Abstract
Concerns around comprehension and recall of consent information by research participants have typically been associated with low health and research literacy levels. In genomics research, this concern is heightened as the scientific and ethical complexities of genetics research, such as biobanking, genetic susceptibility, data sharing, and incidental findings may be more difficult for potential research participants to understand. However, challenges to research participants’ comprehension of consent information may be compounded by factors beyond health and research literacy levels. To identify factors that may impact research participants’ understanding and recall of consent information, we designed a qualitative study to explore whether participants enrolled in a tuberculosis genetics study (TBGEN-Africa) in Cameroon understood the objectives of the study, the risks and benefits and certain key aspects of the study such as biobanking and data sharing. The results showed that research participants had limited understanding and/or recall of the TBGEN-Africa study goals and methods. Some participants were of the opinion that TBGEN-Africa was not a genetics study because tuberculosis is not an inheritable condition. Factors that may have hindered understanding and/or recall of study information are diagnostic misconception (research participants consider research as part of medical diagnosis), and information overload and situational vulnerability (consent at a time of physical and emotional distress). There is a need for improved practices to support research participants’ understanding of consent information in genetics studies including designing the consent process in ways that minimize psychological distress and diagnostic/therapeutic misconception.
Keywords
Introduction
Research participants’ understanding of study information is a major ethical issue in genomics research in Africa (Kengne-Ouafo et al., 2016; Ngwenya et al., 2020; Tindana et al., 2012; Traore et al., 2015). Generally, low levels of genetic literacy amongst study populations in African countries have been associated with limited comprehension of consent information in health research and genetics studies (Kengne-Ouafo et al., 2016; Mboowa and Sserwadda, 2019). On the other hand, diagnostic misconception (i.e. where potential research participants consider research as part of medical diagnosis) is also emerging as a common and troubling phenomenon in genomics research (Masiye et al., 2017; Mweemba et al., 2019). Put together, these studies suggest that complex scientific concepts, low literacy levels and in certain cases, some immediate research benefits, may be contributing to the limited understanding of consent information by research participants in genetics studies in Africa. Given that informed consent is the cornerstone of the ethics of health research involving human participants, it is important that researchers ensure that potential participants have an adequate understanding of the information provided during the consent process.
International health research ethics guidelines require that information about a research study should be provided to potential research participants in a manner that is clear and comprehensible, allowing them to make an informed decision on whether they would like to be part of the study or not. These guidelines also stipulate the sorts of information that should be provided to potential research participants, namely: the purpose of the study, the research procedures, anticipated risks and benefits of participating in the study, how confidentiality will be maintained and the rights of a research participant. Only after receiving and understanding such information are potential participants expected to willingly accept or decline to be part of the research. Comprehension of research information is therefore a necessary requirement for informed consent in health research. However, reports of limited comprehension, misunderstanding and recall of study information amongst research participants are not uncommon (Mystakidou et al., 2009; Pietrzykowski and Smilowska, 2021). Empirical studies across Africa regarding informed consent have highlighted that the complexity (concepts and terminology) of a study, low research and health literacy levels, and cultural factors may influence research participants’ understanding of consent information, as well as their decision to participate in the study (Afolabi et al., 2014; Molyneux et al., 2004). It has also been reported that persons who are: socio-economically disadvantaged, have limited access to healthcare, feel very sick, or just received a diagnosis of the disease under study, tend to be vulnerable when invited to be part of a study (Aliyu, 2011; Kalabuanga et al., 2016). This might be the case even when, as a group, they are not considered a vulnerable population or classified as such in national and international research ethics guidelines.
To identify ways by which informed consent for genetics research could be improved, we conducted a qualitative study (Glaser and Strauss, 2017) to explore factors that may impact on research participants’ comprehension or recall of study information for a Tuberculosis Genetics (TBGEN-Africa) study in Cameroon. TBGEN-Africa is a multi-site H3Africa project (H3Africa Consortium, 2014) that seeks to understand genetic variation in human susceptibility to tuberculosis. TBGEN-Africa has sites in four African countries, namely Cameroon, Ethiopia, Eritrea, and Sudan. In Cameroon, participant enrollment is taking place in two regions: The tuberculosis treatment centers in Buea, Limbe (Southwest Region) and Douala (Littoral region). The TBGEN-Africa study makes a good case study for exploring research participants’ understanding and recall of study information because as an H3Africa project, TBGEN-Africa used an informed consent template that was developed by the H3Africa consortium following engagement with different stakeholder groups (de Vries et al., 2015; Ramsay et al., 2014). The template (H3Africa ELSI WG, 2018) includes simplified text that researchers can use to explain concepts in genomics and biobanking.
