Abstract
A polygenic risk score (PRS) is a genomic test that can reveal the risk of developing common diseases, such as type 2 diabetes. Before the testing, the participant must have sufficient understanding to give informed consent to participate in genomic testing, which may include a lot of new information and concepts. An additional challenge comes from understanding incidental findings of testing, although these are limited in the context of PRS. The purpose was to explore and describe participants’ perceptions of informed consent and research participation in the context of polygenic risk score testing for type 2 diabetes. The Research Participation and Informed Consent scale was part of a multi-scale study in the DIGIDIA feasibility pilot project. Recruitment targeted participants at increased risk of type 2 diabetes, those with pre-diabetes, or those diagnosed with diabetes in the previous year. After providing informed consent, participants (n = 60) completed baseline surveys. This article focuses on the data of the consent process collected as part of baseline surveys. Statistical tests, inductive content analysis, and mixed methods were employed. The findings show that the process of informed consent was clear and understandable from a consent perspective, but the purpose of PRS testing raised some ambiguity. Expertise in PRS was not seen as limited to just one profession, but many healthcare professionals were seen as potential experts. Participants emphasized a need for information, especially regarding incidental findings of genomic testing. The participant’s genomic literacy and understanding should be verified before genomic testing and informed consent. It should also be considered whether the possibility of incidental findings ought not to be brought up when the primary aim is solely PRS testing.
Keywords
Introduction
The use of polygenic risk score in type 2 diabetes
A polygenic risk score (PRS) is a genomic test that can reveal an individual’s genetic predisposition to various diseases. PRS is obtained by summing up an individual’s risk genes, weighted by the impact of these genes on that particular disease (Yanes et al., 2020). Type 2 diabetes, for instance, is a strongly polygenic disease with several hundred underlying gene variants (Grant, 2019). By calculating these gene variants present, an individual PRS score is obtained. Also noteworthy is that the gene variants have been identified through population-based genome-wide association studies, so the PRS should be interpreted in relation to the disease risk present in that population (Yanes et al., 2020).
Although PRS as an individual numerical score is precise, it is often clinically interpreted as different levels of risk such as “high risk” or “low risk” (Torkamani et al., 2018). When communicating disease risk, other risk factors and lifestyle should always be taken into consideration, as a healthy lifestyle is significantly associated with a lower risk of developing type 2 diabetes, regardless of genetic risk level (Li et al., 2020). PRS information could be utilized in therapeutic interventions, disease screening, or life planning (Torkamani et al., 2018), but its use is not yet established in public healthcare.
Informed consent and participation in genomic testing
Informed consent is a cornerstone of ethical practice, particularly in the context of genomic testing, where it carries significant importance due to its potential implications for participants’ health, and privacy. Informed consent consists of three key elements: threshold (including competence and voluntariness), information (encompassing disclosure, recommendation, and understanding), and consent (involving the patient’s decision and authorization). The process of informed consent is rooted in the four main principles of clinical research and healthcare ethics, which are autonomy, non-maleficence, beneficence, and justice (Beauchamp and Childress, 2019). Informed consent is based on ethical principles, but legal and regulatory norms and standards must also be considered (World Medical Association, 2024). Legal aspects include, among other things, how to store, and use genomic samples in the future, as well as protecting privacy (McGuire and Beskow, 2010). The recently revised Declaration of Helsinki (2024) particularly strengthened the autonomy and voluntariness of informed consent, introducing the concept of “free and informed consent” (World Medical Association, 2024).
In clinical research, it may be impossible to disclose all the risks and benefits within the time available or in an understandable framework. Therefore, the requirements for disclosure have been understood more as providing adequate information (Beauchamp and Childress, 2019). This challenge is particularly evident in genomic testing, due to the scope and complexity of the genomic information. Koplin et al. (2022) emphasize that the goal should be appropriate rather than fully informed consent, since it is not necessary to understand all the information related to genomic testing. Instead, ethical principles should guide the process, helping individuals to make decisions that align with their own values (Koplin et al., 2022).
Risks and benefits of genomic testing
Highlighting the risks and benefits, genomic testing is generally viewed positively by the public (Haga et al., 2013; Haukkala et al., 2018; Taylor, 2011). Reasons for participation may include the possibility of personal benefit (Berrios et al., 2018), such as improving one’s health (Malen et al., 2016), learning about healthy behaviors (Mills et al., 2014), and understanding genetic risk (Godino et al., 2021; Mills et al., 2014) as well as benefiting family and relatives (Berrios et al., 2018; Godino et al., 2021). Additionally, participants hope that the information will be clinically useful (Godino et al., 2021) and help others (altruism). Trust in the institution and provider is also mentioned (Berrios et al., 2018).
Discrimination (Haukkala et al., 2018; Mallow et al., 2016), data protection (Mallow et al., 2016; Samuel and Farsides, 2018), and effects on health insurance (Armstrong et al., 2012; Mallow et al., 2016) have been highlighted as the possible risks of genome testing. Also, the nature of the results may pose challenges. The participant should be able to interpret the results of the genomic test, but the results may be inaccurate (Tandy-Connor et al., 2018). The result of predictive genomic information can cause a psychological burden (Houdayer et al., 2019) or feelings of fatalism (Markowitz et al., 2011). The information can also create uncertainty about the future, but at the same time, it provides an opportunity to deviate from the prediction of the genomic information through preventive measures (Houdayer et al., 2019). However, very few participants believe that knowing their genetic risk factors would have adverse psychological effects (Malen et al., 2016), and the reported genetic risk of disease has not been associated with changes in depression or anxiety (Hartz et al., 2015).
Ethical issues of genomic testing relate to consent, security and reliability of data, commercial enterprises, and the handling of incidental findings (Samuel and Farsides, 2018). Privacy and data security are significant to the participant (Trinidad et al., 2010). For example, in biobanking, ethical evaluation and privacy were valued more than the pursuit of health benefits (Critchley et al., 2017). However, Trinidad et al. (2010) highlighted participants’ understanding and acceptance of sharing de-identified genotypic and phenotypic data for the sake of science, research, and ultimately health benefits. From another point of view, participants may perceive genomic information as familial rather than private information, and one cannot own their genes (Dheensa et al., 2016).
Challenges in conveying genomic information: the role of informed consent and understanding
Genomic literacy is part of health literacy, enabling individuals to understand and interpret their personal genomic information (Erby et al., 2008; Nakamura et al., 2017). The challenge lies in providing sufficient information and assessing participants’ competence to ensure autonomy (Varkey, 2021), because it is common for the participant to have no prior knowledge of genomic testing (Godino et al., 2021). Limited genomic literacy and insufficient understanding may be attributed, among other factors, to the conceptual novelty, complexity, and extensive scope of genomic information (Saelaert et al., 2020). However, literacy level appeared to have very little effect on understanding genomic research, but the language and time used had a greater impact on understanding informed consent (Bukini et al., 2020). Insufficient time before testing can therefore result in uninformed consent (Saelaert et al., 2020).
