Abstract
Members of research ethics committees (RECs) face a number of ethical challenges when reviewing genomic research. These include issues regarding the content and type of consent, the return of individual research results, mechanisms of sharing specimens and health data, and appropriate community engagement efforts. This article presents the findings from a survey that sought to investigate the opinions and attitudes of REC members from four Arab countries in the Middle East and North Africa (Egypt, Morocco, Sudan, and Jordan) toward these ethical issues. Our findings suggest that efforts are required to better familiarize REC members with the requirements for ethical biobank research. Additionally, we recommend that further research is undertaken with REC members regarding the main items that should be present in the Material Transfer Agreements /Data Transfer Agreements in their corresponding countries and the type of consent that should be used in genomic research.
Introduction
Advances in genomics have promoted the development of large biobanks at local and national levels. These biobanks collect and store biospecimens and associated health data that can be distributed to investigators for future genomic research. Recently, biobanks have been established in low- and middle-income countries (LMICs). The development and management of biobanks raise several significant ethical and legal challenges (Budimir et al., 2011; Coppola et al., 2019). These challenges include the content and type of informed consent, appropriate return of the individual return of results, data transfer agreements (DTAs) and material transfer agreements (MTAs) (Tindana et al., 2020), and methods to engage the community (Budimir et al., 2011; Coppola et al., 2019).
Consequently, members of research ethics committees (RECs) face challenges when reviewing genomic research (Huanhuan et al., 2021). For instance, RECs in LMICs might lack the necessary expertise (Ochieng et al., 2020) and fail to realize the proper recognition of their exact roles and levels of oversight when reviewing genomic research (Azahar et al., 2023). Finally, the lack of national regulatory frameworks governing biobank research in LMICs might lead to inconsistency among RECs in reviewing such research (Vodosin et al., 2021).
Issues regarding informed consent procedures present other challenges. For example, REC members can hold different views regarding the type of consent used in genomic studies. One study showed that many REC members believe that broad consent is suitable for research in LMICs (Tindana et al., 2020). However, broad consent can present difficulties in discussing with participants concepts regarding specimens and data sharing for future unspecified research, especially in communities with low to average literacy (Cheah et al., 2018; Petrini, 2010). This concern can be addressed by using other types of consent, for example, tiered consent, specific consent for each new study (Mikkelsen et al., 2019), or dynamic consent (Steinsbekk et al., 2013; (Abdul Aziz and Mohd Yusof, 2019). Another challenge for informed consent is deciding precisely what information should be disclosed regarding whether and to what extent results are returned to participants participating in genomic research (Pereira et al., 2016; (Vears et al., 2021).
The nature of biospecimen and data-sharing agreements (MTAs and DTAs) also present challenges (Mahomed and Labuschaigne, 2019). Participants from LMICs in a recent global forum agreed that while more efforts are needed to encourage data sharing, international guidelines are necessary to protect the interests of LMICs (Bull and Bhagwandin, 2020).
Finally, efforts at engaging communities can be complex due to cultural beliefs, scientific literacy, and the appropriate interaction with the community. For example, investigators need to tailor a unique model to help empower the community to act as agents of change (Staunton et al., 2018). Moreover, Tindana et al. (2020) found that REC members believed that efforts should be made to keep community members updated on the status of research conducted on their stored samples to sustain trust relationships.
Few studies have investigated stakeholders’ perspectives on biobank research in the Middle East. One study explored the perspectives of biobank managers (Abdelhafiz et al., 2022) and several surveyed research professionals (Alahmad et al., 2015, 2020; Al-Ebbini et al., 2020; Alkaraki et al., 2022). In addition, one paper explored the Islamic perspectives regarding biobanks (Alahmad and Dierickx, 2018). However, we are unaware of any investigations that examined the viewpoints and practices of REC members regarding their review of biobank research. Accordingly, we aimed to obtain data about REC members’ perspectives and practices regarding various issues of genomic research. Specifically, our objectives included: (1) to determine the informational elements that are included in genomic research informed consent forms and the opinions of REC members regarding the information that should be included in these consent forms; (2) explore REC members’ opinions of the type of informed consent required for biobank research; and (3) investigate the attitudes of REC members regarding aspects of biobank research (specifically, the return of incidental research results, information in MTA/DTAs, and the type of efforts to engage communities).
Methods
Study Design: We conducted a cross-sectional questionnaire study involving local research ethics committees in four countries in the Arab region of the Middle East: Egypt, Jordan, Morocco, and Sudan. We recruited participants between July to September 2020.
Study tool: We developed a questionnaire by identifying the ethical issues related to the management of biobanks. All questions were closed-ended and in the English language. A multidisciplinary team of experts in research ethics from the countries included in this study developed the individual questions based on the existing literature. We assessed the content validity of the initial version of the questionnaire by having five biobank experts review the content of the questions. The final version of the questionnaire was developed based on these comments.
The questionnaire opened with definitions of the following common biobank concepts: “biobank,” “stored tissue samples,” “secondary research,” and Material Transfer Agreements (MTAs) and Data Transfer Agreements (DTAs). Subsequently, the questionnaire was organized into the following sections: (a) demographic and background information; (b) current practice regarding the information provided in consent forms currently used by RECs; (c) participants’ opinions regarding the degree of importance of elements in informed consent for biobank research; (d) current practice regarding the type of consent forms that their REC has reviewed for future unspecified research; (e) participants’ opinions regarding the preferred type of consent form to be used in future unspecified research; (f) participants’ attitudes regarding issues involved in the return of individual research results; (g) participants’ opinions regarding information items that should be included in a material transfer agreement; and (h) participants’ attitudes regarding methods of community engagement.
