Abstract
The Joint Research Centre (JRC) is the European Commission’s in-house science and knowledge service, employing a substantial staff of scientists devoted to conducting research to provide independent scientific advice for EU policy. Focussed on various research areas aligned with EU priorities, the JRC excels in delivering scientific evidence for policymaking and has published numerous science-for-policy reports and scientific articles. Drawing on a scientific integrity statement, surveys among JRC’s research staff, and thematic discussions with JRC’s research leaders, the JRC has developed a comprehensive Scientific Integrity and Research Ethics (SIRE) framework, including instruments, procedures, and guidelines to ensure high standards and independence in its research. Key components of the SIRE framework include a Scientific Integrity Officer, an Editorial Review Board, a Research Ethics Board, and guidelines for responsible conduct of research. This article provides an overview of the JRC’s SIRE framework and how it was developed, emphasising the importance of maintaining independence, integrity, and ethics in scientific research that supports EU policy. The article also discusses potential gaps in the framework and where additional efforts may be needed, comparing with the recent U.S. National Science and Technology Council report on Protecting the Integrity of Government Science.
Keywords
Introduction
The Joint Research Centre (JRC) is the in-house science and knowledge service of the European Commission. It has around 2800 staff members who provide independent, evidence-based knowledge and science to support European Union (EU) policies. JRC scientists work in various research areas, including nuclear physics, energy, mobility, climate, sustainable resources, civil security, migration, health, food security and safety, economics, and social sciences. The JRC operates five research sites with experimental facilities and laboratories in Italy, Germany, Belgium, the Netherlands, and Spain, with its headquarters in Brussels. Annually, the JRC publishes about 400 science-for-policy and technical reports, as well as 1000 scientific articles in peer-reviewed journals. It also directly contributes to policy documents and produces around 500 technical reports annually for internal use in the policy development process (EU Science Hub, 2023; JRC Publications Repository, 2023).
Given its role as a provider of scientific evidence for EU legislation, it is crucial that the JRC conducts its research with high integrity, research ethical standards, and independence from external influences that could compromise the validity or objectivity of its findings. JRC researchers collaborate closely with policymakers, EU Member States experts, politicians, non-governmental organisations, industry representatives, and other stakeholders, exposing the researchers to various external influences. This, coupled with the complex global political and public environment, increases the risk of disinformation and misinformation, potentially leading to public distrust in JRC research. Additionally, researchers working on science-for-policy face time pressure to align with the policy agenda, which may conflict with responsible conduct of research. Furthermore, they often need to collaborate with colleagues outside their field of specialisation, requiring adaptability and interdisciplinary approaches. While JRC researchers generally have strong support and resources, the unique nature of science-for-policy work presents distinct scientific integrity challenges compared to those in academic institutions (Šucha and Sienkiewicz, 2020; Topp et al., 2018).
In 2016, as part of the development of the JRC 2030 strategy (JRC 2030 Strategy, 2016; Revitalising the JRC Strategy 2030, 2022), the JRC updated and consolidated its procedures on scientific integrity and research ethics into a single framework. This involved conducting surveys and thematic sessions with JRC researchers to identify common issues and challenges they face during their research. The resulting framework, called the JRC Scientific Integrity and Research Ethics framework (SIRE), is now in operation. This article explains how it was developed and the various instruments it includes. The article also examines potential gaps in the framework and the need for further efforts, comparing it to the recent U.S. National Science and Technology Council report on Protecting the Integrity of Government Science (Protecting the integrity of government science, 2021), which is, to our knowledge, the only available comprehensive study on scientific integrity and research ethics, with a focus on science for policy.
Materials and methods
In 2015, the JRC adopted the Singapore Statement on Research Integrity, which establishes principles and responsibilities for research integrity (Singapore Statement on Research Integrity, 2010). The JRC made slight adaptations to the Statement to align with its role as a provider of scientific evidence for policymaking, while retaining the fundamental elements of the original text. To further explore the scientific integrity and research ethics challenges that JRC researchers encounter in their daily work, a survey was conducted, followed by a series of thematic discussion sessions.
Survey
The JRC’s approximately 1500 research staff members were invited in 2018 to answer questions designed as multiple-choice quizzes, covering aspects such as integrity, research methods, authorship, ethical considerations, etc. (see Figure 1). The quiz mode for the questionnaire was chosen as a form of gamification in order to make the questionnaire more appealing. In total, 30 questions organised in three modules with 10 questions each were drawn up by the JRC’s Scientific Committee and adapted to the JRC context. The questions were arranged under the headlines from the JRC Scientific Integrity Statement and quoting the ‘responsibilities’ given in the Statement. The questions were together, as far as possible, covering all the ‘responsibilities’ and were randomly distributed to the respective module. When the quiz was completed, the participant was shown one preferred option for each question, even though sometimes multiple options might have been valid. After each question, respondents were invited to indicate whether the described scenario is relevant to the JRC. Respondents were also invited to give a free text comment following every question.
