Abstract
Global health emergencies often lead to a proliferation of health-related research and resultant data, which is shared across borders to help control the outbreak of disease and support decision-making regarding public health interventions. However, efforts to share data can be hindered by diverse international ethical and legal frameworks. The frameworks aim to govern coordinated processing, sharing and transfer of health data across borders thus placing burdens on researchers who are willing or obligated to share data. In this paper, we investigate ethical values and legal principles that can be applied to ensure a more streamlined approach in the governance of cross-border health data sharing during global health emergencies. A scoping review approach was used to better understand emerging evidence about issues related to the ethical and legal governance of cross-border health data sharing. We first highlight the challenges of sharing health data across borders. We then discuss how the challenges might be overcome through the application of ethical values and legal principles that have been identified in literature. This is followed by a proposal for possible ways of streamlining the governance of health data sharing from ethical and legal perspectives in a bid to better prepare for future global health emergencies and promote the responsible sharing of health data.
Keywords
Introduction
The World Health Organization (WHO) has often repeated calls for rapid access to “both raw and analyzed data or other pertinent research findings” to ensure an effective response to major disease outbreaks (Modjarrad et al., 2016: 2). Disease outbreaks can lead to public health emergencies of international concern if the emergence of the disease is unexpected and leads to serious public health impact (WHO, 2005; Annex 1). In such circumstances, the development of effective communication channels between the health system and other stakeholders within and between different countries is critical to enable rapid responses in emergency situations (Gustavsen et al., 2016). Global health emergencies often lead to a proliferation of health-related research to generate important data. The need for rapid collection and sharing of data during public health emergencies, as witnessed during the Ebola outbreaks in West Africa and the global COVID-19 pandemic have led to data being shared across national borders to help control the outbreak of diseases and support decision-making in public health interventions (Cullinan and Roberts, 2020; Liverani et al., 2018; Tacconelli et al., 2022). Data sharing helps to ensure scientific rigor and creates opportunities for better and more accurate results (Inter Academy Partnership, 2022; Townsend, 2022), and generalizability of research findings (Tacconelli et al., 2022). However, efforts to share data can be hindered by diverse international ethical and legal frameworks. The frameworks aim to govern coordinated processing, sharing and transfer of health data across borders thus placing burdens on researchers who are willing or obligated to share data (Brand et al., 2022; Mahomed et al., 2022). Even within the European Union where the Regulation (EU) 2016/679 (General Data Protection Regulation or the GDPR) was in force, there were challenges in cross-border data sharing due to lack of consensus on the interpretation of the GDPR (Tacconelli et al., 2022). This has led to the proposal for a European Digital Health Space (EDHS) in 2022 to facilitate data sharing across borders (European Parliament, 2022). The challenges highlight the need for guidelines on data sharing to be clear to guarantee the protection of personal information (WHO, 2022) and avert other related potential harms.
Guidelines should also ensure equitable data sharing since norms that maximize the utility of the data, such as rapid and real-time data sharing, currently seem to be prioritized, thus causing tensions with equally important equitable norms such as researcher recognition and equitable access to data (Pratt and Bull, 2021). Failure to ensure equity often leads to exploitation and conflicts of interest (Bull and Bhagwandin, 2020), which in turn causes tensions and inequalities that tend to emerge during public health emergencies. This was seen during the COVID-19 pandemic when low- and middle-income countries (LMICs) that participated in research and provided samples, and other forms of data ended up paying more for the vaccines when compared to their high-income country counterparts (Singh et al., 2022). This demonstrates inequality when it comes to sharing data, samples and the benefits that result from research. It also raises ethical concerns due to differential treatment of LMICs when compared to higher-income countries. These issues could have been provided for in sample and data sharing agreements, if guidelines had been followed.
Current literature has highlighted the challenges of cross-border health data sharing during global health emergencies and proposed possible solutions to overcome the identified challenges (ASSAf, 2021; Tacconelli et al., 2022). However, the proposed solutions “such as federated data analysis (or federated learning) and the adoption of common standards for data encoding and sharing” only partially address the challenges (Tacconelli et al., 2022: 2). It is thus unclear what efforts have been made to streamline the governance of data sharing during global health emergencies and what ethical values and legal principles should be applied to ensure a streamlined approach in the governance of cross-border data sharing within collaborative research projects. This study aims to answer the question “what ethical values and legal principles can be applied to ensure a more streamlined approach in the governance of cross-border health data sharing during global health emergencies?” The specific objectives of the study are:
• To identify the challenges of cross-border health data sharing during global health emergencies.