We calculated a total score for the questionnaire sections regarding MTAs and community engagement. For the section on MTA/DTAs, the scoring point rubric was yes = 3, I don’t know = 2, and no =1. For the section on community engagement, the scoring point rubric was very important = 5, important = 4, slightly important = 3, not important = 2, and neutral = 1.
We calculated the total score for each section by simply adding the responses. The response options’ numerical values for negatively worded statements were reversed. The possible MTA/DTA total score ranged from 10 to 24, and that for community engagement score ranged from 6 to 30.
We summarized the data from the questionnaire as frequencies and percentages for categorical variables and mean ±SD for normally distributed quantitative data. Data were analyzed using the SPSS software analysis package version 24.
Results
We collected complete responses from 83 REC members. Most were from Egypt (65.0%), an equal number were from Morocco and Sudan (13.3% each), and 4.8% were from Jordan. Over half identified as female (55.4%) and were graduates (51.8%). Although most members (82.9%) reported that biobanks did not exist in their institutions, 50% said that their RECs reviewed protocols involving the collection of biological samples for future research. Many reported that their RECs reviewed genetic research protocols (62.2%). More than half (64.1%) had been members of a REC for more than 2 years (see Table 1).
Description of study participants’ characteristics (n = 83).
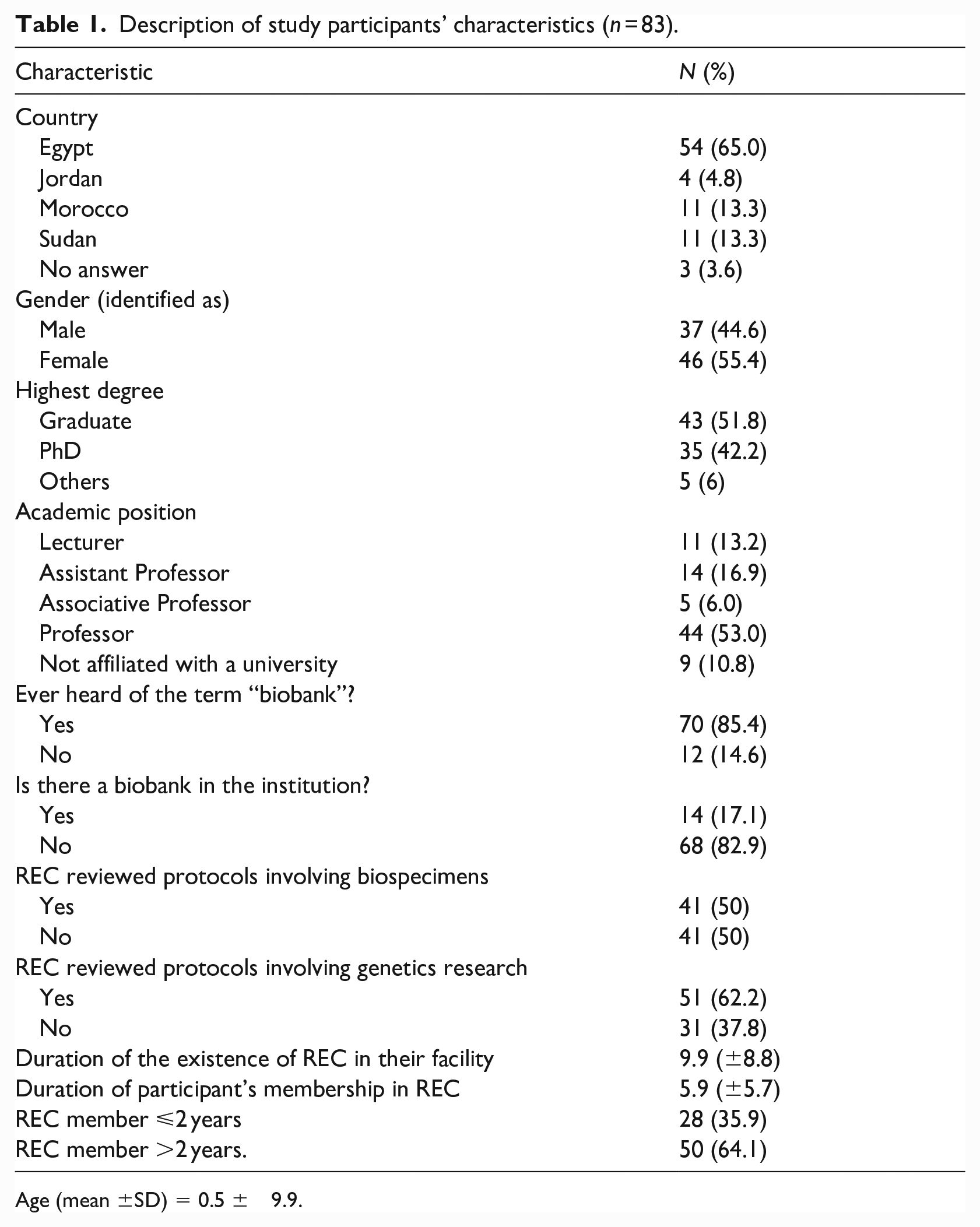
Age (mean ±SD) = 0.5 ± 9.9.
