Abstract
Dynamic consent is increasingly recommended for longitudinal and biobanking research; however, the value of investing in such systems is unclear. We undertook a rapid review of the benefits and challenges of implementing dynamic consent by searching five databases (Ovid Medline, Ovid Embase, Scopus, Web of Science, Cumulative Index to Nursing and Allied Health Literature – CINAHL) for articles published up to May 2023 that report on participants’ or researchers’ experience of dynamic consent. From 1611 papers screened, 12 met inclusion criteria. Guided by thematic analysis with an inductive approach, we synthesised 31 benefits and 8 challenges. Benefits included: enhanced participant experience through improved consent management and tailoring; greater participant engagement and retention through increased autonomy, trust and communication; reduced costs and burden and increased accessibility and inclusivity. Participants and researchers also valued additional features that dynamic consent platforms facilitate such as two-way communication and return of research updates. The main challenges included the digital divide and consent fatigue. The papers gave recommendations to mitigate these challenges, for example by supplementing with other communication tools and allowing a broad consent approach, respectively. Overall, dynamic consent was described as a valuable consent approach with many benefits and some surmountable challenges. Most included literature was qualitative, so further research is needed to quantify the impact of dynamic consent on recruitment, retention, and participant experience. Further long-term investigations are necessary to explore whether participants want to and do change their consent over time, as well as the impact of dynamic consent on participant privacy.
Introduction
For consent to be valid, it must be informed. This is stipulated in legal and ethical frameworks worldwide. For example, the European Union’s General Data Protection Regulation (GDPR) requires consent be ‘freely given, specific, informed and unambiguous’ (Regulation (EU), 2016). Regarding participation in human research specifically, the Australian National Statement on Ethical Conduct in Human Research states that to consent, research participants must understand ‘the purpose, methods, demands, risks and potential benefits of the research’ (National Health and Medical Research Council et al., 2018: 16).
The current practice for consent in open science research and open-ended research projects, such as biobanks, is to use broad consent, whereby participants consent to a general framework allowing their data and samples to be used in future research projects; which may still be unspecified at the time of initial consent (Steinsbekk et al., 2013). However, agreeing to unknown future research arguably does not satisfy the requirements of informed consent (Mikkelsen et al., 2019) whereby participation is founded on sufficient information and understanding of the research and its implications (National Health and Medical Research Council et al., 2018), potentially causing ethical and regulatory issues. Additionally, there has more recently been a shift away from paternalistic top-down governance towards greater participant autonomy and research involvement (Steinsbekk et al., 2013).
Harnessing technological advances to implement a ‘dynamic consent’ approach may address this issue of informed consent in longitudinal studies. Dynamic consent allows participants to review and change their consent choices over time via a personalised digital interface. This may improve researcher-participant communication, and participant autonomy and engagement (Prictor et al., 2020). We note that dynamic consent is more relevant for fulfilling ethical obligations for consent to research participation, compared to consent for processing of personal data, which is likely to remain largely governed under other legal bases such as public interest (Dove, 2018).
Dynamic consent is a fairly new approach, with much grey literature and commentary on the topic, but little empirical research. Consequently, developing a dynamic consent platform may appear daunting for researchers, especially if they are unsure the benefits will outweigh the costs. There is a need to collate evidence on whether dynamic consent is a viable solution, how to implement it, and what challenges may arise.
This rapid review aimed to synthesise literature on the benefits and challenges of dynamic consent as reported by those who have implemented it as researchers or used it as participants. We sought to provide recommendations for researchers of longitudinal studies considering implementing a dynamic consent model.
Methods
Systematic reviews are necessary to synthesise evidence for comprehensiveness, accuracy and research quality; for example, when determining an effect size for clinical guidelines. However, rapid reviews offer timely literature synthesis to aid decision making for stakeholders (Moons et al., 2021). Given the largely qualitative nature of the data we sought on researchers’ and participants’ experiences with dynamic consent, we chose a rapid review approach.
Where applicable, we conducted the review following the 2012 Enhancing transparency in reporting the synthesis of qualitative research (ENTREQ) guidelines (Tong et al., 2012).