• To identify proposals from current literature for overcoming ethical and legal challenges of cross-border data sharing.
• To map the ethical values and legal principles that can inform a more streamlined approach in the governance of cross-border health data sharing during global health emergencies.
In this paper, we discuss the ethical and legal challenges in cross-border data sharing and ethical values and legal principles that have been identified in current literature to overcome the challenges. This is followed by a proposal for possible ways of streamlining the governance of health data sharing from ethical and legal perspectives in a bid to better prepare for future global health emergencies and enable responsible sharing of health data.
Scoping review methodology
A scoping review approach was used to better understand emerging evidence on issues relating to ethical and legal governance of cross-border health data sharing in global health emergencies. Scoping reviews enable researchers to synthesize diverse bodies of knowledge and describe their relevance, range, and nature. We followed the methodological framework of Arksey and O’Mally (2005) for scoping review studies. Arksey and O’Malley outline five steps to conduct such reviews which include: (1) identifying the research question; (2) identifying relevant studies; (3) study selection; (4) charting the data; (5) collating, summarizing, and reporting the results.
Search strategy
Searches were conducted on the Google Scholar search engine and four databases: Pub-med, Web of Science, Scopus, and Medline. Full texts of peer-reviewed articles were located and screened. The search terms used were: “health data AND pandemics OR health emergencies,” “health data AND ethical issues,” “health data AND legal issues,” “health data AND cross-border sharing,” “health data AND governance,” “health data AND streamlining governance.”
Selection process
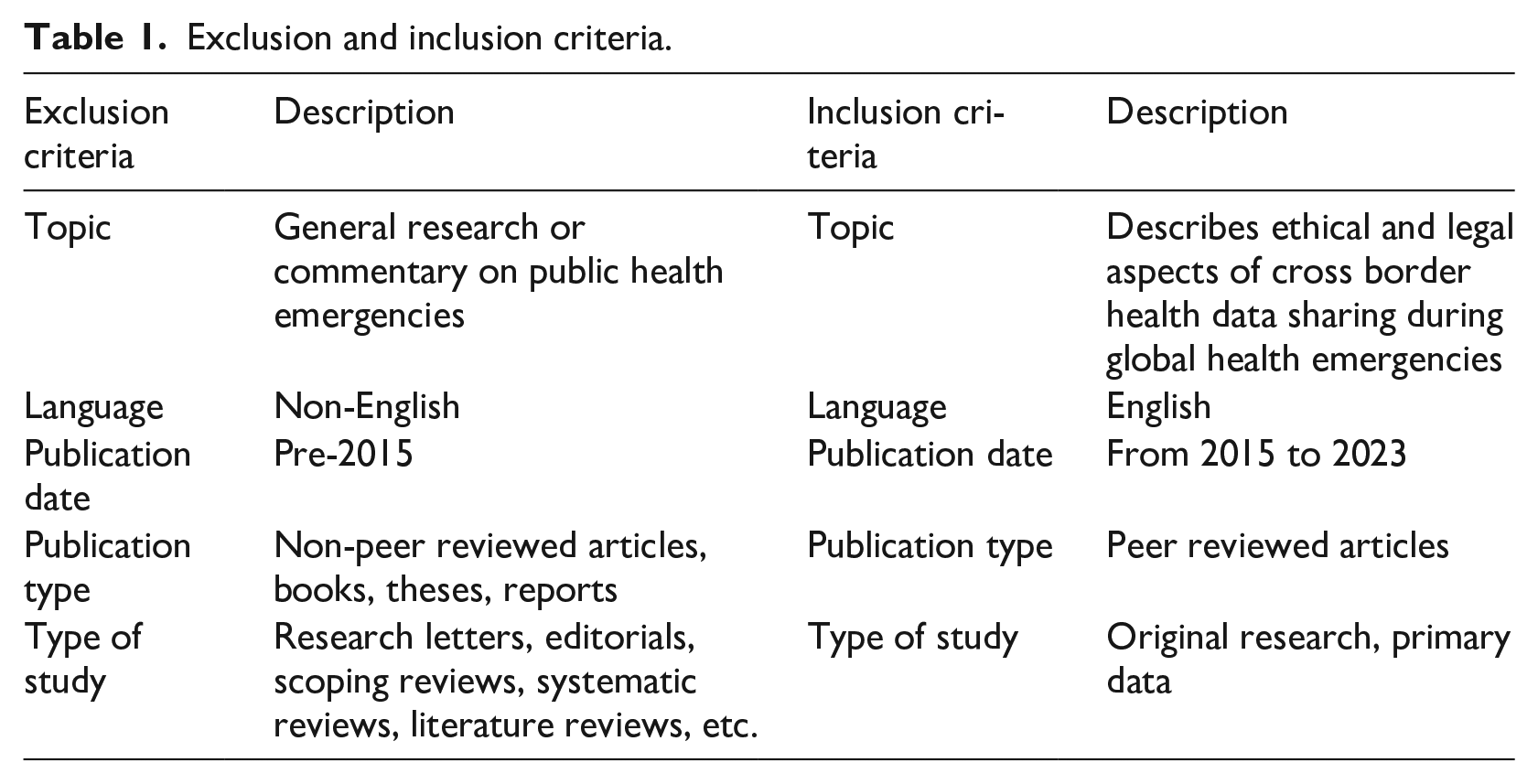
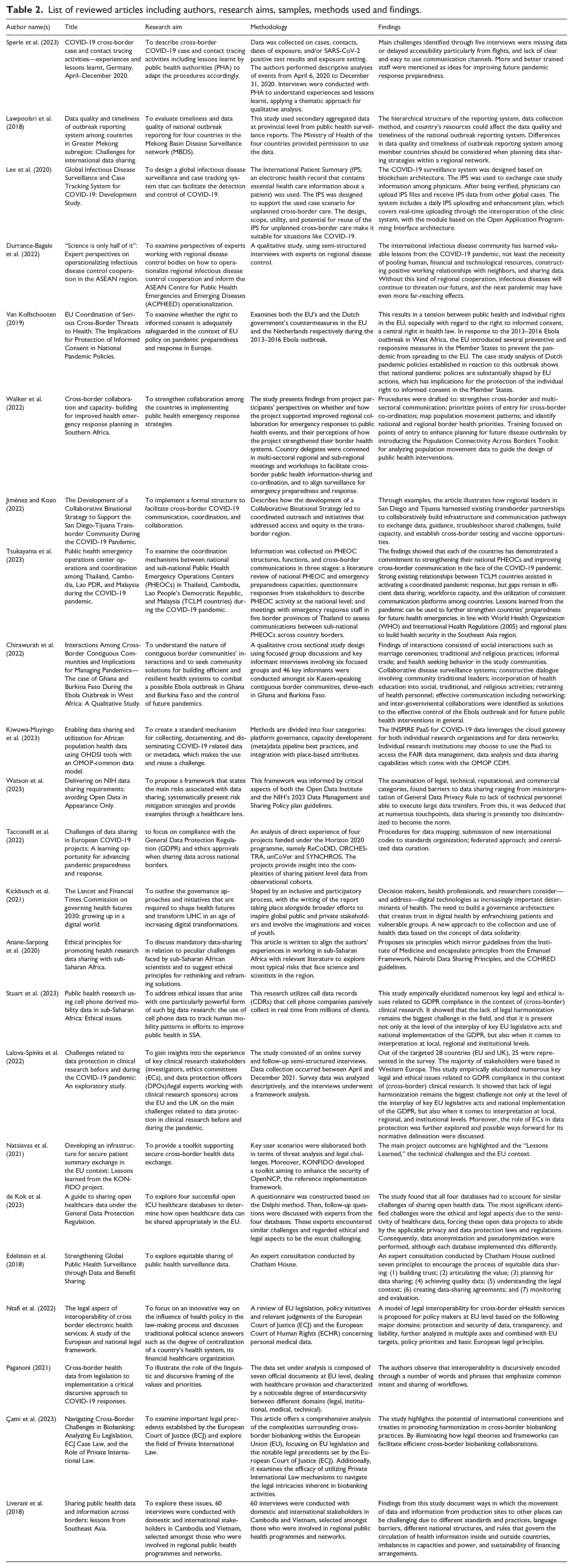
The inclusion and exclusion criteria outlined in Table 1 below were used to assign a value of “include,” “exclude,” or “maybe” to the identified articles to ascertain whether the article should be included in the review or not. Further, Figure 1 elaborates on the strategy that was used to include and exclude the articles in relation to Table 1. In a situation where it was not possible to decide based on the title and abstract alone, the full article was reviewed. When the first reviewer (LM) was unsure of whether to include an article a second reviewer (PA) reviewed the article, and this was resolved by the full article being retrieved and re-read in each case. After removing duplicates and excluding articles that did not match the search criteria, a total of 23 articles were included in the analysis. A summary of the characteristics of the 23 articles selected for review can be found in Table 2.
