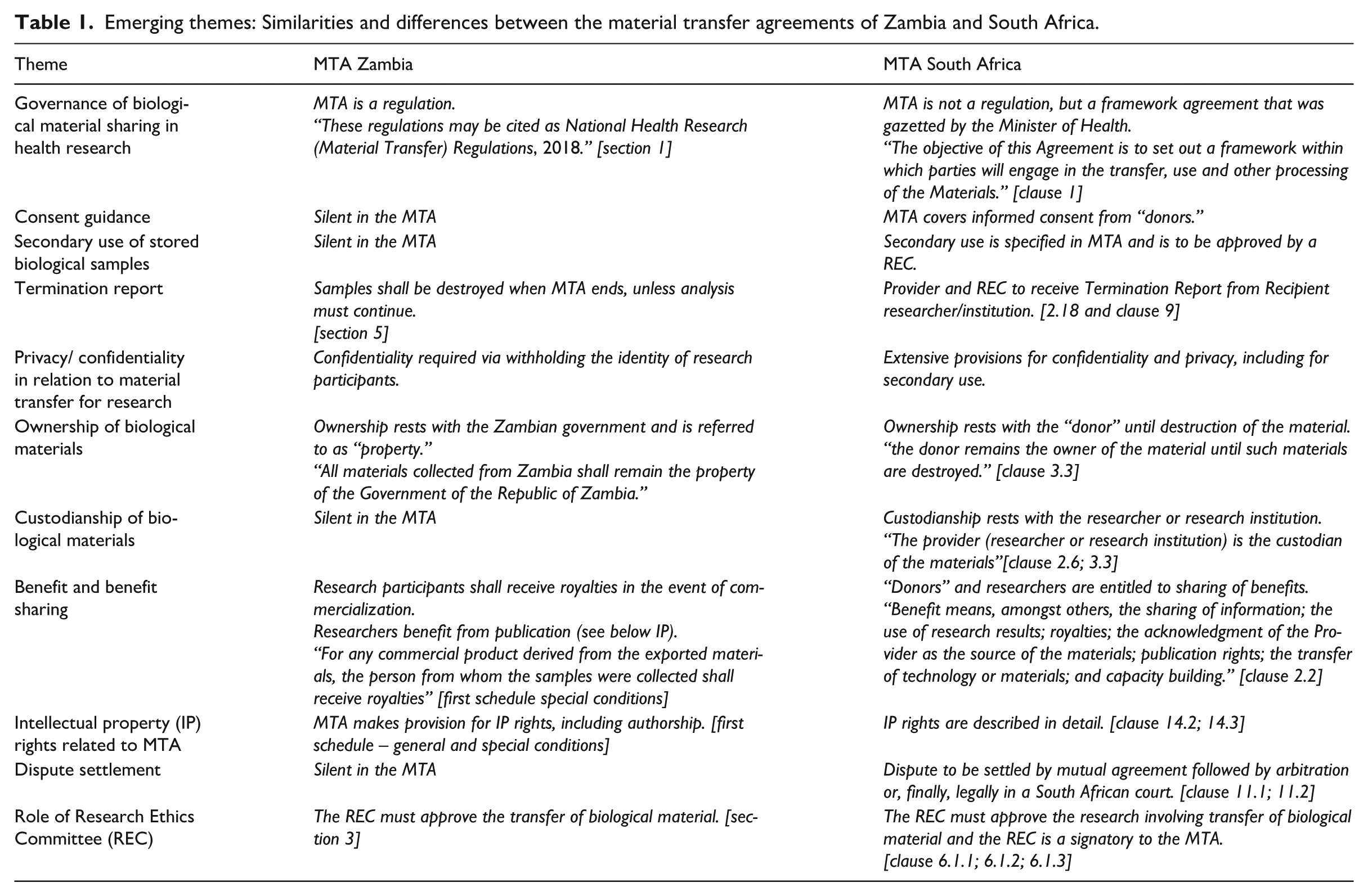
Theme 1: Governance of biological material sharing in health research
In the MTA documents accessed and analyzed, the text for the main reference document from Zambia refers categorically to the Material Transfer Agreement as regulations, and that “these regulations may be cited as National Health Research (Material Transfer) Regulations, 2018.” [National Health Research [Material Transfer] Regulations – S.I. 92/2018]. In contrast, the document from South Africa indicates that it provides a framework, “the objective of this Agreement is to set out a framework within which parties will engage in the transfer, use and other processing of the Materials.” (SA MTA Notice no. 719 in Government Gazette no. 41781 dated 20 July 2018)
The National Health Research Act in Zambia, makes clear reference to provisions that give power in the case of Zambia and guide health researchers when they conduct health research involving material transfer. These include specific requirements including the “power of the board over human and animal research” and powers of the Authority over biological materials.” (National Health Research Act No. 2 of 2013).
