Abstract
The inclusion of animals in research studies involves a great responsibility to ensure animal welfare within the relevant ethical and legal frameworks. This study aimed to review compliance with the ARRIVE Essential 10 requirements and the ethical oversight of animal behaviour and welfare studies in farm animals. Three journals and a total of 133 articles were reviewed for compliance with the ARRIVE Essential 10 items and criteria. Each article obtained a final score according to whether or not each criterion was met within each item. Likewise, ethical declaration in each article was recorded. Chi-square test and linear models were built to assess associations between the ethical statement presentation and the final ARRIVE Essential 10 score with the country, the species, and the journal. We found that 15% of the articles did not present an ethical statement. The journal with the highest impact factor and the countries of the Global South presented an ethical statement more frequently. Regarding the Essential 10, the item with the lowest agreement score was ‘Blinding’ and the one with the highest agreement was ‘Outcome Measures’. Also, significant differences were found between journals in terms of the Essential 10 score. Essential 10 provides relevant information that allows reviewers and readers to identify possible welfare risks and the validity of the results in animal welfare science publications.
Introduction
Animals remain central to biological research, either because they are used as models in biomedicine, allowing medical advances (Festing and Wilkinson, 2007), or for unravelling information about the biology and ecology of species. Nevertheless, there is evidence of a reproducibility crisis in science (Baker, 2016). In particular, the Institute for Laboratory Animal Research (ILAR) has expressed concern about the number of peer reviewed studies containing data from experiments using animals that cannot be reproduced (National Academies of Sciences, Engineering, and Medicine, 2015), often due to an inability to understand exactly how the research was conducted because the reporting is poor (Simera et al., 2010). This situation not only affects reproducibility, but also reduces the value of those studies and increases societal concerns about the welfare of the animals that were involved. It can also lead to a decrease in funding and support of science, in particular of translational research (Pritt and Hammer, 2017).
The inclusion of animals in any research entails a great responsibility for scientists, who must ensure that animal welfare is prioritised and that all actions are carried out within an ethical and legal framework (Tadich et al., 2020). Thus, many countries have developed legislation on the use of animals in research, while institutions, mainly universities, have established animal care and use institutional committees (IACUCs) (Ghasemi and Dehpour, 2009). However, most of the available legislation and guidelines are focused on biomedical research and clinical trials (for a catalogue of guidelines see: Simera et al., 2010) and do not always consider the studies that only include behavioural observations and/or welfare assessments of animals. This is probably because these activities are considered as less invasive, or potentially less harmful to the animal. For example, Directive 2010/63/EU (2010[(2010)]) states that it does not apply to ‘practices not likely to cause pain, suffering, distress or lasting harm equivalent to, or higher than, that caused by the introduction of a needle in accordance with good veterinary practice’, which may create the impression that the Directive does not apply to some observational studies. When there is no legislation in place, the responsibility rests on the IACUCs, who base their decisions on international recommendations and principles, such as the 3Rs principle developed by Russell and Burch (1959). The decisions, reviews, policies, and other actions that IACUCs recommend to scientists, in addition to ensuring the welfare of the animals involved, may also improve research reproducibility (Pritt and Hammer, 2017).
Guidelines for reporting of animal studies are helpful since they provide structured information about what is needed for a complete account of what was done (Simera et al., 2010). Kilkenny et al. (2010) led an initiative to produce guidelines for reporting animal research called ARRIVE (Animals in Research: Reporting In Vivo Experiments) which consisted of 20 items describing the minimum information that should be reported for studies including data from animals. These guidelines were endorsed by funding agencies and by journals. Nevertheless, following inception, there was little improvement in reporting standards (Baker et al., 2014; Gulin et al., 2015; Ting et al., 2015). In 2020, an updated and revised version of the guidelines was published, ARRIVE Guidelines 2.0. For the revised version, information was reorganised and classified into two sets, with the
Methodology
Journals reporting studies with animals that included words such as ‘welfare’ and ‘behaviour’ in the title were eligible for the study; journals that addressed broad topics (e.g. Animal, Animals, PLoS ONE), or that had a biomedical focus (e.g. Journal of Biomedical Science) were excluded. The published articles from three scientific journals 1 , with an aim and scope that mentioned animal welfare and ethology, were reviewed between January and March of 2022: Journal of Applied Animal Welfare Science (AAW, impact factor 1.44), Applied Animal Behaviour Science (AAB, impact factor 2.569), and Animal Welfare (AW, impact factor 2.06), according to the 2021 impact factor (IF) provided by the Journal Citation Report. The inclusion criteria for articles were as follows: articles published in 2021, with farm animals (cattle, sheep, horses, pigs, poultry, fish, goats), and that used live animals for the research. The decision to include only farm animals was based on the areas of expertise of the researchers (authors). Reviews, surveys, and letters to the editor were excluded. Among the three journals, 324 scientific articles were published in 2021, of which 133 corresponded to studies on farm animals (Figure 1).