Exclusion and inclusion criteria.
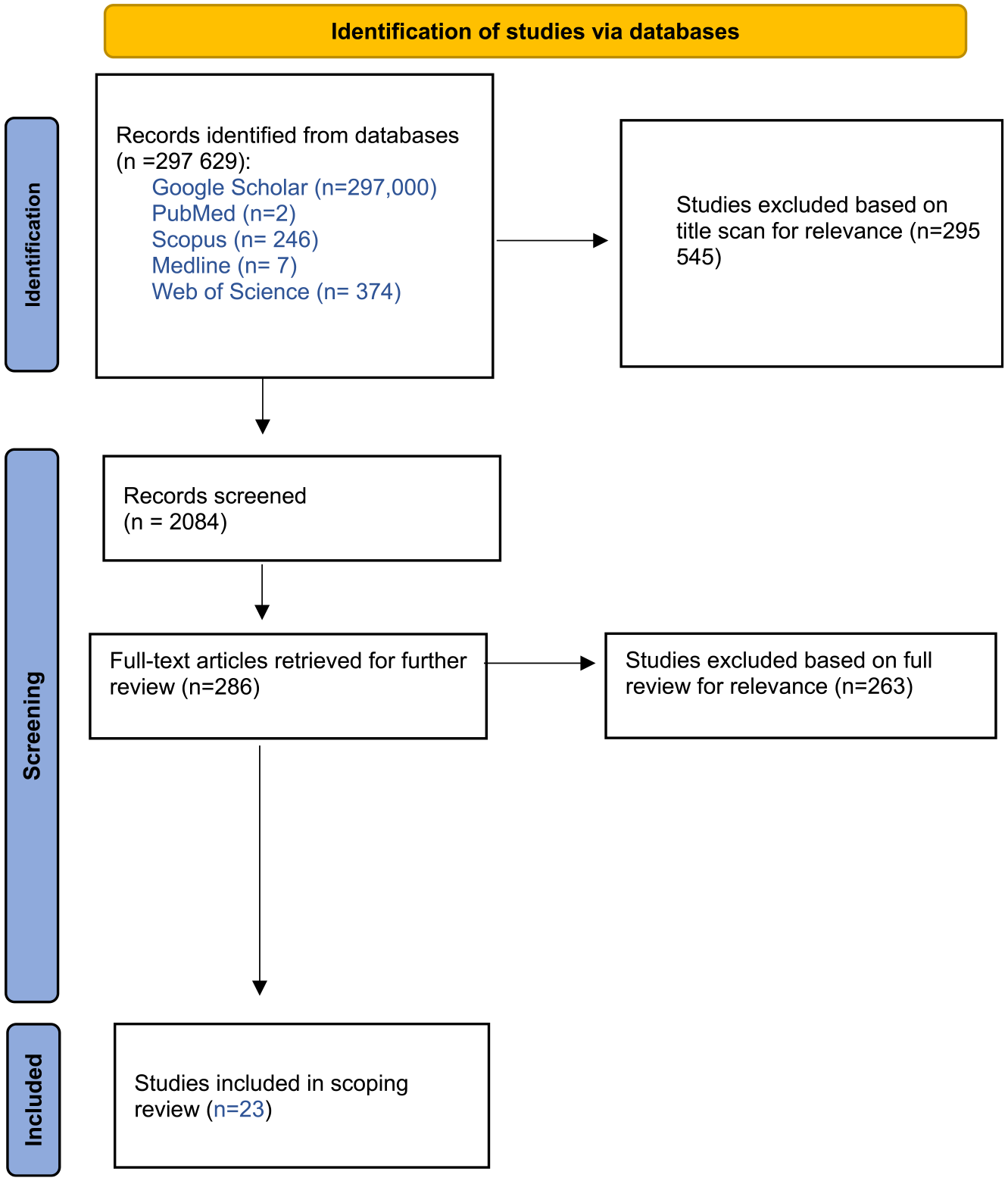
PRISMA flowchart.
List of reviewed articles including authors, research aims, samples, methods used and findings.