Comparatively, the South African National Health Act highlights provisions for the purpose of donation of human biological materials as being for health research and to advance health sciences and gives the Minister powers to make regulations regarding “the importation and exportation of tissue, human cells, blood, blood products or gametes.” [National Health Act, 61 of 2003. In both countries, the documents have provisions that require approval for biological material transfer and approval of material transfer research, “no material shall be transferred for purposes of a health research project that has not been approved by an HREC.” (SA MTA Notice no. 719 in Government Gazette no. 41781 dated 20 July 2018)
Theme 2: Consent guidance
The Zambian MTA is silent on consent guidance related to material transfer; however, this aspect is covered in the law. The National Health Research Act No. 2 of 2013 defines consent as “voluntary agreement to participate in health research by a non-minor individual, capable of understanding potential risks and benefits.” It defines “human participant” as “any living person who consents to participate in health research and further includes deceased bodies as provided in the Human Tissue Act.” The Act prohibits removing tissue, organs, blood, or gametes from a living person without written consent. The Zambian Act further stipulates “A person shall not withdraw blood, blood products, tissue or gametes from a living person for any unspecified future health research activity or unspecified storage.”
The South African MTA describes informed consent as a “a formal agreement signed by a donor with legal capacity to give permission for material donation.” The document elaborates on informed consent, namely, that it “includes an ongoing information sharing process which allows a Donor to consent to participate and determine whether and how their Materials will be utilized in the Project, as approved by the HREC from time to time,” allowing donors to reconsider consent and determine usage. “Researchers must document consent in relevant forms and inform donors of developments in the project.” The SA MTA further guides that the provider must submit the informed consent form for secondary uses of the material to the HREC should the need arise for secondary use. As such, informed consent in South Africa can be viewed as a form of broad consent, with an element of dynamic consent given that ongoing participant involvement is included. The South African National Health Act of 2003 in section 71, outlines the process for obtaining consent for health research or experimentation with humans.
Theme 3: Secondary use of stored biological samples
The MTA in Zambia is silent on this important aspect related to material transfer studies. However, Section 47, subsection 2 of the National Health Research Act of 2013, states that “a person shall not withdraw blood, blood products, tissue, or gametes from a living person for any unspecified future health research activity or unspecified storage.” Hence the Act permits secondary use of samples for specified future health research.
The South African MTA, on the other hand, provides for secondary use of biological samples in future research that extends beyond the original research question. Secondary use is defined in clause 2 as the “use of the materials for health research purposes other than the uses determined in the approved protocol.” The South African MTA states in clause 6 that secondary use of biological samples applies to health research, as contemplated in section 1 of the National Health Act, 2003. Furthermore, all HRECs are required to review and approve health research proposals and protocols, including for the secondary use of biological samples.
Theme 4: Termination report
In Zambia, the MTA refers to termination as part of the general conditions of an MTA and the regulations (National Health Research [Material Transfer] Statutory Instrument No. 92 of 2018) guide what happens at the termination of the agreement. It stipulates in section 5, subsection 3 that “where an MTA has been terminated, the parties shall destroy the samples unless the analysis is one that cannot be abandoned.” The National Health Research Act and regulations do not clearly state the need for a termination report.
