Abstract
Many countries, including Australia, have established a national scheme that supports the recognition of a single ethical review for multi-centre research conducted in publicly funded health services. However, local site-specific governance review processes remain decentralised and highly variable. This short report describes the ethics and governance processes required for a negligible risk national survey of physiotherapy-led airway clearance services in Australia. We detail inconsistencies in research governance document preparation and submission (platforms, processes, forms and signatories) and report the time cost and likely impact of these inconsistencies on health services research outcomes. Processes and strategies that facilitated success in this project are identified and summarised as helpful hints to other researchers looking to embark on negligible risk research in public health facilities.
Keywords
Ethical review for human research is based on the degree of risk involved to the participants (National Health and Medical Research Council (NHMRC) et al., 2007 (updated 2018)). Negligible risk research, with no foreseeable risk of harm or discomfort to the participant beyond inconvenience, such as participating in a de-identified survey, may be exempt from ethical review (Scott et al., 2020). In an effort to reduce unnecessary duplication, many countries, including Australia, have established a national scheme that supports the recognition of a single ethical review for multi-centre research conducted in publicly funded health services (Canadian Institutes of Health Research, 2018; NHMRC, 2011; Scott et al., 2020). Publicly funded health services can require site-specific governance review for research protocols, a process that remains decentralised and highly variable.
This short report describes the ethics and governance processes required to undertake a national survey of physiotherapy-led airway clearance services in Australia. There is currently no register or government data that details the location and models of care for these services. This project contained two phases to address this gap: the first, a scoping review that aimed to report on the availability of web-based information about public and private airway clearance services in Australia (Cooper et al., 2019) and the second, a national survey of identified services to describe current models of care (Cooper et al., 2023).
The survey involved a single pre-identified participant (respiratory physiotherapist working in airway clearance identified in the scoping review) completing an online questionnaire of 15 minutes duration. The survey sought information on the estimated number of clients in past year, referral pathways and programme description (staffing, scheduling, interventions offered). The respondent site was not required to access individual patient records or health service data, collect individual client data or provide summary analysis of data.
Started out well (July 2018)
For each service identified online in the scoping review, contact was planned to confirm currency of information. Ethics approval was provided by the host university Human Research Ethics Committee (HREC) for initiating contact with sites to confirm currency of information, and to disseminate the proposed national survey. Both phases of this project were considered as ‘negligible risk’ research (NHMRC et al., 2007 (updated 2018)).
First challenge (November 2018 to May 2019) ‘Cannot confirm currency of web-based information without approval from my HREC provider’
Most sites (291/296, 98%) contacted as part of the scoping review confirmed currency of their service provision and contact details based on the ethical approval provided by the host university HREC. Five health sites (including all of Queensland Health) contacted during the scoping review requested ethical approval from a National Mutual Acceptance (NMA)-recognised ethics committee or their local equivalent in order to provide this information. To ensure all states and territories were represented in the sample for the proposed national survey, NMA was sought and gained from the host public health facility HREC. This permitted ethical approval under the national recognition scheme of the two phases of the research project across applicable Australian jurisdictions (three quarters of the eight national States and Territories were covered by this scheme at that time).
With NMA ethics approval, the service contact details of all 296 identified web-based services were confirmed. Multiple attempts resulted in direct contact being made with 234 of these health services via telephone or email (confirmation in one attempt 111/234 (47%), two attempts 72/234 (31%), three attempts 51/234 (22%)). A total of 176 sites confirmed airway clearance services across 76 local health networks.
Second challenge (June to October 2019) ‘But what about site specific governance?’
Each of the 176 confirmed services required site-specific research governance approval for a public service health professional to participate in the survey. Initial contact was made with the research governance officer (RGO) assigned to each local health network (n = 76). Figure 1 summarises the processes navigated from the point of approaching health networks for site specific governance, to the outcomes achieved.