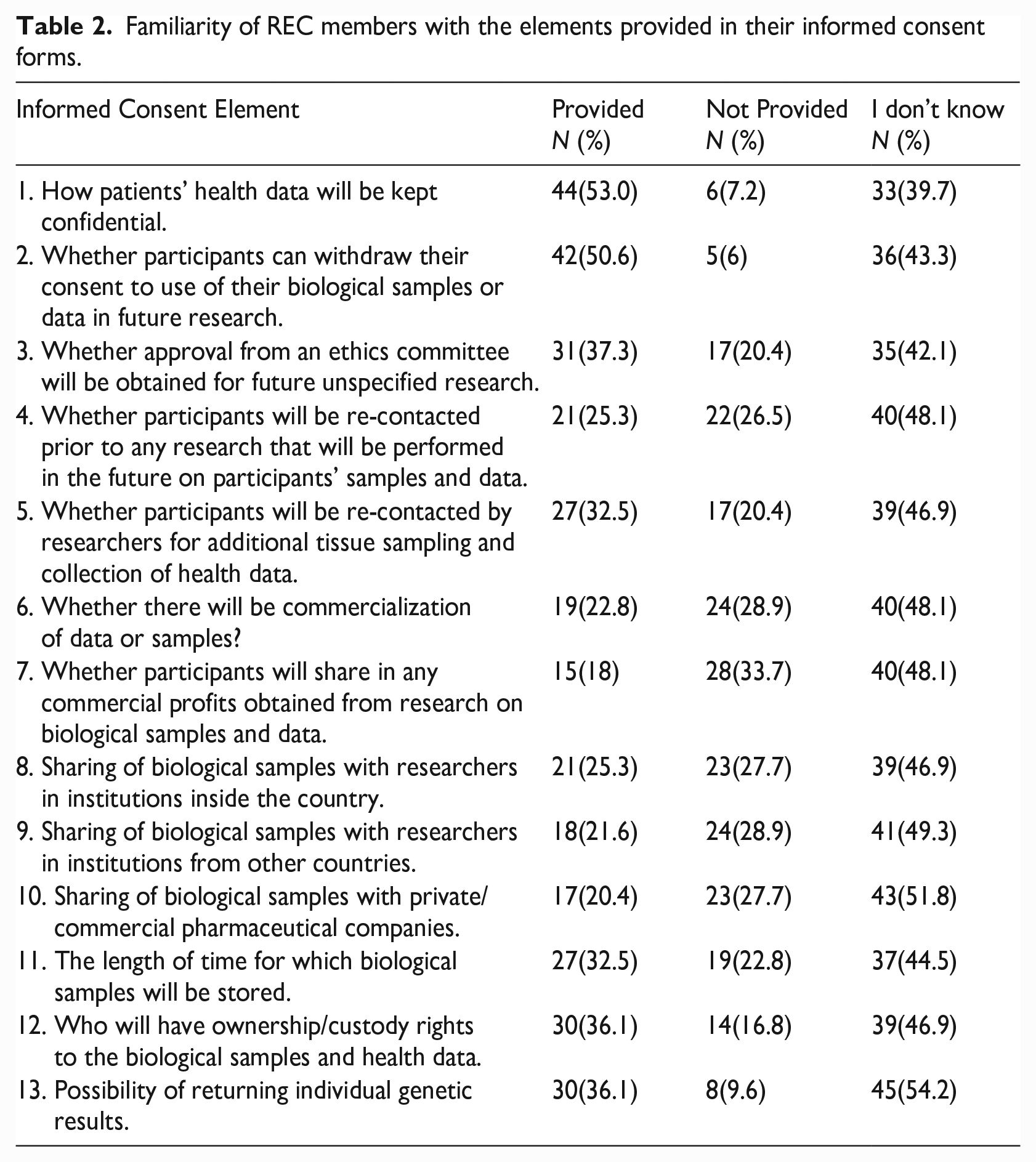
Regarding the REC members’ familiarity with the elements of information provided in their informed consent forms, approximately 40-55% of the REC members revealed that they did not know whether specific elements were included. About 20-50% of the REC members knew of several elements of information that were provided. These included elements involving confidentiality of health data, withdrawal of consent for the use of biological samples or data in future research, re-contact for additional tissue sampling and collection of health data, commercial profits obtained from research on biological samples and data, sharing of biological samples with researchers from other countries, and sharing of biological samples with private/commercial pharmaceutical companies. (See Table 2 for additional details).
Familiarity of REC members with the elements provided in their informed consent forms.