Three scientific journals with the number of issues and articles published during 2021, and the number that met the inclusion criteria. AWW = Journal of Applied Animal Welfare Science, AAB = Applied Animal Behaviour Science, AW = Animal Welfare.
Three researchers (the authors) reviewed the journal articles. Before beginning, the researchers read two articles together to discuss and combine standards for evaluation of each article. The articles were assessed according to the ARRIVE Essential 10 (Figure 2) and the inclusion of an ethical statement. The agreed protocol was to read the abstract first, then the introduction to search for the aim and hypothesis, and finally, to the materials and methods section to check each criterion of every item of the Essential 10.
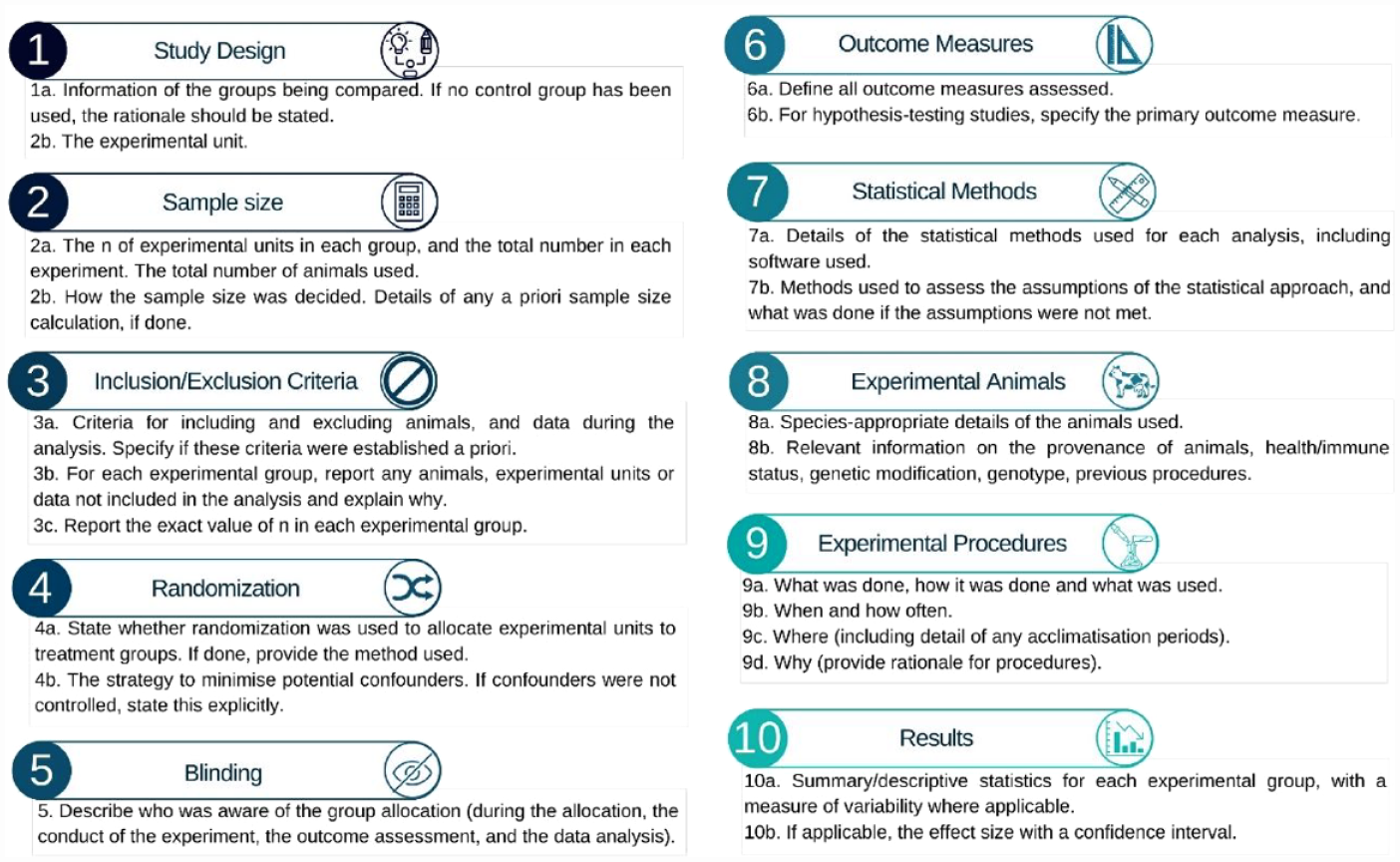
Items of ARRIVE Essential 10 and criterion within each item.