Eligibility criteria
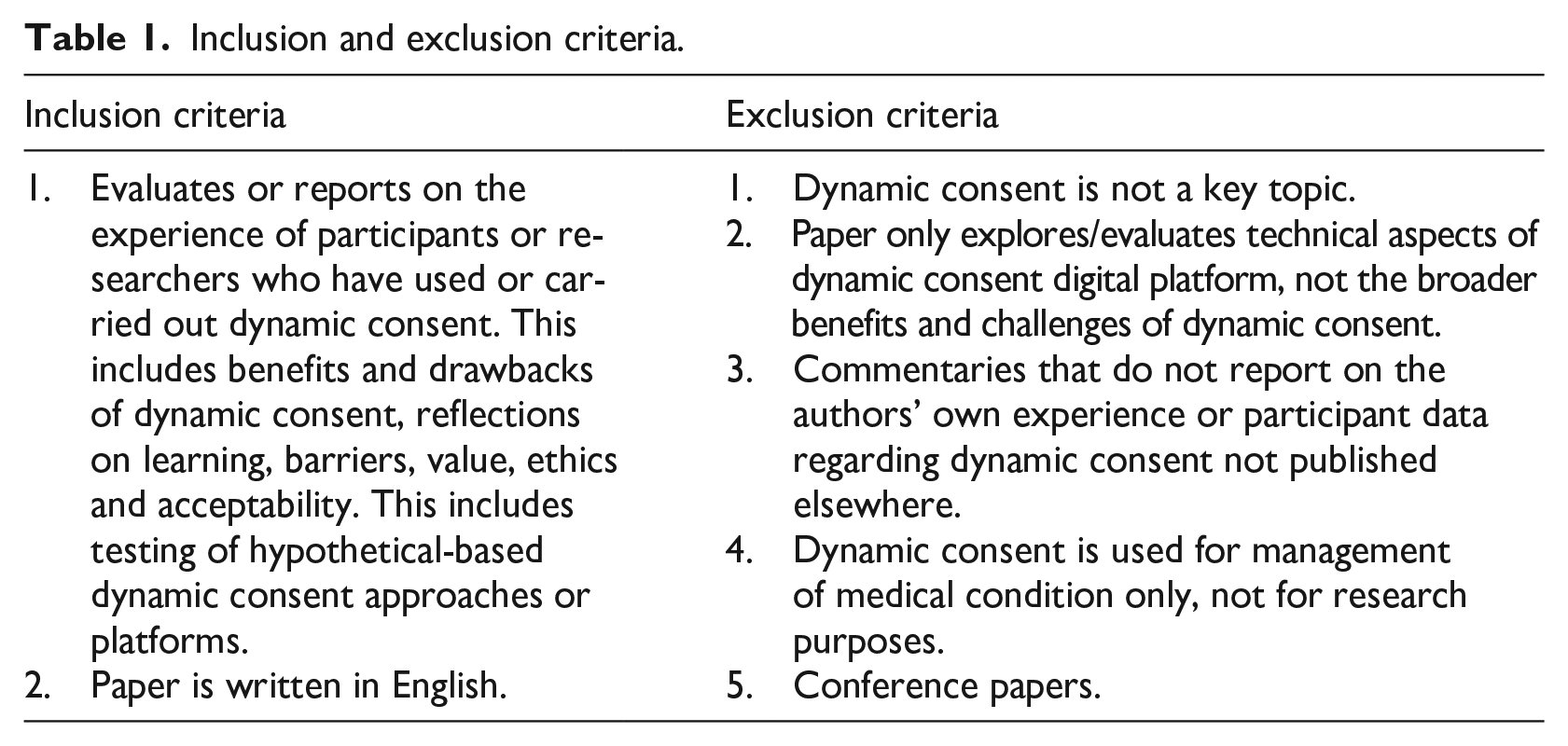
The inclusion and exclusion criteria are outlined in Table 1. As there is much commentary published but little empirical evidence, we chose to only report on the experience of participants and researchers who had used or implemented dynamic consent. Due to the small number of studies using dynamic consent in practice, we included papers reporting on indirect evaluations such as focus groups where community members trialled and gave feedback on a dynamic consent interface and workshops seeking researchers’ experiences implementing/maintaining dynamic consent. We refer to these collectively as hypothetical-based studies. We excluded papers reporting primarily on technical aspects of a dynamic consent platform (as these would not generalise to other studies) and papers reporting on dynamic consent used only to manage medical conditions. Conference presentations were excluded due to insufficient detail to extract benefits and challenges.
Inclusion and exclusion criteria.
Information sources and search strategy
The first author conducted the search, screening, extraction and data synthesis stages. The second author contributed to screening, extraction and data synthesis as described below. The literature search was completed on May 21–22, 2023, using Ovid Medline, Ovid Embase, Scopus, Web of Science and Cumulative Index to Nursing and Allied Health Literature (CINAHL) for their broad coverage of human research fields. Results were limited to English, with no date restrictions. The search was pre-planned using comprehensive search strategies. Full search strategies for all databases including Boolean operators are listed in the supplemental material.
Selection process
All retrieved articles were imported into Covidence (Veritas Health Innovation, 2023) and duplicates were removed.
Inter-rater agreement was assessed using Cohen’s kappa statistic on Idostatistics (Scarpellini, 2022), and interpreted according to Landis and Koch (1977). The first author screened all papers by title and abstract, and the second author dual screened 20% of papers, with 81.8% agreement (Cohen’s
Data collection
The first author extracted study characteristics, benefits and challenges of dynamic consent from each paper. The second author checked the data extraction for accuracy and completeness, resolving conflicts through discussion with all authors.
Synthesis methods
Data synthesis was guided by thematic analysis with an inductive approach (Mihas, 2023) to extract themes. The first author utilised open coding with a line-by-line approach (Strauss and Corbin, 2004). Extracted data for each open code were then pooled and compared to refine, combine or subdivide existing codes and create new codes. For example, initial codes for overcoming geographical barriers and translation into different languages were combined into ‘accessibility and inclusivity’, which was then grouped with other codes into the overarching theme of Information Technology.
Results
Search results
Figure 1 outlines the search results. The search yielded 1611 articles. After removing duplicates, 792 articles remained. Titles and abstracts were screened for eligibility, leaving 62 articles for full text review, with 12 articles ultimately included in this review.