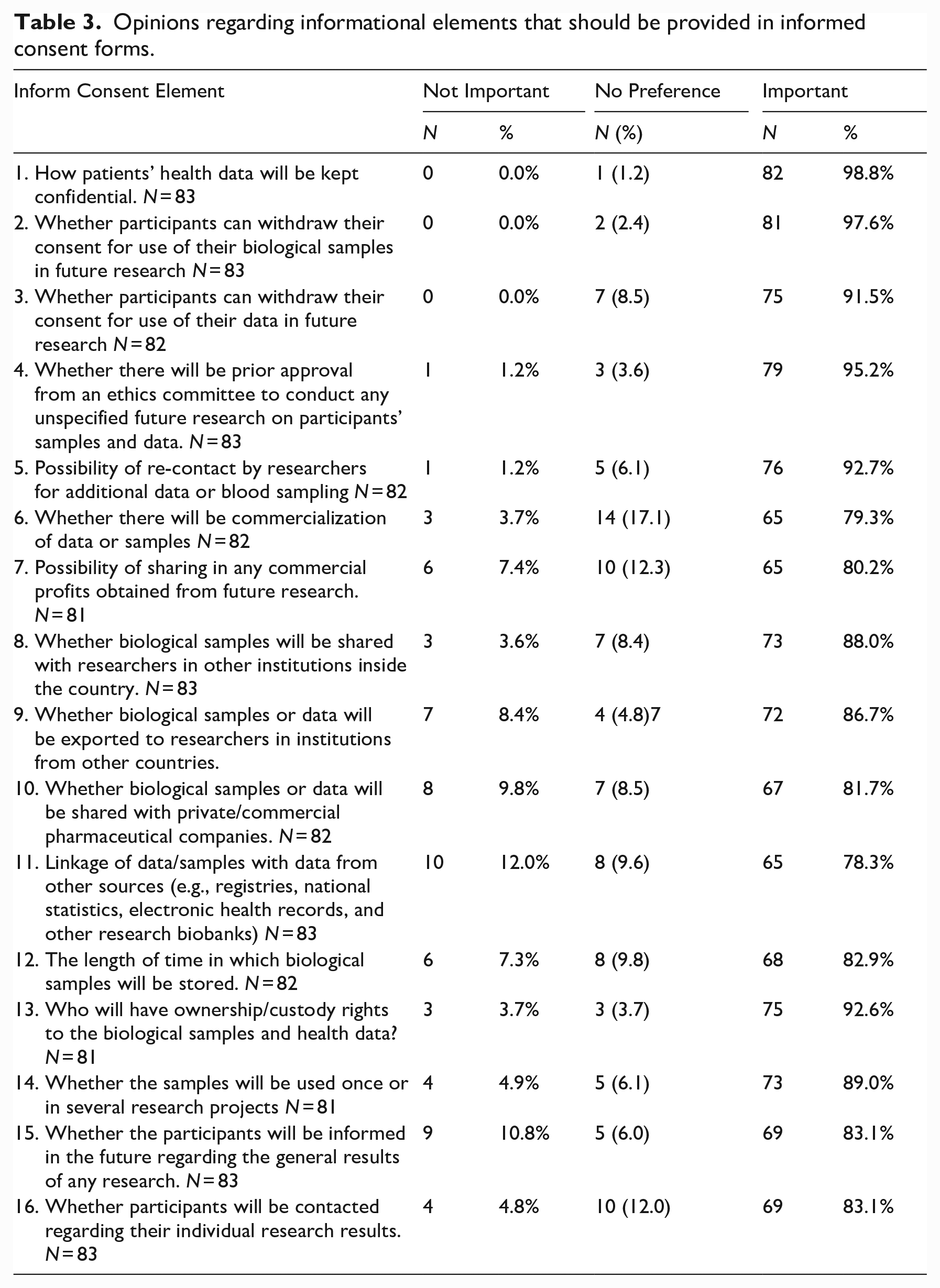
Concerning REC members’ opinions about the elements of informed consent specific to genomic research that should be provided, more than 80% believed that many (14/16) of the listed elements should be included in consent forms (see Table 3).
Opinions regarding informational elements that should be provided in informed consent forms.
When asked about the types of consent used previously for future unspecified research, a significant majority (70%) stated: “broad consent.” See Figure 1 for more details.
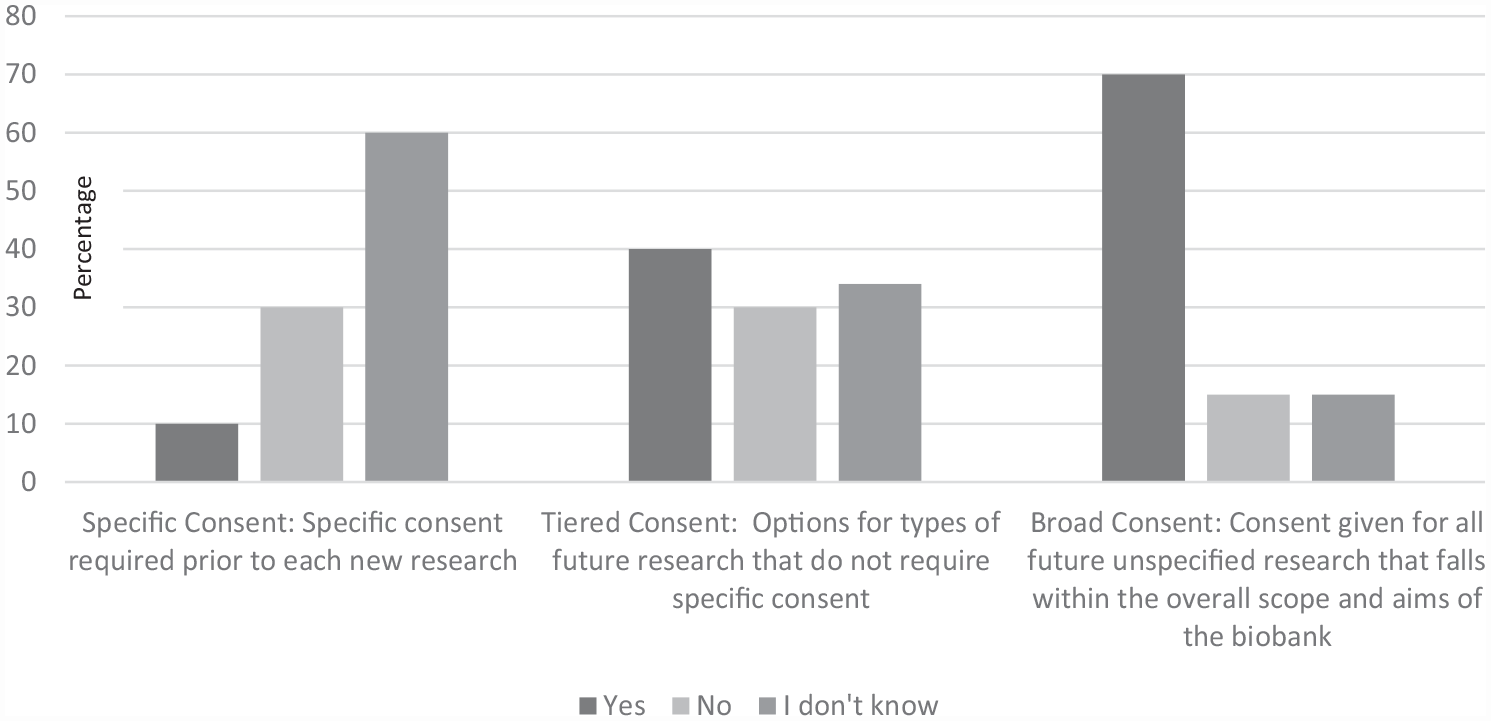
Frequency of types of consent forms previously approved by the research ethics committees.
Regarding REC members’ opinions on the type of consent that should be used for the collection of biospecimens for future research, approximately a quarter chose broad consent (26.8%), specific consent (25.4%), or dynamic consent (23.9%). A lower number of participants favored tiered consent (16.9%). (See Table 4).