For analysis of compliance with the Essential 10, each item was assigned a score of between 0 and 1; the score varied according to the number of criteria within the item that were met. If an item had multiple criteria, 1 was divided by the number of criteria. For example, the ninth item ‘Experimental Procedures’ contains four criteria, so each had a possible score of 0.25 for meeting the criterion or 0 if it was not met. Each article was assigned a total score containing the Essential 10 (22 criteria). If a criterion was not applicable, it was awarded the maximum score.
For the presentation of the ethical statement, the complete document was reviewed, since some articles include it within the methodology and others at the end of the manuscript. If a licence or approval number was included, this was also recorded. Finally, information was also collected on the species studied, which were classified as follows: ruminants (cattle, sheep, goats), equines (horses, donkeys), pigs, poultry (laying hens, broilers, turkeys), and fish. The country where the study was conducted was also recorded according to the location of the animals described in the methodology section. Countries were then classified as Global North or Global South according to Odeh (2010). The classification does not necessarily relate to geographic location, rather it is a grouping of countries along socioeconomic and political characteristics. Global North includes Australia, Canada, Israel, Japan, New Zealand, South Korea, and the United States, Europe, Russia. The Global South is made up of Africa, Latin America and the Caribbean, Pacific Islands, and Asia, excluding Israel, Japan, and South Korea (Odeh, 2010).
Two statistical analyses were carried out using R 4.1.0 (R Development Core Team, 2021):
Information about the presentation of the ethical statement (yes or no) was used for evaluation of associations with the species studied, the country, and the journal. The articles that did not declare an approval number were also considered in the group of articles with an ethical statement. This was done using the Chi-square test or Fisher’s exact test (when sample size <5).
The final compliance score for each article was used to evaluate its association with the species studied, the country, and the journal. This was done through linear models using
Results
General description of the reviewed articles
The total number of articles reviewed was 133 from the three scientific journals. The percentage of studies reviewed according to the journal was 10 publications (7.5%) from journal AAW, 112 (84.2%) from journal AAB, and 11 (8.3%) from journal AW. The distribution of the papers reviewed by continent is shown in Figure 3, with Europe contributing the largest number of publications and Africa the smallest. According to the distribution of countries based on the Global North-South divide, 67.7% of the articles reviewed corresponded to the Global North and 32.3% to the Global South.

Distribution of the articles (
According to the species studied, most of the reviewed articles reported work with bovines (either dairy or meat), and the species least used was the donkey (Figure 4).
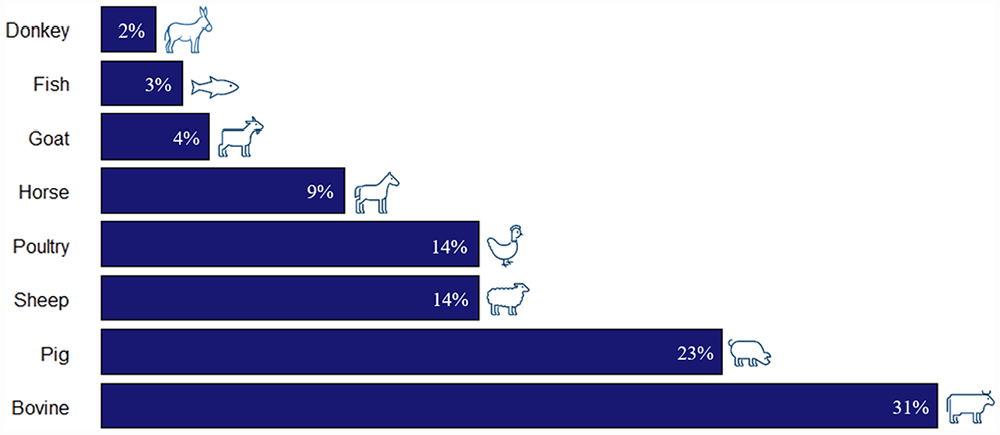
Distribution (%) of publications (
Ethical statement
Most of the reviewed publications presented an ethical statement (84.96%, 113 articles); of those, 26 publications did not deliver an ethics approval number from an ethics committee or regulatory body. The rest (15.04%, 20 articles) did not mention the ethical statement, although there were three studies in which the lack of ethical approval was justified:
The percentage of articles without an ethical statement per journal was 40% for AAW, 10.7% for journal AAB, and 36.3% for journal AW. Fisher’s test showed statistical differences between the journals and the inclusion of an ethical statement. Proportionally, journal AAB had more articles with an ethical declaration compared to journal AAW (
According to the North-South divide, 20% of the publications from countries in the Global North did not present an ethical statement compared to 4.6% of the articles from the Global South. Fisher’s test revealed that the countries of the Global North presented an ethical statement less frequently compared to the countries of the global south (
According to the species, the percentage of publications that did not present an ethical statement was 29% for equine studies, 25% for fish studies, 14% for poultry, 8% for pigs, and 12% for ruminants. Nevertheless, the statistical analysis did not reveal significant differences between the animal species and the presentation of the ethical statement.