Genome sequencing can reveal more than just the intended testing outcomes. The term “incidental findings” refers to results that are not related to the original purpose of the test (Hegde et al., 2015). “Secondary findings” are also not of the original intent but are purposefully found through analysis (Miller et al., 2023) while in general, additional findings may be an appropriate term from the participant’s perspective (Tan et al., 2017). In this article, the term “incidental findings” is used, indicating that no other findings were intentionally sought.
It is impossible to disclose these potential incidental findings before the test, which can lead the participant to have unrealistic expectations. Therefore, it is not only about a “right to know” or “right not to know” (Varkey, 2021), but the inability of healthcare to provide adequate information to support decision-making. Berrios et al. (2018) pointed out that participants trusted the provider or institution would make the “right” decision about which results to return, and whether to report results of uncertain significance (Berrios et al., 2018). This kind of trust can reduce autonomy in informed consent if the participant accepts the test based on general trust in healthcare or research, rather than making their own decision based on the information received. For secondary and incidental findings in clinical exome and genome sequencing, both opt-in and opt-out consents have been discussed. Opt-in means the participant gives consent, while opt-out allows the participant to also indicate refusal (Ormond et al., 2019).
There might be unrealistic expectations and hopes regarding the testing (Tomlinson et al., 2016). According to Godino et al. (2021), 69.6% (n = 307) of participants wanted to know any variants, 27.1% only those associated with preventable diseases, and only 3.3% the variants for which the test was undertaken (Godino et al., 2021). Based on this, a desire to know all the variants can lead to an incorrect assumption and hopes about the possibilities of the test.
Informed consent and counseling before a genomic test should be guided by what information is eventually returned to the individual (Grove et al., 2014). However, reporting incidental findings is not that simple. If a carefully selected gene panel test is used, it is unlikely to reveal incidental findings. The likelihood of incidental findings increases as the number of tested genes grows (Hegde et al., 2015). When testing a PRS for type 2 diabetes (as in this study), potential secondary findings are not actively sought as with extensive whole-exome or genomic sequencing for diagnostic purposes. PRS is always combined with phenotypic data, and other individual markers are not sought, so nothing else is likely to be found. Communicating such uncertainty to the participant can be challenging in the informed consent process.
The purpose of the research was to explore and describe participants’ perceptions of informed consent and research participation in the context of polygenic risk score testing for type 2 diabetes.
Methodology
Recruitment
Recruitment targeted people 18–64 years old, with an increased risk of type 2 diabetes, those with pre-diabetes or those diagnosed with diabetes in the previous year, living in one city in Finland with 250,000 inhabitants. More detailed inclusion criteria were diabetes risk scores above 12 (Lindström and Tuomilehto, 2003), pre-diabetes as per WHO definition of impaired fasting glucose (IFG) defined as fasting plasma glucose (FPG) of 6.1–6.9 mmol/L or impaired glucose tolerance (IGT) defined as 2-hour plasma glucose of 7.8–11 (Alberti and Zimmet, 1998), or diabetes diagnosed by a physician. Participants had to be able to speak Finnish and be able to answer the questionnaires.
Participants were recruited during their visits to the diabetes risk nurse due to their known diabetes risk or while at the health center for other reasons. During these visits, they were identified as suitable for the study based on their diabetes risk. In addition, one public research bulletin was shared in a Facebook group maintained by the residents of the city. The recruitment phase began in June 2022 and ended in December 2022, when the targeted feasible number of 60 participants was met for this pilot study. From this point forward, the informed consent process was managed exclusively by one public health nurse (research nurse).
Research context
The research nurse provided standardized information about the study using a written information sheet (Supplemental File 1) and explained the concept of polygenic risk score (PRS) using illustrative images (Figures from Supplemental File 2 and Figures 1 and 3 from the article by Perola et al. (2019)). All participants received the same information in a structured session, which lasted approximately 15–20 minutes. Participants were given the opportunity to ask questions during and after the session.
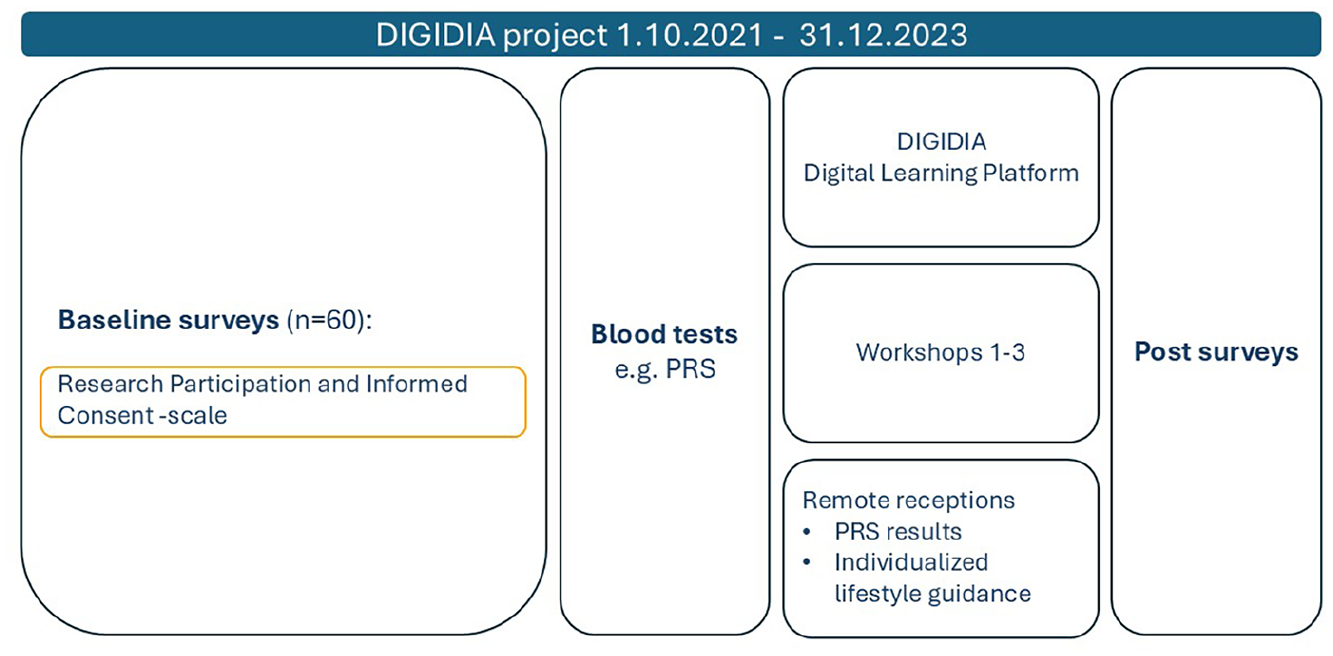
Research participation and informed consent scale as part of baseline surveys in the DIGIDIA project.