Opinions regarding the type of informed consent to use for the collection of biospecimen for future unspecified research, n = 71 (only participants who gave one choice).
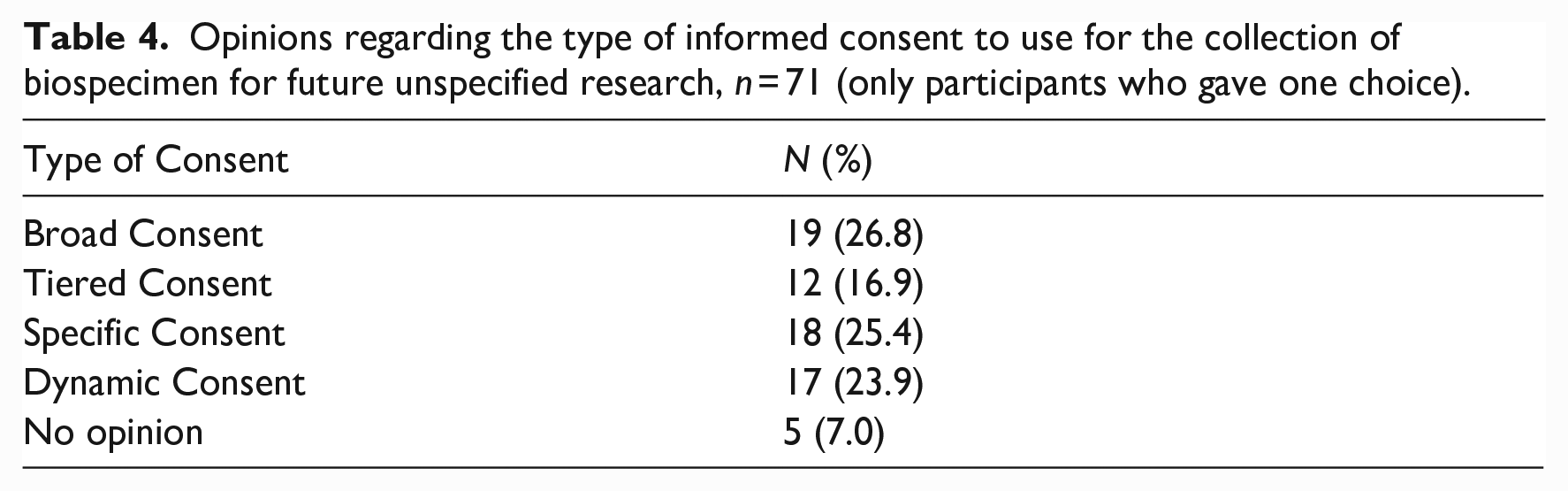
Table 5 shows the REC members’ attitudes regarding the return of individual research results to participants. A slight majority of the REC members (51.8%) held that individual research results should be returned to the participants if the medical intervention could address the genetic abnormality. A significant majority (38.3%) thought results should be returned regardless of whether an action could be taken, and only 9.9% percent stated that genomic results should not be returned to the participants.
Attitudes regarding return of individual research results to participants.

Concerning the information that should be included in MTAs or DTAs, the mean total score was 19.61 (maximum possible score = 24), indicating that REC members favored many of the listed elements that should be included in these agreements. (See Table 6).