The ARRIVE Essential 10
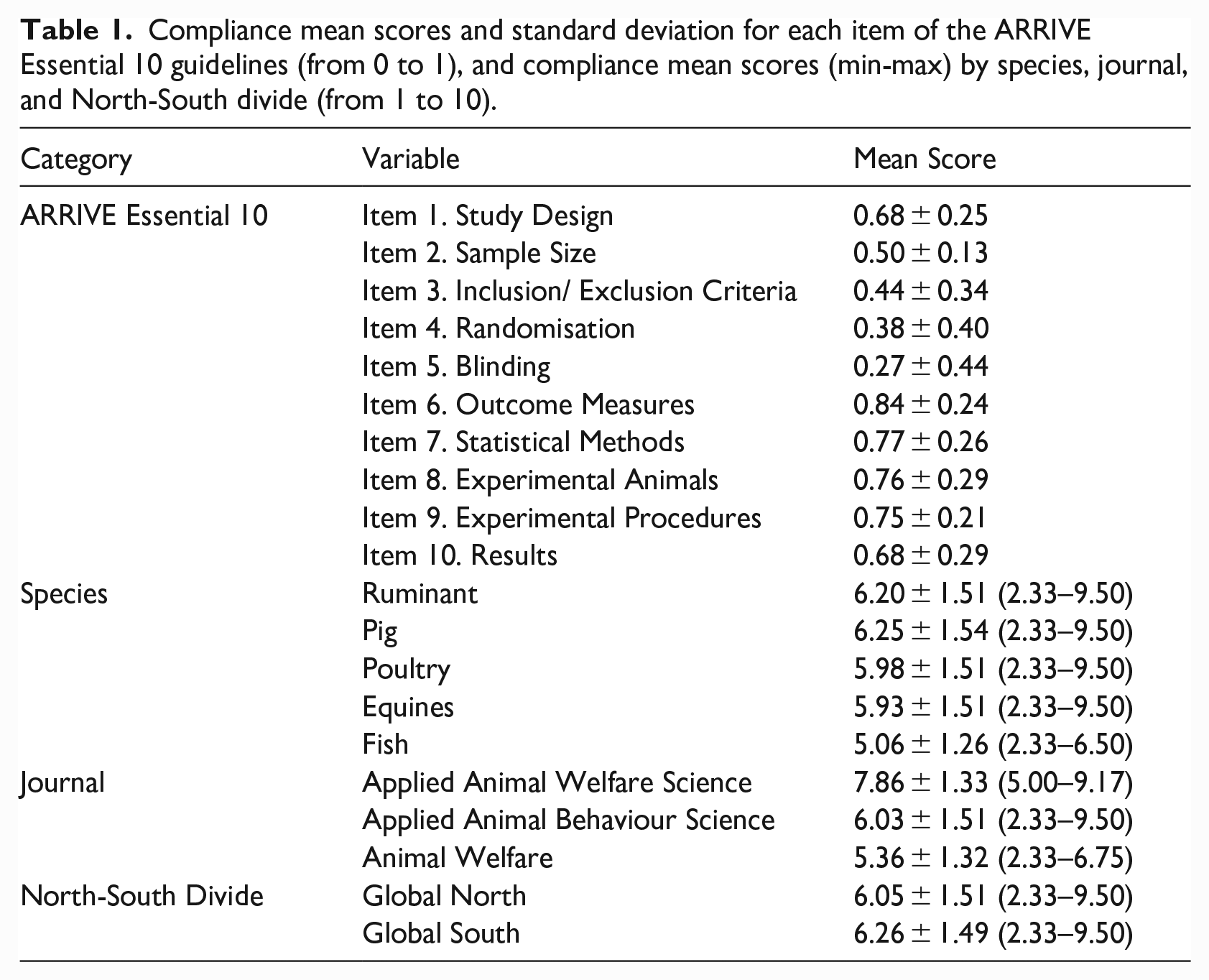
Overall, the item with the lowest compliance score was ‘Blinding’ and the item with the highest compliance score was ‘Outcome Measures’. The criterion that the largest number of articles did not meet was ‘Explain how the sample size was decided?’ from the item ‘Sample Size’. The criterion that the largest number of articles met was ‘What was done, how it was done, and what was used?’ of the item ‘Experimental Procedures’. Descriptive statistics of the compliance for species studied, North-South divide, and journal are shown in Table 1. Figure 5 shows the percentage of adherence to each criterion of the 10 items of the ARRIVE Essential 10 guidelines.