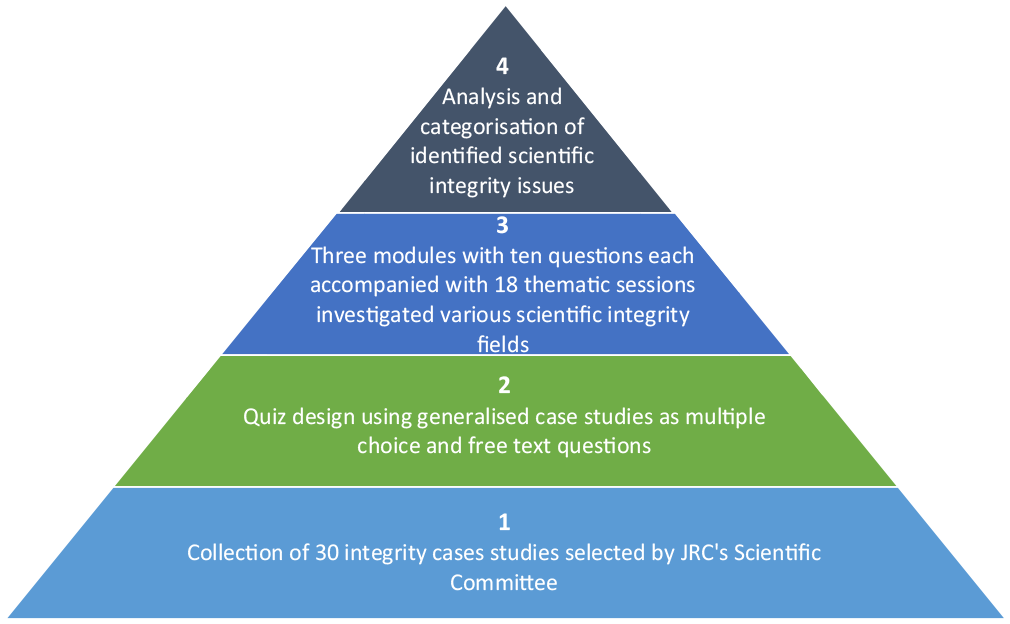
The approach used to design the quiz survey to investigate potential scientific integrity issues in the JRC.
The full list of questions is provided in Annexe. In total respectively 523, 315 and 342 JRC scientists responded to the invitations to the three modules.
Thematic discussion sessions
The thematic sessions were organised in 2018/2019 following the quizzes and consisted of 18 two-hour discussions led by the JRC’s Chief Scientist and JRC’s Scientific Integrity Officer. The participants included heads of research units, their deputies, research project leaders, members of the JRC Scientific Committee, and a small number of selected staff members, such as research advisors. A total of 457 staff members were initially invited, with 141 finally participating in a session, resulting in a 31% turnout.
The agenda for all sessions was as follows:
Introduction to scientific integrity in the JRC, covering: i. Scientific misconduct, for example plagiarism, authorship disputes, fraud, etc. ii. Intrinsic integrity of science, related to, for example, traceability, accountability, documented uncertainty, clarity about assumptions, etc. iii. Extrinsic integrity of science, related to for example politically motivated requests, bias, conflict of interest, withholding findings, time pressure, etc.
2. Summary and explanation of the results of the scientific integrity quiz, discussed above.
3. Open discussion in approximately 75 minutes on topics arising from the introduction and the quiz.
Each session was followed by optional closed individual meetings where participants could, in full anonymity and confidentiality, talk about and discuss scientific integrity cases they had personally experienced.
Ethical considerations
Survey participants were provided with clear and concise information about the questionnaire, including its purpose. The respondents were able to choose not to answer questions they found uncomfortable or irrelevant. The survey was voluntary and used the EU survey tool (DPR-EC-01488, 2022; EU-Survey, 2024) in anonymous mode. No additional personal data were collected that could breach the agreed confidentiality and anonymity. The aggregated data were presented in thematic sessions to senior scientists and project managers with the aim to improve research practices and promoting scientific integrity within the organisation. The thematic sessions did not conduct research on human participants but analysed pre-existing data from the survey. The participation in thematic sessions as well the sharing of experiences was voluntarily. The meeting was not recorded and any information that was shared was considered as confidential. The organisers did not seek an ethical assessment for the survey or thematic sessions. These activities were internal efforts to increase awareness, led by the JRC’s Chief Scientist and JRC’s Scientific Integrity Officer, with oversight from the organisation’s management.