Data analysis
All articles were read and re-read to explore and understand raw data. Further, it was important to be as inclusive as possible given the limited evidence currently available on legal and ethical issues on cross-border health data sharing during public health emergencies. In this regard, we included original research articles where authors had used different methods to conduct their studies. A total of 23 articles were selected for review. While individually drawing from each article was important, it was also essential to compare themes and to synthesize the results.
Results
We located 23 original articles that described research on the legal and ethical challenges relating to cross-border health sharing data during global health emergencies. The selected articles were published between 2015 and 2023. We chose this period to enable us to include articles that focus on the most recent global public health emergencies that raised data sharing concerns. Most (14) of the articles were published during and after the COVID-19 pandemic. The studies presented work undertaken in different regions, North America (Jiménez and Kozo, 2022), Africa (Anane-Sarpong et al., 2020; Chirawurah et al., 2022; Kiwuwa-Muyingo et al., 2023; Stuart et al., 2023; Walker et al., 2022); European Union (de Kok et al., 2023; Holub et al., 2020; Lalova-Spinks et al., 2022; Natsiavas et al., 2021, 2022; Sperle et al., 2023; Tacconelli et al., 2022; Van Kolfschooten, 2019); and Asia (Liverani et al., 2018; Tsukayama et al., 2023). Some of the articles presented data from more than one region (Durrance-Bagale et al., 2022; Edelstein et al., 2018; Kickbusch et al., 2021; Lee et al., 2020; Watson et al., 2023). The published articles collected data using a range of methodologies including questionnaires, interviews, review of policy documents, screening health records and bio-banking research infrastructure.
Themes
The four primary themes that emerged from the analysis of 23 articles are outlined below.
Theme 1. Data quality, security, and risk
Despite expectations to use and share public health data, it is difficult to attain (van Panhuis et al., 2014). The analyzed articles identified different challenges pertaining to data quality during public health emergencies, including management, data interoperability as well as monitoring and analysis. The absence of datasets and information in standardized formats presents major challenges to the effective management of public health interventions. Moreover, sharing health data across borders raises security concerns and the need to minimize breaches can become challenging (Li et al., 2022). The development of information technology has increased the network security risks to both infrastructure and to data in general. In addition, the growing use of new technologies such as artificial intelligence also contribute to increased risks (Berger and Schneck, 2019). Yet, some public health emergencies, such as the COVID-19 pandemic, are time sensitive and therefore this information may need to be accessed immediately. Tsukayame et al. (2023) noted that there were differences in IT capacity and systems, which complicated data sharing within the Asian region. They noted that lack of standardized communication methods led to inconsistency in information sharing between countries (Tsukayama et al., 2023). Lawpoolsri et al. (2018), on the other hand, reported inconsistent data formats and missing variables, which compromised the quality of data in the Greater Mekong subregion. Similarly, Sperle et al. (2023) reported the need for good quality data to come up with a generalizable sample size in order to mitigate risks during health emergencies. Additionally, for good quality data to be collected, financial, human, and technical resources are required. Some studies also underscored the need for human resources such as better trained staff who collect good quality data (Sperle et al., 2023; Walker et al., 2022; Watson et al., 2023).
Theme 2: Ethics and protection of privacy in health data sharing
It was also evident from the analyzed articles that technologies can be used to discover and assess pathways of diseases during health emergencies (Kiwuwa-Muyingo et al., 2023), and expedite health interventions and treatment in ways that adhere to ethical standards. These standards include obtaining patients’ informed consent and protecting privacy by de-identifying data (Van Kolfschooten, 2019; Watson et al., 2023). Vital for the handling of personal data is that data processing must be lawful, clear, and fair (Mulder and Tudorica, 2019). Personal information is regarded as sensitive and confidential data when it might possibly impact an individual’s fundamental rights and freedoms. Van Kolfschooten (2019) highlights the importance of the right to informed consent and the need to safeguard this in the context of the European Union and international policy, particularly in pandemic preparedness and responses in the EU region. Similarly, Watson et al. (2023) reiterate that patients sometimes share their personal information with little to no gain. Health data is sensitive since it relates to the data subjects’ health and engagement with health services. The data can also comprise population-level aggregated data regarding burdens of disease and the usage of health services (World Health Organization, 2021). Consequently, the correct balance should be struck between safeguarding the rights of data subjects and the sharing of data responsibly during global health emergencies to facilitate data driven approaches to healthcare and research. The need for this balanced approach has led to increased calls for standardized governance frameworks for data sharing (Bennett et al., 2020; IAP, 2022; Seastedt et al., 2022) to ensure data access and interoperability of heterogeneous health-related data (Tacconelli et al., 2022). This leads to the next theme of data sharing agreements.