Methods
This was a qualitative research study that used in-depth interviews and participant observation. The study was designed and led by TBGEN-Africa researchers as part of capacity building efforts (postgraduate training). All, but one (EGE), of the researchers are familiar with the study site (demographics, culture and way of life) either through their research activities, training and/or current residency. One of the authors (EY) is not directly involved in the TBGEN-Africa but was the thesis supervisor of the first author (AIM), allowing for an outside perspective and critique of the study, in particular its design, conduct, analysis and interpretation of the results.
Nineteen semi-structured, one-on-one in-depth interviews were conducted with participants who had been enrolled in the TBGEN Africa project site in Cameroon. The goal of the interviews was to explore participant’s understanding of the TBGEN-Africa study objectives, study procedures (including recall of information on sample and data sharing), and to identify factors that may influence their understanding of the consent information. The interviews were supplemented by direct observation (Phillips, 1996) of the consent process. Interviews were conducted at the Buea and Limbe enrollment sites, as these were the only sites that were enrolling participants at the time. Data collection was undertaken from 5th July to 15th September 2020.
Interviewees for this study were purposively selected (Ames et al., 2019; Emmel, 2013) from the larger group of consented TBGEN-Africa research participants. The criteria for selection were based on either our observation of the consent process and/or availability for an interview. The final sample size was guided by the principle of information saturation (Burmeister and Aitken, 2012; Guest et al., 2006). Interview questions were open ended (Rapley, 2001) and related to the information provided during the consent process. This included questions about their knowledge and awareness of the goals of research, understanding of genetics, TBGEN-Africa study objectives and procedures including sample and data sharing, risks and benefits of the study and concerns about study procedures. Interviews were conducted in English and Pidgin (a lingua franca widely spoken in the Anglophone regions of Cameroon). All interviews were audio-recorded and transcribed verbatim. Transcripts were checked for quality by one of the authors (AIM). Observation of the consent process was undertaken using a predefined observation guide with information about how the team explained the study objectives, study processes and procedures to the prospective participants.
Data analysis
NVivo-12, a qualitative data analysis software (QSR International, 2018), was used to facilitate thematic analysis (Braun and Clarke, 2012) of the interview transcripts. Data analysis started after the first two interviews. This was to allow for the identification of themes and patterns that were emerging from the data and to fine-tune the interview guide as necessary (Creswell and Poth, 2016). Following the first three interviews, a coding framework was developed by AIM through inductive thematic coding (Saldaña, 2016). The quality of the coding framework was checked by ABC. Discrepancies were resolved with two members of the research team (DEF and GHI). The established thematic framework was then used to code all 19 interview transcripts. New codes were added to the coding framework based on analysis of subsequent interviews. To describe the overall study findings, we used verbatim anonymized quotes from the interviews to support the narratives.