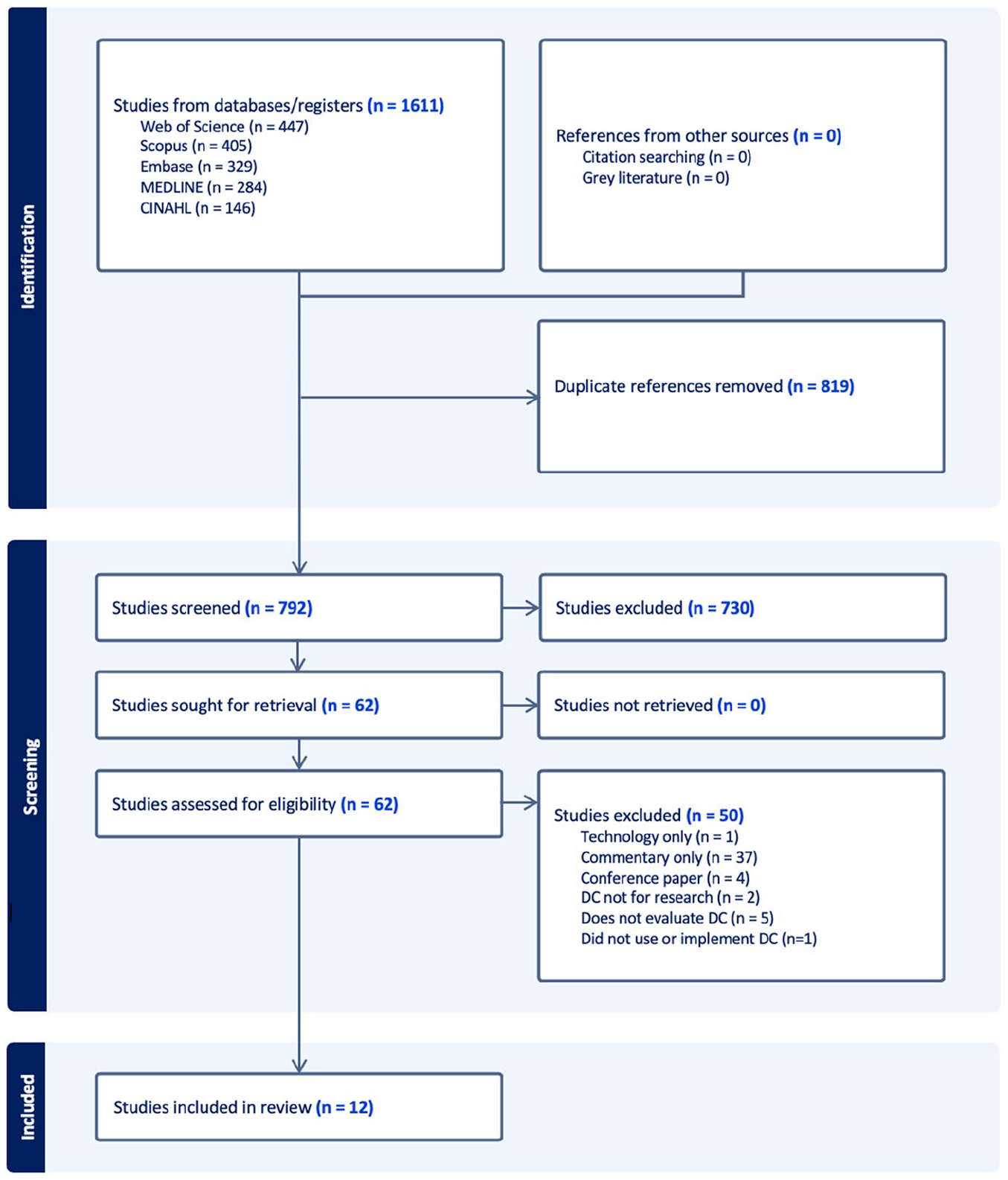
PRISMA diagram.
Study characteristics
Table 2 collates the characteristics of included papers. Publication dates ranged from 2015 to 2022. The 12 papers were published from seven countries, with five from the UK.
Study characteristics.