Compliance mean scores and standard deviation for each item of the ARRIVE Essential 10 guidelines (from 0 to 1), and compliance mean scores (min-max) by species, journal, and North-South divide (from 1 to 10).
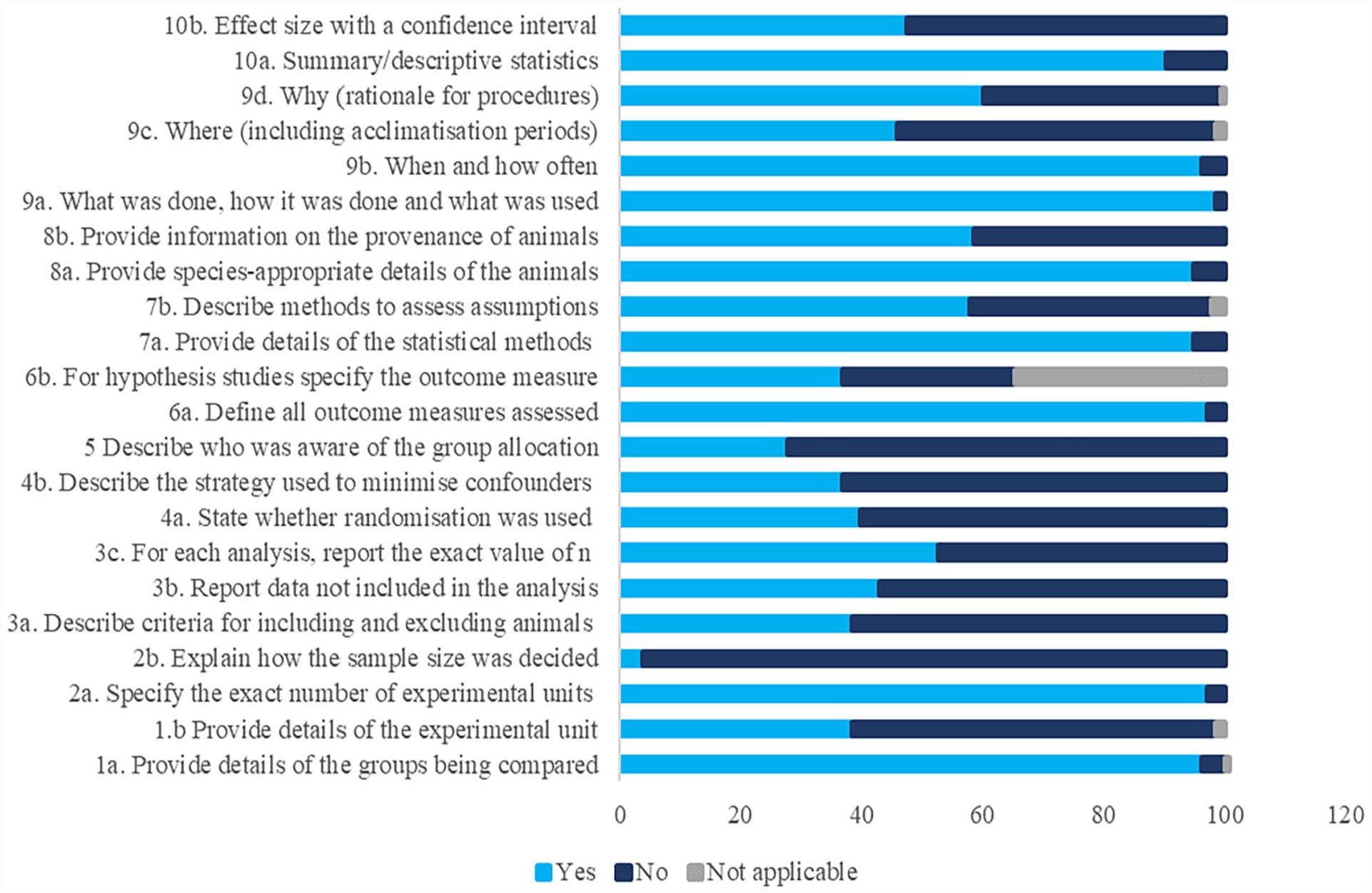
Percentage of adherence to the ARRIVE Essential 10 guidelines according to each criterion in 133 reviewed articles on farm animals.