Participants’ opinions regarding the information that should be in the Material/Data Transfer Agreement.
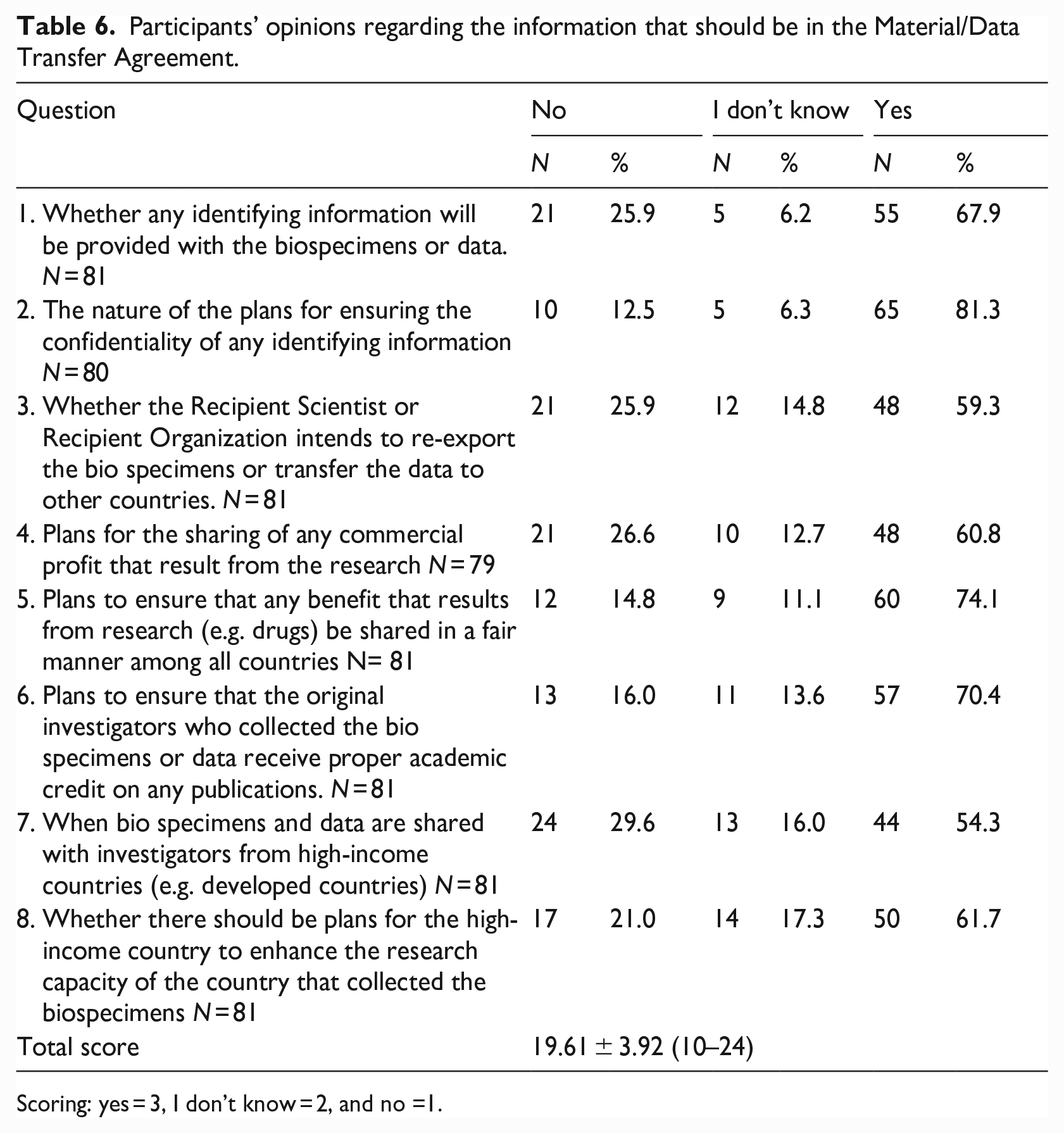
Scoring: yes = 3, I don’t know = 2, and no =1.