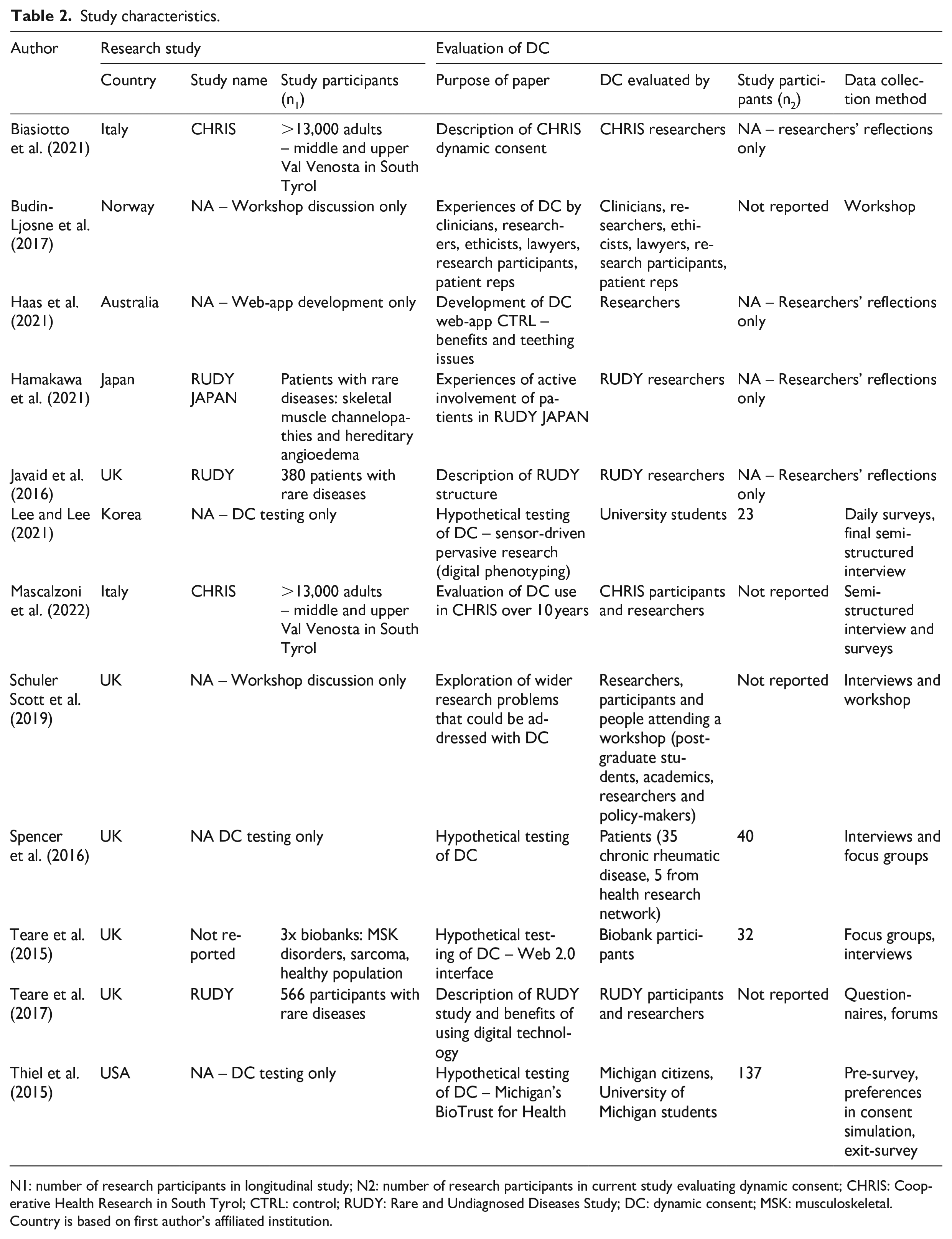
N1: number of research participants in longitudinal study; N2: number of research participants in current study evaluating dynamic consent; CHRIS: Cooperative Health Research in South Tyrol; CTRL: control; RUDY: Rare and Undiagnosed Diseases Study; DC: dynamic consent; MSK: musculoskeletal.
Country is based on first author’s affiliated institution.
Five articles applied dynamic consent in a longitudinal study (Biasiotto et al., 2021; Javaid et al., 2016; Hamakawa et al., 2021; Mascalzoni et al., 2022; Teare et al., 2017). The remaining articles were hypothetical-based, and provided attitudes and perceptions as opposed to empirical evidence: four were hypothetical user testings (Lee and Lee, 2021; Spencer et al., 2016; Teare et al., 2015; Thiel et al., 2015), two reported on general experiences of researchers and/or participants who had used dynamic consent (Budin-Ljosne et al., 2017; Schuler Scott et al., 2019) and one article described developing a dynamic consent web-app (Haas et al., 2021). Data collection methods included interviews, surveys/questionnaires, focus groups, workshops, forums and preferences inputted into the consent simulation. Perspectives were gathered from: researchers and participants of studies using dynamic consent, university students, the public, existing biobank participants trialling dynamic consent, clinicians, ethicists, academics, lawyers and patient representatives.
Benefits and challenges
Table 3 displays the benefits and challenges by theme. Notably, studies often evaluated different dynamic consent platforms, hence, the reported benefits and challenges may be specific to each platform.
Benefits and challenges by theme.

Indicates that the benefit or challenge is an additional feature of the digital platform used for dynamic consent.
Besides managing and modifying participants’ consent, the dynamic consent platforms were often used for other functions not integral to dynamic consent; hereon referred to as ‘additional features’ (asterisked in Table 3). Inclusion of these features depends on the platform used and the study’s design and available resources. Examples include return of individual results and data interoperability, which would require allocated staff time and budget, or technical software alignment and data formats respectively.
Participant experience
Consent management
The most common benefit reported was participants’ ability to make granular and dynamic consent choices (Biasiotto et al., 2021; Budin-Ljosne et al., 2017; Haas et al., 2021; Hamakawa et al., 2021; Javaid et al., 2016; Lee and Lee, 2021; Mascalzoni et al., 2022; Spencer et al., 2016; Teare et al., 2015, 2017; Thiel et al., 2015). Participants valued the possibility of changing their consent and many had a strong preference for active consent. It was also noted researchers could benefit from easier consent for secondary use of data or samples in further studies, improving the recruitment process and increasing participant involvement and engagement (Biasiotto et al., 2021; Haas et al., 2021; Javaid et al., 2016; Spencer et al., 2016; Teare et al., 2015, 2017).