Comparatively, the South African MTA gives a detailed account of the termination report, which in clause 2, “means a report prepared by the recipient and submitted to the provider on the termination of the project.” The South African MTA provides the details of what to report on, safeguarding the materials, and specific responsibilities of the recipient. Further, upon termination of the project, the recipient is guided to immediately discontinue using the material for any purpose whatsoever. Destruction or return of the biological samples to the provider, transfer of materials, or any other arrangements made will be undertaken by the recipient, with the express approval of the HREC. For South Africa, the National Health Act of 2003 does not provide for termination reports.
Theme 5: Privacy and confidentiality in relation to material transfer for research
In Zambia, the MTA of 2018 in section 5 uses anonymity as a means of maintaining confidentiality. It further states that “the parties to an MTA shall ensure that confidentiality is maintained by withholding the identities of participants.” This is backed up by the National Health Research Act, section 53, which states that “the Minister shall, in consultation with the Authority, prescribe and specify the requirements and contents of an MTA and shall provide for the terms and conditions regarding intellectual property rights, publication rights, the various uses of the material, including reproduction and replication, confidentiality of information and transfer to third parties and liabilities.” The National Health Research Act in Part III section 20, further stipulates that failure to uphold privacy and confidentiality constitutes research misconduct.
On the other hand, the SA MTA stipulates in clause 13, the safeguards for confidentiality and requires that the “recipient shall always keep the identity of the donor(s) and the materials secure and confidential. Confidentiality includes, but is not limited to, the properties, characteristics, content, composition, potential secondary uses or methods of use of the material. The provider and the recipient shall treat all the information relating to the nature and processes of the research in whatever form as confidential.” The National Health Act of South Africa provides only general confidentiality, as it applies in healthcare settings.
Theme 6: Ownership of biological materials
Zambia, according to the MTA terms and conditions set out in form IV, provides for ownership of materials and the relevant rights. The MTA stipulates that “all materials collected from Zambia shall remain the property of the Government of the Republic of Zambia. For any commercial product derived from the exported materials, the person from whom the samples were collected shall receive royalties.” The National Health Research Act, section 53, states that “the Minister shall, in consultation with the Authority, prescribe and specify the requirements and contents of an MTA and shall provide for the terms and conditions regarding ownership of the material, including any of its derivatives and modifications.”
The SA MTA states in clause 3, that “the donor remains the owner of the material until such materials are destroyed.” According to the National Health Act of 2003, there is no explicit mention of ownership of donated biological material.”
Theme 7: Custodianship of biological materials
In Zambia, neither the MTA nor the National Health Research Act make reference to custodianship.
In contrast, the SA MTA stipulates that “the provider (researcher or research institution) is the custodian of the materials, and the donor (human participant) remains the owner of the material until such materials are destroyed.” This provision is legally disputed since it conflicts with the provisions of the NHA. The South African National Health Act of 2003 does not mention custodianship, and it does not explicitly define ownership of biological material.
Theme 8: Benefits and benefit sharing
In Zambia, the MTA states “for any commercial product derived from the exported material, the person from whom the samples were collected shall receive royalties. In the National Health Research Act section 35 highlights the issue of benefits, stating that “the Minister, in consultation with the Authority, establish mechanisms for involving communities in health research and make regulations for the protection of interests of stakeholders and the sharing of risks, benefits and outputs in health research programmes and activities.”
In contrast, the SA MTA in clause 2 indicates that “benefit means among other things, the sharing of information; the use of research results; royalties; the acknowledgment of the Provider as the source of the materials; publication rights; the transfer of technology or materials; and capacity building.” It further defines “benefit sharing as a process or act of sharing benefits that derive from the Project in a manner that is fair and equitable.” In clause 7, the South African MTA provides for benefit sharing and encourages that the sharing of benefits should be discussed and negotiated between the provider and recipient before materials are transferred to the recipient. On the other hand, the South African National Health Act, 2003, indicates that “A person from whose body, human biological material is withdrawn may only be reimbursed for reasonable expenses incurred by him or her in order to effect the donation concerned” as defined in section 60(4) of the Act.