Informed consent (Supplemental File 3) was obtained in writing before any study procedures. After consent was given, participants completed the questionnaires, including the “Research Participation and Informed Consent” scale, which took about 20 minutes on average to complete.
PRS results were planned to be communicated personally to participants during a scheduled video call (see Figure 1). However, the exact timing of results delivery could not be guaranteed, as analysis required the collection of a larger batch of samples. It is important to note that PRS results did not affect the baseline data collection described in this study.
The information emphasized a preventive perspective, as regardless of the genetic risk level, maintaining a healthy lifestyle is crucial. Therefore, the information given was not entirely new, but constructed based on the participant’s comprehension of risk factors and the overall risk associated with type 2 diabetes, while PRS was a new concept for many. This article focuses on the data collected at baseline survey after informed consent (Figure 1).
Measures
Quantitative and qualitative data were collected using the Research Participation and Informed Consent scale. The scale was based on Halkoaho’s dissertation (Halkoaho, 2012), developed with Kuopio Birth Cohort (Kuopio Birth Cohort, 2012), and brought into the context of PRS. The scale consisted of the following components:
Open-ended questions were used to explore
Data analysis
Data were described using frequencies for categorical variables and medians (min, max) for skewed continuous variables. Items for Informed Consent and Research Participation were combined into sum variables. Informed Consent sum variable ranged from 13 to 52, and Research Participation sum variable ranged from 11 to 44. The sum variables were assessed as skewed. The Mann-Whitney U test was used to compare a continuous skewed variable between two independent groups, the Kruskal-Wallis test was used for a continuous skewed variable with more than two independent groups, and the Spearman correlation was used to assess association between two continuous skewed variables. The chi-squared (χ2) test was used for categorical variables when the assumptions were met: all expected counts were at least 1, and 80% of the expected counts were at least 5. The statistical significance level was set at p ⩽ 0.05 (Gray and Grove, 2021). Analyses were done using the statistical program SPSS version 29.
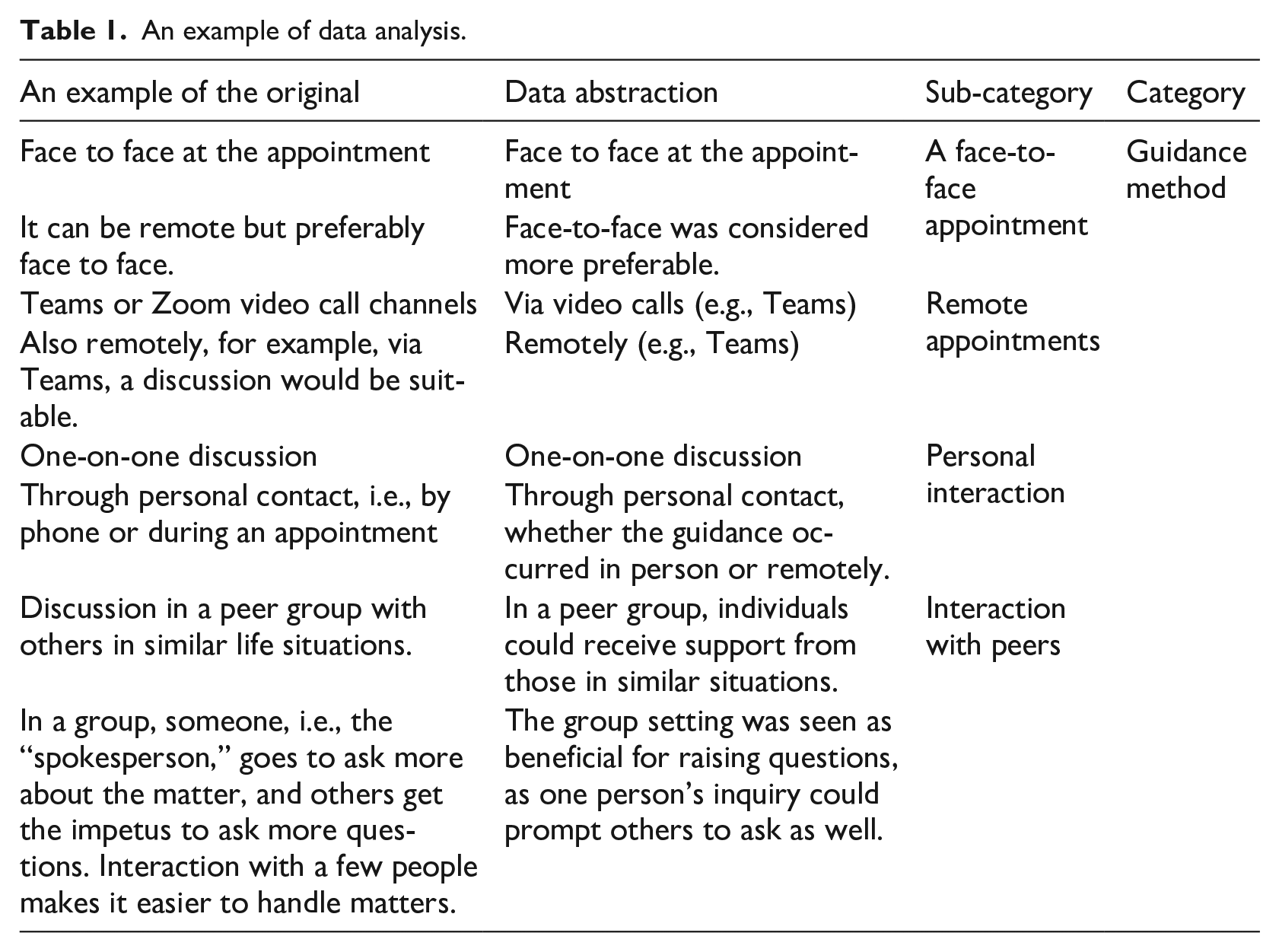
Qualitative open-ended questions were analyzed using inductive content analysis, consisting of data abstraction, coding, and grouping codes into sub-categories (Kyngäs, 2020). In the results, the sub-categories are highlighted in bold text. An example of the data analysis is presented in Table 1. The participants’ quotes and analyzed data were translated from Finnish to English.
An example of data analysis.