Mascalzoni et al. (2022) reported their dynamic consent platform improved participant understanding of research, using interactive questions to test comprehension prior to consent. The digital platforms also provided a record of consent for participants to check what consent preferences they had set (e.g., who can access their data, use their data/samples, and how the data are being used) and to revisit and remind themselves about the study information (Javaid et al., 2016; Mascalzoni et al., 2022; Spencer et al., 2016; Teare et al., 2015, 2017).
Teare et al. (2015) reported a perceived advantage of controlling the setting and timing of providing consent. This was contrasted with paper-based consent approaches whereby patients may be asked for consent in stressful periods, such as right before a surgery or in a clinic.
A few articles recognised the likely benefits of a more nuanced approach to consent withdrawal (Lee and Lee, 2021; Mascalzoni et al., 2022; Schuler Scott et al., 2019). Instead of a binary approach with broad consent, participants can, for example, choose what data continues to be collected, whether already collected data can continue to be used, or whether any communications continue. Relatedly, it was noted that dynamic consent could allow participants to change their consent preferences regarding how their data would be handled if they die or become incapacitated (Mascalzoni et al., 2022).
Several papers cited that dynamic consent allowed participants to maintain their autonomy by offering transparency and the ability to change consent over time (Biasiotto et al., 2021; Javaid et al., 2016; Mascalzoni et al., 2022) or would likely do so (Budin-Ljosne et al., 2017; Lee and Lee, 2021; Spencer et al., 2016; Teare et al., 2015). This autonomy in combination with receiving research updates may empower individuals to feel like active participants.
In two hypothetical-based studies, participants with low level privacy concerns preferred not to use the dynamic consent platform or to use it passively (Lee and Lee, 2021; Spencer et al., 2016). Lee and Lee (2021) suggested that participants felt constrained in their consent decisions due to concerns that revoking consent would affect research quality or cause personal financial loss. Participants felt obligated to maintain their initial consent, potentially reducing their autonomy to make consent choices based on their true preferences.
Tailoring
Although papers were cognisant of consent fatigue as a potential challenge of dynamic consent, only one included paper, a hypothetical-based study, reported this (Lee and Lee, 2021). Consent fatigue describes a cognitive burden due to having excessive granular consent choices to make (Kaye et al., 2015). In Lee and Lee’s (2021) sensor-driven digital health research, participants struggled trawling through a list of data items to select the data collection types they wanted to deactivate. Contrastingly, three hypothetical-based articles reported that their digital platforms allowed participants to select from pre-determined consent guides or to use broad consent (Haas et al., 2021; Teare et al., 2015; Thiel et al., 2015), reducing the burden of too many consent choices.
A few articles cited the benefit of using their platform to tailor information to meet the literacy, interest levels and motivation of individuals, including providing participants with the right amount of information at the right time (Mascalzoni et al., 2022; Teare et al., 2015, 2017). Some articles reported that their platform allowed participants to control their contact preferences (e.g. method, frequency, alerts and contact reasons) as a beneficial additional feature (Biasiotto et al., 2021; Haas et al., 2021; Mascalzoni et al., 2022; Teare et al., 2015, 2017).
Engagement and retention
Most articles noted the possibility of ongoing communication between participants and researchers as a beneficial additional feature of the digital platform (Biasiotto et al., 2021; Budin-Ljosne et al., 2017; Haas et al., 2021; Hamakawa et al., 2021; Javaid et al., 2016; Mascalzoni et al., 2022; Teare et al., 2015). This two-way ongoing communication may allow more specific responses or information than traditional paper materials, may not be limited by space or time and may improve participant engagement and trust.
Several articles reported an additional feature whereby platforms facilitated updating participants on research outcomes to which they had contributed (Biasiotto et al., 2021; Javaid et al., 2016; Mascalzoni et al., 2022; Teare et al., 2017). Similarly, many hypothetical-based studies also noted the perceived advantage of updating participants on research outcomes (Haas et al., 2021; Spencer et al., 2016; Teare et al., 2015). Overall, these updates helped improve participant interest, engagement, and trust.
Several researchers noted that the dynamic consent platform, and its additional features, improved transparency and trust (Biasiotto et al., 2021; Hamakawa et al., 2021) or would be likely to do so (Haas et al., 2021; Lee and Lee, 2021; Spencer et al., 2016). Participants in one study (Spencer et al., 2016) noted a lack of transparency and awareness around data use with current paper-based approaches, and researchers framed this as dynamic consent opening the ‘black box’ around consent and reuse of data, helping to mitigate concerns around institutional trustworthiness (Spencer et al., 2016: 8). Separately, Biasiotto et al. (2021) and Hamakawa et al. (2021) noted that the communication on the dynamic consent platform led to increased trust.
The digital dynamic consent platform may improve participant retention (Budin-Ljosne et al., 2017; Mascalzoni et al., 2022). Mascalzoni et al. (2022) credited this to the granular consent choices enabling participants to stay involved in research, instead of withdrawing from the study completely when dissatisfied with specific consent aspects. Meanwhile, Budin-Ljosne et al. (2017) attributed this to the ongoing communication enabled by the digital platform. A further potential additional feature of the platforms was allowing participants to update their contact details. Haas et al. (2021) also noted that dynamic consent platforms could improve participant scientific literacy.