Results
Survey
For the survey, the distribution of the number of ‘preferred’ answers for all three modules was almost Gaussian and centred around 5 questions, with a standard deviation of about 1.5. Only about 5% of the respondents chose eight or more ‘preferred’ answers, suggesting a moderate level of agreement or understanding overall. Crucially, the relatively modest average score is an artefact of the binary scoring system used in the multiple-choice options, rather than a true reflection of participants’ awareness of the topics. This key consideration, along with further insights, is more thoroughly detailed in the section on the thematic discussion sessions below.
In the survey, each question was paired with an additional question on the issue’s perceived relevance to the JRC (see Figure 2).
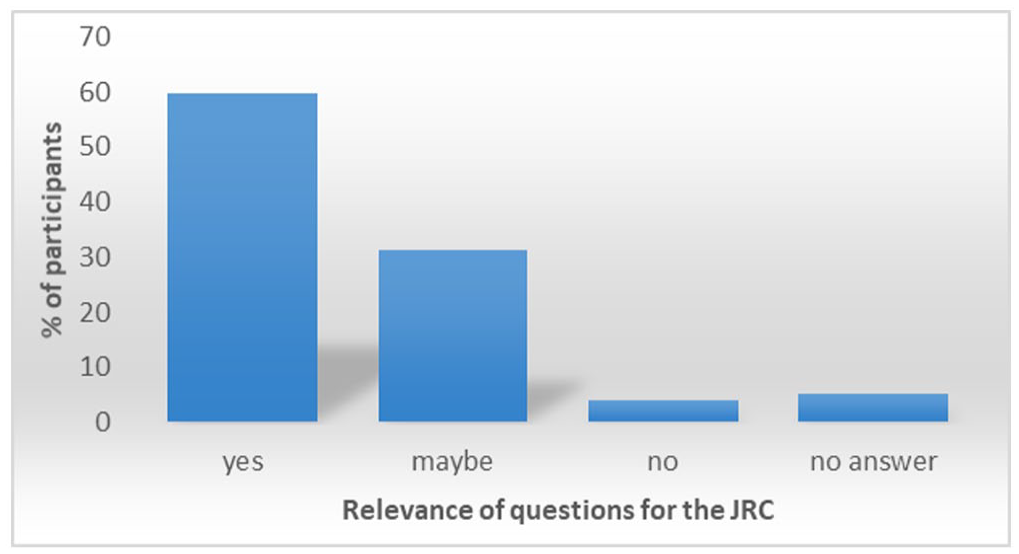
The majority of participants confirmed the relevance of questions for the JRC. Only 4% of the participants did not find the questions relevant for the JRC.
The response rate indicated that 60% of the questions were perceived as of relevance to the JRC. It should be noted that some situations may be experienced as more relevant because scientists are more often exposed to the particular situation, whether there is an issue or not. Moreover, since the questionnaire addressed a limited number of issues with no given time window for when an issue would have occurred, it only dealt with the matters included in the questions and for an unknown period of time and therefore could not be considered to give the full picture of the current situation.
Issues reported most frequently, by over 20% of respondents, included:
• Authorship of scientific publications, including ignoring or refusal of authorship among scientists, ignoring technicians as co-authors, and authorship claims by staff hierarchy like heads of unit and research project leaders who have not participated in the actual research work.
• Authorship of policy documents, with different practices and a lack of guidance on who should be listed as a co-author, including on presentations in policy meetings, workshops, and conferences.
• Unclear rules for duplicate publishing, referring to the manner in which a study that has already been published in a publicly available policy report can be (re-)published in a peer-reviewed scientific journal, and vice versa.
• Policymakers show a tendency to view scientific work through the lens of consultancy rather than as independent research and not anticipating and acknowledging that research may take time.
Situations with lower reported occurrences, between 10% and 20% of respondents, included:
• Unclear rules for self-plagiarism, such as different practices on paraphrasing text sections from previous publications, translation of publications to other languages, and parallel publishing of results.
• Research based on wrong assumptions; sloppy and relaxed research.
• Scientists presenting or publishing colleagues’ data without consent, references or falsely claiming originality.
• Delaying publications without clear justification, by co-authors, management, and internal reviewers.
Situations with even lower reported occurrences, less than 10% of respondents, included:
• Problems with conflict of interest and appraisal of research ethics.
• Copyright issues; breaching professional secrecy as a reviewer.
• Deliberate plagiarism of others’ research.
• Disputes among colleagues on research methods; discrediting colleagues’ research work.
• Colleagues overstating/understating research studies/knowledge in conferences, in contact with policy officers, and to stakeholders.
Thematic discussion sessions
In the thematic discussions, the conversations developed broadly in the same directions for all sessions, though each one had its distinct focus. The moderate agreement with the quiz’s preferred answers emerged as an initial talking point, eventually becoming an important piece of feedback. Indeed, participants misinterpreted their lower-than-expected score as a sign of insufficient knowledge about how to act with integrity.