Theme 9: Intellectual property rights related to material transfer agreements
In Zambia, the MTA stipulates, under its general and special conditions, that publications coming out of an MTA should acknowledge the source of the material. The MTA should include terms related to “intellectual property rights and patents; rights to authorship; benefits to the health researcher, community or individual(s), where applicable; and access to the data generated from the study.” The National Health Research Act of 2013, under section 53, highlights the issue of intellectual property rights and stipulates that “the Minister shall, in consultation with the Authority, prescribe and specify the requirements and contents of an MTA and shall provide for the terms and conditions regarding intellectual property rights, publication rights, the various uses of the material, including reproduction and replication, the confidentiality of information and transfer to third parties as well as liabilities of these parties.”
In contrast, the SA MTA provides for intellectual property rights and stipulates that they are dealt with through relevant laws related to the applicable protocol and underlying third-party agreements, as far as these are relevant. In the SA MTA, clause 2.11, intellectual property rights are defined as meaning “statutory and other proprietary rights resulting from outputs created by the human mind, such as copyright, patents, scientific works, discoveries, and trademarks.” Clause 14 of the South African MTA states that, “where the recipient wishes to publish any information concerning the project (in either oral or written form), the provider must be notified and provided with a copy of the publication at least ten (10) days prior to submission of the proposed publication. The provider must be supplied with a final copy of the publication before it is published by the recipient. The recipient must acknowledge the provider’s contribution of the material unless otherwise requested by the provider.” In South Africa, the National Health Act does not refer to intellectual property rights.
Theme 10: Dispute settlement
In Zambia, neither the MTA nor the National Health Research Act refers to dispute settlement.
Conversely, in South Africa, the MTA, clause 11, deals with dispute settlement and specifies in clause 11.1 that “Should a dispute arise between the Parties in connection with this Agreement, the Parties must, within a period of fourteen (14) days after the date on which the dispute arose (the Dispute Date) meet to discuss the dispute and endeavor to resolve the dispute amicably, by mutual agreement.” The MTA guides that, should the dispute fail to be resolved, it must be taken to court, which would be a costly exercise. In clause11.3, it stipulates that “If senior management of the respective Parties are unable to resolve the dispute within a period of thirty (30) days after it has been referred to them, either Party may institute action in accordance with South African laws, in a South African court, unless the Parties agree to resolve such dispute by arbitration in terms of a separate arbitration agreement.” The National Health Act is silent on this issue.
Theme 11: Role of research ethics committees in material transfer agreements
In Zambia, the MTA is silent on the role of the HREC as regards material transfer studies. However, according to the National Health Research Act of 2013, HREC means a committee registered and accredited by the Board under section eighteen. Further, section 17 of the Act stipulates that “all proposals for health research under this Act shall be reviewed by the Board or any other accredited health research ethics committee, as may be prescribed, and shall be approved by the Board.” This was further supported by the text highlighting that “transfer of biological materials without approval is punishable.” [National Health Research Act, 2013] Specifically in the Zambian MTA, there is guidance that “application for material transfer is made to Authority.”
In contrast, the SA MTA in clause 3.2 indicates that “the Parties agree to conduct themselves hereunder in compliance with South African laws and policies, that no Materials shall be transferred for purposes of health research project that has not been approved by an HREC.” To this effect, there is a footnote 2 that further guides that “as all health research ethics committees are required to review and approve health research proposals and protocols (footnote 3), approval of secondary uses of Material must be obtained from a health research ethics committee.”
Further, the NHA similarly defines “health research ethics committee” to mean any committee registered in terms of section 73. In section 72, the Act provides for the establishment of the National Health Research Ethics Council (NHREC) and its supervisory role over HRECs including, in subsection 6b, to register and audit HRECs. In section 73, the Act stipulates that the “health research ethics committee must (a) review research proposals and protocols in order to ensure that research conducted by the relevant institution, agency or establishment will promote health, contribute to the prevention of communicable or non-communicable diseases or disability or result in cures for communicable or non-communicable diseases; and (b) grant approval for research by the relevant institution, agency or establishment in instances where research proposals and protocol meet the ethical standards of that health research ethics committee.”