The digital nature of the platform improved recruitment through a lower burden on research staff, as well as interoperability between organisations and nations (Javaid et al., 2016); and facilitated engagement through the ability to change consent and provide research updates (Biasiotto et al., 2021; Mascalzoni et al., 2022).
Information Technology (IT) platforms and systems
While one article noted that initial start-up costs for dynamic consent platforms may be high (Budin-Ljosne et al., 2017), they speculated these would decrease as online solutions become normalised. Indeed, together with four other papers, they noted that overall, dynamic consent could reduce costs and/or increase time efficiency (Budin-Ljosne et al., 2017; Javaid et al., 2016; Mascalzoni et al., 2022; Teare et al., 2015, 2017). Budin-Ljosne et al. (2017) noted that, with study information available online, less time may be required for one-on-one discussions with potential participants. Similarly, Teare et al. (2015) posited that patient driven participation with dynamic consent may decrease the costs of re-contact for biobank research. Javaid et al. (2016) reported a reduction of staff hours spent on recruitment and data entry, lowering research administration costs.
Multiple researchers noted a dynamic consent platform can improve accessibility and inclusivity as a beneficial additional feature, through overcoming geographical, language and time barriers (Hamakawa et al., 2021; Javaid et al., 2016; Mascalzoni et al., 2022; Teare et al., 2017). It may reduce travel time and costs for participants by allowing access from any location, and aid language translations for linguistically diverse communities, potentially increasing participant recruitment, including internationally. The digital platform may also enable processes and communication free from time constraints (e.g., time zones or scheduling).
As technology is always progressing, Javaid et al. (2016) noted the additional feature of their dynamic consent platform being built to support future developments. Four articles noted the additional feature of the digital platform allowing a multimedia approach for educating participants on the research studies (Haas et al., 2021; Mascalzoni et al., 2022; Teare et al., 2015, 2017).
Two papers noted the digital nature of dynamic consent may make way for the additional benefit of interoperability between organisations in different sectors or nations, enabling research collaborations (Budin-Ljosne et al., 2017; Teare et al., 2017). Teare et al. (2017) noted their electronic dynamic consent platform would allow groups in different countries to modify and implement the software to reach wider populations, exemplified by the translation of RUDY (Rare and Undiagnosed Diseases Study) into RUDY JAPAN (Hamakawa et al., 2021).
Some articles noted that dynamic consent can provide more reliable data storage and a tracked overview of consent changes (Budin-Ljosne et al., 2017; Hamakawa et al., 2021). Similarly, a few papers noted the electronic nature of dynamic consent may facilitate the beneficial additional feature of easier data transmission or sharing (Budin-Ljosne et al., 2017; Hamakawa et al., 2021; Javaid et al., 2016). Budin-Ljosne et al. (2017) noted the digital platform may link to laboratory information systems or electronic patient records to automatically update consent choices across systems for electronically tagged data/samples. Likewise, Javaid et al. (2016) noted that linkages can be made with other research databases. Hamakawa et al. (2021) noted that the digitisation of this data would likely surpass the speed and reach of paper-based models.
A few papers cited the ‘digital divide’ as a challenge (Hamakawa et al., 2021; Spencer et al., 2016; Thiel et al., 2015). The ‘digital divide’ describes a gap between those who access IT and those who do not, due to various factors including material (lack of devices and internet), poor digital literacy and social and economic determinants (Srinuan and Bohlin, 2011). Spencer et al. (2016) found 25% (10/40) of participants expressed apprehension using a digital consent platform due to limited device access or low confidence using IT. Meanwhile, Thiel et al. (2015) found that approximately 2% of participants were reluctant to use a digital platform due to lack of fixed internet access. Hamakawa et al. (2021) noted some participants had difficulty completing registration due to IT unfamiliarity, but this was overcome using other communication tools (e.g. letters, telephone).
Another challenge observed was difficulty verifying participant identity if there is no face-to-face interaction (Javaid et al., 2016; Thiel et al., 2015). Thiel et al. (2015) found that questions they used to verify participant ID, including social security number and banking details, were received poorly by participants. Javaid et al. (2016) found digital systems made it difficult to confirm participant ID and diagnosis, as few clinicians returned participant diagnosis information.
Health data records
Most papers mentioned the additional feature of the digital platform being designed to collect participant health data (Biasiotto et al., 2021; Budin-Ljosne et al., 2017; Haas et al., 2021; Hamakawa et al., 2021; Javaid et al., 2016; Lee and Lee, 2021; Teare et al., 2017), including through questionnaires or sensor-driven wearable devices (Lee and Lee, 2021). These offer faster and easier alternatives to paper-based collection (Hamakawa et al., 2021; Teare et al., 2017); however, self-reported data may not match the quality of standardised measures administered by trained staff (Teare et al., 2017). Platforms may provide the option to record participants’ family history (Teare et al., 2017) or for individual medical care in consultation with physicians through self-recorded symptoms, medication, clinical events or management as beneficial additional features (Budin-Ljosne et al., 2017; Hamakawa et al., 2021; Javaid et al., 2016; Teare et al., 2017).
A few articles noted the return of individual clinical results to participants as a beneficial additional feature of their digital platform (Biasiotto et al., 2021; Haas et al., 2021; Mascalzoni et al., 2022).