Findings
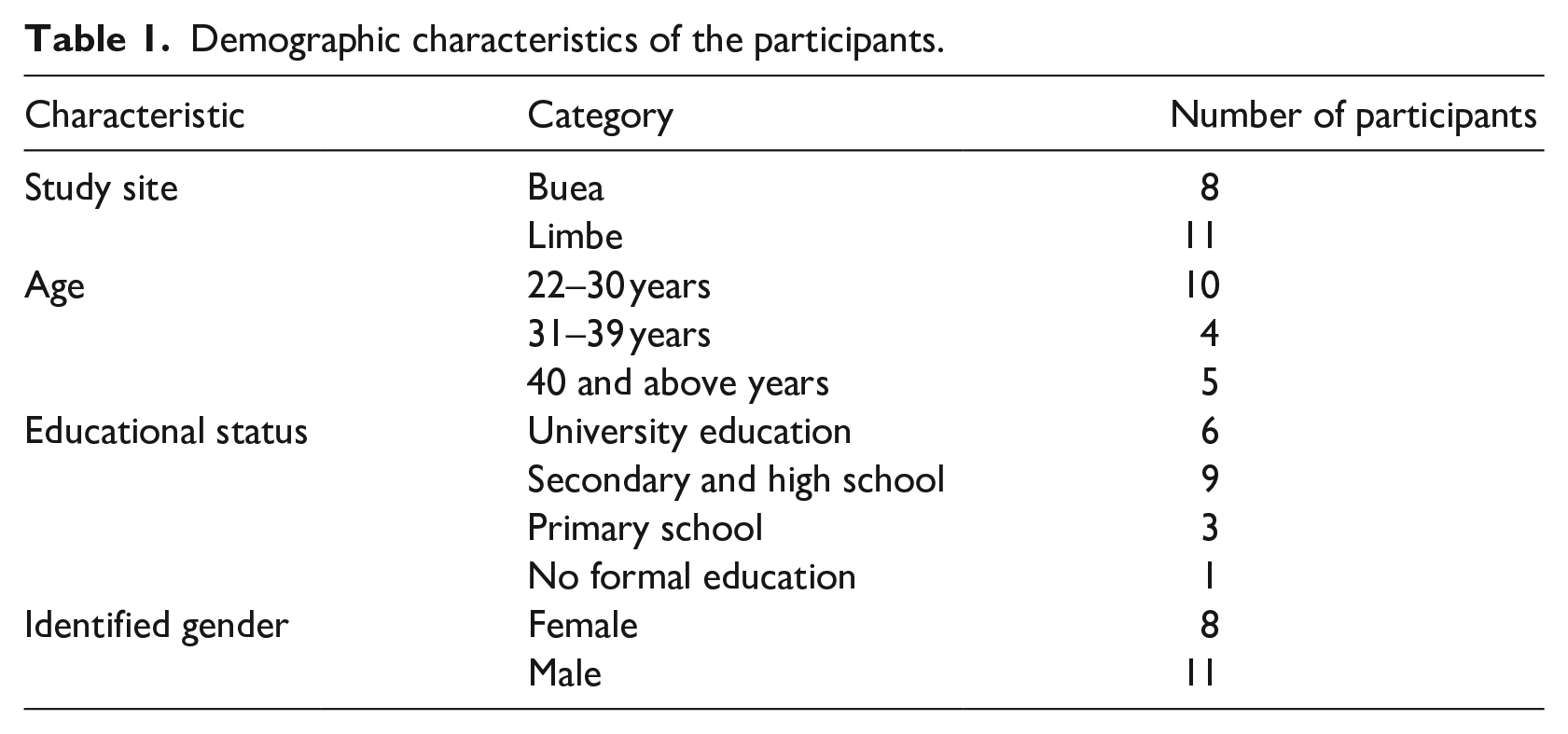
Most of the participants (see Table 1) were between the ages of 22 and 30 years and had completed secondary school education (about 12 years of formal education) and indicated that it was their first time participating in research.
Demographic characteristics of the participants.
Understanding of the objectives of TBGEN
The main objective of TBGEN-Africa is to investigate the influence of host genetics and pathogen factors on susceptibility to tuberculosis. All participants acknowledged that they had been informed of the TBGEN objectives. However, when asked say or explain what the objective of TBGEN is, the majority said it was to identify or develop new treatments or vaccines for TB.
“They want to find drugs for TB patients better than the ones we are taking now. This one is for six months but they want to reduce the duration, may be into 2 or 3 months. Rather than taking the drugs for six months, taking and getting cure in 3 months will be good improvement. So, they are doing the research for that purpose” (P16).
Only one interviewee gave an explanation that was close to the objectives of TBGEN-Africa, although they did not mention genetics in their explanation. When probed on what enabled their understanding, they explained that when they had received their TB diagnosis, a medical doctor explained to them that certain factors may make some individuals more prone to getting TB compared to others.
“Patients get more prone to TB when they are HIV positive. You are likely to be prone to a particular TB like that. The research will help them to understand how to prevent TB in the society. And also, they can have a better understanding of the cause” (P15).