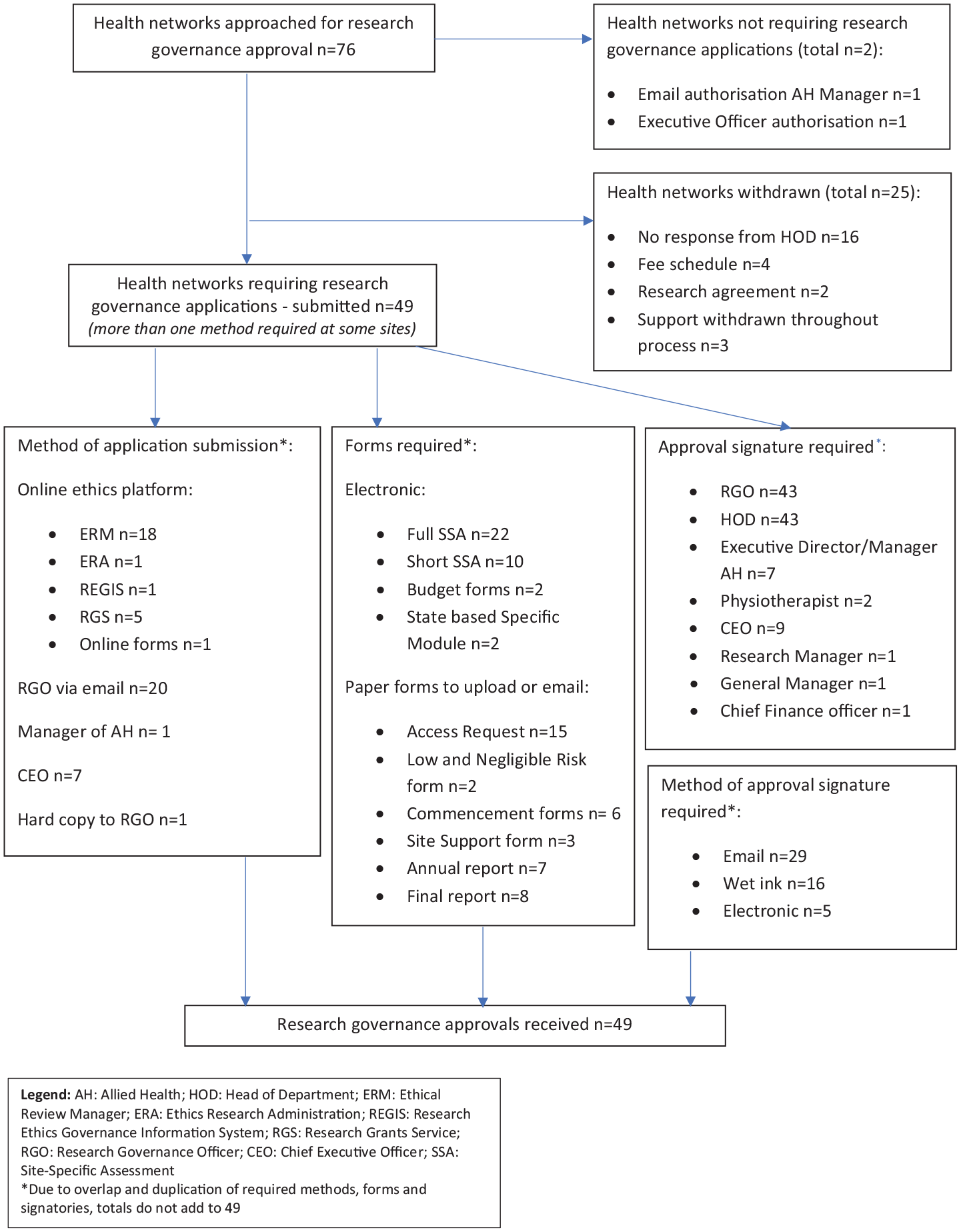
Public health network research governance processes and outcomes for a negligible risk survey study.
Each health network had site-specific information about governance requirements on their webpages. While there was relative uniformity in the online information available, local RGOs within the same State/Territory often requested varied application forms.
Research governance applications were prepared for all 76 health networks and each required support from the relevant Head of the Physiotherapy Department. Applications were unable to be progressed at 25 sites, due in most cases to lack of response from the Head of Department (16/25, Figure 1). Two sites requested a collaborative research agreement, but as sites were not taking responsibility for the overall conduct of the research, this was not able to be negotiated. Five networks required a fee (AU$220 to $440) to consider new governance applications. As the proposed research was unfunded, sites were approached for a waiver of the fee schedule. One network waived the fees and applications were discontinued for the remaining four that were unwilling to waive the fee.