Theme 3: Data sharing agreements
Some of the reviewed articles identified strong cross-border coordination mechanisms that lead to positive results when health data is shared across borders (Durrance-Bagale et al., 2022; Jiménez and Kozo, 2022; Tsukayama et al., 2023). Walker et al. (2022) discussed the development of legal frameworks in the Southern African Development Community (SADC) region outlining the roles and responsibilities of countries, the conditions under which the countries share data, as well as the way the data is used. This allowed a transparent process, which built trust and solidarity amongst stakeholders and partners. Durrance-Bagale et al. (2022) elaborated on how the building of partnerships and collaborations is key to the success of regional networks and further builds trust and transparency. This is essential when data is being shared across borders as legal frameworks can be well organized to allow easy access to data (Durrance-Bagale et al., 2022; Walker et al., 2022). Existing governance frameworks that are insufficiently equipped to deal with the complexities of international data sharing are a concern since they tend to perpetuate inequalities due to piece-meal decision-making, which does not consider the roles of multiple stakeholders in the data ecosystem (Kaye et al., 2018). These governance structures are designed to protect personal data within national borders, which can slow down data sharing due to the additional bureaucratic layers that a national focus creates. Kaye et al. (2018) observe that applying territorial governance to broader areas perpetuates existing inequalities as it obscures the contributions of different stakeholders to the data stream, thus underlining the importance of suitable data agreements.
Theme 4: Political commitment, transparency, and accountability
Maintaining transparency and accountability for data sharing processes, usage and intended outcomes is crucial to ensure that trust amongst all stakeholders is maintained. The need for trust and transparency includes matters relating to political, social, and organizational factors, and citizens’ engagement (Abboud et al., 2021). One of the challenges of maintaining transparency emerges from sharing data through informal networks, thus restricting other users from accessing the data (Pisani et al., 2018). This problem has been addressed by institutions and research groups through the establishment of data repositories and Data Access Committees that transparently regulate access and enable cross-border data sharing (Parker et al., 2019). Further, some of the articles underscored the importance of political commitment and data sharing. In the interviews conducted by Durrance-Bagale et al. (2022) with key experts, some respondents highlighted the importance of politicians being involved from the beginning with the associated and administrative support, as this helps to ensure that every stakeholder clearly understands the process and outcome.
Discussion
Proposals for overcoming ethical and legal challenges in cross-border health data sharing
It is clear from this scoping review that there is no one solution for overcoming the identified ethical and legal challenges in cross-border health data sharing during public health emergencies. The challenges ranging from lack of quality safety guidelines, data quality, differences in IT capacity within countries, limited budgets for collecting quality data, lack of memoranda of understanding for sharing information, and human resources capacity building have been noted (Holub et al., 2020; Sperle et al., 2023; Tsukayama et al., 2023; Watson et al., 2023). These challenges show that there is a need to streamline the existing systems to facilitate cross-border health data sharing. Current developments in information and communication technology have also improved ways of sharing information rapidly through user-friendly systems, which are available globally (Fegan et al., 2011). Accordingly, governance frameworks need to be more efficient and effective in enabling cross-border data sharing in ways that comply with ethical and legal standards.
Quality data should be shared rapidly during public health emergencies (Sane and Edelstein, 2015), yet the barriers that are identified in this review may impede the sharing of data. These barriers mainly include insufficiently equipped governance frameworks, lack of cultural sensitivity, inequitable data-sharing practices, lack of enabling frameworks for sharing data, and concerns relating to security, transparency, and accountability.
Issues relating to data ownership, data interoperability, different guidelines and loss of trust tend to complicate sharing health data during public health emergencies. Furthermore, missing details in the collected data, lack of training and inadequate preparation can raise ethical concerns as elaborated by Sperle et al. (2023). In many cases, during health emergencies, there are no plans for long-term use of the collected data, but this position is changing as noted by Holub et al. (2020) with the European biobanking infrastructure; during the COVID-19 pandemic biobanks were able to preserve valuable biological materials complemented by detailed health-related data for future research purposes (Holub et al., 2020). Pre-existing relationships are key to ensuring respect and well-functioning data sharing systems amongst professionals.