Regarding the association between the agreement scores and the journal, the linear model was statistically significant (
Discussion
This study aimed to understand the level of compliance with the ARRIVE Essential 10 guidelines and the inclusion of an ethical statement in animal behaviour and welfare studies published in three scientific journals during 2021. Of the 133 articles included in the assessment, most had been published in AAB journal. This is because this journal publishes 12 issues each year, with an average of 20 articles per issue, while journals AW and AAW publish four issues per year with an average of 11 and 8 articles per issue respectively. Furthermore, most articles originated in Europe, with 67.7% being classified as undertaken in the Global North. This is not surprising since the bibliometric study by Freire and Nicol (2019) also reported that the countries of origin of most animal welfare studies, published between 1988 and 2017, were European countries, Canada, and the United States of America. This higher rate of scientific articles published by researchers from the Global North countries has been reported as a possible bias that could be based on institutional prestige, a shared background of editors and reviewers, geo-origin of editorial boards, language and networks, and accessibility (Baruch, 2001; Gallo et al., 2022; Patel and Sumathipala, 2001). Another reason could be a lower submission rate of manuscripts from researchers in the Global South related to a smaller percentage of the GDP of these countries being invested in science (Gallo et al., 2022; Valenzuela-Toro and Viglino, 2021).
When the articles were classified according to the farm species studied, the most frequent were with bovines, similar to the findings of Freire and Nicol (2019) and Gallo et al. (2022) for animal welfare articles. This is probably because cattle are an important source of milk and meat; when kept in intensive commercial systems, more scientific questions about cattle behaviour and welfare may arise. On the other hand, donkeys were the least studied species included in the articles assessed. This could be related to their largest populations being located in countries from the Global South and kept mainly in extensive non-commercial systems (Norris et al., 2021).
Regarding the inclusion of an ethical statement within the articles, we found that over 80% of analysed articles reported this information. This result is in accordance with other animal research studies. For example, Uiterwijk et al. (2020) reported a high percentage of articles (94%) included information concerning ethical statements in studies of pulmonary valve implantations in animals. Ting et al. (2015) found 22% of articles with no statement concerning ethics approval in animal studies in the field of experimental rheumatology. Taken together, these results indicate that, regardless of the discipline, there are a number of researchers who do not provide the information necessary to assess whether the inclusion of animals in an experimental study has been overseen by an IACUC. Thus, there is no means to assess whether basic principles like the 3Rs (Russell and Burch, 1959) and minimum welfare standards were in place, which in turn can affect the quality and validity of these studies involving animals.
All three assessed journals explicitly request the reporting of an ethical statement in their guidelines for authors. Nevertheless, our findings revealed differences between the three journals. The AAB was the journal with the highest number of articles including an ethical statement compared to the other two journals. AAB encourages researchers and reviewers to use ethical guidelines to assess the acceptability of manuscripts submitted to the journal. For this, the journal itself has conformed an Ethics Committee that has prepared its ethical guidelines and complementary material that advises researchers and reviewers (International Society For Applied Ethology, 2022). Similarly, the journal
While AAB had a greater number of articles that presented an ethical statement, articles from this journal obtained a lower average score in the Essential 10 than those from the AAW journal. This finding is in line with the lack of association found between the total score for the Essential 10 and the presentation of the ethical statement. It is important to remember that the ethical statement, which was initially part of the 20 items of the ARRIVE guidelines (Kilkenny et al., 2010), is no longer within the Essential 10 in its new version (Percie du Sert et al., 2020). Thus, authors may not necessarily be checking for these other methodological requirements.