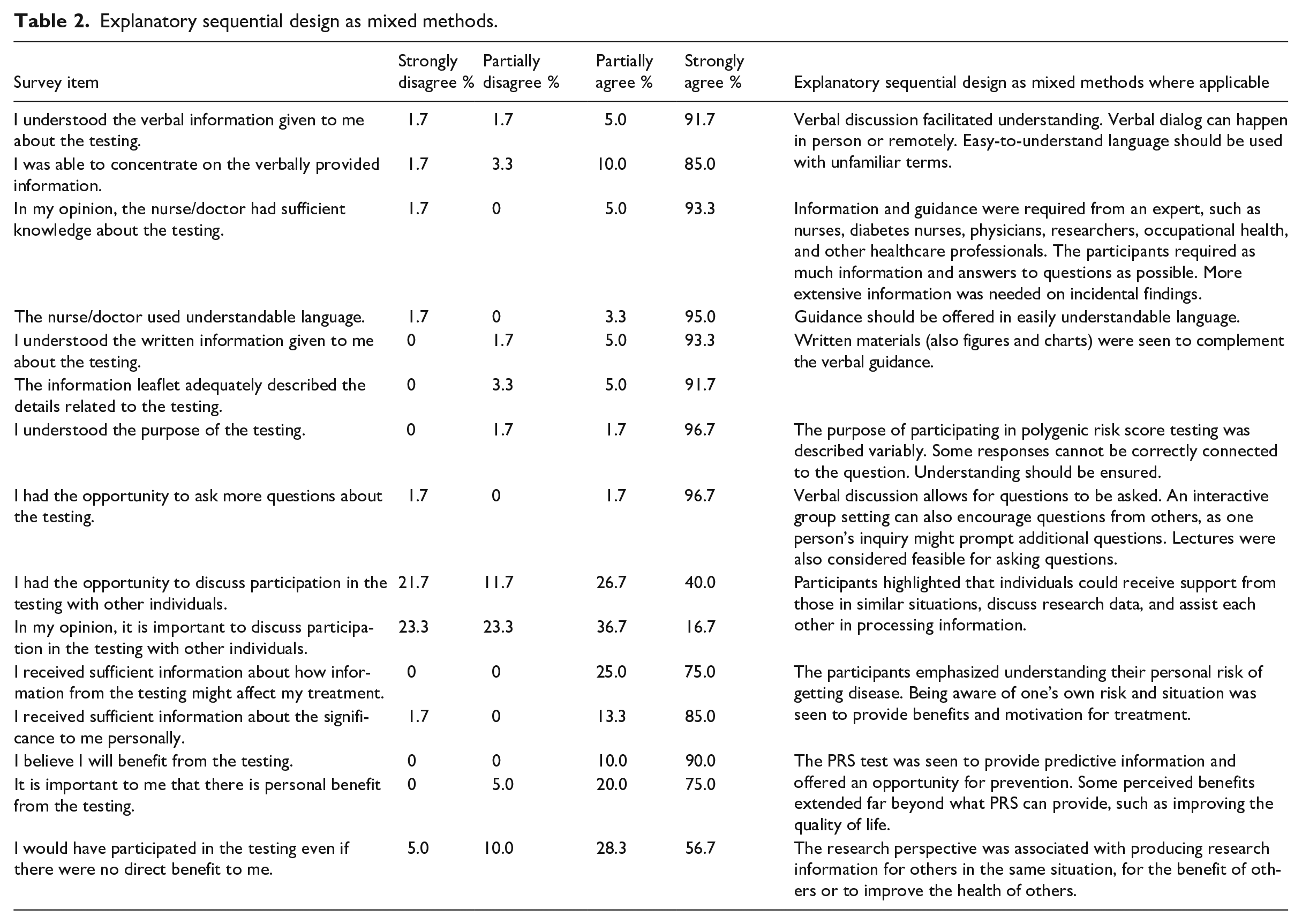
As the study encompasses both quantitative and qualitative components, Table 2 integrates data showing an explanatory sequential design as mixed methods where applicable. Quantitative data was analyzed first, followed by qualitative data. A mixed methods approach was employed with the qualitative section explaining the quantitative results (Creswell and Plano Clark, 2017). The report utilized the Good Reporting of A Mixed Methods Study (GRAMMS) checklist (O’Cathain et al., 2008).
Explanatory sequential design as mixed methods.
Participants
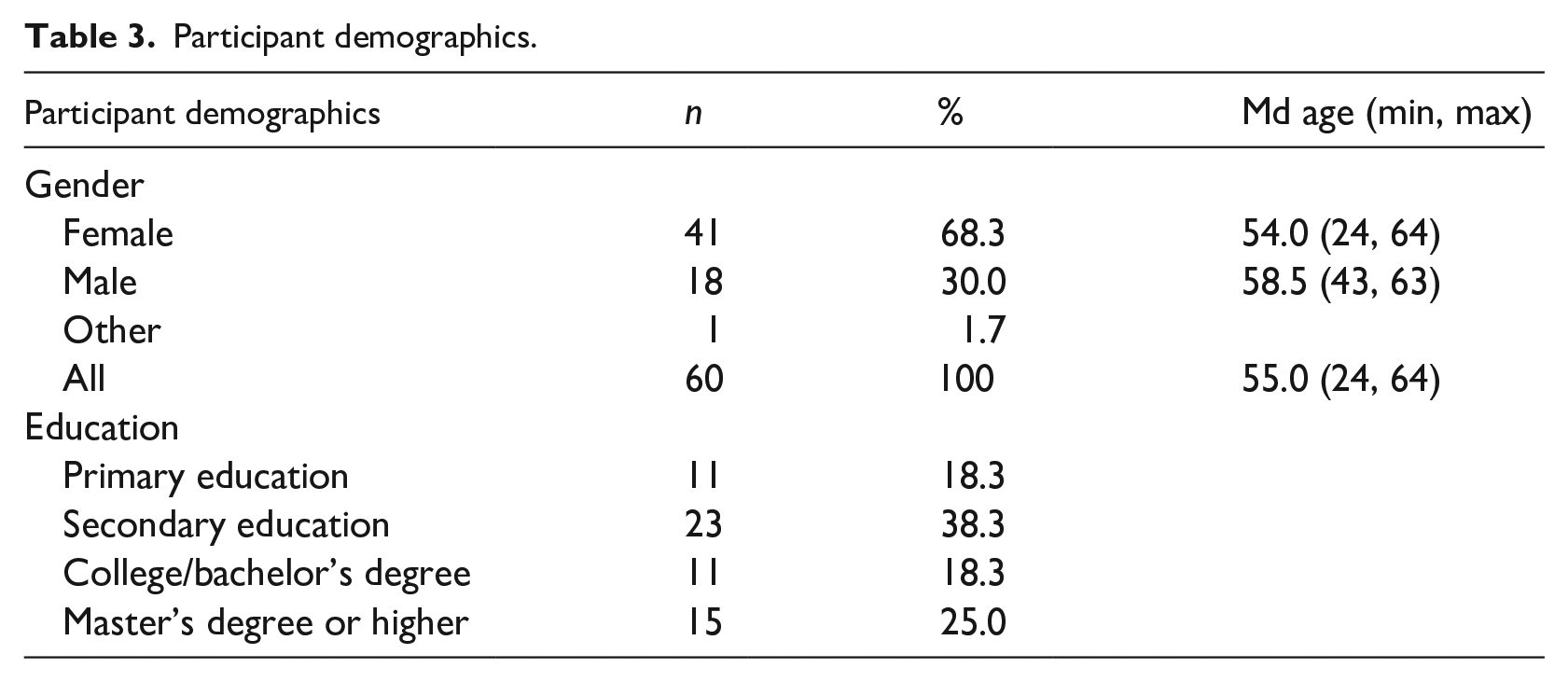
The sample (n = 60) comprised of 41 women, 18 men, and one classified as other. The average age was 55 years old (min 24, max 64). In terms of education, 81.7% (n = 49) had education beyond the basic level, which is slightly less than in the general population, as in the Finnish population aged 25–64 in 2022, the proportion of individuals who had completed a post-primary education qualification ranged from 84.0% to 87.9% across different age groups. Also, in Finland the percentage of individuals aged 25–64 with a secondary education qualification varied from 37.9% to 50.7% across age groups (Statistics Finland, 2022), while in this sample, 38.3% had completed a secondary education qualification and likewise indicates the lower end of the range (Table 3).