Technical barriers, as mentioned in some articles (Lawpoolsri et al., 2018; Tsukayama et al., 2023), are a huge concern. Inadequate data collection and lack of standardization, as well as shared protocols across agencies and surveillance locations, means that there is varying data quality and incompatibility in the different databases. Consequently, data is not always comprehensive or sometimes lacks variables that are important. Furthermore, language barriers have also been reported (Abboud et al., 2021) as contributing to barriers in cross-border data sharing.
Enabling regulatory frameworks for data sharing
The standardization and automation of data would make sharing health data easier and cheaper as this would be less demanding to clean up and help to ensure that the quality of the data is up to standard (Abboud et al, 2021; Lawpoolsri et al, 2018; Sane and Edelstein, 2015). It is important to note that data sharing also entails the ability to collect high-quality data, thus the need for a capable and skilled workforce.
Legal barriers can also impede health data sharing due to strict personal data protection laws that prevent data from being shared, as well as unclear legal frameworks in which exceptions to data protection on health grounds are not explicit (Kaplan, 2016; Sane and Edelstein, 2015). These gray areas become problematic as public health institutions are uncertain about sharing health data. Furthermore, lack of official or unofficial data sharing agreements across borders, and proprietary claims over data, obstruct transparent cross-border health data sharing. The main concerns raised about the regulatory frameworks relate to “lack of well-defined guidelines for data sharing, knowledge of legal regulations or technical resources to standardize and share data” (Tacconelli et al., 2022: 6). In such scenarios, legal frameworks have the possibility of overcoming technical barriers by promoting capacity building. The SADC region, for example, has developed the SADC Model law, which guides Member States on how to develop and draft data protection laws (Musoni, 2022). Additionally, the EU Decision on cross-border threats has developed public health capacity by ensuring member states’ compliance with regional standards (European Union, 2013). The regulation on the creation of the European Health Data Space, for example, has led to recommendations that can support an enabling environment for seamless data sharing to promote healthcare and research (European Parliament, 2022; Zoi et al., 2021). The European Health Data Space (EHDS) incorporates federated interrogation networks that allow remote analysis of data by researchers without data being transferred, thus ensuring protection of data subjects’ privacy (Zoi et al., 2021). Additionally, the EHDS has led to specific proposals for “common governance mechanisms (policies, guidelines, code of conduct and agreements)” that are useful for moving toward a streamlined governance system to overcome legal and ethical barriers in cross-border data sharing (Zoi et al., 2021: 8). These regional initiatives are a step in the right direction for developing a more streamlined approach, which is necessary for addressing the concerns that are identified in this paper. The values and principles that can be used for the envisaged approach are summarized below.
Streamlining the governance of health data sharing
Compliance with ethical values and the FAIR principles
The values of equity, trust, reciprocity, and accountability can be used to streamline the governance of data sharing by encouraging stakeholders to comply with the FAIR principles, namely making data findable, accessible, interoperable, and reusable (Wilkinson et al., 2016), which can be challenging during public health emergencies. A streamlined approach that is based on the aforementioned values should have the attributes of being able to maximize the use of data and ensuring productive cross-border data flows and the protection of data subjects’ rights (Bitange and Thegeya, 2022). Moreover, the approach can ensure an equitable framework by being fair, efficient, and fostering accountability. Although the FAIR principles have been endorsed by many data sharing stakeholders, there are still challenges regarding data reuse, especially in health-related research, which may arise from the need to ensure confidentiality and privacy. Commercial interests and sensitivities should also be handled carefully. To facilitate compliance with these principles, support is needed to develop capacity to generate good quality data and ensure interoperability to enable data sharing (Anane-Sarpong et al., 2020; O’Connell and Plewes, 2015).
Responsible data sharing
The WHO guidelines (2017) oblige all parties who are involved in public health surveillance to share data in a timely fashion (Guideline 15). This calls for responsible data sharing, which requires stakeholders to allow the sharing of as much data as openly as possible and in a timely fashion while maintaining public trust (RDA Guidelines, 2020: para 6.4). Data trusts have been proposed as tools for ensuring trust due to their potential for representing diverse stakeholders’ interests and being more flexible to accommodate rapid evolution in technologies (Milne et al., 2022; Smith, 2022). Moreover, stakeholder participation and trust are essential for any programmes that promote data sharing (Smith, 2022). Notably, data sharing itself is said to promote “trust, integrity and completeness in science” (Anane-Sarpong et al., 2020: 87). To this effect data sharing should be promoted while ensuring that data is rapidly available and accessible, ethical, equitable, and reliable.