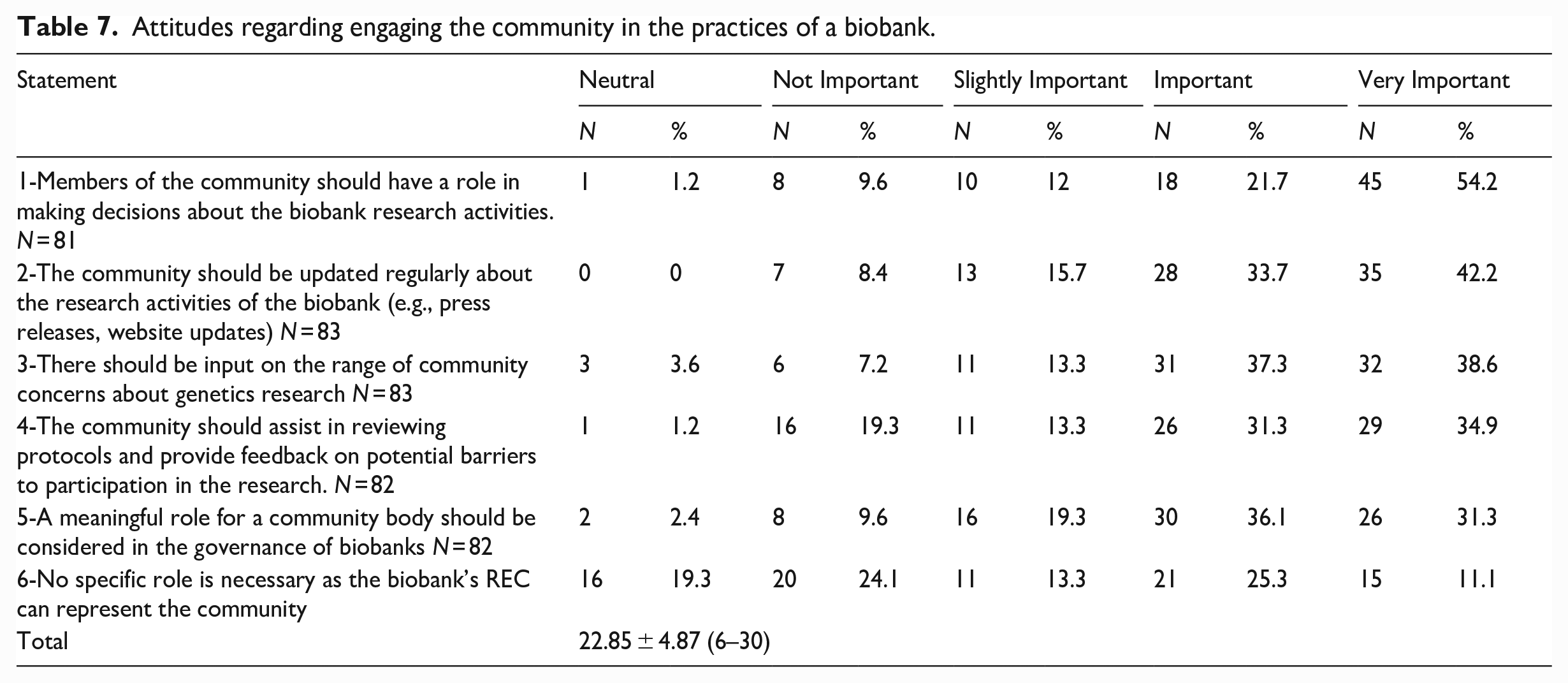
Regarding the attitudes about engaging the community, the mean total score was 22.85 (maximum possible score = 30), indicating that REC members held affirmative attitudes about engaging communities. Specifically, a sizable majority of the members thought it would be “important” or “very important” for the community to have roles that included making decisions, reviewing protocols, and having a meaningful in the governance of the biobank. (See Table 7).
Attitudes regarding engaging the community in the practices of a biobank.
Discussion
Our findings reveal critical perspectives and practices of REC members regarding ethical issues associated with biobank research. These issues revolved around informed consent, the return of individual research results, material and data transfer agreements, and community engagement.
Regarding informed consent elements, only about one-quarter of the participants acknowledged awareness of the elements relevant to biobank research was mentioned in the consent forms. As we did not access the actual information sheets and consent forms, our results might be due (at least in part) to faulty memories. However, our results do support other studies that have reviewed consent forms and show that many biobank elements relevant to genetic research are missing forms (Alkaraki et al., 2020; Al-Riyami et al., 2011; Nair and Ibrahim, 2015; White and Gamm, 2002). A study performed in Jordan indicated that 36% of researchers did not explain to participants the possibility of reuse of samples in the future, and a substantial percentage did not explain the possibility of data sharing with other researchers inside the country (42%) or outside the country (46%) (Alkaraki et al., 2022).
The absence of critical elements regarding genetic research might reflect REC members’ lack of awareness of the required guideline recommendations regarding the necessary critical information. Unfamiliarity with the regulations, a lack of research ethics-trained staff, and a lack of a governance framework have been identified by others as challenges for biobank research in LMICs (Barchi et al., 2015; Bull and Bhagwandin, 2020; Ezzat et al., 2022; Nansumba et al., 2019; Yakubu et al., 2018).
Bull and Bhagwandin (2020) maintain that failure to provide participants with necessary information regarding genetic research threatens the protection of participants, fails to respect persons, and can compromise trust in relationships. Regarding the significance of trust, Ngwenya et al. (2020) concluded that participation in biobank research is based on trust, regardless of the purposes of the study, especially in communities with high rates of poverty and illiteracy.
A sizable majority of the REC members in our study believed that all the recommended elements relevant to genetic research should be included in the consent forms. These results align with previous studies showing that REC members from LMICs strive to protect participants by providing them with as much relevant information as possible in the informed consent forms (Barchi et al., 2015; Bull and Bhagwandin, 2020).
Participants revealed that different types of consent had been used in their review of research, with broad consent being the most reported (more than 70%). However, participants’ opinions regarding the type of consent that should be used were evenly split between broad, specific, and dynamic consent. Broad consent might be more feasible for settings with limited resources to re-contact participants, while specific consent is more respectful of autonomy and building trusting relationships (Moodley and Singh, 2016). A South African study (Mwaka and Horn, 2019) revealed that most investigators believed that re-consent is obligatory if they wish to investigate an unrelated condition or when genetic analysis is added to the original research. Furthermore, surveyed participants from LMICS prefer to remain engaged in the future use of their specimens (Luque et al., 2012). Regarding dynamic consent, while the digital communication aspects of dynamic consent can enhance participants’ autonomy and control regarding the specimens and data and increase engagement between participants and biobank managers, implementing dynamic consent can be resource-intensive and might lead to participants deciding to withdraw, which can negatively impact research quality. Furthermore, the “digital divide” might exacerbate inequities among marginalized communities (Steinsbekk et al., 2013; Teare et al., 2021).