Before the interviews, we had hypothesized, based on previous studies, that research participants’ understanding of TBGEN objectives may be hampered if they had limited basic knowledge of genetics. Therefore, in the interviews we asked the participants if they had come across the word genetics, before and if so, what it means to them and if they thought TBGEN study had anything to do with genetics. Only a few participants defined genetics and when they did, they referred to inherited diseases or physical traits that are common in a family.
“Yes, it [gene] is something you can pick it from your family or their blood. You inherit it from your family. I know that particular family members go through the same type of disease. Mother gets ill of a disease and then that pass to the children and to their children’s children. It continuous like that and that is the inheritance issue” (P09).
In our observation of the consent process, we noted that research staff used the term “inheritance” when they explained the study objectives. However, all but one participant expressed certainty that TBGEN was not a genetics study because TB is not inheritable.
“No, the study was about TB only. Isn’t it? They told me so. I don’t even know what you mean by genetics” (P16).
Understanding and recall of TBGEN research procedures about bio-banking and data sharing
A core component of TBGEN-Africa is sample and data sharing. Samples from the TBGEN Cameroon site will be sent to Ethiopia for genetic sequencing and long-term storage in a biobank. Genetic data will also be deposited in the European Genome Archive as is required of all H3Africa projects. To this effect research participants must provide consent for (1) their samples to be sent to the TBGEN-Africa Hub in Ethiopia, (2) long-term storage of their samples in a biobank in Ethiopia, and (3) samples and data sharing with other researchers. All these were included in the consent form. Also, in our observations of the informed consent process, the TBGEN enrollment team often explained to potential participants that samples would be sent to Ethiopia for further analysis. However, in responding to the interview questions, many interviewees expressed surprise that their samples, and associated data, would be shared with researchers outside of the Cameroon site as well as sent to the TBGEN hub (in Ethiopia) for genetic sequencing and biobanking.
“To share how? Outside Buea? I did not take that into consideration, and they did not tell me that they will send it [the samples] to Ethiopia, or anywhere outside Buea. I should be aware that they can take my sample outside the country.. . . It was good to tell me that they want to take it somewhere else, out of the country. It is only now that you are mentioning to me, and I am hearing from you that they may send it to another country which not everyone likes it” (P12).
When participants expressed surprise that their samples were to be sent to Ethiopia for genomic sequencing and biobanking, we asked if they would have been reluctant to consent to the study if they were aware that their samples would be sent out of Cameroon, and also shared with other researchers. Some of the participants had an overall positive attitude toward sample and data sharing, while others were indifferent. Those who expressed support for data and samples sharing said it was important for the common good.
“They can research on it for whatever sickness or for whatever years. For example, if their lab does not have enough equipment, they can do it with another site. They said they will send it [the samples] to Ethiopia, they can send. They can also research on it whatever they want. What matters is they should find out what really, they need from it. And that findings can help many other people” (P17).
Interviewees who did not want their samples to be shared did not provide specific reasons other than that it was a personal choice.
“They [researchers] told me that they will process the samples in Buea. But now you are telling me that the study is among four countries and that they can send my samples outside Buea, or the country [Cameroon]. Mine should be conserved. It shouldn’t be sent to any country; it is only because of my personal reservations” (P15).
In terms of the study risks and benefits, it is stated in the informed consent forms that the main risk to potential participants is that in rare instances, the study outcomes may inadvertently reveal information on TB susceptibility for ethnic groups, potentially leading to ethnic group harm or stigma. When asked about the risk of participating in the study, all participants said there was no study-related risk and that even if there was a risk, the clinicians would have advised against participating in the study.
“I don’t see any risk or side effect, but the medical doctors know more than me, and they don’t allow any risk to their patients.” (P09).
A major worry for participants, however, was the volume of blood (25 ml) that was being collected for the study.
“I was scared when I saw the blood, they took from me, because as I said earlier, I was vomiting blood for weeks and then they collected that much blood. I said hey!! I have been vomiting blood and now they took this much’. I fear these days of running short of blood. The next day the nurse came, and she said your blood level is 10, but two days before it was 12. So, I was scared” (P14).