Discussion
Based on a detailed review and analysis of the MTAs of Zambia and South Africa, the results are discussed thematically comparing and contrasting aspects presented in the results section above.
Governance of biological material sharing in health research
The study established that both Zambia and South Africa have specific laws and guidelines that have been implemented to regulate and guide the use of biological materials in research. In Zambia, it is a legal requirement to sign MTAs before health research involving the transfer of biological materials is undertaken as stipulated in the Health Research Act and the regulations.
In South Africa, the SA MTA provides ethical guidance for parties engaging in health research involving biological material transfer. However, the legal status of the SA MTA has been challenged by some scholars (Esselaar et al., 2024; Kamau et al., 2024; Thaldar and Shozi, 2021, 2023) who argue that the SA MTA is not a regulation in terms of the NHA, which implies that it is not subordinate legislation that could be enforced. There are further arguments that the MTA refers to itself as a framework that was published for public notice, implying that the substantive provisions of the MTA could be changed by parties who want to establish an MTA and that the published MTA seems to be a guide and not a legal blueprint. Future revisions of the South African MTA should resolve any ambiguities for researchers and HREC members alike.
Consent guidance
Generally, there is no uniformity in models for informed consent, with different settings adopting models that address their unique challenges (Grady et al., 2015; Krogstad et al., 2010). While the Zambian Health Research Act promotes specific consent for the research question under study and any future research, the South African MTA makes provision for a broad consent approach including the secondary use of biological samples obtained during research. Specific consent is provided by research participants after being adequately informed about the aims, benefits and burdens of a particular study before each new study is undertaken, and the consent only covers the specific study in question (Maseme et al., 2023; Wiertz and Boldt, 2022). The Zambian National Health Research Act, refers to specific consent as it prohibits unspecified future studies and unspecified storage. (Grady et al., 2015). The Zambian Act prohibits collecting samples without consent and material sharing without specific consent. In South Africa, informed consent is emphasized through broad consent, which requires that a HREC will approve future/secondary research projects. While a broad consent model is more suited to research involving biological samples, in South Africa, there is contention around broad consent and specific consent, as the latter is supported by the Protection of Personal Information Act 4 of 2013 (American Bar Association, 2021). Although the SA MTA also alludes to a dynamic form of consent with continual engagement of research participants/donors, this may not be feasible if samples are anonymized. Likewise, geographic mobility in SA may make it challenging for sample donors to be contacted years after the original study. It is also possible that donors may be deceased several years after the original study. Dynamic consent often requires access to digital technology or devices for continual long-term engagement with participants. In SSA, research participants or donors may not have access to such technology or access to the internet to engage on the digital platforms required for dynamic consent.
In line with the consent guidance, both countries require consent for participants with legal capacity, and have special provisions for consent when minors are involved as research participants in all types of research. There are no special provisions where biological samples are collected during research on minors (Wolf and Lo, 2004). In the literature and in practice there has been a shift to promote broad consent which creates flexibility and efficiency (Maloy and Bass, 2020; Mungwira et al., 2015). It is a considered alternative to traditional specific consent where participants agree to a particular study only, as is the situation in the Zambian context.
Secondary use of stored biological samples
Secondary use of biological material is encouraged as has been provided for in the SA MTA. This is because, it reduces the recruitment burden on new participants for every research question. Secondary use of samples is also useful to validate some research findings. Despite the value that secondary use of biological samples holds, the Zambian Health Research Act lacks reference to secondary use of research material. The Zambian approach could be due to other provisions that guide on prohibition of unspecified future health research and unspecified storage. The inherent uncertainty of future research could possibly explain why the Zambian Act, regulations and guidelines are silent on secondary use of biological samples. Revisiting the Zambian Health Research Act to explicitly discuss the secondary use of research material could be an important next step (Mungwira et al., 2015).