Participant demographics.
Ethical considerations
PRS raises several ethical issues, such as those related to the interpretation of results and the uncertainty in risk communication, largely due to the limited explanatory power of the tests (Andreoli et al., 2024). Informed consent in genomic testing also includes ethical issues, for example, when it comes to incidental findings, or variants of uncertain significance (Berrios et al., 2018). This further emphasizes the role and ability of healthcare professionals to explain and provide accurate information before testing, and challenges individuals’ understanding, as adequate comprehension is needed before informed consent, and testing.
Participants gave written informed consent to participate in a multi-scale pilot study, DIGIDIA project (Figure 1) aimed at increasing digital competence and health literacy. The consent stated that participant information would be handled confidentially. In connection with the research, the data may be transferred or disclosed in coded form, securely to parties outside the research group, or used in scientific publishing. Participants were also asked if they want to know potential incidental findings if there is treatment or prevention available. However, it is worth noting that any other findings were not actively sought. Through these preconditions and information, the participants gave their informed consent. A genome test was conducted to calculate the PRS of type 2 diabetes during the project. The study received ethical approval from the Regional Ethics Committee of the Expert Responsibility area of Tampere University Hospital (approval no. R22018).
Results
The quantitative section
Informed consent and research participation
The respondents scored a median of 49 points (min 19, max 52) in the “Informed Consent” section (Figure 2), indicating that the entire process of informed consent was generally perceived as clear and understandable by the participants. Items: “I had the opportunity to discuss participation in the testing with other individuals” and “In my opinion, it is important to discuss participation in the testing with other individuals” raised more variance (Table 4). More than half of the participants (53.4%) deemed it important to discuss their participation with others. At that very moment, participants did not have the possibility to discuss things with others, but if more time for decision-making had been needed, it would have been arranged. On the other hand, almost half of the participants did not feel the necessity to discuss things with others (Table 4), and were able to make decisions voluntarily without conversing with others (Table 5). There were no statistically significant differences in gender (p = 0.727) or education level (p = 0.249) with “Informed Consent,” and age and “Informed Consent” did not correlate with each other (p = 0.342; Table 6).
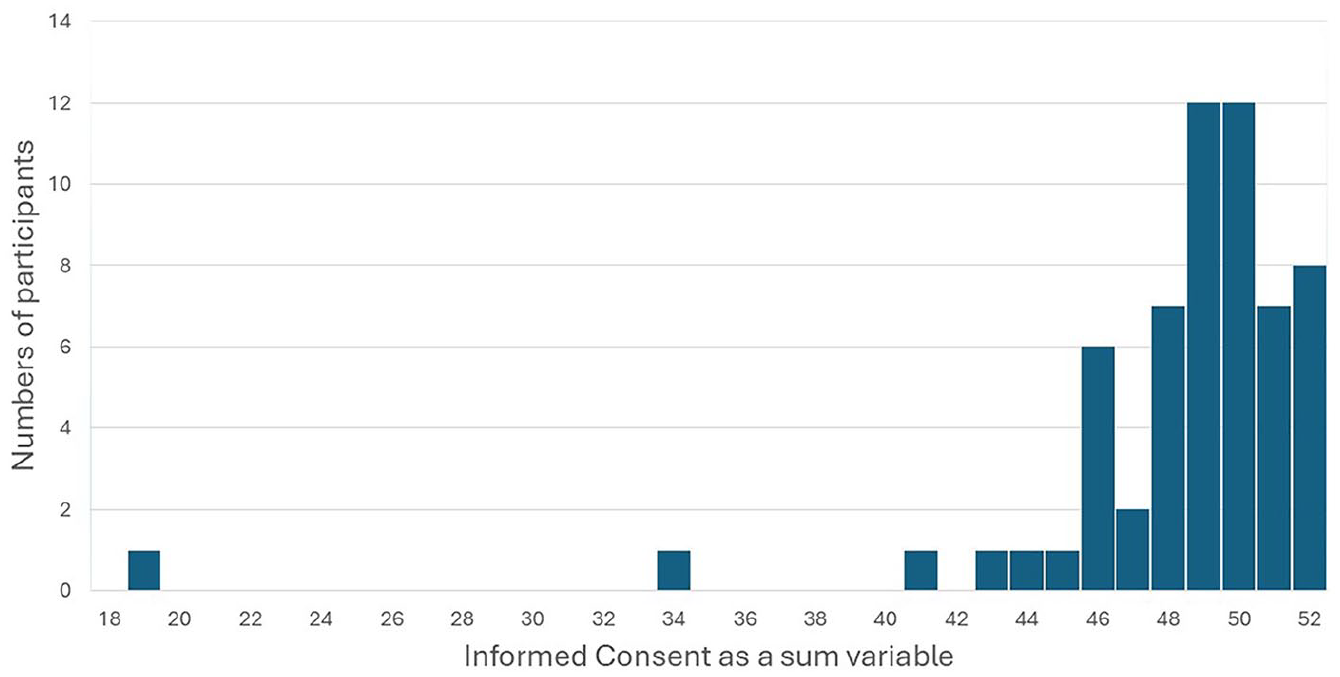
Participants’ informed consent sum variable prior to a polygenic risk score genomic test for type 2 diabetes.
Informed consent prior to a polygenic risk score genomic test for type 2 diabetes.