During observation of the consent process, we noticed that the sample collection tubes were pre-filled with reagents or stabilization fluid. Therefore, when the collected blood samples were transferred to the tubes, it may have given research participants the impression that the volume of blood collected was high. This concern was communicated to the enrollment team, leading to a change of strategy whereby the healthcare workers show participants the pre-filled tubes prior to drawing their blood.
Factors that affected participants’ comprehension and/or recall of TBGEN study information
The TBGEN-Africa consent forms are written in relatively simple English and most of the participants we interviewed had at least a secondary/high school-level qualifications. At the time of enrollment, the study was also explained to potential participants in either English or Pidgin. We noticed that TBGEN-Africa staff also asked potential participants if they understood the study objectives and had any questions. Many participants confirmed that they understood and, in some cases, asked questions mainly around the amount of blood to be collected. In the interviews, we wanted to gain insights into factors that might have contributed to the limited understanding or recall of the study objectives and procedures amongst participants. Three main themes emerged (Table 2) from the data analysis: diagnostic misconception, information overload and situational vulnerability.
Factors affecting comprehension of consent information in the TBGEN study in Cameroon.

Diagnostic misconception
Diagnostic misconception can occur when potential research participants consider their participation in research as an opportunity to receive medical diagnosis of their health conditions (Knerr and Ceballos, 2015; Masiye et al., 2017). In the TBGEN-Africa study, prospective participants were adults who had received a positive TB diagnosis. They were aware of their TB diagnosis at the time they were invited to join the study. Nonetheless, some mistook TBGEN-Africa as either part of their clinical care or an opportunity to get more information about their TB diagnosis and other diseases. In these cases, they were not aware that TBGEN was a research project. As one participant described, the project was helping patients to check for diseases other than tuberculosis.
“They take the samples to check it if I have other sicknesses [other than TB]. They will test it in their lab and know what sicknesses are disturbing the people . . . so they want to help the patients by checking their samples” (P19).
Another participant said that a key motivation for consenting to the study was the opportunity to get cost-free laboratory testing for other diseases “They [the research staff] took the samples to examine in their lab. They will examine the blood to find out if there are any bacteria, parasite or worms. And they said all the lab test is for free. So, they will examine my all sample for free. If they discover anything in my blood, they will tell me. This is one of the reasons for why I decided to accept to participate” (P07).
The few participants who were aware that TBGEN-Africa was a research project were also able to distinguish between the goals of health research and clinical care. Most of the participants described research as discovery based and healthcare as treatment-oriented. For example, one participant explained that in routine healthcare you will get a specific diagnosis and treatment prescription. While in research, “Researchers do not give you treatment, but they can tell you more about yourself from the sample they collected from you, what problems you have” (P15).
“In the hospital, a doctor may tell you that you have malaria and typhoid, but researchers can discover more beyond that, on the same particular patient. That is what I understand by research” (P07)
Information overload
A second factor that may have hindered participants’ comprehension or recall of the consent information is information overload. When we observed the consent process, we noticed that at any one time, the TBGEN-team were simultaneously collecting multiple samples for other studies that were different from or sometimes related to the parent study (TBGEN-Africa). For instance, in addition to the blood and sputum samples collected for the study, some staff or students were, in parallel, collecting additional samples for use in other studies about malaria and filaria. These studies were mainly for postgraduate students, some of whom were part of TBGEN-Africa. Therefore, during the consent process, participants were given information for different studies at the same time, and those who agreed to participate signed separate consent forms for each of the studies. This resulted in information overload and may explain why some interviewees said that TBGEN-Africa was a study about malaria or filaria.
“The researchers collected many samples: blood, cough [sputum], stool and urine to discover if I have worms, filaria and malaria” (P06, male, 40).
On the other hand, malaria and filaria are endemic in Cameroon and researchers at the University of Buea, including some members of the TBGEN team, have, over time, been involved in several community-based studies on filaria and malaria. This might also have contributed to perceptions that TBGEN was a study on malaria and/or filaria.
Situational vulnerability
Situational vulnerability was the third major factor that affected research participants’ comprehension of the TBGEN-Africa study information, and to an extent, their decision to participate in the study. Situational vulnerabilities are health and other social factors that may influence one’s ability to make free and informed decisions (Biros, 2018; Bracken-Roche et al., 2017). In the interviews, some participants mentioned that at the time they were approached to join the TBGEN-Africa study, they were still struggling to accept their positive TB diagnosis and therefore were psychologically stressed. Consequently, it was difficult to understand the information in the consent documents and even to follow-up on the verbal explanations provided by the TBGEN staff.