It is also interesting to highlight that only one (AW) of the three journals assessed are listed as endorsers of the ARRIVE guidelines according to the ARRIVE guidelines website (https://arriveguidelines.org/supporters/journals). This may result in authors not checking the structured guideline when reporting their methodology and relying only on the author’s instructions, which generally are not that detailed. A study by Novak et al. (2022) revealed that the guidelines to authors from British and veterinary medical journals contained more words associated with animal welfare, the 3Rs, and the ARRIVE guidelines than those from eight other countries and other disciplines. Editors and reviewers are responsible for checking that manuscripts contain enough information to allow reproducibility of the research but assessing the quality of peer review is out of the scope of the present study, making it impossible to draw firm conclusions. It would be interesting if future studies could examine, for a larger sample, the quality of the review process and compliance with the ARRIVE Essential 10. Nevertheless, we encourage editors to review their guidelines for authors and reviewers to be attentive to the information provided that can ensure animal welfare and bioethical standards of the manuscripts.
When analysing the Essential 10, we found that items associated with blinding, sample size, and randomisation were poorly reported in the articles assessed (Table 1). These results are in accordance with Ting et al. (2015), where only 17.1% of the 41 articles reporting interventional animal studies in rheumatology reported randomisation, and 29% reported blinding. However, most importantly, none of the articles reported how the sample size was estimated. Similarly, in Uiterwijk et al. (2020) sample size calculation and randomisation were poorly reported (less than 20% of the articles assessed). Sample size calculation is crucial to ensure adequate power but also reflects that the principle of reduction, developed by Russell and Burch (1959) might be under consideration. A proper sample size calculation is indispensable for assessing the model validity and robustness of results (Percie du Sert et al., 2020), but also avoids small sample sizes that do not allow the application of proper statistical methods, or excessive sample sizes that result in wastage and raise ethical, scientific, and economic issues (Ding et al., 2022; Kang, 2021). Furthermore, a lack of reporting of these items may be associated with low awareness of scientists about reporting guidelines. For example, Reichlin et al. (2016) reported that less than half of their survey participants were aware of the ARRIVE guidelines, and of those that did know them, 15.2% declared not to adhere to them and 34.8% only occasionally.
The item with the highest average score was ‘outcome measures’, as found by Ding et al. (2022). Reporting outcome measures is vital to avoid selective outcomes reporting bias. This type of bias is more frequently observed in studies that only report significant results. Unfortunately these studies tend to be more frequently found in the experimental animal literature (Dwan et al., 2008; Ting et al., 2015; Tsilidis et al., 2013). The high score could also be associated with the number of studies that did not state a hypothesis, and thus the reporting of primary outcomes (criterion 6b, Figure 5) was not applicable, receiving the complete score.
This study found an association between the average Essential 10 score and the journal assessed. This result contrasts with the study by Uiterwijk et al. (2020), who did not find a significant correlation between agreement with the item and the 5-year impact factor of the journal in which the studies were published. Our finding that AAW journal showed significantly higher compliance scores than AAB and AW must be treated cautiously because of the differences in sample sizes. Nevertheless, it might be attributed to authors, editors, or reviewers’ awareness of ethics requirements, an aspect that cannot be evaluated within the frame of our results. We found no association between the final reporting Essential 10 compliance score and the Global North/South classification, or the species studied.
The Essential 10 items of the ARRIVE 2.0 guidelines emphasise the methodology and statistics used. This contributes to bias reduction and assesses the appropriateness of the methods used together with the validity and reliability of the results (Kang, 2021). These factors increase transparency and allow us to consider the appropriateness of the use of animals. Nevertheless, Katsnelson (2020) admits that the ARRIVE guidelines have failed to improve reporting by scientists but ARRIVE 2.0 may solve some problems by simplifying reporting for authors and reviewers. Although Ting et al. (2015) and Uiterwijk et al. (2020) published their study before the ARRIVE 2.0 guidelines, which aims to simplify reporting (Percie du Sert et al., 2020), it seems that the same items continue to be poorly reported by scientists. For example, recently, Ding et al. (2022) showed that 98.5% of the articles associated with animal models of bone defects and fractures, did not report how sample size was decided, together with 92.4% failing to report blinding and only 53.2% reporting randomisation.