The misunderstanding about the scoring highlighted the true purpose of the quiz, which was to challenge participants to navigate complex real-life ethical dilemmas. The quiz also emphasised the importance of providing a variety of plausible options in multiple-choice questions related to ethical scenarios, rather than just one obvious answer. However, giving full points only to one preferred answer led to lower scores for those who chose realistic but less optimal options. This demonstrates the benefit of a grading system that acknowledges the complexity of these choices, which would have been a better approach in hindsight.
While the quiz initially caused confusion, it effectively sparked discussions among participants about scientific integrity and research ethics. It also underscored the need for clear communication about the quiz’s objectives and how scores should be interpreted.
Besides the general criticism related to the scoring in the quiz, the matters most raised and discussed by participants are grouped under three headings: basic scientific integrity issues, the dual role of the JRC in providing science for both policy and the scientific community, and a proposed internal JRC Editorial Review Board and a JRC Research Ethics Board. We discuss each of these in turn.
Basic scientific integrity issues
The sessions highlighted several basic scientific integrity issues at the JRC, including authorship, plagiarism, and practices for duplicate publishing, that is, where the same research is published as a science for policy report and in a journal article. Concerns were raised regarding the variability in authorship practices across different units within the JRC, particularly in determining who should be listed as a co-author. Suggestions were made to address the issues of ‘guest writers’ and ‘ghost writers’ in guidelines. The question of whether external service consultants should be included as authors was discussed, emphasising the need for clearer JRC specific guidelines.
Plagiarism was a recurring topic, with discussions focussing on the definition of plagiarism, the use of plagiarism-detection software, whether self-plagiarism is a problem at all, and the importance of addressing incidents where external individuals publish ideas or data from the JRC without consent or acknowledgement. The discussions also touched on the issue of duplicate publishing, noting the differing practices among journals and the need for clear guidelines at the JRC.
Overall, the sessions highlighted the importance of establishing clear and detailed guidelines on authorship for both scientific publications and policy documents, as well as the need for plagiarism detection software and a central service for addressing incidents of unauthorised use of JRC data. Participants also expressed a desire for guidelines on duplicate publishing.
The dual role of the JRC
The discussions emphasised the unique challenges faced by researchers at the JRC, which serves a dual purpose of supporting EU policies but also advancing scientific knowledge. This dual role creates complexities, as the priorities and expectations for scientific research differ between the scientific community and policy support. For instance, authorship is crucial in scientific publications, but holds less weight in in-house policy reports. The time pressure and the need for approval from policy writers also differ between the two types of publications and there may be challenges in publishing content that may be inconvenient for policy purposes. In addition, the JRC needs to be clear that they cannot discover whatever policy writers are asking for and provide precise answers to every question. In any case, JRC researchers will insist on applying methods/models which are scientifically correct.
The sessions highlighted the need for clear guidelines and statements about scientific limits to navigate the dual responsibilities. It was suggested that an Editorial Review Board could help reconcile the conflicting demands and responsibilities. The dual purpose of JRC’s scientific work has implications for its organisation and approach, particularly in relation to authorship, duplicate publications, and plagiarism. In the discussions it was also suggested that the work processes and procedures applied at the JRC need to be systematically reviewed to address these complexities.
JRC editorial review board
During each session, the potential implementation of a JRC Editorial Review Board to ensure scientific quality in publications was discussed extensively. The decision to seek a new publication review and approval process at JRC was generally well-received, with the Editorial Review Board representing an improvement in the review approach. The previous JRC hierarchical workflow system lacked differentiation between scientific and policy-support publications, and the new process was suggested to address this. However, there were concerns about the mobilisation of competent in-house reviewers, with some scientists being uncomfortable reviewing topics outside their expertise and the need for recognition and time credit for reviewers. Overall, the implementation of an Editorial Review Board was expected to enhance the quality and approval criteria of publications across the JRC.
JRC research ethics board
The need of the JRC Research Ethics Board was only briefly discussed during the sessions, but was a frequent topic of discussions in other JRC forums. In the past decade, the JRC research portfolio has expanded to encompass projects that require consideration of ethical aspects. Examples include studies aimed at understanding human behaviour, toxicological experiments utilising human cells and tissues, handling sensitive personal data or big data, and research with potential dual use, often conducted in JRC laboratory facilities where stringent safety regulations are necessary to safeguard humans and the environment. In addition to emerging research activities with potential ethics issues in the JRC, Horizon Europe framework research programmes mandate that all research projects adhere to ethical standards which led to the decision of the JRC senior management to establish a permanent Research Ethics Board (REB). In 2021, the REB was fully operational and supported project leaders with a systematic screening of JRC projects in order to identify potential research ethics issues that necessitate an ethics appraisal. Projects with ethics issues are reviewed by the REB in accordance with how the ethics issue will be handled in the project.