Concerning the return of individual research results, our study showed that only a slight majority of REC members agreed that it is essential to inform participants of their results only if a medical intervention can be given to address the genetic abnormality. In contrast, a sizable minority believed that all results should be returned, regardless of whether a treatment or intervention could be offered. A recent systematic literature review revealed that professional stakeholders generally prioritized the return of individual results that could potentially change clinical management (Vears et al., 2021).
Regarding MTA/DTAs, most REC members believed that much of the recommended information for MTA/DTAs should be included. However, a sizable minority of the members did not think that information regarding data export to other countries or information about intentions to use for commercial profit is essential for the MTA. Similarly, Sather and colleagues (Sathar et al., 2014), in their review of genetic proposals, found several critical items missing from the MTAs.
Most REC members in our study agreed with the importance of including detailed aspects about sharing data/ samples with researchers locally or outside the country and with pharmaceutical companies. A survey of biomedical researchers in Jordan showed that 61.7% of Jordanian researchers shared samples locally, while 47.2% shared biosamples with colleagues from outside Jordan (Alkaraki et al., 2022). Participants in the Global Forum on Bioethics in Research (GFBR) explored ethical issues associated with data sharing in LMICs and agreed that data sharing should be encouraged to decrease inequity between HICs and LMICs (Bull and Bhagwandin, 2020). Furthermore, data sharing is critical for reproducing previous study results, discovering new medicines, and enhancing studies of rare diseases by combining different data sets (Kodra et al., 2018). However, a significant concern is that LMICs with limited resources receive little in return for sharing data, while HICs receive valuable data/ samples (Singh et al., 2022), raising issues regarding the exploitation of vulnerable communities in Africa (Moodley and Singh, 2016).
We found that most REC members held affirmative attitudes with many issues regarding community engagement, including having a role in the governance of biobanks. This result aligns with the prevailing opinion that community engagement enhances trust between professional stakeholders and community members (Adhikari et al., 2020); (Kerasidou, 2017). Furthermore, participants at the previously mentioned Global Forum held in Africa recognized the importance of implementing practical approaches to community and public engagement around data sharing and biobanking in LMICs, which go beyond providing information and facilitating the consent process (Bull and Bhagwandin, 2020). Finally, engaging the community can ensure that the proposed research serves the community’s priorities (Ngwenya et al., 2020).
Limitations
We acknowledge several limitations to our study. First, we recognize the existence of sampling bias due to the self-selection nature of recruitment and the potential for exclusion of some (for instance, due to network connectivity issues). Second, we failed to reach the calculated sample size. This might have been due to several factors, including that the pool was smaller than predicted, inaccurate email addresses and WhatsApp information, or individuals being uninterested /too busy.
Additionally, many responses (27% of total responses) required exclusion from our analysis due to incompleteness. This lack of a representative sample and an incomplete sampling frame limits the generalizability of our results.
Conclusions and recommendations
Our results demonstrate that efforts are required to better familiarize REC members with the requirements for ethical biobank research. While our study showed REC members’ positive opinions toward the provision of detailed information relevant to genomic research during the informed consent process, only a minority were able to confirm that the relevant component elements were provided in their consent forms.
Similarly, regarding MTA/DTAs, most REC members believed that much of the recommended information for MTA/DTAs should be included. However, a sizable minority of the members agreed about the need to include some information required for fair and ethical practice (for instance, when data are shared with investigators from high-income countries).
While a sizeable majority (approx. 80%) of our participants believe the involvement of communities in biobank research activities is necessary, a significant minority (more than 40%) thought that the RECs could represent the community interests.
There is also a need for further discussion and agreement about the type of consent most appropriate in different circumstances; our REC members’ opinions were evenly split between broad, specific, and dynamic consent.
We recommend further research is warranted with REC members regarding the main items that should be present in the MTA/DTAs in their corresponding countries and the type of consent that should be used in genomic research.
Footnotes
Authors’ contributions
Z.M. collected data from participants, contributed to the statistical analysis of the data, and wrote the first draft, F.A. collected data from participants and revised the first draft. H.J.S conceived the idea of the study, and substantively revised the work. All other authors were involved in the acquisition of data and revised the work. All authors approved the submitted version
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]() This work was supported by Award No. R25TW007090 of the Fogarty International Center at the National Institutes of Health, USA.
This work was supported by Award No. R25TW007090 of the Fogarty International Center at the National Institutes of Health, USA.
Ethical approval and consent to participate
The study was approved by the Research Ethics Committees of Suez Canal University, Egypt (Reference# 3982); Cairo University, Egypt (No. 19/10/15); Sidi Mohamed Ben Abdellah University, Morocco (Avis Du Comite D’Ethique Hospital-Universtaire Fes No 13/19); Jordan University Hospital, Jordan (Ref 67/209/5766), University of Khartoum, Sudan (Ref: FM/DO/ED); and the University of Maryland, USA (HP-00095620).
Availability of data
The dataset used in the current study is available from the senior author on reasonable request.