Termination report
Termination reports have been adopted in some local material transfer templates (Witshealth.co.za, 2025). The South African MTA guides what happens at termination of a research project, regarding project status and materials. Other similar guides exist in line with the termination report; for example, in the United Kingdom, the Lambert MTA template mandates sample care and destruction at the donor site (Service.gov.uk, 2025). Destruction of samples is also an important aspect of privacy protection. The Zambian Act does not refer to this important aspect of research and the MTA lacks detail. This represents an area for future development.
Privacy and confidentiality
Confidentiality is crucial for human participant protection in health research (Bos, 2020) encompassing informational and physical privacy. Researchers must consider participant interests in Institutional Review Board (IRB) or REC protocol applications, ensuring they adhere to laws that outline confidentiality. This includes controlling access to personal information. Both countries’ health research laws provide a description of confidentiality.
Ownership of biological materials
Both countries’ laws highlight that ownership of materials and their derivatives is not in the hands of the researchers. The Zambian government owns biological materials in terms of the Zambian Act, while there is no similar provision in the South African National Health Act. In as much as the SA MTA clause 3, has some provision for a donor to be the owner, this stipulation is disputed since there is no clear legal basis in the NHA. This is furthermore in conflict with Regulation 26 of the Regulations regarding the General Control of Human Bodies, Tissue, Blood, Blood Products and Gametes (No. R.180 dated 2 March 2012), which stipulates that the recipient of donated material gets exclusive rights in such material, thus also ownership. This position is supported by Thaldar et al and Esselaar et al who argue, that the legal use of the term "donation" in the NHA necessarily entails a transfer of ownership to the recipient/institution. Furthermore, the NHA mandates that such donations must be made to specified institutions, such as universities. This legal framework contradicts the SA MTA’s claim that research participants retain ownership of donated material (Esselaar et al., 2024; Thaldar, 2023; Wismayer (2024)). Ownership of biological materials remains controversial in Africa and globally (Petrini, 2012; Thaldar, 2020). In the United States of America, cases like Moore versus the University of California, Greenberg versus Miami Children’s Hospital Research Institute, and Washington University versus Catalona took a legal position that a research product developed from human tissue is distinct from the original tissue, becoming the property of the researcher or recipient (Passmore et al., 2024). It should, however, be noted that these cases occurred in jurisdictions outside Africa and therefore do not create any legal precedent in Zambia or South Africa.
The South African National Health Act does not include an explicit provision for ownership of donated material but specifies that the donor cannot be given any financial or other reward for biological material or organs. It is, however, not clear if this stipulation also includes research samples. While most participants support providing samples for research, concerns about future use, benefit sharing, and exporting samples are raised (Moodley and Kleinsmidt, 2020). Participants may be more willing to participate if they feel more in control of the use of their samples (Piasecki and Cheah, 2022).
To provide remedial considerations, Piasecki et al. explored ownership issues in data transfer, highlighting pitfalls and suggesting a compromise through a data governance structure like a data access committee or a subcommittee of HREC or a National Ethics Committee for fair access (Thaldar et al., 2020).
Custodianship of biological materials
The Acts in both countries lack custodianship provisions. However, the South African MTA, clause 3, clarifies that the provider is the custodian. This stipulation is disputed since there is no clear legal basis in the National Health Act, and it is also in conflict with Regulation 26 of the Regulations regarding the General Control of Human Bodies, Tissue, Blood, Blood Products and Gametes (No. R.180 dated 2 March 2012), which stipulates that the recipient of donated material gets exclusive rights in such material, thus also ownership.
Benefits and benefit sharing
The Zambian Health Research Act and the South African MTA provide guidance on accruals and benefits for research, but the latter does not specify who receives them. The Zambian Health Research Act defines benefits as financial, health, or other advantages gained from research. However, the South African MTA’s benefit-sharing provision is broad (Bedeker et al., 2022). To this end, the Zambian MTA guidelines which were developed to operationalize the statutory instrument, simplify the sharing of benefits, allowing them to be given to the health researcher, institution, community, or individual. Despite the potential impact individual royalties might have on inducement, none of the national laws have provisions to deal with inducement. This is clearly an oversight.