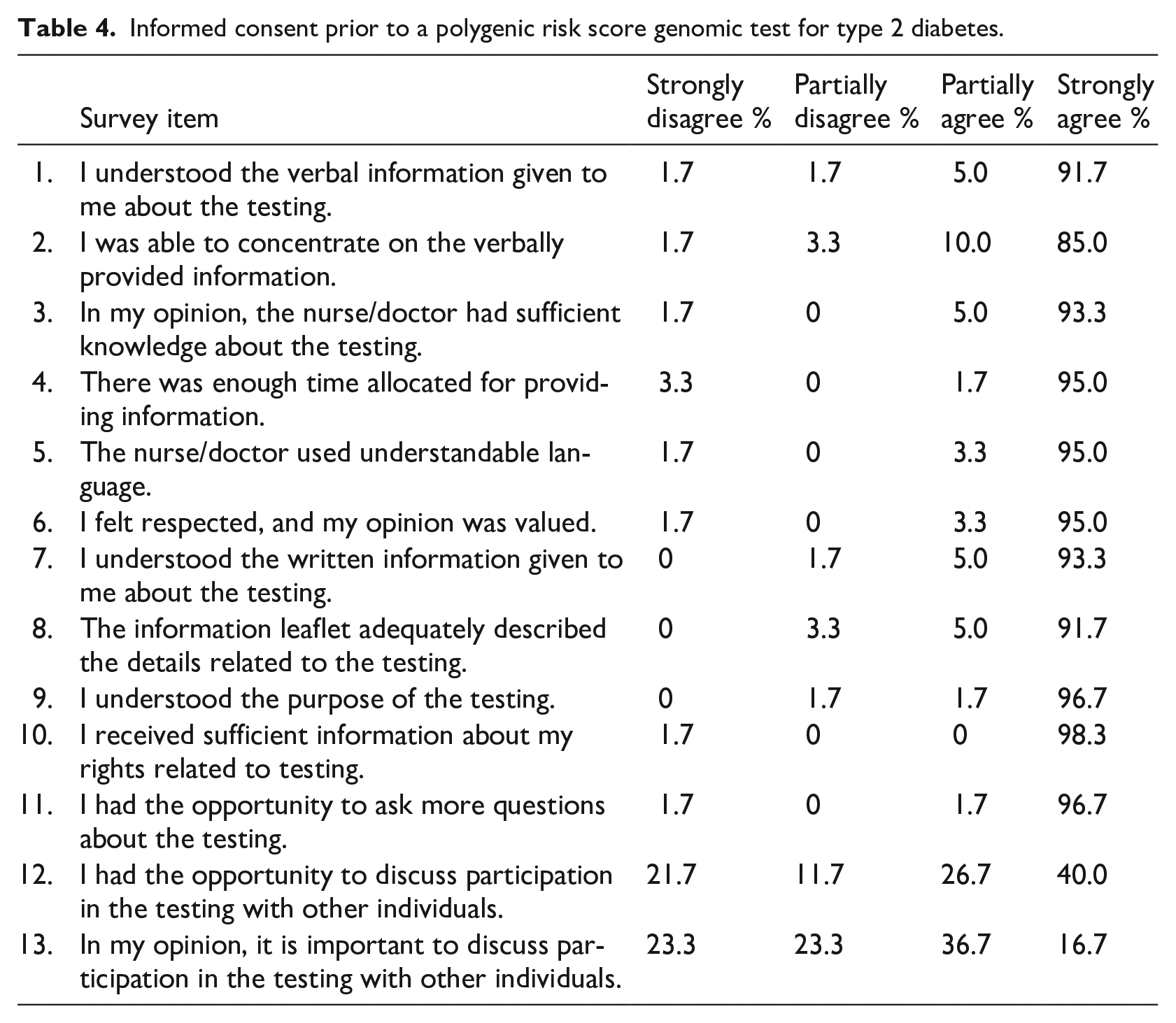
Participation in the polygenic risk score genomic test for type 2 diabetes.
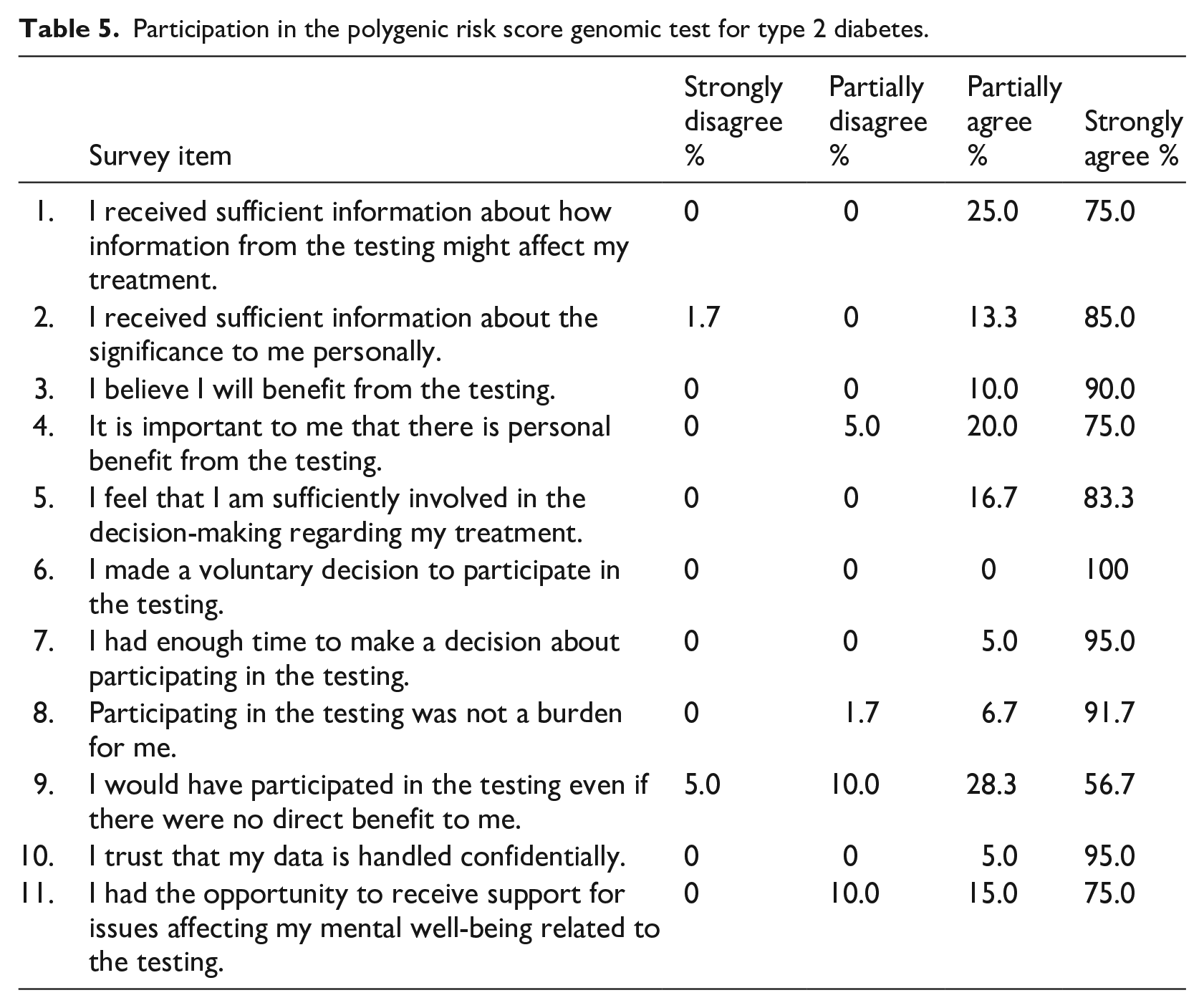
Informed consent, research participation, and overall implementation in relation to the background variables.
Mann-Whitney.
Kruskal-Wallis.
Spearman correlation.
Pearson Chi2.