Applying ethical principles and implementing animal welfare strategies is a prerequisite for successful animal experiments. Researchers should design studies considering the reduction of adverse events that can induce poor animal welfare states but should also implement refinement strategies that promote good animal welfare (Jirkof et al., 2019). Many items of the Essential 10 can have a direct impact on animal welfare. For example, reporting item number nine, associated with the experimental procedures, is fundamental to understanding how refinement measures are being implemented. In addition, in studies that induce pain in animals, information about pain management strategies (how, when and, what) is a methodological concern that directly impacts animal welfare and the reproducibility of the study (Carbone and Austin, 2016).
We found that the articles from countries of the Global South presented ethical statements more frequently and did not differ significantly in compliance with the ARRIVE Essential 10 criteria from those of the Global North. These results are in contradiction with the assumption that research studies in low- and middle-income countries (LMIC) are not subjected to the same scrutiny, ethical review processes, data monitoring, and feedback as countries in the Global North. The prejudice that international journals have about studies carried out in the Global South (mainly LMICs) is well-known and documented in several areas of scientific research (Keiser et al., 2004; Niriella et al., 2021; Tyrer, 2005; Victora and Moreira, 2006). For instance, English-speaking countries and northern European nations (e.g. Denmark, The Netherlands) have the highest publication rates in prestigious medical journals, with English proficiency being a factor for publishing in high-ranking journals (Man et al., 2004). Manuscripts in which the main author is from a non-English speaking country tend to have negative comments about language from reviewers, despite having been previously checked by a native English speaker (Valenzuela-Toro and Viglino, 2021). This leads to practices such as including a co-author from a developed country so that the study originating in a LMIC is viewed more favourably in the eyes of the reviewers (Niriella et al., 2021). Likewise, Global South authors’ manuscripts are commonly rejected for not being of general interest, or as more appropriate for a local journal, while manuscripts from high-income countries are published despite the circumscription of the topic or place of study (Niriella et al., 2021; Victora and Moreira, 2006). Also, the number of editors and members of international journals from countries of the Global South can be minimal (Keiser et al., 2004; Victora and Moreira, 2006). In the case of AAB, this has been overcome by including a more diverse (in terms of geographical origin) Advisory Editorial Board. We believe that our results may illustrate this prejudice by demanding (or questioning) more of authors from the Global South and being more tolerant of the lack of ethical statements in articles from the Global North. As mentioned by Niriella et al. (2021) in their letter on racism in academic medical publishing, manuscripts from non-Western emerging economies tend to be reviewed with greater rigour and scrutiny by journals compared to those from Western countries. This, we believe, extends to all countries of the Global South and non-English speaking countries (e.g. see barriers for Latin-American researchers, Ciocca and Delgado, 2017; Valenzuela-Toro and Viglino, 2021).
Some limitations of the present study include the small number of journals (three) included, together with an overrepresentation of one of the due to the higher total number of articles published per year by AAB when compared to AAW and AW. Additionally, the limited period studied could influence results, and future studies might include a longer period, together with more journals that publish studies with farm animals. As concern for bioethical and reproducibility standards of science has increased, one would expect the indicators studied to improve over time. This study only included journals that have a scope on animal welfare and behaviour, which incorporate studies that are usually less invasive or purely observational. In some countries, this type of study may not be submitted for ethical oversight due to low invasiveness, which could also be affecting the results. Nowadays, it is known that observational studies can induce stress by the sole presence of observers (McMann and Paterson, 2003), and hopefully institutional animal care and use committees will request ethical oversight for these studies.
Conclusion
Research can impact animal welfare, and although the Essential 10 are not the only factors that can affect welfare, they provide relevant information that allows reviewers and readers to identify possible welfare risks and assess the validity of the results. There could be cases where not meeting the ARRIVE Essential 10 criteria results in information that is not valid nor reproducible, cases in which it would not be able to morally justify the use of animals.
Adhering to higher ethical standards when conducting animal experiments implies, among other things, duly informing agreement with guidelines and regulations regarding the use of animals for scientific purposes. Likewise, improving and standardising the criteria used in the peer review evaluation is of great relevance for ensuring the quality, validity, and transparency of the studies involving farm animals. Our study showed that, although journals have ethical guidelines for reviewers and authors, it is not wholly effective. Therefore, we encourage editorial committees and reviewers to be stricter about the acceptability of manuscripts when the researchers do not report basic criteria, such as the ethical statement.
Footnotes
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]()