Ethics and governance
Some articles reported that dynamic consent could ensure compliance with ethical and/or regulatory requirements for research participation (Biasiotto et al., 2021; Haas et al., 2021; Mascalzoni et al., 2022), for example, by ensuring ‘specific consent and renewed consent for new studies’ (Mascalzoni et al., 2022: 1392). Similarly, one paper noted that dynamic consent may improve privacy management (Lee and Lee, 2021); however, Budin-Ljosne et al. (2017) posed the potential challenge that research ethics committees may be less familiar with dynamic consent approaches.
Discussion
This review found value in implementing a dynamic consent approach in longitudinal research, with researchers and participants reporting many benefits to be gained from dynamic consent. While challenges exist, these are not overwhelming and there are ways to mitigate them, as described below.
Benefits
Overall, for participants, dynamic consent may facilitate the ability to make dynamic and granular consent choices, have a record of consent, be in control of their contact preferences and have greater transparency of their data and sample uses; equipping them to feel more informed, involved, empowered and trusting in research.
Dynamic consent may afford researchers the ability to obtain consent for further studies, provide nuanced withdrawal approaches and improve recruitment and retention. The digital nature of dynamic consent may increase time efficiency, reduce administration costs and allow researchers to reach a higher standard for the ethical and legal requirements of informed consent, not realised by broad consent.
Furthermore, participants and researchers appreciated the additional features that a digital dynamic consent platform may facilitate (e.g., data collection, return of research updates and ongoing communication). Consequently, we recommend researchers explore implementing platforms including these additional features.
We note that the value of some benefits depends on the mode of initial consent; that is, whether it is a traditional face-to-face consent or via the dynamic consent platform without any requirement for face-to-face interaction. For example, if initial consent is face-to-face, this may negate some benefits in timing, accessibility, efficiency, and cost; while conversely, enhancing ID verification processes.
Challenges
While several challenges were reported, these were not insurmountable, and several authors posed ways to anticipate and mitigate these, as discussed below.
Digital divide
The digital divide results from poor accessibility for people without IT devices or knowledge (Chang et al., 2004). This may be due to a range of social, geographic and economic determinants (Bezuidenhout et al., 2017) or personal choice to not engage (Goulden et al., 2017). Although the digital divide is a commonly identified potential challenge of dynamic consent (Prictor et al., 2018; Steinsbekk et al., 2013), we note that as research shifts toward digital formats, this issue becomes more prevalent and is not exclusive to dynamic consent. Our review found that researchers were cognisant of this and implemented mitigation strategies; resultingly, only three groups observed this issue, two being hypothetical-based studies (Spencer et al., 2016; Thiel et al., 2015). Several papers recommended reducing the impact of the digital divide and exclusion of participants by supplementing with traditional methods of consent and communication (e.g. paper-based consent, mail/newsletters, phone calls, in-person support) (Biasiotto et al., 2021; Budin-Ljosne et al., 2017; Haas et al., 2021; Hamakawa et al., 2021; Javaid et al., 2016; Mascalzoni et al., 2022; Spencer et al., 2016). While maintaining a hybrid approach between digital and other methods may require more resources, we recommend it to ensure accessibility and inclusivity.
Consent fatigue
While consent fatigue from the burden of excessive consent choices is often cited as a potential challenge of dynamic consent (Prictor et al., 2018; Steinsbekk et al., 2013), only one (hypothetical-based) study found this to be a challenge (Lee and Lee, 2021). Mascalzoni et al. (2022) argued that when properly applied, dynamic consent respects participants’ choices and contact preferences rather than overwhelming them. We recommend researchers utilise the ‘tailoring’ properties of dynamic consent to mitigate consent fatigue; by providing broad consent or pre-determined consent guides as options and allowing contact preference selection.
Other challenges
While passive usage of the platform was framed as a challenge by some, it could also be viewed as participants exercising their autonomy (i.e. choosing broad consent) and only an issue when participants feel obligated toward passive use or are unable to use the platform. However, researchers could promote active consent management by leveraging multimedia features to induct participants and support their autonomy and proper use of the platform for both dynamic consent and additional features such as data collection and communication.
While initial costs for implementing dynamic consent may be high, they are expected to decrease as online solutions become more common, and platform sharing or national dynamic consent platforms emerge (Budin-Ljosne et al., 2017). Additionally, costs may be offset by lower research administration costs later (Javaid et al., 2016). Likewise, as dynamic consent becomes more common, ethics committees will gain familiarity and can collaborate with researchers to establish approval criteria (Budin-Ljosne et al., 2017).
Other considerations
In this section, we note important considerations for implementing dynamic consent that did not clearly emerge as benefits or challenges in the included literature.
Privacy and security
While only Lee and Lee (2021) reported increased privacy as a benefit, and none reported it as a challenge, many highlighted the importance of data privacy and security in dynamic consent implementation. Researchers and participants expressed concerns about inappropriate data sharing or leakage (to third parties, employers or insurance companies) in research generally (Schuler Scott et al., 2019; Spencer et al., 2016; Teare et al., 2015; Thiel et al., 2015). However, participant privacy concerns were generally reported as low (Lee and Lee, 2021) and were outweighed by the benefits of data sharing for medical research (Spencer et al., 2016). Further, dynamic consent offers participants control over the level of risk they are comfortable with (Schuler Scott et al., 2019; Spencer et al., 2016; Teare et al., 2015). Based on their findings regarding hesitancy with ID verification, Thiel et al. (2015) proposed that researchers need to balance security/privacy with ease of use and accessibility. While not reported as a challenge in this review, researchers must ensure the dynamic consent platform has the necessary security to safeguard the privacy of patient information and their consent options.