Benefit sharing in research is often unequal due to lack of regulatory frameworks (Wiertz and Boldt, 2022) and neglected implementation of benefit sharing plans (University of the Witwatersrand Human Research Ethics Committee Medical, 2014).. However, as is seen in the South African MTA and its local adaptation by a local institution, benefit sharing should be fair and equitable, involving engagement between the provider, recipient, and research participant (University of the Witwatersrand Human Research Ethics Committee Medical, 2014). The provisions of the MTA are, however, subject to the stipulations in the National Health Act, which in section 60(4), only allows reimbursement for reasonable expenses related to the donation of biological samples. Thaldar et al, provide an analysis of legal restrictions on benefit sharing, and are of the opinion that South African law does not allow benefit sharing with research participants who provide biological material (Thaldar and Shozi, 2023). Furthermore, the SA MTA, as subsidiary legislation, cannot override the NHA’s primary legislative authority as has been earlier discussed. Additionally, Kamau et al, offer a broader examination of benefit sharing in South African law, beyond the context of research participants, in which it is demonstrated that the issue of benefit sharing is multifaceted underscoring the complexity of its legal governance (Kamau et al., 2024).
Benefit sharing remains contentious because it is viewed differently from legal, ethical and community perspectives (Kamau et al., 2024). From the perspective of the research ethics principles of fairness and justice, those who bear the burdens of research, should benefit (Office for Human Research Protection, 1979). Likewise, communities have expressed wishes to share in the benefits of research (Moodley et al., 2014). Benefit sharing also does not necessarily imply “payment” for biological samples. Individuals and communities involved in research may benefit in various ways beyond payment. For instance, the San Code of Research Ethics, also referred to as the TRUST code, developed in 2017, refers to a range of non-monetary research benefits including co-research opportunities, skills sharing and research capacity building development (Schroeder et al., 2019).
Intellectual property rights related to MTAs
According to the World Trade Organization, intellectual property rights are the rights given to people over the creations of their minds (Saggi, 2016). The Zambian Health Research Act mandates the Authority to specify intellectual property rights requirements, while South African law does not. However, the South African MTA references existing laws and protocols related to intellectual property rights. The Zambian Health Research Act protects intellectual property rights related to health research, ensuring full dissemination of information and benefits. The South African MTA mentions sharing of information, research results, publication rights, technology transfer, and capacity building. It should be emphasized that from the ethical point of view, there should be equitable benefit sharing including those related to intellectual property rights (Cervo et al., 2016).
Dispute settlement
Dispute resolution is crucial for resolving conflicts between parties (Burton, 1990). The Zambian Health Research Act lacks this, but the South African MTA outlines the process for nonjudicial or court settlements, addressing potential conflicts in the MTA model. Since these are not legally binding provisions, it is, however, possible for parties to an MTA in South Africa to explore other dispute resolution options available in South African law since the National Health Act is silent on this matter. That said, the SA MTA’s dispute settlement provision has been extensively critiqued by Thaldar et al., particularly regarding the financial burden of arbitration compared to litigation (Thaldar et al., 2020). Given the cost constraints in many African jurisdictions, Swales et al, proposed online arbitration using expedited arbitration rules as a more cost-effective alternative (Swales et al., 2023).
Role of health research ethics committee
Both Zambia and South Africa have provisions for HRECs to review and approve protocol applications. However, the SA MTA, includes the REC as a signatory to the MTA which has raised controversy amongst REC members (Mahomed and Labuschaigne, 2019). Some REC members argue that the REC is not a juristic body and hence has no legal standing to be a party to the MTA. Andanda et al cite other mechanisms of control for countries that may not have regulatory authorities yet utilize national agencies to approve the export of samples (Andanda and Govender, 2015). However, for countries with established systems of health research governance, it can be argued that HRECs should play their mandated role in overseeing the ethical aspects of material transfer in research particularly when they involve human biological materials. RECs should ensure that the transfer of materials is conducted ethically, protecting the rights, welfare and autonomy of research participants (Mahomed and Labuschaigne, 2019). However, this does not necessitate the REC being a signatory to the MTA.