Implementing scientific integrity and research ethics (SIRE)
The outcome of the thematic sessions and the quizzes underlined the significance of adhering to globally recognised standards on scientific integrity (COPE, 2023; European Code of Conduct for Research Integrity, 2023). Based on this, the JRC’s senior management decided to update the JRC’s existing processes and introduce a set of instruments in order to establish a robust JRC Scientific Integrity and Research Ethics Framework (SIRE). It was argued that this would also support a management ambition to demonstrate the JRC’s commitment to scientific integrity, not only because it instils trust among research colleagues and policymakers, but also because it helps the JRC manage risks both within and outside science.
The different elements of SIRE and their functions as adopted so far include:
A JRC Scientific Integrity Officer;
A JRC Editorial Review Board;
A JRC Research Ethics Board;
Guidelines, information, and training on responsible conduct of research.
The SIRE Framework is today incorporated into the JRC’s Integrated Management System and the European Commission’s Internal Control Framework (Revision of the Internal Control Framework, 2017) through a contribution to the Commission’s Anti-Fraud Strategy (European Commission Anti-Fraud Strategy, 2023).
The elements of the SIRE are presented in more detail below, as well as a discussion on where more efforts may be needed.
The scientific integrity officer
The JRC Scientific Integrity Officer advocates proper conduct of research according to standards of integrity. The Officer is the person in the JRC whom staff members can consult and to whom they can report incidents of possible non-compliance with the principles of scientific integrity, in confidence and guaranteed anonymity if they so wish. The Officer can also advise on what to do in cases of doubt on whether there is a breach of scientific integrity and has the mandate to investigate potential cases of misconduct.
The Scientific Integrity Officer is supported by the JRC’s Research Integrity Advisory Group (RIAG), which is comprised of seven members from the JRC’s Scientific Committee. The RIAG assists the Officer in investigating specific cases of potential research misconduct and violations of the JRC Scientific Integrity Statement. Investigations are conducted in a timely manner, with complete confidentiality and transparency for the parties involved, while managing potential conflicts of interest among RIAG members. The RIAG may also propose strategies and actions to promote and maintain a high level of scientific integrity at JRC. The Scientific Integrity Officer reports directly to the Director General and senior management on an annual basis, and also when specific cases require the Director General’s attention and decision.
A total of 37 cases were addressed to the Scientific Integrity Officer during 2014 to 2022. The majority concerned milder integrity issues that are common also in academic research institutions. Authorship dispute cases were the most common (15), followed by conflicts in research collaborations (3), guest/ghost/gift author cases (2), and questionable research practices (2), the latter concerning a research subject questioned by management and a claim of non-credible research methods. Three cases (3) involved the premature dissemination of non-peer-reviewed research to the public without indicating that the results were preliminary findings. Only one (1) instance of traditional research misconduct, in this case plagiarism, was reported. However, also minor forms of research misconduct and procedural errors can consume significant time and energy to resolve, disrupt collaborations and research, and damage the institution’s reputation.
In addition, the Scientific Integrity Officer received 11 complaints from researchers whose articles were refused publication by JRC’s management for various reasons. As a science provider for policy, the JRC likely experiences this issue more frequently than, for example, universities. Articles may be blocked due to quality concerns, as expected in an academic institution, but also if management, policy developers, or stakeholders perceive the content or results as sensitive, policy-biased, or premature, given ongoing policy development. Management may be concerned that publishing certain research data potentially disrupts policy progress, especially as policymakers strive to balance stakeholder needs across society and consider EU Member States’ expert opinions. Moreover, JRC research also extends into security-related areas, where sensitive data and results have to remain classified. Nevertheless, information may be in a grey zone where management, scientists, and others disagree on whether to disclose data, including those with the potential for dual use.
In any case, all 37 cases received have been successfully resolved in consensus, where some required consultations with JRC external partners and publishers. The Scientific Integrity Officer has thus acted as a service that picks up and resolves situations that potentially could develop into more serious conflicts and even research misconduct.
The JRC editorial review board
The JRC adopted an Editorial Review Policy in 2020, setting out an editorial review of JRC research publications. To implement this policy, the JRC established a JRC Editorial Review Board (JERB) chaired by a full-time Editor-in-Chief. The JERB ensures that JRC scientific publications and the knowledge contained therein meet quality and integrity standards.
The JERB’s Rules of Procedure details its operations, the role of the nine Editors and nine Associated Editors appointed by the JRC’s senior management, the guidelines for reviewers and the integration of the JERB procedure in the authorisation workflow for publications. Prior to the editorial review, the responsible JRC head of unit approves the submission of a publication. The final approval of the publication is given by the responsible Director, following a positive assessment by the JERB.