“That day, it was not really a right time for me to read all the information. I was even refusing to accept the disease. I didn’t read the paper. For me at that particular day, I was even a bit sad. Because I was told that my sample was TB positive, I mean that I have TB. So, I was just rushing on things to finish and go, though, I gave my samples voluntarily” (P09).
Some participants also reported that they were experiencing physical and emotional pain because of their ill-health and therefore could not focus on specific details. As a result, although the researchers may have explained the details of TBGEN, the timing was not good and this may help to explain why they could not remember information about the study.
“The day they [the research field staff] came to me, I did not like to talk to anybody. My mind, at that time, was not working well. I was feeling pain on my chest. So, I could not really understand what the lady [the researcher] was explaining entirely. I just focused on some points that interested me” (P07).
The above quotes offer examples of specific situations that may make potential participants vulnerable when consenting to a study.
Discussion
Inadequate understanding of informed consent by research participants has been reported in several studies and remains a major concern for initiatives that aim to promote the ethical conduct of health research (Afolabi et al., 2014; Tam et al., 2015). Our findings raise similar concerns. The findings of this qualitative study show that some research participants in a TBGEN Africa study in Cameron had limited understanding and/or recall of the study objectives and procedures. Participants’ comprehension of consent information was influenced by diagnostic misconceptions, information overload and situational vulnerability. While diagnostic misconception is emerging as a common phenomenon in genetics research studies (Masiye et al., 2017; Nobile et al., 2013), the impact of situational vulnerability and information overload on participants’ comprehension of is largely unexplored. The findings of our study suggest that persons should not be invited to participate when they have just received a diagnosis for a serious medical condition. In addition, further research would be helpful for achieving balance between the presentation of certain study benefits (e.g. free laboratory testing) and diagnostic and therapeutic misconception.
To overcome the challenges of consent comprehension in genetics research in Africa, initiatives such as the Human Heredity and Health in Africa (H3Africa) consortium (H3Africa Consortium, 2014) have suggested that researchers may explain genetics using inheritance or common traits in families. This suggestion seems appropriate as many participants in our study also explained genetics using inheritance. However, it may not apply to concepts like genetic susceptibility to infectious diseases. In the TBGEN-Africa study, some participants mentioned that tuberculosis was not inheritable and therefore TBGEN-Africa could not possibly be a genetics study. Similar studies in Ghana (Tindana et al., 2012) and Mali (Traore et al., 2015) have reported that research participants’ understanding of genetic susceptibility to infectious diseases can be challenging and using words like inheritance (a more familiar term) to explain genetics in the consent process may be more confusing. A study in Ethiopia about genetic susceptibility to Podoconiosis recommended the use of analogies such as “why do some people get sick with common cold while some others do not, despite living in one household?” (Addissie et al., 2016; Tekola et al., 2009). This approach could be adopted and tested for genetics studies about infectious diseases. There is also a need to develop a repertoire of phrases that could be used in explaining genetic susceptibility to infectious diseases.
The challenge of information overload in the TBGEN-Africa may have been perpetuated by simultaneous consenting for other sub studies nested within the Cameroon site. In resource limited settings, it is not uncommon for research teams to accommodate mini projects in a single field study or research enrollment drive. Most times this is unavoidable as research teams have to make maximum use of limited research resources to support postgraduate students. While this may be a cost effective approach to supporting postgraduate research, it may affect consent comprehension especially if consent for several projects are taking place in parallel. There is a need to seek other ways of supporting project-related postgraduate research without compromising consent procedures. A more feasible approach might be to organize the process such that defined mini projects are covered in the research ethics clearance and informed consent for the main project. While we understand that students and early career researchers need to seek separate research ethics clearance for their respective projects, research ethics committees should consider opportunities for postgraduate students to request waivers for seeking consent if sample and data collection for their projects is already covered by research ethics clearance and approved consent documents for the main study.