Engagement over consent choices
Participants valued dynamic consent, but the reviewed papers lacked sufficient data to quantify how frequently participants vary their consent. Mascalzoni et al. (2022) reported that only 22 participants changed their consent from 2011 to 2018, however it remains unclear whether this represents all changes among the >13,000 participants. Instead, many studies alluded to participants valuing the engagement provided by the dynamic consent platform over the ability to change their consent preferences (Spencer et al., 2016; Teare et al., 2015). Future research should quantify the frequency of consent changes over time.
Governance
Soulier (2019) criticised dynamic consent for its potential to impair biobank governance. However Biasiotto et al. (2021) and Mascalzoni et al. (2022) argued that oversight bodies (researchers, ethics boards and access committees) maintain authority over evaluating research value and ensuring regulatory compliance; and participants’ increased autonomy through dynamic consent operates within these clear governance frameworks. Our review found no evidence that dynamic consent threatens governance structures.
Strengths and limitations
To our knowledge, this is the first review of studies of implemented dynamic consent approaches (including hypothetical-based studies), contrasting with papers that have theorised on possible benefits and challenges (Prictor et al., 2018; Steinsbekk et al., 2013). Our review employed a more comprehensive search (covering five databases) and applied stricter inclusion/exclusion criteria than previous research available.
Another strength is the inclusion of longitudinal studies, not just biobanks (where dynamic consent approaches have been most prominent), giving good representation of the contexts in which dynamic consent could be applied. Our study also followed ENTREQ reporting guidelines when applicable (Tong et al., 2012). Critical appraisal was excluded due to the heterogeneity of study types, and only partial dual screening means we may have missed other insights into dynamic consent.
Several limitations of this review and the included studies must be acknowledged. Firstly, the use of different dynamic consent platforms across studies likely influenced the reported benefits and challenges, especially for platforms with additional features. While including longitudinal studies of real-world dynamic consent was a strength, studies using other methodologies (e.g. user testing, workshop discussions and web-app development) provided perceptions and attitudes to dynamic consent as opposed to empirical evidence. User testing studies were also conducted in clinical settings rather than real-life home environments potentially influencing findings. Additionally, most studies (5 of 12) were from the UK, some studies had small samples sizes while others had significantly larger participant cohorts, all of which may impact the generalisability of findings.
Finally, most studies reported qualitative attitudes towards dynamic consent, except Thiel et al. (2015) who investigated user experience and factors related to participants’ willingness to join the biobank. Studies quantifying the impact of dynamic consent on recruitment, retention and other project success indicators are needed.
Future directions
We encourage researchers planning to implement dynamic consent to consider the benefits reported here and how to maximise those that may be of value to them. They should also consider applying the suggested strategies to mitigate reported challenges.
We further encourage researchers to proactively evaluate and report on their implementation and use of dynamic consent, quantifying its impact on factors such as recruitment, retention, withdraws, participant experience and research quality. This evaluation should be transparent and reproducible, encouraging different research groups to apply the same methodology where applicable. Establishing a standard evaluation approach of the impacts of dynamic consent would reveal the generalisability across different contexts and the contextual factors that influence this.
Further studies investigating dynamic consent used in practice are necessary, as they provide empirical evidence and hold more value than hypothetical-based studies.
Further investigation is needed on the impacts of dynamic consent models on privacy and security, and frequency of use. Studies should report quantitative data on how often participants change their consent and whether they are more likely to opt-in (provide consent) or opt-out (withdraw consent). Such analysis should consider the nature of the initial and subsequent research: as socially or scientifically controversial research may be expected to yield lower uptake and greater withdrawal and opt-outs regardless of the dynamic platform model. Once sufficient evidence is available, these findings can be compared to other studies without dynamic consent implemented.
Conclusion
This review found numerous benefits of dynamic consent platforms that not only include improved participant experience and consent management, but also additional features that the same digital platform can facilitate, for example, two-way communication and data collection. Furthermore, the challenges identified, such as the digital divide and consent fatigue, were relatively few and strategies were reported to mitigate these; for example, by supplementing with traditional forms of consent or providing tailoring options such as pre-determined consent guides or contact preference selection, respectively. Overall, we consider dynamic consent to be a valuable consent approach to implement.
Supplemental Material
sj-docx-1-rea-10.1177_17470161241278064 – Supplemental material for A rapid review of the benefits and challenges of dynamic consent
Supplemental material, sj-docx-1-rea-10.1177_17470161241278064 for A rapid review of the benefits and challenges of dynamic consent by Winnie Lay, Loretta Gasparini, William Siero and Elizabeth K Hughes in Research Ethics
Footnotes
Acknowledgements
We thank librarian Poh Chua for their support in developing the search terms and two masked examiners who provided feedback on an early version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research at the Murdoch Children’s Research Institute is supported by the Victorian Government’s Operational Infrastructure Support Program.
Ethical approval
The authors declare that research ethics approval was not required for this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