The Board presents an Annual Progress Report with an analysis of the reviewed manuscripts and other indicators collected for monitoring purposes. In its second year of operation, the Board returned over 60% of the submitted reports to the authors with requests for minor or major revisions. For journal articles, the Board’s role is limited to a preliminary verification, leaving the detailed content review to the peer review process of the journal. Nonetheless, revisions were requested for 12% of the reviewed articles.
As mentioned above, one of the most common concerns among JRC researchers regarding the review procedure was the potential delays that this additional quality control step would entail. Therefore, the JRC’s senior management tasked the JERB to handle the review of reports within 21 days and articles within 7 days. In practice, the JERB handles publications on average within half of these targets.
In 2022, the JERB involved over 300 in-house reviewers across the JRC, generally selected from a different research unit to that of the authors. In doing so, it also played an important role in the diffusion of knowledge within the JRC and, as a positive by-product also fostered collaboration and exchange of ideas. A satisfaction survey showed that most JRC scientists, middle and senior managers are satisfied with both the content of review comments and the timeliness of the service offered by the JERB.
To further improve the JRC’s approach to publication, the JERB engaged in developing guidelines and instructions on publication-related matters as well as the set-up of support services and tools, including, for example, enhanced proofreading support and (future) access to plagiarism-checking software. A content analysis of all reviewer comments identified common challenges in producing JRC publications, which can further inform remedial actions.
The JRC research ethics board
JRC’s senior management appointed 10 REB members with complementary scientific expertise from across the JRC to develop a working instruction mirroring the principles of the ethics screening in Horizon Europe (EU Grants - Ethics self-assessment, 2021; Horizon Europe, 2024). The REB implements and administrates the research ethics appraisal and follow-up procedure for all JRC research projects. In doing so, the REB has the mandate to grant ethics clearance to JRC projects and to reject projects on ethical grounds. The REB reports on its activities to the JRC Director-General and provides advice to assist JRC researchers on how to comply with Horizon Europe research ethics standards. Reports from the REB are not publicly available.
In 2021–2022, JRC projects were, for the first time, systematically analysed for ethics issues. Research project leaders received an introduction into ethics issues in research and were asked to screen their project for potential ethics issues. With the help of predefined questions in eight research ethics fields, the project leaders could quickly recognise whether a project contained an ethics component. In total, 386 project representatives participated in the ethics assessments. They declared various research ethics issues, such as human studies including the involvement of vulnerable groups such as children, the use of personal data including health data, and laboratory studies that could potentially harm the environment and human health. A number of projects also used artificial intelligence (AI) in their scientific work, see Table 1. However, no ethics issues were identified that led to the cancellation of the project in question.
Number of projects with potential ethics issues as declared by the project leader.

Responsible conduct of research – Guidelines, information, and training
The JRC Scientific Committee has developed a set of guidelines on responsible conduct of research based on conclusions drawn from the quizzes and the thematic sessions. These guidelines address also JRC specific concerns on best practices and different approaches, including issues related to ‘Authorship’, ‘Plagiarism’, ‘Duplicate publication’, ‘Dissemination of non-peer-reviewed research’, ‘Use of AI and AI-assisted technologies in writing scientific manuscripts’, as well as ‘JRC quality standards for scientific publications’, the latter intended as reviewer’s guidelines developed by the JRC Editorial Review Board. Other guidelines are under consideration, for example on ‘Reproducibility in research’ and ’Selection of journal for publishing’. The guidelines are consistent with international standards (see e.g. COPE, 2023) but go beyond what is available elsewhere by including JRC-specific issues. For example, the guidelines on authorship also cover authorship of policy documents.
In addition, the JRC has developed an in-house training course on scientific integrity and research ethics for JRC scientists, which is used predominantly for the induction of newcomers. The course presents the JRC’s scientific integrity statement, the SIRE framework, the role of the Scientific Integrity Officer, publishing procedures and research ethics appraisal at JRC, and available JRC guidelines on responsible conduct of research.
Discussion
For a research organisation involved in science-for-policy support, ensuring scientific integrity and research ethics is crucial for maintaining the quality and credibility of scientific work. This responsibility extends beyond the research process itself and encompasses the communication and use of the research outcome in policy making processes. In a report titled ‘Protecting the Integrity of Government Science’ by the Scientific Integrity Fast-Track Action Committee of the U.S. National Science and Technology Council, NSTC (Protecting the integrity of government science, 2021), the authors emphasise the importance of addressing scientific integrity and research ethics issues in science for policy, not only during the planning and execution of research but also when managing research, communicating research findings, and integrating science into policy.
There was a general agreement of the participants in the thematic sessions and survey that authorship disputes, ways of collaborating both inside and outside the European Commission, choosing research approaches for policy support, and general matters pertaining to research ethics and scientific integrity are of relevance for the JRC. They confirmed the necessity of a framework that addresses those issues in the various phases of science-for-policy cycle, which we will discuss here together with areas where we believe further work is required.