Conclusion and recommendations
Informed consent is the centerpiece of ethical research involving human participants; it is predicated on an adequate understanding of what the research is about and on the genuine freedom and willingness of participants in the process. It is therefore important that researchers ensure that these minimum requirements are met when they seek consent to enroll in a study. Extra precautions are also required when the study involves vulnerable populations and/or populations who may be medically or socially disadvantaged. Nonetheless, vulnerability is a broad concept (Grady, 2009; Schroeder and Gefenas, 2009) and our study shows that while the TBGEN- Africa study population are not generally categorized as vulnerable, the timing for enrollment (right after obtaining a distressing diagnosis of TB) may have induced vulnerability for some individual participants. We recommend that researchers, when designing and conducting their studies, consider that while a study population may not be defined as vulnerable, certain instances may introduce situational vulnerability, which may in turn have an impact on the informed consent process. For example, as well as diagnostic or therapeutic misconception, inviting patients to join a study just after they have been diagnosed with the health condition under investigation might lead people to participate without adequate knowledge of the research and associated risks.
Consent is considered valid if potential participants are adequately informed of the aims, methods, risks, benefits, funding, any conflicts of interest, affiliations of the researcher, relevant aspects of the study and that their participation is voluntary. This raises the question of whether consent is still valid if a separate research study, undertaken after participant enrollment, reveals that some participants may have misunderstood the goals of a study, do not remember what the research was about, and are unable to recall essential components of the study such as data and sample sharing. In the TBGEN-Africa study, we consider the consent provided by participants to be valid, even though our study showed limited understanding or recall of consent information. This is because the TBGEN-Africa consent form contained all essential information about the study and was easy to understand; the information was explained verbally to all potential participants; and prospective participants were only enrolled into the study after they confirmed they had understood the information and were willing to participate. Nonetheless, given the reports of inadequate understanding of consent information in health research, there is a need for improved practices to support participants’ understanding of study information. For example, persons responsible for obtaining consent could ask potential participants open ended questions about the study objectives and procedures to verify understanding of the study information. Another approach might be to use a “teach-back” technique whereby the research participants are asked to explain the information provided to a member of the study team. While we acknowledge that these approaches may be time and resource consuming, it is vital that every effort is made support research participants’ understanding of consent information in ways that minimize psychological distress and diagnostic/therapeutic misconception.
Footnotes
Acknowledgements
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported through H3Africa [H3A-18-003]. The Human Heredity and Health in Africa (H3Africa) is a program of the Alliance for Accelerating Excellence in Science in Africa (AESA) platform. AESA is a funding, agenda-setting and program management initiative of the African Academy of Sciences (AAS), the African Union Development Agency (AUDA-NEPAD), founding and funding global partners, and through a resolution of the summit of African Union Heads of Governments. H3Africa is supported by the National Institute of Health (NIH), Wellcome Trust, and African Society of Human Genetics (AfSHG). NSM receives support from an NIH-National Heart, Lung, and Blood Institute grant to the Sickle Africa Data Coordinating Center-SADaCC [grant number: U24HL135600]. The views expressed herein are those of the author(s) and not necessarily those of the funders, AESA and her partners.
Availability of data and materials
The data presented in this article are available from the corresponding author on reasonable request.
Ethical approval
Research ethics clearance was obtained from the Ethics Review and Consultancy Committee of the Cameroon Bioethics Initiative (CBI/457/ERCC/CAMBIN). All participants provided written informed consent.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]()
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported through H3Africa [H3A-18-003]. The Human Heredity and Health in Africa (H3Africa) is a programme of the Alliance for Accelerating Excellence in Science in Africa (AESA) platform. AESA is a funding, agenda-setting and programme management initiative of the African Academy of Sciences (AAS), the African Union Development Agency (AUDA-NEPAD), founding and funding global partners, and through a resolution of the summit of African Union Heads of Governments. H3Africa is supported by the National Institute of Health (NIH), Wellcome Trust, and African Society of Human Genetics (AfSHG). NSM receives support from an NIH-National Heart, Lung, and Blood Institute grant to the Sickle Africa Data Coordinating Center-SADaCC [grant number: U24HL135600]. The views expressed herein are those of the author(s) and not necessarily those of the funders, AESA and her partners.