The respondents scored a median of 42 points (min 36, max 44) in the
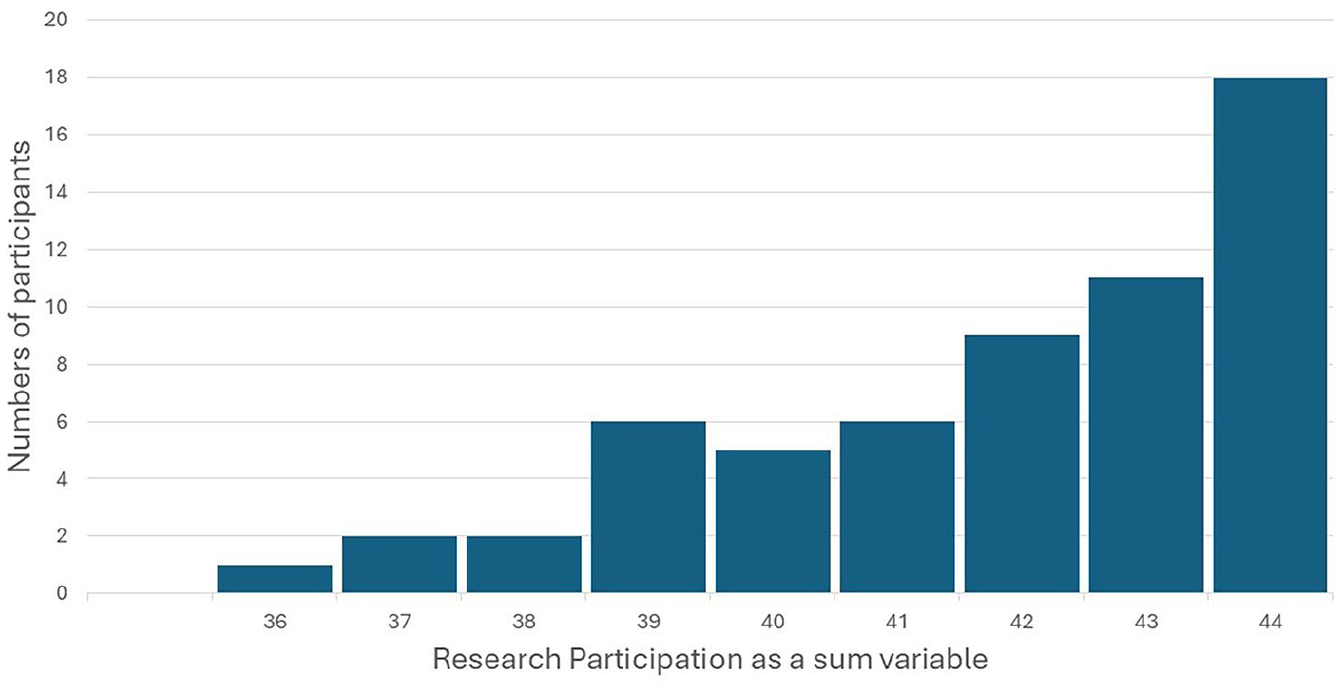
Participants’ research participation sum variable in the polygenic risk score genomic test for type 2 diabetes.
All participants were quite satisfied with the
The qualitative section
Way to discuss issues related to polygenic risk score testing
Participants highlighted aspects related to the guidance method, guidance technique, and expertise. Participants found it beneficial to discuss PRS-related issues in
“It can be remote, but preferably face-to-face.” (Man, code 35) “Through personal contact, i.e., by phone or during an appointment.” (Woman, code 22)
Some respondents emphasized the importance of
“In a group, someone, i.e., the ’spokesperson,’ goes to ask more about the matter, and others get the impetus to ask more questions. Interaction with a few people makes it easier to handle matters.” (Woman, code 14)
“If necessary, I believe verbal communication is the best way, as written communication (e.g., chat, etc.) may cause terminology misunderstandings. Verbally, it is easy to ask for more information and ensure both parties understand each other. I don’t see a difference whether the verbal communication is by phone, face-to-face, or, for example, via a Teams conversation.” (Woman, code 34)
“I believe the best way is to discuss face-to-face, but at the same time receive written information to support what was heard, so that the matter can be internalized.” (Woman, code 57)
Participants preferred to receive information and guidance from
“This conversation with the diabetes nurse was a very good way to have a discussion.” (Woman, code 25)
The participants required as much information and answers to questions as possible. More extensive information was needed on incidental findings. There was also a desire for a broader societal visibility of the topic, including talk shows and newspaper articles.
“As much information as possible.” (Woman, code 36)
Purpose of participating in polygenic risk score testing
Participants described the purpose of participating in the PRS testing as finding out the
The responses varied from “I don’t know” to a well-structured answer that revealed an understanding of genetic susceptibility, the formation of the risk score, personal risk, and the interpretation of PRS compared to the population: “The risk score test is used to map out the genes inherited by an individual that increase their predisposition to type 2 diabetes. The predisposition given by these genes is added together to give a number describing the sum of the risk, known as the risk score. By comparing this number to previous research data, an individual can be informed about the magnitude of their risk of developing type 2 diabetes compared to others.” (Woman, code 60)
“To find out if there is a hereditary risk of developing type 2 diabetes.” (Man, code 40)
The PRS test was used to assess the
“In order for a person to better understand their risk of developing an illness and become motivated to engage in treatment, it is important that they are aware of their risk.” (Woman, code 28)
The purpose of the PRS was to provide
“To gain knowledge for the future and thereby improve the treatment of the disease going forward.” (Woman, code 1)
The PRS test was regarded as providing an
“To find out how I can prevent developing type 2 diabetes in the future and to learn how high my risk of developing it is.” (Woman, code 24)
Some participants highlighted
“To know at least some other possible diseases.” (Man, code 49).
Also, there were participants who did not know the purpose of participating in the PRS test, answering “I don’t know,” which is quite similar to “disagree” answers (3.4%) appearing in the Informed Consent scale (“I understood the verbal information given to me about the testing”; see Table 4).
The
“To obtain information about inherited risk factors and simultaneously advance research for others who might be in the same situation.” (Woman, code 46)
In addition, the participants brought up the purpose of the DIGIDIA project as the development of a digital intervention, building an online environment for diabetes prevention and guidance.
“To create an online environment for the prevention of diabetes.” (Woman, code 58)
The participants also described
“Improving the patient’s quality of life.” (Woman, code 21) “To achieve the longest possible healthy life.” (Woman, code 27)
Discussion
The quantitative part of the research revealed that the process of informed consent was clear and understandable from a consent perspective, but qualitative questions revealed there was some ambiguity in the understanding of PRS. Participants described the PRS as providing hereditary and personal risk, with predictive information and opportunities for prevention. They also described the purpose as including the benefits of participating, as well as benefits for research.
In previous studies, in addition to benefiting oneself, benefits for others and trust in the institution have been mentioned (Berrios et al., 2018; Godino et al., 2021). Data security is also a common concern when processing genomic data (Critchley et al., 2017; Samuel and Farsides, 2018), but in this study participants trusted the confidential handling of their data, which may have further enhanced their willingness to participate in genomic research, even though they could not necessarily describe the purpose of participating in the PRS test.