Study limitations
This study has both strengths and limitations. In comparing the MTAs of two research intensive countries in sub-Saharan Africa, this paper raises awareness of the conceptual, ethics and legal controversies implicit in the contractual documents that are concluded when biological samples are shared. It therefore serves to advance the debate on aspects of ownership, custodianship, benefit sharing and consent. Such deliberation, could enhance understanding of MTAs for researchers and regulators. However, the paper has some limitations. This study used the gazetted South African MTA as the legal comparator. It does not take into account potential revisions to the South African MTA as the process is incomplete and controversial. Regulatory documents from two countries with a similar regulatory context have been compared and contrasted. Other countries within sub-Saharan Africa that may have different regulatory systems or lack specific laws in line with health research have not been included. There is therefore the possibility that, had this study included other countries, a more divergent perspective on the sufficiency of the legal framework on material and data transfer might have emerged. Further, the findings are limited to the provisions and clauses of the two countries specific laws that deal specifically with health research. Furthermore, the methodology used did not provide end-user perceptions on the sufficiency of various laws and regulations.
Conclusion
Notwithstanding the controversies surrounding the legal status of the SA MTA, the emerging themes from the thematic analysis of the existing laws and regulations in Zambia and South Africa related to MTAs, provide an indication of the adequacy of the legal frameworks for MTAs by catering for issues related to ethical conduct and protection of research participants. These themes reflect provisions that at the minimum provide a basis for local or regional MTAs, reflecting how each theme speaks to ethical principles around autonomy (consent, data privacy/confidentiality, custodian/ownership,), beneficence (benefit sharing, secondary use and termination reports), and justice (existing regulations, MTA as a framework, intellectual property rights, dispute settlement).
However, it was observed that while there were variations between the two countries in the context of ownership, custodianship and benefit sharing, the two countries combined had provisions that can ensure ethical conduct and a better protective outlook for research participants in sub–Saharan Africa.
Interestingly, there were also differences between the two southern African countries in terms of the content and detail to which these protective elements were addressed. The Zambian MTA regulations had four elements missing and these led to silence on consent models, secondary use, termination reports and dispute settlement. This calls for immediate reflection to ensure that gaps are bridged and harmonized for the ultimate ethical conduct and protection of human participants. This equally applies to the South African MTA, which should be reviewed to address concerns and controversies. Given that the South African MTA is contested on some legal grounds, there is a need for legal clarity in South Africa about MTAs in health research. Such clarity is also required for researchers and HREC members.
Reaching consensus on provisions in the MTA in both countries is a research ethics imperative especially in the context of collaborative research involving biological samples.
Footnotes
Acknowledgements
Special thanks to Ms Siti Kabanda from the Faculty of Health Sciences, Stellenbosch University, South Africa, who supported the first author with becoming acquainted with Atlas Ti software and guided the analysis.
List of abbreviations
DAC Data Access Committee
EDCTP European and Developing Countries Clinical Trial Partnership
GGR Genetics and Genomics Research
HREC Health Research Ethics Committee
MTA Material Transfer Agreement
UBMTA Uniform Biological Material Transfer Agreement
WHO World Health Organization
Ethical Consideration
The Zambian National Health Research Authority provided the authorization to conduct the research. Ethics approval was obtained from the University of Zambia Biomedical Research Ethics Committee (REC) REF. NO. 1420-2020, and Stellenbosch University HREC Ethics Reference No: S20/10/278 (PhD).
Consent to participate
The approach to the study was such that there was no interaction or intervention with human participants.
Consent for publication
Not applicable.
Author contributions
VC contributed to the conception, design and to the acquisition of data for the study as part of his PhD work; VC also did the data analysis and drafted the article; KM and TR revised it critically for intellectual content and also gave final approval of the version to be published. DB provided legal input and revised several drafts of this manuscript for its legal content and consistency with South African law. He has also checked it against the South African MTA.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully acknowledge funding support from an NIH Fogarty grant: D43 TW01511-01- Advancing Research Ethics Training in Southern Africa (ARESA): Leadership Program.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data is provided within the manuscript and is publicly accessible as these documents are in the public domain