In planning and execution of research
Scientific integrity and research ethics encompass the entire research process, from managing research work to publishing in peer-reviewed scientific journals. These issues have been extensively documented and discussed in various academic papers and forums, such as conferences and platforms, and are also commonly included in university courses for PhD students (Armond et al., 2021; The Embassy of Good Science, 2024; WCRI, 2024).
In order to address questions related to scientific integrity and research ethics in the planning and execution of research, the JRC has created a number of guidelines and procedures. Some of these tools are directly incorporated into the JRC project management cycle. For instance, every scientific project is screened for possible ethics issues during the project’s planning stage. Following the screening process, scientific activities involving ethical considerations are reviewed by the REB before starting with any research. During the execution of the project, members of the REB assist the project teams when needed. While the REB focuses on obligations of researchers to their research subjects, the JERB ensures integrity and objectivity in science-for-policy reports through a rigorous peer review process of the presented research methodology, findings, and conclusions. If scientists have concerns about quality and compliance with integrity standards, they may seek advice from the Scientific Integrity Officer or follow the JRC’s responsible conduct of research guidelines on authorship, plagiarism, duplicate publication, communication of non-peer reviewed research and the use of AI in publications.
The JRC also supports the implementation of proper research conduct through its rigorous assessment process when recruiting scientists, with a preference for those with documented doctoral and post-doctoral research experience. Newcomers are offered an introductory course outlining the expectations for responsible research conduct. Early-career scientists benefit from experienced supervisors and can seek advice from a mentor who acts as a mediator between research fellows and management if needed.
In managing research
In academia, research leaders, such as supervisors and professors, are typically expected to be highly qualified researchers with strong scientific education and experience. However, in a science-for-policy organisation like the JRC, heads of unit are also responsible for managing the science-policy interface. This means that scientific qualifications are not always the top priority in the recruitment of managers. According to the ‘Protecting the Integrity of Government Science’ (Protecting the integrity of government science, 2021), scientific integrity may be compromised when managers and supervisors exert scientifically unjustified influence on science for policy research. Therefore, the JRC should assess the extent to which management-related scientific integrity challenges have occurred in the past and consider institutional norms and procedures to prevent or mitigate potential challenges in the future.
The JRC’s management is well aware of this challenge and endorsed a scientific integrity statement in 2015, leading to the establishment of the SIRE framework. The JERB supports the management of scientific work by systematically reviewing comments to JRC reports made by policymakers, particularly focussing on the potential inclusion of unsubstantiated statements suggested by policymakers or report authors. Scientists can raise concerns about scientific misbehaviour or misconduct, including by their managers with the Scientific Integrity Officer, their supervisor, or mentor. As a last resort, the European Commission has a whistle-blower procedure in place, which includes incremental steps depending on the hierarchical level of the concerned manager, linking to the Commission’s anti-fraud strategy and corrective procedures, including a protection assurance programme for the whistle-blower. The head of unit is formally defined as the project ‘owner’ and is responsible for the implementation of guidance and relevant legislation related to ethics issues identified in the screening process of scientific activities. Additionally, research project managers serve as liaisons between science and policy and are responsible for notifying decision-makers of any possible ethical concerns related to emerging research techniques and cutting-edge technologies.
The survey and thematic sessions suggest that JRC scientists face difficulties, including getting acceptance of research results that challenge established policy visions, requests to take specific stances conflicting with research findings, requests not to share results, or to delay or suppress scientific publications and policy reports containing unwelcome results for the dominant policy option. Moreover, science-for-policy researchers are often asked to work in emerging or unfamiliar fields, which requires time to build the necessary skills and networks. Time pressure could affect the quality of the research, especially when managers are not familiar with the scientific process. Again, the survey and thematic sessions suggest these challenges remain areas for reflexion at the JRC in fostering scientific integrity and research ethics in managing research.
In communicating research
Proper and effective communication of research results is crucial for providing credible and reliable scientific evidence to the scientific community, policymakers, the public, and stakeholders.
The peer review process implemented by the JRC Editorial Review Board, the recruitment of qualified researchers and the responsible conduct of research guidelines help to ensure that the publications made by JRC researchers meet scientific integrity standards. These efforts mainly focus on communicating to other scientists and policymakers, however.
When communicating and distributing research, the JRC relies on the principles outlined in its scientific integrity statement and its guidance for responsible research conduct to guarantee that research results are communicated appropriately. In case of breaches of scientific integrity standards in communicating research, the Scientific Integrity Officer and the Editor-in-Chief serve as advisors and points of contact.