Third challenge: ‘Does anyone use the same platform, process, forms or signatures for site specific governance?’
Approval was gained from all 49 governance applications submitted. There were significant variations and duplications in the required methods of document preparation and submission, as we describe below.
In general, smaller regional health networks did not have access to electronic platforms or dedicated RGOs assigned to co-ordinate governance processes. In seven regional networks, the health network Chief Executive Officer (CEO) was contacted directly via email to seek clarification on local governance requirements and processes. After provision of all relevant study documentation, in each case, the CEO provided timely written approval to disseminate the survey throughout their organisation.
At sites without electronic platforms (n = 23), health networks had separate paper forms for access requests versus low and negligible risk studies. Eight health networks had ongoing reporting requirements throughout the life of the project (Figure 1).
Where electronic platforms required electronic signatures (n = 25), only five signatories were able to sign the forms electronically due to difficulties with registering on their local platform and/or confusion over the order the electronic signatures were obtained, where the incorrect order invalidated the application. For the remaining 20 cases, copies of electronic documents were sent to the signatory for a wet ink signature, returned to the applicant, and then uploaded into the electronic platform as an additional document.
Survey disseminated (November 2019)
The survey was disseminated to 126 public health sites across 51 networks and was open for 1 month for each site. A total of 91 unique responses were available for analysis.
Summary
There were 17 months from the original host university ethics approval to dissemination of the survey. It took seven of these months to gain research governance approval at 51 Australian public health networks for their sites to participate in this negligible risk research (average of 50 [39] business days; median [interquartile range], range = 5–94 days). Much of this time was taken up with determining individual site requirements and then preparing and submitting documents (21[37] days, range = 2–74 days). Our approach achieved national representation with a high proportion of rural and regional responders (53/91, 58%). However, at sites where barriers in governance processes contributed to lengthy time frames and site disengagement, we were unable to progress to data collection creating a potential source of bias in the survey responses.
Compared to previous research that has described variation in the time taken to obtain governance review (Duplancic et al., 2019) and complete negligible risk studies (Rush et al., 2018), this survey achieved excellent representation from a high number of health services in a shorter space of time. Strategies that made this process as efficient as possible were: searching for information commonalities on governance websites; frequently communicating with RGOs regarding site-specific governance requirements; and keeping a detailed and up-to-date database of all communication and site-specific requirements. Applying for NMA HREC for five sites was unanticipated but we chose to re-apply in order to have representation from as many States and Territories as possible. Applying to an NMA HREC initially, asking RGOs for known site-specific variations and seeking acknowledgement of the application at a state-wide level were useful lessons learned. In the absence of any clear site-based governance directive, contacting the CEO of the health organisation directly had its advantages but is not a desirable nor sustainable method of obtaining approval.
Despite identification of the barriers and challenges and frequent past calls for change (Clay-Williams et al., 2018; Glasziou et al., 2021; Haas et al., 2019; Parliament of Australia, 2021; Samir et al., 2022; Scott et al., 2022), our experience reflects that inconsistencies in governance processes are still problematic, impacting on study outcomes, researcher time burden, and potentially influencing decisions to conduct future studies. We encourage efforts working towards consistent and streamlined research governance processes to support high quality, representative conduct of clinically relevant health services research.
Footnotes
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]() LC is supported by an Australian Government Research Training Programme (RTP) Fee Scholarship (fee waiver). The funding body played no role in the design of the study; collection, analysis, and interpretation of data, nor in writing the manuscript.
LC is supported by an Australian Government Research Training Programme (RTP) Fee Scholarship (fee waiver). The funding body played no role in the design of the study; collection, analysis, and interpretation of data, nor in writing the manuscript.
Ethics approvals
University of South Australia HREC (Approval No. 201308 / July 16, 2018).
Southern Adelaide Clinical Human Research Ethics Committee (Approval No. 10.19/March 7, 2019)