Two values which underpin responsible data sharing are solidarity and transparency. These values are useful for avoiding punitive actions against stakeholders who share data transparently, as happened to Southern African countries when they shared the genomic sequencing data of the omicron variant of COVID-19 (B.1.1.529) with the global community (Moodley et al., 2022; Petersen et al., 2022). The travel bans that were imposed on Southern African countries after they shared data were based on unfounded fears as the bans did not stop the spread of the variant across the globe.
Cultural sensitivity and privacy regulation
Cultural differences and privacy regulations in cross border health management can bring about huge challenges if not considered (Austin et al., 2021). These challenges can be mitigated by understanding the different cultures and jurisdictions in different regions. Cultural sensitivity can be ensured through stakeholder engagement and trust, which are essential for any programme that promotes data sharing (Smith, 2022). When one is aware of these differences, proper planning can be done so that novel ethical and human rights challenges are dealt with effectively. The main barrier that emerges from privacy regulations relates to the varying approaches to determining the appropriate legal basis for processing data, which can lead to divergent advice from research ethics committees (RECs) that review protocols, thus making cross-border data sharing challenging. The determination of an appropriate legal basis is generally agreed to be the responsibility of the data controllers who are accountable for safeguarding data subjects’ rights (Lalova-Spinks et al., 2022). Nonetheless, RECs are expected to assess risks when reviewing protocols and the mandate of RECs in data protection requires further clarification to ensure that the application of different legal bases in cross-border studies does not create inequalities between participants or data subjects from different countries (Lalova-Spinks et al., 2022). Another layer of complexity emerges from the assessment of the compatibility of additional processing of data with the initial purpose for which the data was collected. It is generally agreed that compatibility should be assessed in each case based on the appropriate safeguards that are in place (Wiewiorówski, 2020). A discrepancy exists in this regard when partial compatibility assessment is conducted based on the expectations of the data subjects instead of the existence of appropriate safeguards (Lalova-Spinks et al., 2022).
Policy and practice recommendations
Our findings suggest that policymakers and governments must prioritize the harmonization of legislation for cross-border health data sharing. Legal frameworks must be put in place to support cross-border health data sharing and to safeguard fundamental rights, maintain consistent adherence to ethics standards, enhance security and to protect privacy in the health data ecosystem since health data is sensitive data.
Further, to ensure consistent and high level of data protection, governments should establish clear interoperability policies, which can guide stakeholders in cross-border data sharing based on suitable data sharing agreements.
Researchers need to play an active role in supporting cross-border data sharing while ensuring that high ethical standards are maintained. They can play a crucial role in developing templates for informed consent, which reassure research participants that their personal information will be protected.
The study’s strengths and limitations
In this study we considered information from different regions including Africa, Europe, Asia, North America. However, a limitation of the study is that we relied on literature that has been published in English. This may have excluded valuable literature in other languages.
Conclusion
There is a need for health systems to transform the way health data is shared across borders to better prepare for global health pandemics, respond effectively to the changing patterns of diseases, and satisfy multifaceted health care needs while taking into account limited resources. Addressing ethical and legal challenges requires collaborative efforts from communities, researchers, governments, the healthcare industry, and international organizations to ensure fair and responsible cross-border data sharing that is respectful of cultural diversity and individual rights. A streamlined global framework that is informed by the current regional best practices such as the European EHDS may be a feasible approach in dealing with unfairness in data sharing. This could facilitate timely access to data during public health emergencies. The process for developing this framework could be led by an international organization, such as the WHO, to ensure that member states are fully committed to sharing quality data and maintaining high levels of integrity. Moreover, all involved stakeholders should state and acknowledge the different interests they have in the data sharing process. The values, which are identified in this paper can be embedded in the framework to ensure mutual understanding, and address issues relating to risks and benefits of sharing data with all stakeholders, as well as any expectations. This will help to build solidarity, trust, and goodwill in all involved stakeholders. Additionally, regional and sub-national legislation and policies that govern the key elements of data sharing mechanisms should be streamlined to eliminate fragmentation of policies that govern cross-border health data sharing in countries. This will ensure flexibility and development of comprehensive data sharing policies that promote good science.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Not applicable since the research did not involve data collection from human participants.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]() .
.
This research has been funded by the PREPARED project (HORIZON-WIDERA-2021-ERA-01-90) under grant agreement number 101058094.