In addition, the European Commission’s new human resource strategy offers even more chances to execute a successful communication plan as it incorporates job-specific training packages. Staff members that are involved in communicating research findings can now benefit from specialised courses and trainings on best practices for disseminating research findings. The JRC’s guidelines for authorship in scientific papers are emphasised, as well as the appropriate timing and manner of providing readers with information on the status of the research, such as whether the work has been published in peer-reviewed journals.
However, it is recognised that further efforts are needed. While scientists are expected to be well-versed in publishing their research in peer-reviewed scientific journals, in science-for-policy activities research results and scientific information must also be conveyed in a way that is easily understood by non-scientists, including those unfamiliar with research, the limitations of research and the ethical standards governing the publication of research. It is therefore important to educate all parties, from researchers to policy writers and dedicated communication staff, about the role of peer review, the importance of validating research results, how different publishing platforms function, and scientific publication procedures in general.
In decision- and policymaking
There is no one-to-one relation between scientific research and policy development and the JRC recognises that scientific research can at best inform the policy making process. However, policymakers and stakeholders cannot be assumed to have a scientific background ensuring the correct interpretation of research findings in the conception, implementation, and evaluation of policies. Scientific processes and language, for example related to uncertainties, statistics, and probability functions, could easily be misinterpreted. Scientific integrity can also be jeopardised in the policy development process by bias, misunderstandings, cherry-picking of results, disregarding relevant scientific information, drawing wrong conclusions, etc.
The JRC tries to remedy this problem by collaborating closely with policymakers and being involved in the entire policy cycle. When appropriate, JRC scientists participate in European Commission technical working groups, inter-service groups and consultations, and the comitology procedure characterising some EU law-making. Nevertheless, experiences show that also for policymakers, there is a need for training and provision of information material on requirements for responsible conduct of research, as well as for the use of research results in policy. While the training of policy makers in this respect may not be the sole responsibility of the JRC, it is developing activities exploring how the scientific literature of policy makers can be improved through for example, the development of competency frameworks (Smart4Policy, n.d). Nevertheless, additional efforts are needed to ensure that research results are effectively and appropriately implemented and used throughout the whole policy cycle, for example by means of information materials on scientific procedures and integrity standards, targeting policymakers and involved stakeholders.
Conclusion
As the JRC operates at the intersection of science and policy, it is imperative to demonstrate its commitment to conducting high-quality research with the utmost scientific integrity, in an ethical manner, and free from unwelcome external influences. Following staff surveys and thematic sessions involving discussions with JRC scientific staff in 2018–2019, the JRC has revised its procedures for ensuring scientific integrity and research ethics, and implemented the comprehensive framework SIRE for scientific integrity and research ethics.
The thematic sessions and survey identified key issues relevant to the JRC:
Authorship disputes.
Collaboration methods inside and outside the European Commission.
Choosing research approaches for policy support.
General concerns about research ethics and scientific integrity.
These findings underscore the need for a comprehensive framework to tackle these issues throughout the science-for-policy cycle.
This framework includes a Scientific Integrity Statement as the foundational document, a Scientific Integrity Officer who serves as the contact person for JRC researchers on research integrity issues, an Editorial Review Board for peer-reviewing and quality control of JRC scientific manuscripts, a Research Ethics Board for the ethical screening of projects, as well as guidelines and informational materials on responsible research conduct.
While most of the efforts have so far been focussed on conducting and managing research, further exploration is needed to determine if additional efforts are required in the areas of communication, and policymaking. This could involve increased training for communicators and policymakers, the development of informational materials on scientific procedures and integrity standards for policymakers and stakeholders, and the monitoring and follow-up on how research is implemented and utilised throughout the entire policy cycle.
We believe the JRC has made substantial progress in establishing its Scientific Integrity and Research Ethics framework, which is well-documented. Other organisations operating at the science-for-policy interface are invited to compare this approach with their own practices.
Supplemental Material
sj-docx-1-rea-10.1177_17470161241243001 – Supplemental material for Fostering scientific integrity and research ethics in a science-for-policy research organisation
Supplemental material, sj-docx-1-rea-10.1177_17470161241243001 for Fostering scientific integrity and research ethics in a science-for-policy research organisation by Göran Lövestam, Susanne Bremer-Hoffmann, Koen Jonkers and Pieter van Nes in Research Ethics
Footnotes
Acknowledgements
The members of JRC Scientific Committee, the JRC Editorial Review Board, and the JRC Research Ethics Board are acknowledged for their support in developing the SIRE framework and setting up the different parts of the SIRE.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]() .
.
Ethics approval
This study was previously discussed and approved by the scientific committee and the senior management of the JRC. Survey participants were asked for their informed consent and provided with clear and concise information about the questionnaire including its purpose. The data were collected anonymously and treated confidentially. The survey was supervised by the JRC scientific integrity officer. The authors choose not to publish the data because they were aware of the sensitivity and ethical concerns associated with doing so.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