Participants highlighted that they considered it important (53.4%) to discuss their participation with others. Additionally, peer groups were seen as beneficial when discussing PRS. The opportunity for discussion with others should be better considered in the informed consent process.
Personal interaction with a healthcare professional, as well as interaction with peers, were emphasized when asked about the best way to discuss issues related to PRS testing. Verbal discussions, easy-to-understand language, and written materials were seen as helpful in understanding. It is worth noting that expertise was not perceived as being limited to just one profession, but many healthcare professionals were seen as potential experts. Andreoli et al. (2024) pointed out in a systematic review that as the use of PRS increases in primary care, health providers who already treat common diseases would need more genomics expertise. Also, the research should focus on the connection between PRS and behavior change (Andreoli et al., 2024). The genomic competence and lifestyle counseling skills of research nurses should be emphasized in the informed consent process. As in this study, research information was provided by a public health nurse (also a registered nurse) who communicated about PRS from a preventive perspective on type 2 diabetes.
The question “Describe in your own words the purpose of participating in the polygenic risk score genetic marker test for adult-onset type 2 diabetes” could have been understood in different ways: the purpose of the PRS test or the purpose of participating. Additionally, for example, the purpose was seen as finding out lifestyle factors, but even though lifestyle factors were included in the individual guidance, the PRS test itself does not assess a person’s lifestyle, but genetic risk. Some participants highlighted investigating other potential diseases, although it was not the purpose of the testing. Also, when participants understood the project’s purpose, their responses did not accurately address the question. Some perceived benefits extend far beyond what PRS can provide, and some of these benefits were reasons for participation, but they did not answer the specific question. In the long run, health may improve, and physical activity may increase if the PRS provides enough motivation, but not solely because of the PRS test.
Participants were looking for as much information as possible, and incidental findings were especially mentioned. Previous research indicates that individuals might expect to receive more information than the test provides (Godino et al., 2021; Tomlinson et al., 2016). Such a misconception can lead to incorrect motivations for participation. The ability to explain the limitations of testing and ensure understanding is challenging when incidental findings or their likelihood in the context of PRS are unknown. When forming context-appropriate, informed consent from an ethical perspective, one should weigh and evaluate the participant’s desire to know, the limitations of the test, and the practical resources available.
Limitations
There are some limitations in this study. The open-ended question “Describe in your own words the purpose of participating in the polygenic risk score genetic marker test for adult-onset type 2 diabetes” could have been understood in two ways: highlighting the purpose of the polygenic risk test or the purpose of participation. Therefore, while it cannot be said that some participants answered incorrectly or wrongly, the question indicates the participant’s understanding. The variability in responses may stem from several reasons, such as genomic literacy level or comprehension, excessive surveys at the time of recruitment, or a misinterpretation of the question. Also, the open-ended questions could be further developed to better integrate quantitative and qualitative aspects for utilizing a mixed methods approach and provide a better understanding of the informed consent process, which may not be revealed through quantitative questions alone.
The sum variables could be examined in more detail in the following studies. The scale was piloted in the Kuopio Birth Cohort project (Kuopio Birth Cohort, 2012), but there has been no international publication about its use. In this study, the Research Participation and Informed Consent scale was tested for the first time on a small group as a feasibility study. Future studies should involve a larger sample size.
Conclusions
• Participants’ understanding of the purpose of the test varied. Therefore, participants’ genomic literacy and understanding should be verified with additional questions before informed consent and genomic testing. The consent process should ideally include personal interaction, easy-to-understand language, and written materials to support verbal guidance.
• The opportunity for discussions with others should be better integrated into the scheduling of the informed consent process.
• Since expertise was not seen as limited to just one profession, genome expertise and competence could be increased among professionals who already do preventive work and lifestyle counseling. PRS information would be a natural addition to risk communication and lifestyle counseling.
• Participants highlighted, among other things, the research perspective as the purpose of the study. The motivation to participate may stem from a desire to advance research and contribute to the common good. Regardless of the participant’s voluntariness, with such a background, special attention should be paid to the participant’s comprehension.
• In healthcare and health research, it should be discussed whether incidental findings should not be disclosed when the purpose of the testing is to utilize only the PRS results. The issue is not about opt-in or opt-out, but rather about the ethical concerns related to applying this option in the context of PRS testing. Participants should receive more information about possible incidental findings and the limitations of the test before providing informed consent.
Supplemental Material
sj-docx-1-rea-10.1177_17470161251358513 – Supplemental material for Informed consent for polygenic risk score test in type 2 diabetes: A pilot study
Supplemental material, sj-docx-1-rea-10.1177_17470161251358513 for Informed consent for polygenic risk score test in type 2 diabetes: A pilot study by Elisa Airikkala, Marja Kaunonen, Elina Pimiä and Arja Halkoaho in Research Ethics
Supplemental Material
sj-docx-2-rea-10.1177_17470161251358513 – Supplemental material for Informed consent for polygenic risk score test in type 2 diabetes: A pilot study
Supplemental material, sj-docx-2-rea-10.1177_17470161251358513 for Informed consent for polygenic risk score test in type 2 diabetes: A pilot study by Elisa Airikkala, Marja Kaunonen, Elina Pimiä and Arja Halkoaho in Research Ethics
Supplemental Material
sj-pdf-3-rea-10.1177_17470161251358513 – Supplemental material for Informed consent for polygenic risk score test in type 2 diabetes: A pilot study
Supplemental material, sj-pdf-3-rea-10.1177_17470161251358513 for Informed consent for polygenic risk score test in type 2 diabetes: A pilot study by Elisa Airikkala, Marja Kaunonen, Elina Pimiä and Arja Halkoaho in Research Ethics
Footnotes
Acknowledgements
Thanks to Tiina Wahlfors for bringing up the geneticist’s perspective on PRS, incidental findings, and informed consent in our discussions. Thanks go to Niina Sallinen, who with great understanding guided the participants regarding informed consent, the PRS, and the entire project.
Ethical considerations
This study was approved by the Regional Ethics Committee of the Expert Responsibility area of Tampere University Hospital (approval no. R22018) in June, 2022.
Consent to participate
Participants provided written informed consent for participation.
Consent to publication
Participants provided written informed consent for scientific publications.
Author contributions
EA: Formal analysis, Visualization, Writing—original draft, Writing—review & editing. MK: Writing—review & editing, Supervision. EP: Funding acquisition, Data Curation, Writing—review & editing. AH: Funding acquisition, Writing—review & editing, Supervision
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The survey was conducted with the DIGIDIA—Promoting digital skills in the low labor market-driven diabetes patients—project. European Social Fund (ESF) 2014–2020, S22669. Tampere Universities funded open access publication fees. No funding was used in the writing process of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
A detailed analysis of the data is available from the authors upon special request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
